Abstract
Four healthy adult dogs (Golden Retrievers aged 6 years and 9 years, Dalmatian aged 13 years, and Mastiff aged 5 years) developed clinical signs of acute respiratory disease and died within 2 to 7 days of onset of clinical signs. The lungs of the 3 dogs submitted for necropsy were diffusely and severely reddened due to hyperemia and hemorrhage. Microscopic lesions in all dogs were suggestive of acute viral or toxic respiratory damage and varied from acute severe fibrinonecrotic or hemorrhagic bronchopneumonia to fibrinous or necrotizing bronchointerstitial pneumonia. Necropsied dogs also had hemorrhagic rhinitis and tracheitis with necrosis. Virus isolation, transmission electron microscopy, and polymerase chain reaction were used to confirm the presence of canid herpesvirus 1 (CaHV-1) in the lung samples of these dogs. Lung tissues were negative for influenza A virus, canine distemper virus, canine parainfluenza virus, canine respiratory coronavirus, and canine adenovirus 2. Canid herpesvirus 1 has been isolated from cases of acute infectious respiratory disease in dogs but has only rarely been associated with fatal primary viral pneumonia in adult dogs. The cases in the current report document lesions observed in association with CaHV-1 in 4 cases of fatal canine herpesvirus pneumonia in adult dogs.
Canid herpesvirus 1 (CaHV-1), a member of the subfamily Alphaherpesvirinae, genus Varicellovirus, causes fatal systemic disease with characteristic necrosis and hemorrhage in neonatal puppies and mild upper respiratory, genital, and ocular disease in adult dogs. 10 Necrosis and hemorrhage in puppies are typically multisystemic, with most obvious involvement of liver, lung, kidney, spleen, small intestine, and brain; however, similar lesions may be encountered in adrenal glands, eye (including retina and uvea), heart, pancreas, stomach, and omentum. CaHV-1 is host specific, infects canids, and grows only on canine cell cultures. The host specificity has been attributed to the presence of unidentified virus-specific receptors on the permissive host cells. 18 CaHV-1 is temperature sensitive (optimum <37°C), susceptible to common disinfectants, and not stable outside the host cell, yet it is ubiquitous in nature and has a worldwide distribution. 4,10
Under natural conditions, CaHV-1 is transmitted transplacentally in utero, during passage through the birth canal at parturition, or through direct contact with oronasal secretions from an infected animal and, rarely, through fomites. 4,10 CaHV-1 pathogenicity is age dependent, and neonatal puppies aged between 1 and 6 weeks are most susceptible. 8,10,15 Transplacental infection during mid to late gestation may result in abortion, stillborn puppies, or weak puppies that die within a few days of parturition. 4,10 Neonates less than 2 weeks of age may become infected during or soon after parturition; the incubation period of the virus is 6 to 10 days. 4 Clinical signs include persistent crying, lethargy, depression, anorexia, loss of body weight, low rectal temperature, abdominal pain, mucopurulent nasal discharge, widespread petechial hemorrhages on mucous membranes, and mild vesicular vulvovaginitis. Puppies usually die within 24 to 48 hours of onset of clinical signs. 4,10 In older puppies (>3 or 4 weeks of age), CaHV-1 infection may result in upper respiratory infections such as tracheitis and bronchitis, which may progress to systemic infection. 10 In puppies that survive the primary infection, CaHV-1 undergoes latency in the neurons of the trigeminal, lumbosacral, and vestibular ganglia, where it may become reactivated in adult animals and undergo a lytic cycle and result in mild upper respiratory tract infection and ocular and genital lesions. 8 Systemic primary infection in adult animals is rare. In adult dogs, primary CaHV-1 infection is usually contracted through venereal and oronasal secretions, and similar to recrudescent infection, primary infection also results in localized or regional, mild, self-limiting upper respiratory tract infection; keratoconjunctivitis; and vesicular genital lesions. 4,12,15
CaHV-1 causing fatal infections in adult dogs is rare, and only a few cases have been reported. Most cases in adult animals occur in immunosuppressed dogs undergoing cancer chemotherapy 15 or dogs under stressful conditions such as corticosteroid medication, radiation therapy, or surgery. 11 The present report describes 4 cases of fatal CaHV-1 infections in 2 adult Golden Retrievers (aged 6 years and 9 years), one 13-year-old Dalmatian, and a 5-year-old Mastiff. CaHV-1 was confirmed using polymerase chain reaction (PCR), virus isolation, and transmission electron microscopy. No other bacterial or viral etiologies were identified.
Materials and Methods
Case Histories
Cases 1 and 2
Two female Golden Retriever dogs, aged 6 years (dog No. 1) and 9 years (dog No. 2), were presented to the University of Georgia Veterinary Diagnostic Laboratory (UGA-CVM) for necropsy after death subsequent to clinical pneumonia. The 2 dogs were part of a group of 4 adult Golden Retrievers from the same household that were boarded together in a kennel. All 4 of the Golden Retrievers developed clinical signs of respiratory disease within a span of 0 to 2 days after boarding. The severity of signs ranged from coughing that resolved to marked febrile illness and dyspnea that was diagnosed as pneumonia via auscultation and radiographs. The 6-year-old dog (dog No. 1) had clinical signs of respiratory disease when the owners picked her up from the boarding kennel and died 2 days later. The 9-year-old dog (dog No. 2) developed clinical signs after she was brought home and died 5 days after developing clinical signs. The 2 dogs that died had been hospitalized and treated with antibiotics. All 4 dogs from the household were current on vaccinations.
Case 3
A 13-year-old male Dalmatian (dog No. 3) from a breeding facility in Birmingham, Alabama, was presented to Mississippi State University College of Veterinary Medicine Diagnostic Laboratory Services (MSU-CVM) for necropsy. The dog was presented to the referring veterinarian with symptoms of anorexia, lethargy, hypersalivation, and ocular discharge, which progressed overnight to severe acute respiratory distress followed by cardiac arrest and death. The dog was clinically and radiographically diagnosed with bronchointerstitial pneumonia; a grade 5/6 systolic heart murmur was also noted. The complete blood counts were within normal limits. Treatment included antibiotics, fluids, antiemetics, antidepressants, and nonsteroidal anti-inflammatory drugs. The dog was current on vaccinations.
Case 4
Tissues from a 5-year-old female spayed Mastiff (dog No. 4) were submitted to the biopsy service of the University of Georgia Veterinary Diagnostic Laboratory. The dog presented to the referring veterinarian with sneezing, coughing, nasal discharge, and poor appetite a week after being boarded. The dog was afebrile and started on oral antibiotics (doxycycline). The other dog in the household presented with milder symptoms, was treated similarly, and recovered. Two days after initial presentation, the patient re-presented to the referring veterinarian in worsened condition and was treated with intramuscular antibiotics (enrofloxacin), fluids, and nebulization but died in less than 12 hours. Radiographs were declined. Both dogs were current on vaccinations, including Bordetella bronchiseptica.
Gross Pathology and Tissue Collection
A complete necropsy was performed on dog Nos. 1 to 3 at their respective institutions. Representative tissue samples from heart, trachea, nasal turbinates, lung, liver, spleen, kidney, urinary bladder, esophagus, stomach, small intestine, large intestine, tracheobronchial lymph nodes, and brain (dog Nos. 1–3) and also thyroid, adrenal glands, pituitary, and bone marrow (dog No. 3) were collected in 10% neutral buffered formalin. For dog No. 4, submitted formalin-fixed tissues included lung, liver, lymph nodes, spleen, and kidney. Formalin-fixed samples were routinely processed, embedded in paraffin, sectioned at 4 µm, stained with hematoxylin and eosin, and examined by light microscopy. Special stains were used as required (Lillie-Twort and Giemsa for dog Nos. 1–2). Fresh tissue samples from lungs were submitted for bacteriology and virus isolation in all 4 cases. Representative tissue samples from lungs were frozen at –80°C for future analyses (dog Nos. 1–3). In addition, fresh lung tissue samples were fixed in yellow fixative (2% glutaraldehyde, 2% paraformaldehyde, 0.2% picric acid, 0.1 M cacodylate-HCl buffer) for transmission electron microscopy (dog Nos. 1, 2, and 4).
Virus Isolation
Virus isolation was performed at 2 independent laboratories at UGA-CVM (for dog Nos. 1, 2, and 4) and MSU-CVM (dog No. 3). Standard protocols were followed. Briefly, Madin-Darby canine kidney (MDCK) cells (ATCC, Manassas, VA) were grown in Dulbecco’s modified Eagle’s medium (DMEM; Sigma-Aldrich, St Louis, MO) supplemented with 10% fetal bovine serum (FBS) (Sigma-Aldrich), penicillin (200 U/ml), and streptomycin (200 mg/ml). The lung tissue samples were homogenized (cold phosphate-buffered saline [PBS]), centrifuged, and filtered using 0.22-µM filters. Filtered supernatant (200 µl) was inoculated onto the cultured MDCK cells (over 75% confluence) and maintained at 35°C and 5% CO2. Characteristic herpesvirus cytopathic effects (CPEs) were observed at 3 days postinoculation (dpi). Cells were harvested at 3 dpi; DNA was extracted and tested for CaHV-1 using conventional PCR and previously published primers (specific for canid herpesvirus 1 glycoprotein B gene) and conditions. 3 Amplified PCR products were sequenced and confirmed by searching against NCBI databases using the BLASTn program and determined to be 100% similar to HQ846625, DQ983328, AY582737, and JX907999.
Transmission Electron Microscopy
Transmission electron microscopy (TEM) was performed on dog Nos. 1, 2, and 4 on the inoculated MDCK cell culture suspensions and on the fresh lung tissues fixed in yellow fixative (dog Nos. 1 and 2) and fresh lung tissue fixed in formalin (dog No. 4). Briefly, yellow fixed tissues were postfixed in osmium tetroxide and en bloc with 2% aqueous uranyl acetate. Tissues were embedded in Epon-Araldite. Thin sections (55–70 nm) were cut and stained with methanolic uranyl acetate and Reynolds lead citrate. Tissues that were fixed in neutral buffered formalin were processed similarly with the exception of treatment preprocessing with 2% paraformaldehyde and 2% glutaraldehyde in 0.1 M phosphate buffer, pH 7.3. MDCK cell culture supernatant was purified and stained with 2% aqueous phosphotungstic acid, pH 7.0. TEM was performed using a JEOL JEM-1210 transmission electron microscope at an accelerating voltage of 120 KeV, equipped with an XR41C bottom-mount CCD camera from Advanced Microscopy Techniques (Woburn, MA).
Results
Gross Findings
All 3 necropsied dogs were in good body condition. In dog Nos. 1 and 2, more than 90% of the lungs were mottled dark red to purple, inflated, wet, and heavy with a slightly rubbery texture. The trachea was diffusely reddened, and the tracheal lumen was filled with stable foam throughout its length. The nasal turbinates were diffusely red to black (Fig. 1). In dog No. 3, the lungs were mottled dark red to purple and inflated (Fig. 2) and had a consistency that varied from soft and slightly rubbery to crepitant. The trachea and bronchi contained a moderate amount of red-tinged, frothy fluid, and similar fluid oozed from cut surfaces. The thorax contained 150 ml of red-tinged translucent fluid. The nasal turbinates were dark red to black. The heart had severe marginal smooth nodular valvular thickening attributed to endocardiosis, left atrial dilatation, left atrial endocardial fibrosis (jet lesions), and focal chordae tendineae rupture.
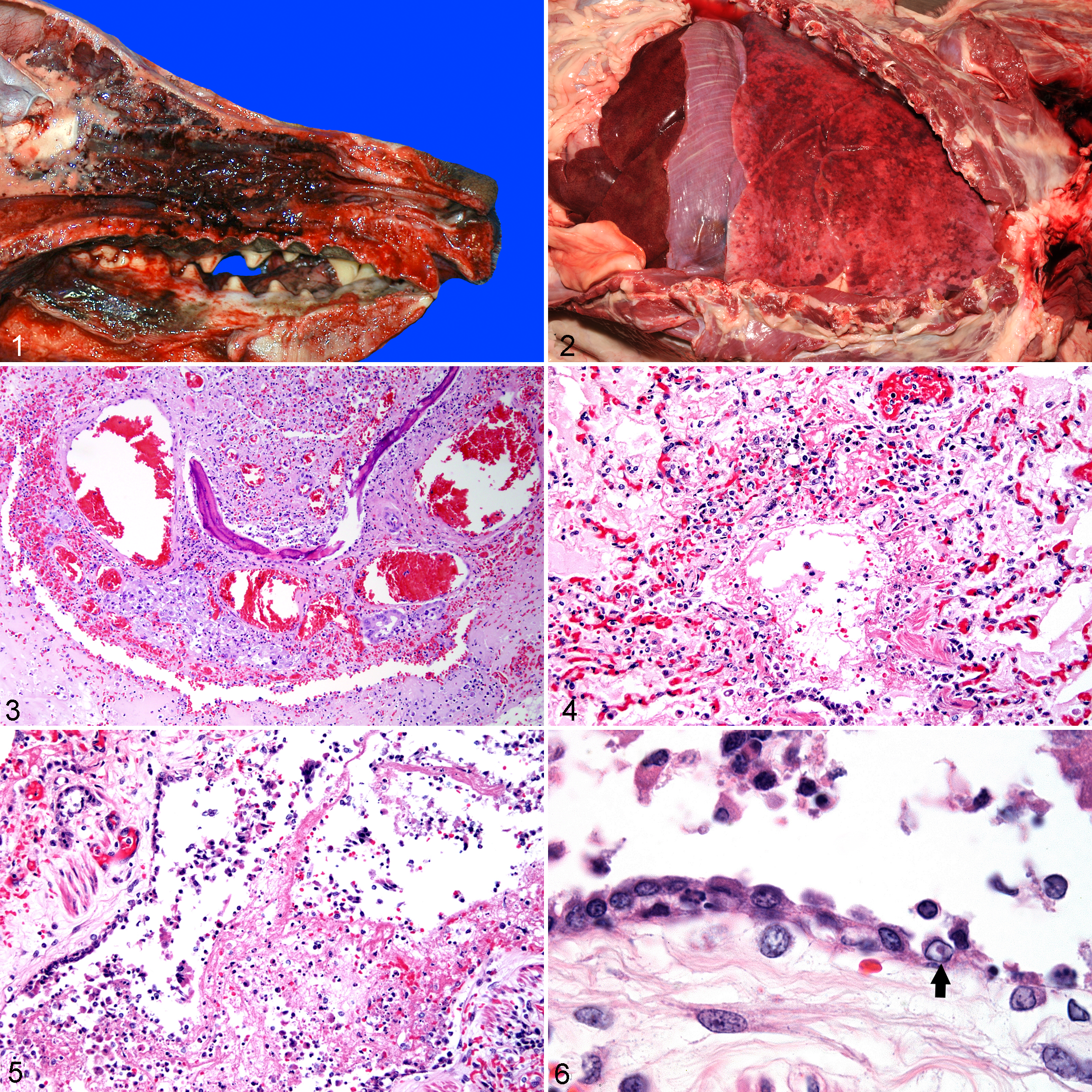
Canid herpesvirus 1, nasal cavity, dog No. 2. Severe hemorrhagic rhinitis with necrosis, fibrin, and hemorrhage.
Histopathology
Overall, the histologic lesions consisted of severe necrotizing hemorrhagic rhinotracheitis and bronchiolitis with scattered edema, fibrin, and septal necrosis. In dog Nos. 1 and 2, the nasal turbinates, trachea, and lungs had severe epithelial damage. In the nasal turbinates, the respiratory epithelium was ulcerated and the lamina propria was covered by a thick mat of fibrin, necrotic cellular debris, and hemorrhage that filled the spaces between the turbinates (Fig. 3). The entire turbinate lamina propria was markedly expanded by edema, fibrin, hemorrhage, and necrotic cellular debris (Fig. 3). Moderate inflammatory infiltrates in the turbinate lamina propria included numerous macrophages and fewer neutrophils, lymphocytes, and plasma cells. Only a few nasal glands remained, and many of these contained necrotic, intraluminal, cellular debris. The tracheal mucosa was ulcerated and the lamina propria was covered by a mat of fibrin mixed with small numbers of macrophages. The tracheal lamina propria had small numbers of scattered perivascular and periglandular lymphocytes and plasma cells. Bronchi and bronchioles had nearly complete loss of the respiratory epithelium, and a mat of fibrin covered the lamina propria. The few airways with remaining epithelium had sloughing of epithelial cells into the airway lumina. The bronchi and bronchioles often contained fibrin and edema that were occasionally mixed with small numbers of neutrophils and cellular debris. Bronchial walls were markedly expanded by abundant fibrin and edema with marked hyperemia. The fibrin and edema extended from larger airways and filled alveoli. Alveoli also contained large numbers of foamy macrophages and variable moderate hemorrhage.
In dog No. 3, the nasal turbinate damage was less severe and included marked hyperemia, necrosis of respiratory mucosal epithelium, lamina proprial edema, and mild infiltrates of lymphocytes, plasma cells, macrophages, and a few neutrophils. Neutrophils, macrophages, and necrotic cellular debris filled the interstices between nasal turbinates. The trachea had multiple small foci of ulceration of mucosal epithelium and mild lamina proprial edema with neutrophils, macrophages, and occasional lymphocytes and plasma cells. Pulmonary changes were less severe than dog Nos. 1 and 2 and occurred in a bronchointerstitial pattern. The lungs had both necrosis of secondary bronchi and bronchioles with loss of epithelium and, sometimes in the same airway, squamous metaplasia of bronchiolar epithelium (Figs. 4, 5). Necrosis with fibrin, mild hemorrhage, and mild mixed infiltrates of neutrophils, macrophages, and some lymphocytes and plasma cells extended into the peribronchiolar alveoli. Larger bronchioles were often covered by a thin layer of fibrin with similar inflammatory cells and cellular debris; this was variably accompanied by similar exudate filling peribronchiolar alveoli and alveolar septal necrosis. Rare bronchial epithelial cells contained intranuclear eosinophilic inclusion bodies with margination of chromatin (Fig. 6). Macrophages with refractile pigment were sometimes in the bronchiolar adventitia. The lung parenchyma peripheral to airways had generalized hyperemia, low protein edema, mild hemorrhage, and occasional intermingled macrophages or neutrophils. No consolidating alveolar inflammatory infiltrates were present. The findings in the lungs were consistent with the generalized mottled red appearance seen on gross examination and were typical of bronchointerstitial pneumonia. The findings in the nasal cavity and trachea were acute rhinitis and tracheitis with multifocal mucosal necrosis. In addition, the liver had widely disseminated foci of centrilobular coagulative necrosis and congestion. Germinal centers in tracheobronchial lymph nodes were moderately depleted of lymphocytes, and some lymphocyte necrosis with macrophage phagocytosis was evident.
Pulmonary histopathology on dog No. 4 was similar to the other cases. Bronchi and bronchioles had necrosis and loss of epithelium with some fibrin and inflammatory cells that included macrophages and neutrophils. Lung parenchyma was markedly hyperemic with low protein edema and frequent alveolar erythrocytes and foamy macrophages and multifocal areas of consolidation. The liver was also congested with multifocal areas of necrosis, fibrin exudation, and mild neutrophilic infiltrate. Lymph nodes had moderate to marked lymphocyte depletion with scattered individual cell necrosis.
Microbiology and Molecular Diagnostic Tests
No bacterial growth was observed on lung cultures from any of the 4 cases. Standardized molecular diagnostic methods, including PCR, real-time PCR (rtPCR), immunohistochemistry (IHC), and fluorescent antibody testing (FA) were used to rule out common canine respiratory pathogens as per respective diagnostic laboratory protocols. Lung tissue samples from dog Nos. 1 and 2 were negative for influenza A virus (PCR, IHC, FA), canine respiratory coronavirus (PCR), and canine distemper virus (FA). Dog No. 3 was negative for influenza A virus (H3N8), canine distemper virus, canine parainfluenza virus, canine respiratory coronavirus, canine adenovirus 2, and B. bronchiseptica by real-time PCR conducted at the diagnostic laboratory at the University of California Davis–School of Veterinary Medicine. Lung tissue from dog No. 4 was negative for canine adenovirus 1 and 2, canine influenza A virus (H3N8), canine distemper virus, canine respiratory coronavirus, B. bronchiseptica, and Mycoplasma spp by real-time PCR conducted at the diagnostic laboratory at the University of Georgia–School of Veterinary Medicine. Amplified PCR products from isolated viral DNA had 100% homology to the CaHV-1 sequences for the canid herpesvirus 1 glycoprotein B gene (167 bp). 3
Transmission Electron Microscopy
Both lungs and MDCK cell culture supernatant were examined by TEM (dog Nos. 1, 2, and 4). The lungs had degenerating bronchiolar epithelial cells, which contained 100- to 110-nm-wide icosahedral intranuclear viral particles that were consistent with herpesvirus nucleocapsids (Fig. 7). The MDCK cell culture supernatant used in virus isolation also contained similar 100- to 110-nm-wide icosahedral nucleocapsids (Fig. 8).
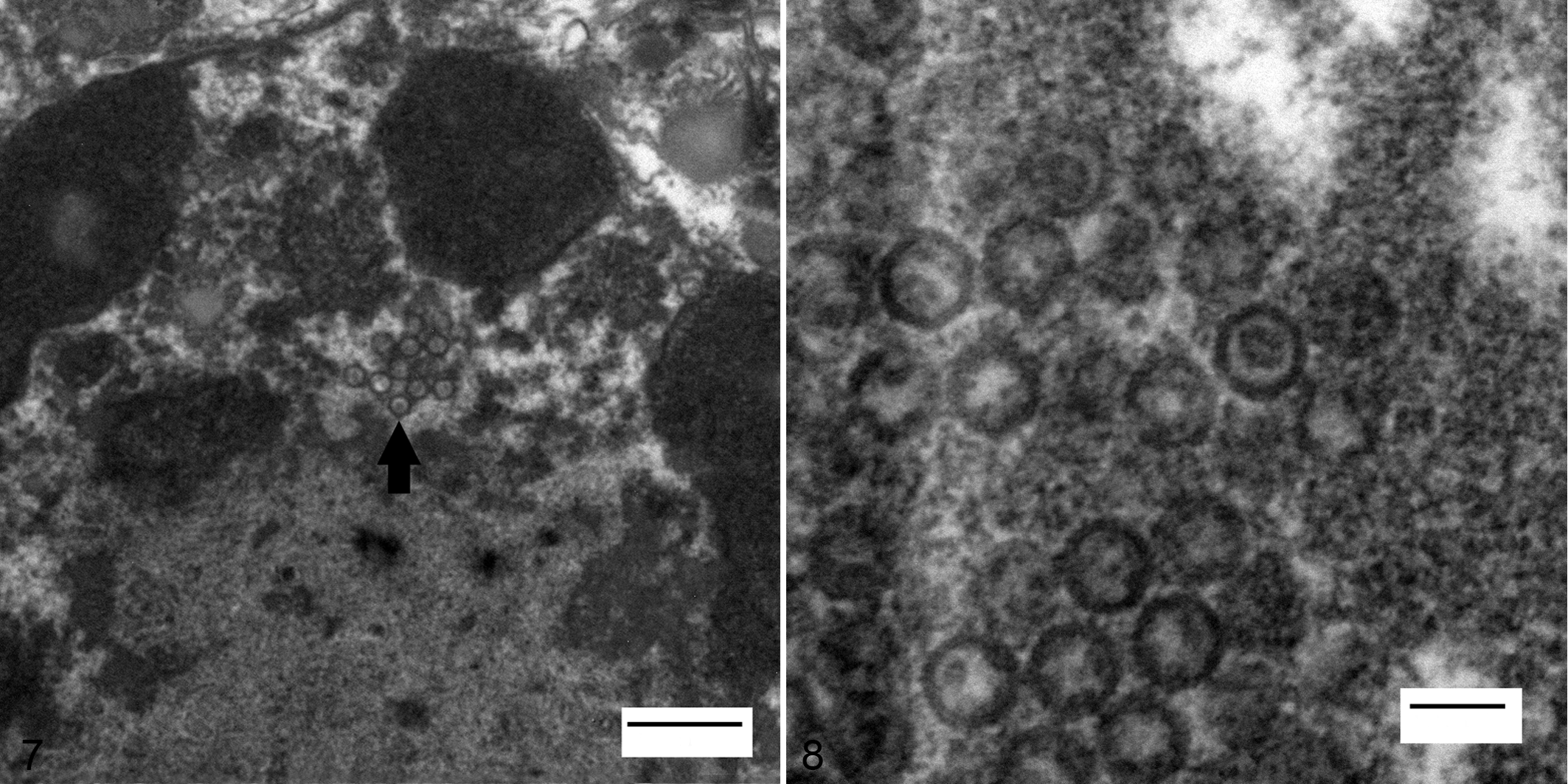
Canid herpesvirus 1, lung, dog No. 2. Transmission electron micrograph of a cell within the bronchiolar lumen with numerous intranuclear 100- to 110-nm icosahedral viral nucleocapsids. Electron-dense line in upper left corner is the nuclear envelope and electron-dense globular material in the nucleus is clumped chromatin. Arrow, viral nucleocapsids. Uranyl acetate and Reynold's lead citrate. Bar, 500 nm.
Discussion
The current report illustrates an unusual presentation of CaHV-1 that manifested as severe acute respiratory disease in adult dogs. CaHV-1 is typically associated with fatal systemic disease in neonatal puppies, mostly younger than 6 weeks. In adult dogs, CaHV-1 infection results in mild self-limiting respiratory infections, ulcerative and nonulcerative keratitis, and mild vesicular lesions of genitalia. CaHV-1–induced fatal systemic respiratory disease in adult dogs is rare. Gadsden et al 9 describe fatal systemic CaHV-1 infection in a healthy adult Bichon Frisé dog; all other reports of CaHV-1–associated systemic infection are related to immune-suppressed conditions 11,15,17,19 or restricted to localized ocular lesions under experimental conditions. 13,14,17
Herpesviruses have coevolved with their hosts over millions of years and thus are extremely well host adapted and have advanced mechanisms for immune evasion and lifelong persistence. Latency is one such strategy, allowing a carrier animal to asymptomatically harbor the virus in the absence of transcription, translation, and production of mature virions. 8 As is typical of alphaherpesviruses, CaHV-1 follows its primary lytic cycle in nasal mucosa, pharynx, and tonsils. 8 Subsequent to viremia, the virus localizes in the neurons of the regional sensory ganglia (trigeminal, lumbosacral, vestibular) 2,9 or lymphocytes of regional lymph nodes (retropharyngeal lymph node) 2,16 and becomes latent. In most cases, this is lifelong latency, and if viral reactivation occurs, infections are subclinical. The specific causes for reactivation from latency are not yet known, although general factors such as stress, pregnancy, chemotherapy, and systemic corticosteroid therapy have been implicated. 9,14,19,20 Following reactivation, the virus moves to the peripheral epithelial cells and a lytic cycle of viral replication and may result in mild subclinical infection or ocular diseases (ulcerative or nonulcerative keratitis with blepharospasm, conjunctivitis, and photophobia) depending on the host immunity. 8 T-cell–mediated immunity likely plays a crucial role in determining the fate of viral reactivation. Recrudescent systemic CaHV-1 infection in adult dogs has been associated with preexisting systemic conditions (diabetes mellitus, malignant lymphoma, and immune-mediated thrombocytopenia), 8 chemotherapeutic drugs (cyclophosphamide or doxorubicin), 8,15 or systemic corticosteroid therapy (prednisolone). 8,12,19,20
In the cases presented here, widespread damage to respiratory epithelium and the presence of CaHV-1 (verified with virus isolation, PCR, and TEM) in the absence of significant bacterial isolates and other respiratory viral diseases strongly support the hypothesis that the cause of the rhinotracheitis and bronchointerstitial pneumonia with necrosis and hemorrhage was CaHV-1. Direct epithelial damage was present in the nasal and tracheal epithelium in 3 cases presented here, as well as in bronchial and bronchiolar epithelium and surrounding alveolar septa in all 4 cases. These findings are typical of acute viral rhinotracheitis and bronchointerstitial pneumonia. The multifocal centrilobular hepatocellular necrosis in the liver in dog No. 3 was attributed to severe hypoxemia due to viral pneumonia, which was further exacerbated by valvular insufficiency due to endocardiosis, acute rupture of a chorda tendineae, and subsequent pulmonary edema. Initial hypoxemia and tachycardia subsequent to viral bronchointerstitial pneumonia likely provoked the mitral valve chorda tendineae rupture.
Previous studies have isolated CaHV-1 from cases of canine infectious respiratory disease (CIRD) complex coinfected with other infectious agents, including canine distemper virus, canine parainfluenza virus, canine adenovirus type 2, canine influenza virus, canine respiratory coronavirus, canine herpesvirus, canine pneumovirus, B. bronchiseptica, Streptococcus equi subsp zooepidemicus, and Mycoplasma spp. 1,5,6,11,21,22 However, it is well accepted that mere identification of CaHV-1 is not confirmation of its causal relationship since a latent herpesvirus infection could be reactivated. The implication is that a latent CaHV-1 infection is reactivated subsequent to inflammatory conditions created by CIRD and the associated glucocorticoid elevation from the stress of prior disease. Erles et al 7 suggested that in adult dogs, latent CaHV-1 may be reactivated during stressful disease conditions such as infectious tracheobronchitis and, once activated, act synergistically with other primary/preexisting causes of bacterial or viral respiratory disease. 11 Kawakami et al 11 reported a nosocomial outbreak of infectious tracheobronchitis among adult dogs in a referral center and concluded that CaHV-1 alone was responsible for the outbreak. Kawakami et al 11 isolated the same CaHV-1 isolate from all affected dogs during the outbreak and, based on the fact that all affected dogs had been under stressful conditions (surgery, corticosteroids, chemotherapy, and radiation therapy), concluded that it was a point source outbreak. The authors speculated that the outbreak might have started as a recrudescent infection in a dog and that the presence of large numbers of dogs under stressful conditions may have facilitated horizontal transmission. 11 These data can be difficult to interpret since CaHV-1 has been reported to have low sequence variability. 23 Regardless, none of these previous studies performed postmortem examinations to correlate lesions with the presence of CaHV-1. The cases presented in this study clearly demonstrate morphologic lesions consistent with a viral etiology with confirmation by PCR, virus isolation, and electron microscopy. Moreover, the cases presented here are 2 independent sets of cases, which rule out a point source of infection. There was no information available about additional cases of respiratory disease in other dogs from the boarding facility (dog Nos. 1, 2, and 4) or the breeding facility (dog No. 3). It is interesting that all cases were purebred dogs (Mastiff, Dalmatian, and Golden Retriever), and all 4 cases originated in the southeastern United States (Mississippi and Georgia). There was no history of immunosuppressive therapy or use of any systemic corticosteroids in any of the cases presented here prior to the development of clinical signs. Interestingly, dog Nos. 1, 2, and 4 had canine housemates with which they were boarded a week before presentation. The housemates developed similar but milder signs of pneumonia and recovered uneventfully, suggesting individual susceptibility is important in disease pathogenesis.
The cases presented in this study are strongly suggestive of a causal relationship between CaHV-1 and acute primary fatal respiratory disease in 4 adult dogs that were not recognized as having clinical disease prior to acute onset of respiratory signs and that did not have concurrent immunosuppressive disease or previous immunosuppressive therapy. This study has one limitation in that recrudescent infection could not be definitively ruled out; however, the chances of 4 apparently healthy adult dogs developing and succumbing to severe acute respiratory disease due to a recrudescent CaHV-1 infection would be very low and highly unusual. In addition, all other recognized canine respiratory viral and bacterial pathogens were excluded by extensive testing. Canid herpesvirus 1 is the probable cause of fatal acute respiratory viral rhinotracheitis and bronchointerstitial pneumonia in the 4 adult dogs described.
Footnotes
Acknowledgement
We thank Mary Ard for her assistance and expertise in the transmission electron microscopic studies performed.
Author Contribution
Conception or design: SK, EAD, AJC. Data acquisition, analysis, or interpretation: KJ, SB, XFW, EWU, JTS, SS, PMK, RJH. Drafting the manuscript: SK, EAD. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy. S. Kumar and E. A. Driskell contributed equally to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
