Abstract
Mx proteins are a group of interferon-induced GTPases whose expression has been demonstrated in a number of human viral infections and in some idiopathic inflammatory diseases. In this study, the expression of Mx protein was evaluated in known viral, nonviral, and idiopathic encephalitides in the dog via immunohistochemistry using an antibody against human MxA. All 12 cases of confirmed viral encephalitis, including 7 cases of canine distemper, 4 cases of canine herpesvirus, and 1 case of rabies, were Mx positive. In canine distemper cases, staining was particularly strong and a variety of cell types were positive, including astrocytes, macrophages/microglia, and neurons. Immunoreactivity for Mx protein was evident in a few cases of nonviral infectious encephalitis, including neosporosis (1/1), Chagas disease (2/3), aspergillosis (1/2), and encephalitozoonosis (1/1). Consistent staining was observed in most cases of idiopathic encephalitis, including granulomatous meningoencephalomyelitis (7/7), necrotizing meningoencephalitis of pug dogs (6/7), and necrotizing encephalitis of the Yorkshire Terrier (3/3) and Maltese (1/1) breeds. Mx staining was negative in 5 normal dog brains; 3 cases of cryptococcosis; and single cases of blastomycosis, protothecosis, and bacterial meningitis.
Keywords
Introduction
Mx proteins are a group of ubiquitous dynamin-like GTPases that are induced by type-I interferons (alpha, beta) 10 and interferon lambda. 16 They were discovered 20 years ago in an inbred mouse strain that exhibited marked resistance to infection with influenza A virus. 12 Mx proteins have been subsequently demonstrated in a variety of cell types in a number of different laboratory and domestic animal species, including rabbits, rats, hamsters, horses, cattle, dogs, cats, pigs, and sheep. 11 Mx proteins have been shown to exhibit antiviral activity against a number of viruses and may be responsible for most of the antiviral effects of type-I interferons. 1 In humans and most other mammalian species, Mx proteins are restricted to the cytoplasm, whereas rodents possess both intracytoplasmic and intranuclear forms. 10 Two types of Mx protein are found in humans, MxA and MxB, with only MxA known to exhibit antiviral activity. 10 In the dog, 2 Mx proteins have been described, Mx1 and Mx2, with Mx1 being 76.9% homologous with human MxA. 27
It has been suggested that MxA expression in humans is potentially a diagnostic marker of viral infection. 4, 19 A wide variety of viruses are inhibited by MxA in vitro, including bunyaviruses, 2, 15, 33 rhabdoviruses, 36 paramyxoviruses, 35 hantaviruses, 14, 17 and orthomyxoviruses. 31 MxA expression has been demonstrated in human patients infected with a number of viruses, including hepatitis C virus, 8, 24 measles virus, 29 adenovirus, 4 cytomegalovirus, 4, 44 human immunodeficiency virus (HIV), 19 and herpes simplex virus. 6 In addition to viral infections, MxA expression has been demonstrated in human inflammatory conditions of unknown or suspected autoimmune etiology. 6, 7, 9, 20, 34, 43
The precise mechanism of MxA's antiviral activity is still unclear. Multiple mechanisms are likely involved, depending on the characteristics of the infecting virus. MxA has been shown to localize in the smooth endoplasmic reticulum and is capable of tubulating lipids. 1 Studies of Crimean-Congo hemorrhagic fever virus have revealed that MxA prevents the production of new infectious virus particles by interacting with the viral nucleocapsid and preventing replication of viral mRNA. 2 MxA inhibits LaCrosse virus by binding and mislocation of the viral nucleocapsid protein into smooth endoplasmic reticulum-associated large perinuclear complexes. 15, 33 Recent studies suggest that MxA's antiviral activity may also involve disruption of endocytosis 13 and regulation of calcium signaling by modulation of calcium channels. 23
Evaluation of Mx protein expression in natural diseases of animals has not been reported. The purpose of this study was to evaluate the presence of Mx protein in viral, nonviral, and idiopathic inflammatory diseases of the canine nervous system via immunohistochemistry, using an antibody against human MxA.
Materials and Methods
The cases used in this study were chosen from archival material at the College of Veterinary Medicine and Biomedical Sciences, Texas A&M University, and the Texas Veterinary Medical Diagnostic Laboratory, College Station, Texas. The MxA antibody used was a mouse monoclonal (M143) against human MxA of undetermined protein concentration (generously provided by Drs. Otto Haller and Georg Kochs, Department of Virology, University of Leipzig, Germany). The tissues were formalin fixed and paraffin embedded. Five-micrometer–thick sections were cut and mounted on silanized slides. Samples were deparaffinized in xylene and rehydrated in graded alcohols. Antigen unmasking was performed by heating slides in a coplin jar containing citrate buffer (pH 6) in a 97°C water bath for 20 minutes. The immunostaining procedure was performed using a labeled streptavidin-biotin system (LSAB2 System-HRP, DakoCytomation, Carpinteria, California, USA). Slides were incubated with the primary antibody (diluted 1°300) for 60 minutes at room temperature. The biotinylated secondary antibody was applied for 20 minutes. The presence of the antigen was detected with streptavidin conjugated to horseradish peroxidase. DAB (3,3′-diaminobenzidine) (DakoCytomation) was used in the color reaction at the site of the target antigen. On each tissue sample, the primary antibody was substituted with a universal mouse negative control. In each batch of cases, both positive control tissue (canine distemper-infected brain) and negative control tissue (normal canine brain) were used.
Results
The results are summarized in Table 1. All cases of confirmed viral infection were strongly immunoreactive for Mx protein. Particularly strong staining was seen in canine distemper cases, in which a variety of cell types were positive, including neurons, astrocytes, and macrophages/microglia. Mx staining was evident in both neuronal perikarya and neuronal processes. In the cerebellum, Purkinje cells and granular layer neurons often stained well (Fig. 1). Areas of greater cellularity, resulting from inflammatory cell infiltration and glial cell proliferation, stained more consistently than did less cellular areas. A similar intensity of staining and range of cell types as the distemper cases was seen in the single rabies case (Fig. 2). In addition to neurons, astrocytes, and macrophages/microglia, ependymal cells and some lymphocytes within perivascular cuffs also stained positively in the rabies case. In the brains of puppies with canine herpesvirus infection, glial nodules within the cerebral cortex were consistently stained (Fig. 3), and there was generally less intense staining of neurons than occurred with the distemper and rabies cases.
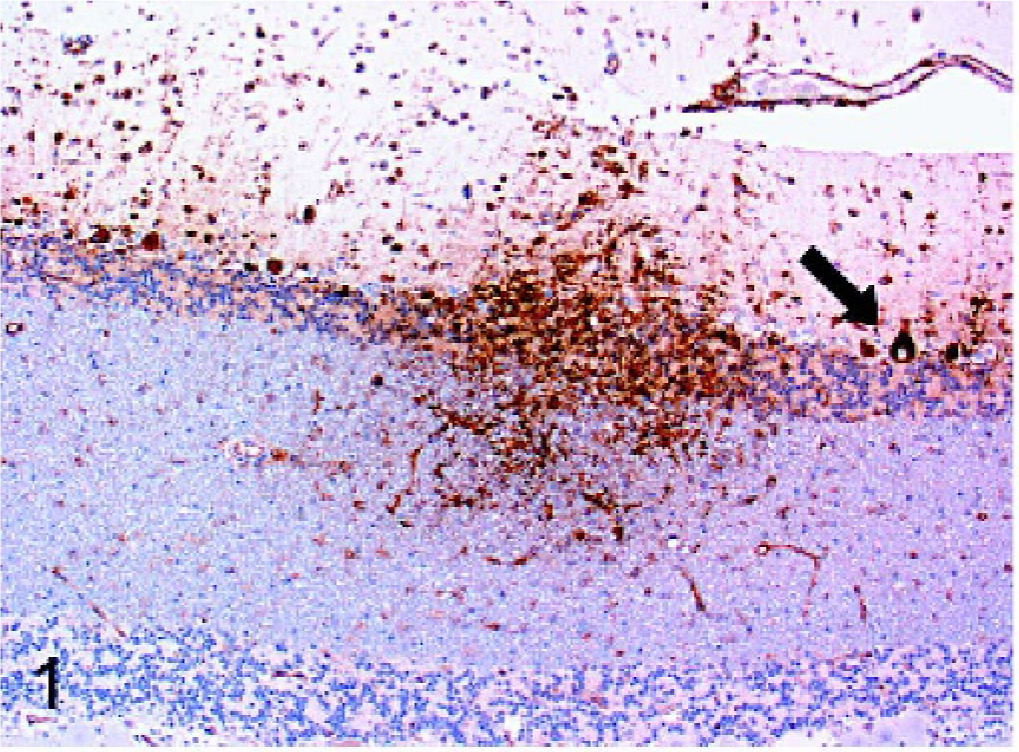
Cerebellum; dog. Intense Mx immunoreactivity is evident within an area of inflammation in the cerebellum of a dog with canine distemper virus infection. Strong intracytoplasmic staining of molecular layer and granular cell neurons is evident. Purkinje cells are also prominently stained (arrow). Strepavidin-biotin-peroxidase method.
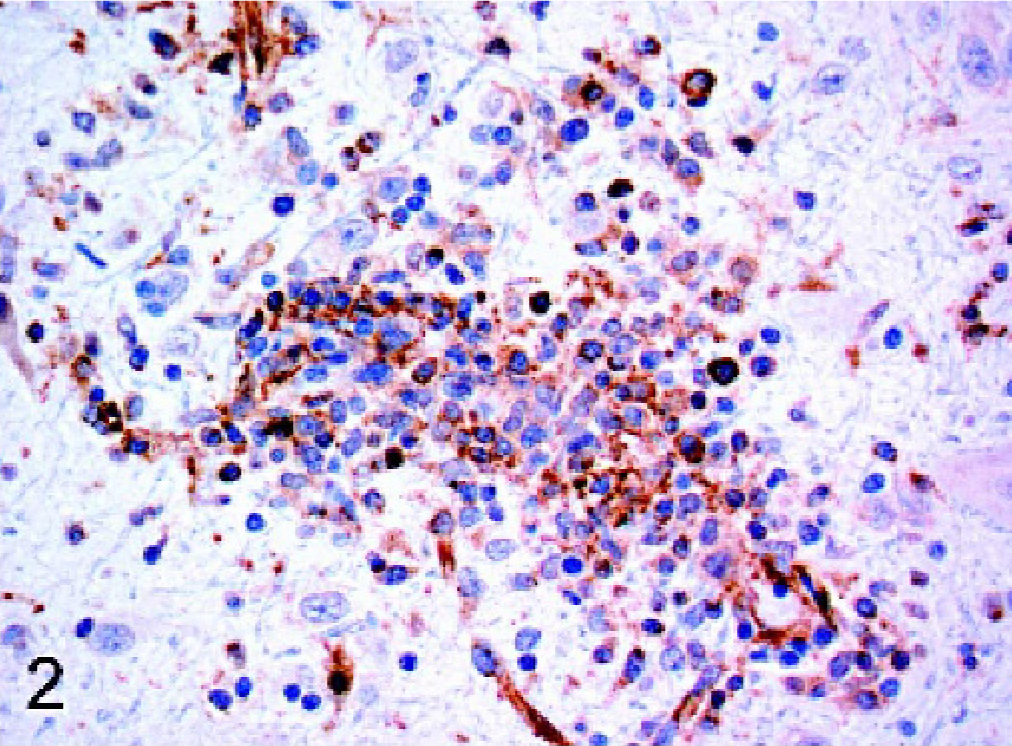
Brain; dog. Within a perivascular cuff in a dog with rabies, the cytoplasm of many cells, including macrophages and lymphocytes, is positive for Mx protein. Strepavidin-biotin-peroxidase method.
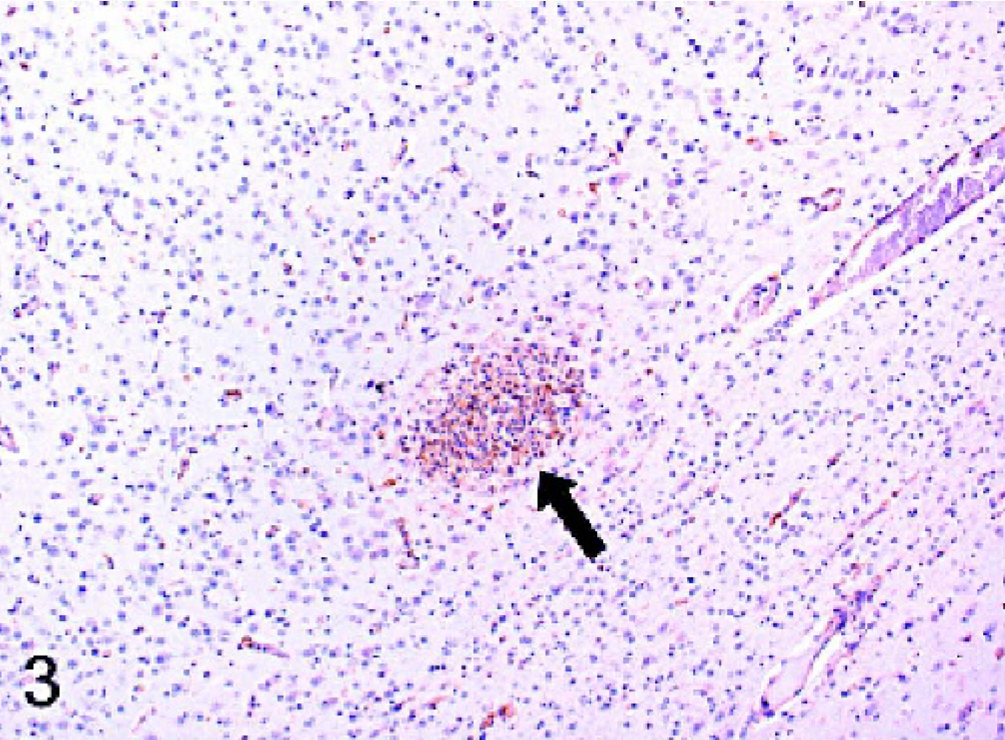
Cerebrum; dog. In the cerebral cortex of a puppy with canine herpesvirus infection, macrophages/microgilia within a glial nodule (arrow) have intense intracytoplasmic immunoreactivity for Mx protein. Strepavidin-biotin-peroxidase method.
Mx protein expression in canine encephalitides.
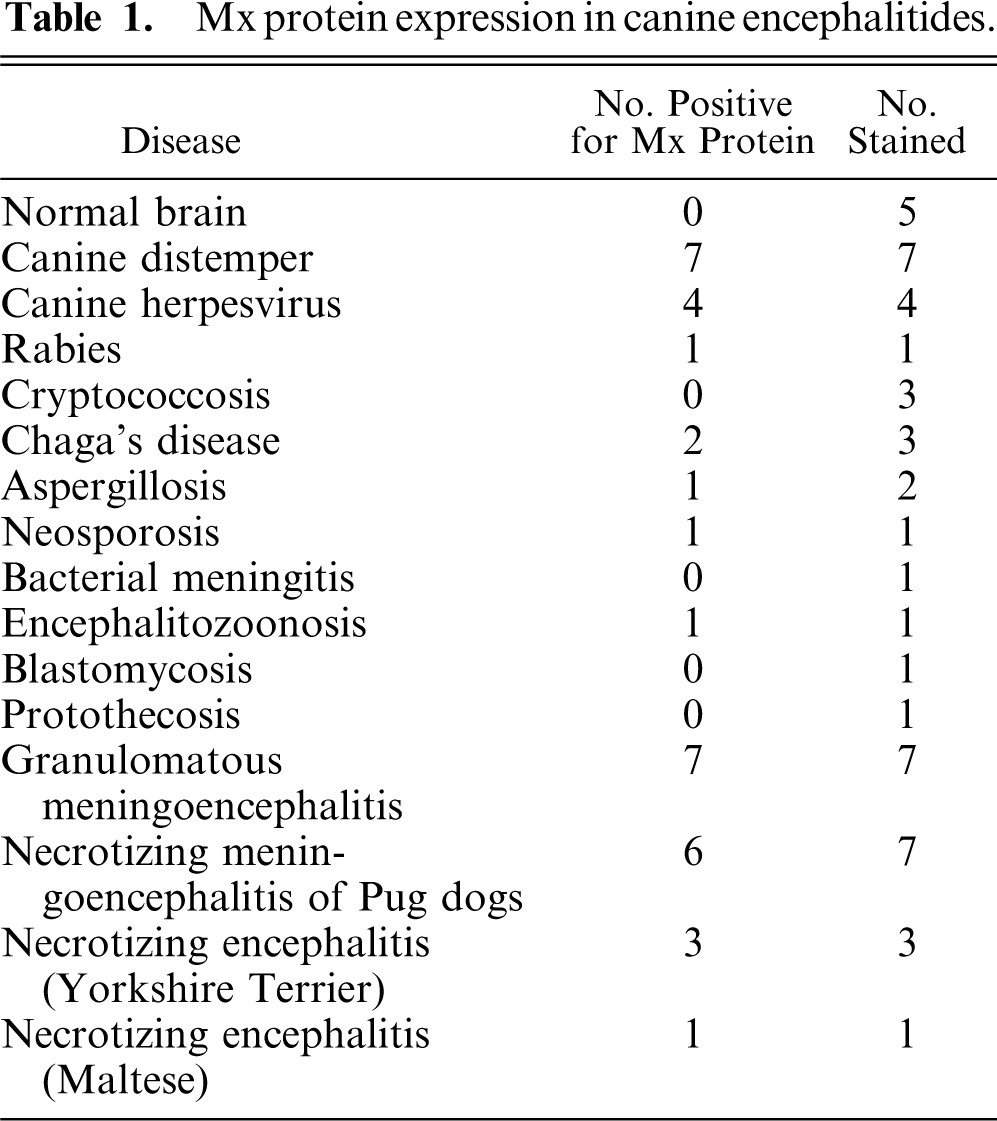
A few cases of nonviral infectious encephalitis were immunoreactive for Mx protein, including neosporosis, Chagas disease, aspergillosis, and encephalitozoonosis. Staining was relatively weak in the 2 positive Chagas disease cases and in the aspergillosis case. Strong staining was observed in the single cases of neosporosis and encephalitozoonosis. The neosporosis case was negative for canine distemper virus via immunohistochemistry. Positive staining was not observed in 3 cases of cryptococcosis or in single cases of blastomycosis, protothecosis, and bacterial meningitis.
Dogs with idiopathic encephalitides were consistently stained for Mx protein, with only 1 case staining negative. The evaluated diseases included granulomatous meningoencephalomyelitis (GME), necrotizing meningoencephalitis of pug dogs (NMEP), and necrotizing encephalitis in both the Yorkshire Terrier and Maltese breeds. In general, the staining in these cases was less widespread and intense than in the cases of confirmed viral infection. All 7 cases of GME were positive, but staining was generally mild and multifocal (Fig. 4). Positive cell types included macrophages within perivascular cuffs, reactive astrocytes, and occasional lymphocytes. All but 1 case of NMEP were Mx protein positive; staining was evident in both gray matter and white matter of the cerebrum and was most prominent in reactive astrocytes (Fig. 5). Neurons and macrophages/microglia also stained in NMEP dogs. The single case of necrotizing encephalitis in a Maltese dog was positive. The staining pattern in this case was similar to the NMEP cases, with conspicuous staining of reactive astrocytes (Fig. 6). All 3 cases of necrotizing encephalitis in the Yorkshire Terrier breed were positive; 1 case had prominent staining of reactive astrocytes within the cerebral white matter, and in 2 cases there was staining of lymphocytes within perivascular cuffs. Endothelial cell staining was observed in most of the cases examined in this study, including some of the normal dog brains.
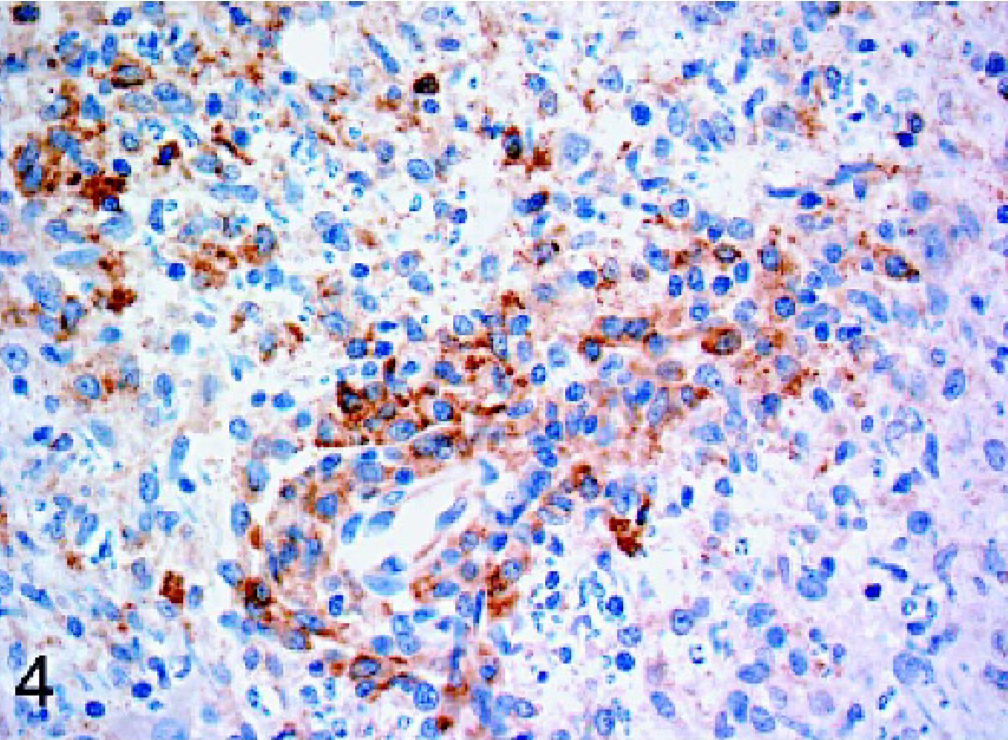
Brain stem; dog. Mx immunoreactivity is evident within the cytoplasm of perivascular macrophages of a dog with granulomatous meningoencephalitis. Strepavidin-biotin-peroxidase method.
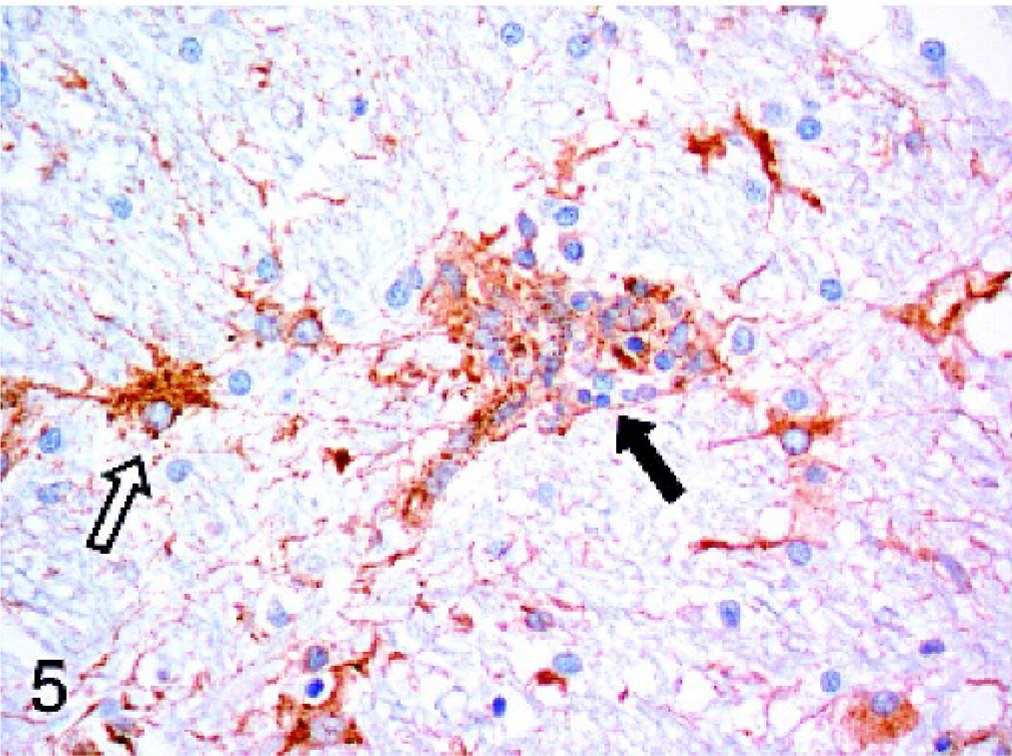
Cerebrum; dog. In the cerebral white matter of a Pug dog with necrotizing meningoencephalitis, macrophages/microglia within a glial nodule (solid arrow) are expressing Mx protein within the cytoplasm. Reactive astrocytes (open arrow) also have intracytoplasmic staining. Strepavidin-biotin-peroxidase method.
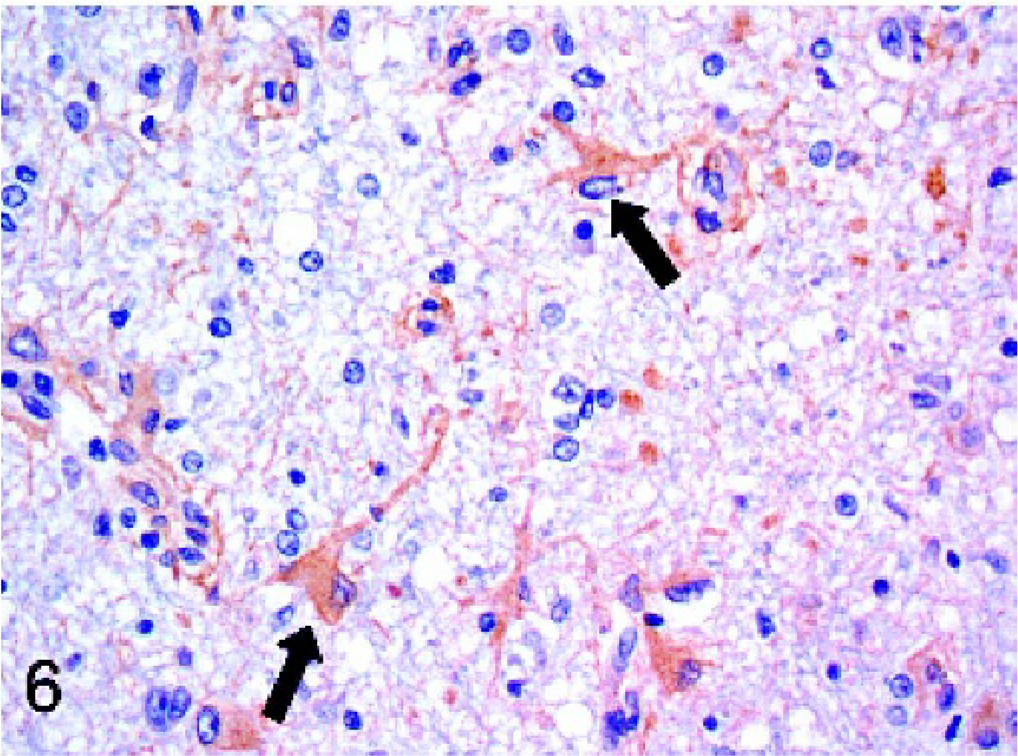
Cerebrum; dog. Immunoreactivity for Mx protein is evident within the cytoplasm of reactive astrocytes (arrows) in the cerebrum of a Maltese dog with necrotizing encephalitis. Strepavidin-biotin-peroxidase method.
Discussion
As expected, cases of known viral infection (canine distemper, rabies, canine herpesvirus) stained strongly positive for Mx protein. The strongest staining was seen in brains infected with canine distemper virus, a paramyxovirus. Demonstration of Mx immunoreactivity has been reported in brains of human patients with another paramyxovirus-induced disease, subacute sclerosing panencephalitis (SSPE), caused by measles virus. 19, 29 Limited studies also suggest that Mx protein production is involved in defense against viruses of Family Rhabdoviridae (rabies, vesicular stomatitis). A single human case of rabies stained positive for MxA, 19 and human MxA has been shown to inhibit mRNA synthesis of vesicular stomatitis virus. 36 Interestingly, canine Mx2 protein appears to exhibit antiviral activity against vesicular stomatitis virus, but canine Mx1, which shares greater homology with human MxA, does not. 27 The positive staining of canine herpesvirus-infected brains is not surprising. Although most studies of Mx proteins have focused on their activity against RNA viruses, herpesviruses and other DNA viruses are also capable of inducing MxA production. 6, 19, 26
The range of cell types that were immunoreactive for Mx protein in this study was greater than in the 2 reports of MxA expression in human encephalitides, including macrophages/microglia, neurons, and astrocytes. In the study by Lampe, Schneider-Schaulies, and Aguzzi, which included cases of HIV, SSPE, and a single case of herpes simplex infection, staining appeared to be restricted to macrophages and microglia. 19 It was shown that astrocytes were the primary cell type expressing MxA in SSPE. 29 In vitro studies have demonstrated MxA expression in mixed glial cell cultures after infection with measles virus, 18 oligodendrocytes, 38 and endothelial cells. 17, 25 Because there was mild staining of endothelial cells in some of the normal dog brains, the specificity of the endothelial cell staining in our study is questionable. Neuronal expression of Mx protein, which was especially prominent in cases of canine distemper infection, has not been previously reported. Receptors for type I interferons are found in a variety of different cell types; 37 therefore, the expression of Mx protein in several cell types in this and other studies is not surprising.
Positive Mx protein staining was seen in several cases of nonviral infectious disease that included aspergillosis, neosporosis, encephalitozoonosis, and Chagas disease. Evaluation of Mx protein expression in protozoal and fungal infections has received little attention, but defense against these infections may also involve type-I interferons. Type-I interferon activity has been documented in human patients with asthma exacerbated by pulmonary aspergillosis, 22 canine kidney cells infected with Neospora caninum, 28 and experimental murine Trypanosoma cruzi infection 41 and toxoplasmosis. 5 The innate immune system can be triggered by binding of pathogens to toll-like receptors (TLRs) on the surface of cell and endosomal membranes, which triggers a cascade of signaling proteins leading to production of interferons alpha and beta and other inflammatory cytokines. 3, 32, 40 For example, at least 1 member of the TLR family, TLR4, can both activate type-I interferon genes 40 and be responsive to both protozoal 30 and fungal 21 infection. In the MxA study by Lampe, Schneider-Schaulies, and Aguzzi, 1 case of human toxoplasmosis was positive for MxA; this patient was also HIV positive. 19 The study also included 3 cases of aspergillosis, 2 cases of cryptococcosis, and 2 cases of bacterial meningoencephalitis, all of which were negative for MxA.
Although it is tempting to speculate that Mx protein expression in idiopathic canine encephalitides, such as GME and NMEP, is evidence of viral involvement in these diseases, Mx protein production is apparently not restricted to viral infections. The known biologic activities of type-I interferons (alpha, beta), originally named for their ability to “interfere” with viral replication, has greatly broadened in recent years. Interferons alpha and beta are now thought to have diverse effects in both the innate and the adaptive immune responses, directly and indirectly influencing the expression and function of a variety of other cytokines and likely playing an important role in autoimmunity. 40 Interferon regulatory factors are a family of 9 interferon-induced transcription factors that have an important role in immune system function. Studies have demonstrated, for example, that knockout mice lacking particular interferon regulatory factors have decreased susceptibility to autoimmune diseases, such as type-II collagen-induced arthritis and experimental allergic encephalomyelitis. 39 Type-I interferons may also promote survival and proliferation of self-reactive T and B cells in both organ-specific and systemic autoimmune diseases. 40 Given the roles that type-I interferons probably play in the development of autoimmunity, it is not surprising that MxA expression has been demonstrated in a variety of human inflammatory diseases of suspected autoimmune etiology, including systemic lupus erythematosus, 42 cutaneous lupus erythematosus, 6, 7, 43 lichen planus, 6, 34 dermatomyositis, 9 chronic inflammatory arthritis, 20 and psoriasis. 6
In this study, Mx protein expression was documented for the first time in naturally occurring animal disease. The demonstration of Mx protein immunoreactivity in viral, nonviral, and idiopathic encephalitides led to the conclusion that Mx protein is not a specific marker of viral infection in the dog. The reason for Mx expression in idiopathic canine encephalitides is unclear, and the pathogenesis of these diseases remains enigmatic. Further evaluation of the role of Mx proteins in the pathogenesis of natural diseases of animals is warranted due to the relative lack of information regarding these proteins in domestic animals. Future studies could include dual immunostaining with cell-type markers to more definitely determine the cell types expressing Mx protein. Dual immunostaining could also be used to study the spatial relationship of viral antigens and Mx proteins in specific viral encephalitides.
Footnotes
Acknowledgements
We thank Dr. Otto Haller and Dr. Georg Kochs, Department of Virology, University of Leipzig, Germany for supplying the MxA antibody and Rosemary Vollmar, Susan Hernandez, and Chaitali Mukherjee for histology support.
