Abstract
According to the WHO, the proportion of people over 60 years is increasing and expected to reach 22% of total world’s population in 2050. In parallel, recent animal demographic studies have shown that the life expectancy of pet dogs and cats is increasing. Brain aging is associated not only with molecular and morphological changes but also leads to different degrees of behavioral and cognitive dysfunction. Common age-related brain lesions in humans include brain atrophy, neuronal loss, amyloid plaques, cerebrovascular amyloid angiopathy, vascular mineralization, neurofibrillary tangles, meningeal osseous metaplasia, and accumulation of lipofuscin. In aging humans, the most common neurodegenerative disorder is Alzheimer’s disease (AD), which progressively impairs cognition, behavior, and quality of life. Pathologic changes comparable to the lesions of AD are described in several other animal species, although their clinical significance and effect on cognitive function are poorly documented. This review describes the commonly reported age-associated neurologic lesions in domestic and laboratory animals and the relationship of these lesions to cognitive dysfunction. Also described are the comparative interspecies similarities and differences to AD and other human neurodegenerative diseases including Parkinson’s disease and progressive supranuclear palsy, and the spontaneous and transgenic animal models of these diseases.
Keywords
Aging in its broadest sense is the gradual progressive impairment or deterioration of normal tissue function and tissue homeostasis. 110,151 The modern advances in technology, health care and nutrition, led to significant increase in the life expectancy of humans and animals. According to the WHO, the proportion of people over 60 years is increasing and expected to reach 22% of total world’s population in 2050. 207
Recent animal demographic studies have shown that the life expectancy of pet dogs and cats is increasing. In the United States, the number of cats and dogs over 10 and 6 years has increased 15% and 6%, respectively, in the last 2 decades. 197 The severity of aging and aging-related deterioration varies greatly between individuals and it varies also in a tissue-specific manner. 55,110 While many biological changes may contribute to this degree of variability, antioxidant status and regenerative capability are probably the most important biological factors that determine the response of a certain tissue or organ to the aging process. 37,172,184 Brain is probably the most vulnerable tissue affected by aging because of its high oxygen requirement, low synthetic capacity of endogenous antioxidants, and limited regeneration capability. 45 Brain aging is associated not only with molecular and morphological changes but also leads to different degrees of behavioral and cognitive dysfunction. Cognitive dysfunction disorders are also described in dogs and recently in cats, however, the correlation between these conditions and age-related neurologic lesions is not completely understood. Age-related neurologic lesions comparable to Alzheimer’s disease (AD) have been described in several species including dogs and primates, but detailed studies of age-related neurologic lesions are lacking and need to be addressed. 23,155,200 A broader understanding and documentation of these lesions are important for veterinary diagnosticians and for future studies on animal models of human aging.
This review is intended to provide the basic material that describe comparative interspecies similarities and differences in neurologic lesions associated with aging and the correlation of these lesions to cognitive deficits. Furthermore, the animal models of human brain aging, AD, and Parkinson’s disease (PD) are discussed.
Common Cognitive Dysfunction Diseases Associated With Brain Aging in Human and Animals
Brain aging is generally associated with declining cognitive functions that cannot be attributed to other medical conditions (eg, sensory decline, toxicosis, infectious disease, or neoplasia). One category of severe cognitive decline is dementia. Dementia is characterized by memory loss and decline in other cognitive functions, resulting in behavioral disturbances and difficulties with normal daily functioning. 156 AD is the most common form of human dementia. 198 In veterinary medicine, cognitive dysfunction syndrome (CDS) is the term used to characterize age-related deterioration of cognitive abilities in aging dogs. The pathology of AD and the cognitive and pathologic phenotypes associated with CDS are described below.
Alzheimer’s Disease
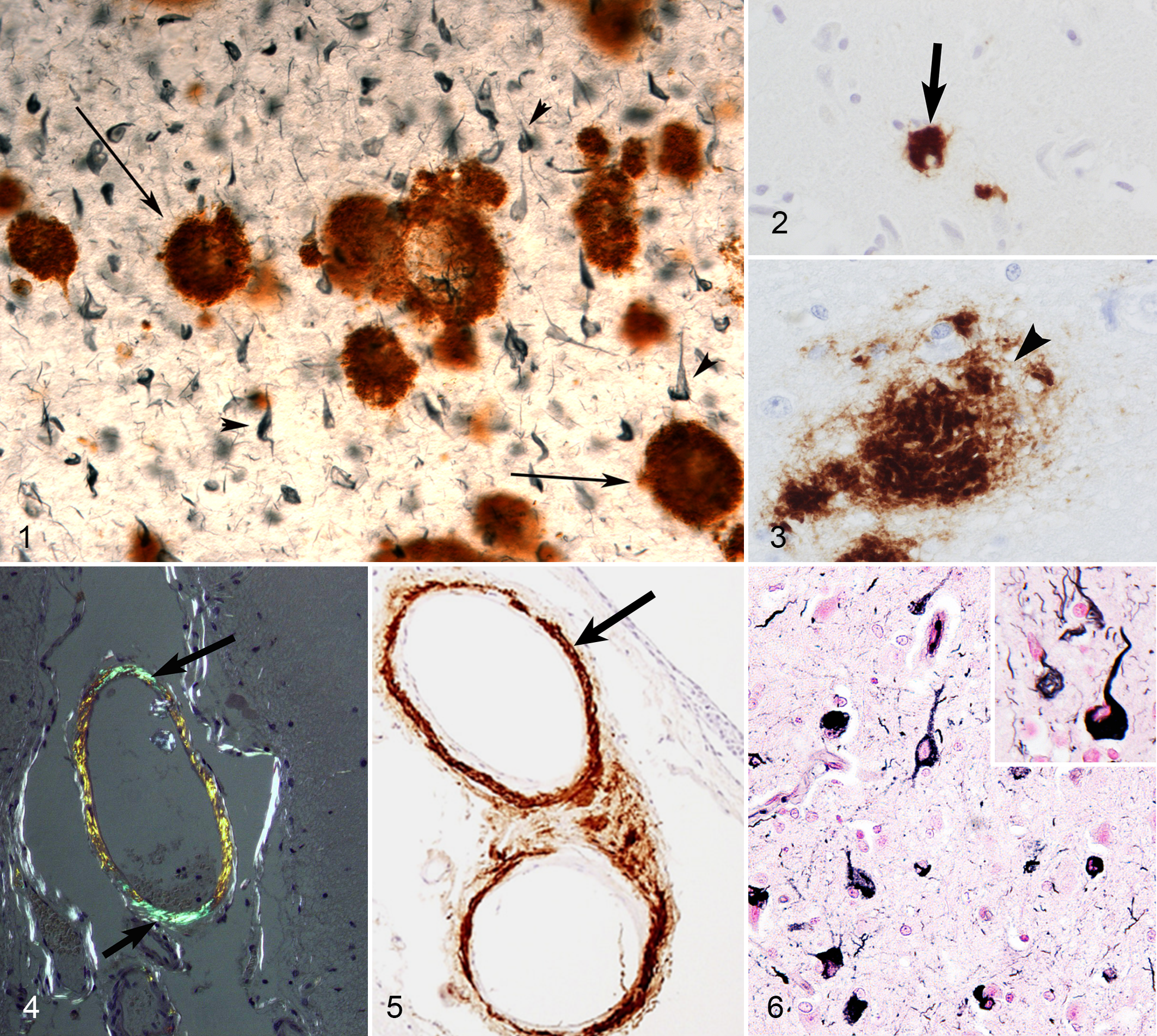
AD contributes for approximately two-thirds of all human dementia cases and affects more than 35 million people worldwide. 97 The 2 pathognomonic lesions of AD are the deposition of extracellular amyloid-β (Aβ) plaques surrounded by dystrophic neurites and the presence of intraneuronal neurofibrillary tangles (NFTs) in the hippocampus, cerebral cortex, and other areas of the brain important for cognitive function (Fig. 1). 78,163 In addition, AD is characterized by severe synaptic loss, inflammation, oxidative damage, astrocytosis, cerebral amyloid angiopathy and neuronal death. The deposition of Aβ and NFT interferes with neuronal homeostasis and disrupts synaptic transmission and plasticity leading to neuronal loss, neurotoxicity, and synaptotoxicity that ultimately lead to cognitive dysfunction. 163 AD has 2 forms, the rare genetically determined familial AD and the sporadic form, which is more common and accounts for up to 98% of AD patients. Familial AD has an autosomal dominant pattern of inheritance and is caused by mutations in 4 genes: amyloid precursor protein (APP), presenilin 1 (PS1), presenilin 2 (PS2), and apolipoprotein E (ApoE). 138,183 These mutations lead to over production and deposition of amyloidogenic Aβ particularly Aβx-42 and ultimately the development of early onset familial AD. The exact etiology of sporadic AD is yet to be determined, however, the “amyloid cascade hypothesis” is the most accepted explanation for its pathogenesis. 4,62 In this hypothesis, the age-dependent accumulation of fibrillar insoluble Aβ peptides in the brain is considered to be the central and triggering event in AD pathology. 4,62
We will next describe the cognitive features of several animal models that may capture some of the phenotype of cognitive decline in patients with AD. These include the dog, the cat, and the horse.
Cognitive Dysfunction Syndrome in Dogs
Dogs may be a good model to study cognitive decline and Aβ contributions to AD. They display patterns of cognitive impairment (decline in learning ability and memory) that are comparable to humans and they also develop amyloid plaques (Figs. 2 and 3) and cerebral amyloid angiopathy (CAA) (Figs. 4 and 5) but not NFTs. 18,200,219 Companion dogs are unique relative to laboratory animals as they share the same environment as humans. Aβ plaques can be seen in clinically normal aged dogs and similar to humans, Aβ plaque load is positively correlated with cognitive impairment in dogs. 34,38,96 Two consistent features of aging in dogs include cognitive domain-specific vulnerabilities as well as significant individual variability in the severity of cognitive decline. Aged dogs show deficits in complex learning tasks assessed in laboratory settings using visual stimuli (eg, size concept learning, 184 oddity discrimination learning, 37,112 size discrimination learning, 67,185 and spatial learning 33 ). In particular, reversal learning and visuospatial working memory, both thought to be measures of prefrontal function decline with age in dogs. 67,173,185 The ability of dogs to remember or choose objects that are closest or furthest from them, known as egocentric spatial learning and reversal is also age-sensitive. 34 Another form of spatial ability, allocentric learning that may also assess spatial attention (landmark task developed in nonhuman primates originally), is also vulnerable to aging. 113,114 Interestingly, not all cognitive functions decline with age and aging dogs can perform similarly to younger dogs on measures such as simple visual discrimination and procedural learning tasks. 111 Thus, sensory deficits are likely not a significant contributor to increased error scores observed on some tasks in old dogs as they can solve easier problem using visual cues.
As mentioned previously, AD is associated with significant memory decline and this can be a feature in a subset of older dogs. Using an object recognition task developed for nonhuman primates, aging dogs show difficulties learning the “rules” of a nonmatching to sample procedure used to test object recognition. 111,115 Based on tests of simple learning, aged dogs deficient on an object recognition memory task may be intact and thus these memory changes are unlikely linked to sensory deficits or the ability to perform the task (ie, move stimulus objects to access hidden food rewards). 66 In systematic laboratory testing procedures, one of the tasks that has shown evidence of being most useful for detecting age impairments is measuring spatial memory ability. Spatial memory can be tested in dogs by requiring animals to first learn the location of a stimulus object (covering a food reward) and then after a delay, to select an identical object but in a different location (nonmatching to position). By increasing the delay interval between the original presentation of an object and the subsequent selection phase, it is clear that older dogs have difficulty with longer delays associated with a greater memory demand. 31,70 Indeed, this task can detect very early decline in memory as early as late middle age in beagles (6 and 7 years). 173 In summary, aging dogs show characteristic age declines in some cognitive domains (memory, executive function) but not all, and some aging dogs remain intact whereas others decline, which is strikingly similar to aging people and those that develop AD.
A key dissociation to establish is who may be showing early signs of AD and what might be considered “normal aging.” 2 It has been a challenge in the human aging literature to identify individuals who show little or no decline in function, and those that show more rapid or significant decline including those with mild cognitive impairment. 141,142 A similar challenge occurs when studying cognitive aging in dogs as some aging animals show cognitive decline whereas others function as well as younger animals with this increasing individual variability becoming notable in middle age (in beagles). 1 Indeed, the variability in cognitive test scores in aging beagles increases with age with older animals falling into 3 clusters. These 3 groups of old dogs can be described as (1) successful agers, (2) impaired dogs, and (3) severely impaired dogs. 73 Increasing individual variability and a tendency to fall into impaired or unimpaired cognitive aging groups has been reported consistently in rodent and nonhuman primate aging 9,106,147 –149 as well as in older people. 163 Although managing a wide range of error scores on a task statistically can be a challenge for group comparisons it can also be a powerful tool. For example, when combining cognitive test scores with subsequent quantification of neuropathology (eg, extent of plaques) one can establish correlations between neuropathological outcome measures reflecting poor cognition that can subsequently lead to testable hypotheses in intervention studies.
Laboratory tests in dogs are labor intensive, time consuming and costly but provide important data and insights into cognitive aging and are highly suitable for testing interventions to improve cognition. In the real-world setting, however, researchers, veterinarians, and companion animal owners acknowledge that functional measures have immense value. As with a long history in rodents, changes in spontaneous behaviors can be easily measured in dogs using a similar open field test in dogs. 65,168,170 A mirror test that was developed for nonhuman primates also provides interesting age associated changes in dogs that can be detected by observing dogs as they react to their reflection. 43,50 Introducing objects into the open field test (for example dog toys) can elicit behaviors that exploratory behavior, which declines with age in some older dogs. 170 A unique aspect of cognition in dogs involving social behaviors cannot be tested in rodents includes tests such as measuring interactions with people, a silhouette of a dog or a model of a dog, the latter 2 measuring conspecific behavior. 66 These rapid 10 minute tests show remarkable sensitivity to aging that on the surface, are consistent with social/behavioral changes in people with dementia. For example, younger dogs actively engage in play with stimulus objects or with a person being in the room. In contrast older dogs show less interest in objects/toys and people but this can be variable; older dogs with cognitive impairments show very little interest in objects or in interacting with a human. 170 Furthermore, aged dogs with cognitive impairments tend to pace and show little directed behavior. Interestingly, aged dogs with cognitive impairments interact with their reflection more than younger dogs and unimpaired aged dogs on a mirror test suggesting a lack of recognition of self, which can occur in patients with AD in more severe stages of the disease. 170
As mentioned previously, laboratory-based tasks are a challenge to translate into the veterinary clinic but several studies report success using informant-based measures. 94 –96 Veterinarians and clinical researches can use questionnaires and have owners report on changes in behavior in the home setting 34,96,145,146 similar in some ways to questionnaires including the Mini Mental State Exam used in human clinics. 18,19 Although there are several published reports of questionnaires sensitive to cognitive and behavioral aging in pet dogs, each appears to have some success in being able to distinguish normally aging dogs from those showing abnormal behavioral changes signaling canine cognitive dysfunction. 96 Several behaviors have been identified as being useful for defining/diagnosing canine cognitive dysfunction and include walking, posture/emotion of expression, elimination behavior, life rhythm, play behavior, exploratory behavior, learned specific behavior, adaptive capabilities, and interactions with other animals or with owners. As with laboratory-based tests, increased variability on scores on these questionnaires are also observed in old dogs. 18
Cognitive Dysfunction Syndrome in Cats
Feline CDS is a relatively newly recognized condition. One study, estimated that cognitive decline affects around 28% of pet cats aged 11 to 14 years and over 50% of cats 15 years of age or older.
61
Characteristic signs of feline CDS are summarized by the acronym DISHAAL, which refers to
Cognitive Dysfunction in Horses
To our knowledge, cognitive dysfunction or age-related behavioral changes in horses has not been described systematically yet, however, there are several nonrefereed reports suggesting that cognitive impairment may occur. Senior horses affected by this condition usually exhibit abnormal behavioral changes and memory loss such as confusion and disorientation in a familiar environment, inability to be led, and failure to recognize a familiar object, however, the correlation of these signs to age-related neurologic lesions is yet to be determined.
Common Age-Related Brain Lesions
Neuronal Lesions
Neuronal loss and brain atrophy
Various age-associated neuronal lesions are found in humans and animals, and some of them are considered to be the hallmarks of several human neurodegenerative disorders such as AD, PD, dementia with Lewy bodies (DLB), and amyotrophic lateral sclerosis (ALS). One of the most common lesions associated with aging is neuronal cell loss that occurs due to necrotic or apoptotic processes, which are induced by mitochondrial or endoplasmic reticulum stress or the disturbance of autophagy or ubiquitin/proteasome systems. 54 Significant neuronal loss will lead to a reduction in neuronal density and ultimately brain atrophy.
Brain atrophy
Brain atrophy is defined as the gradual reduction in brain volume and size after reaching its mature size. Age-related brain atrophy is described in dogs, cats, chimpanzees, rhesus macaques, and humans. 32,96,163 Grossly, the atrophied brain is characterized by decreased volume and weight, being firmer than normal and may exhibits a darker gray-yellow discoloration. The meninges are usually thickened (due to fibrosis and/or mineralization) and become partially adherent to the skull. The gyri or folds are narrowed and wrinkled and the sulci become shallow and widened. The ventricles are mildly to moderately dilated (hydrocephalus ex vacuo). 108 Thinning of the cortex and increasing size of the lateral ventricles have been reported in many studies of aging dogs (Fig. 7). 56,90,175 Not all areas of cortex show thinning at an equivalent rate and some brain regions show more rapid atrophy than others. For example, in aging beagles, the prefrontal cortex shows tissue loss at younger ages (8-11 years) than does the hippocampus (after 11 years). 186 These losses in tissue volume that are region specific also correlate with cognitive decline on measures sensitive to circuits within those regions, 153 similar to elderly humans with AD.
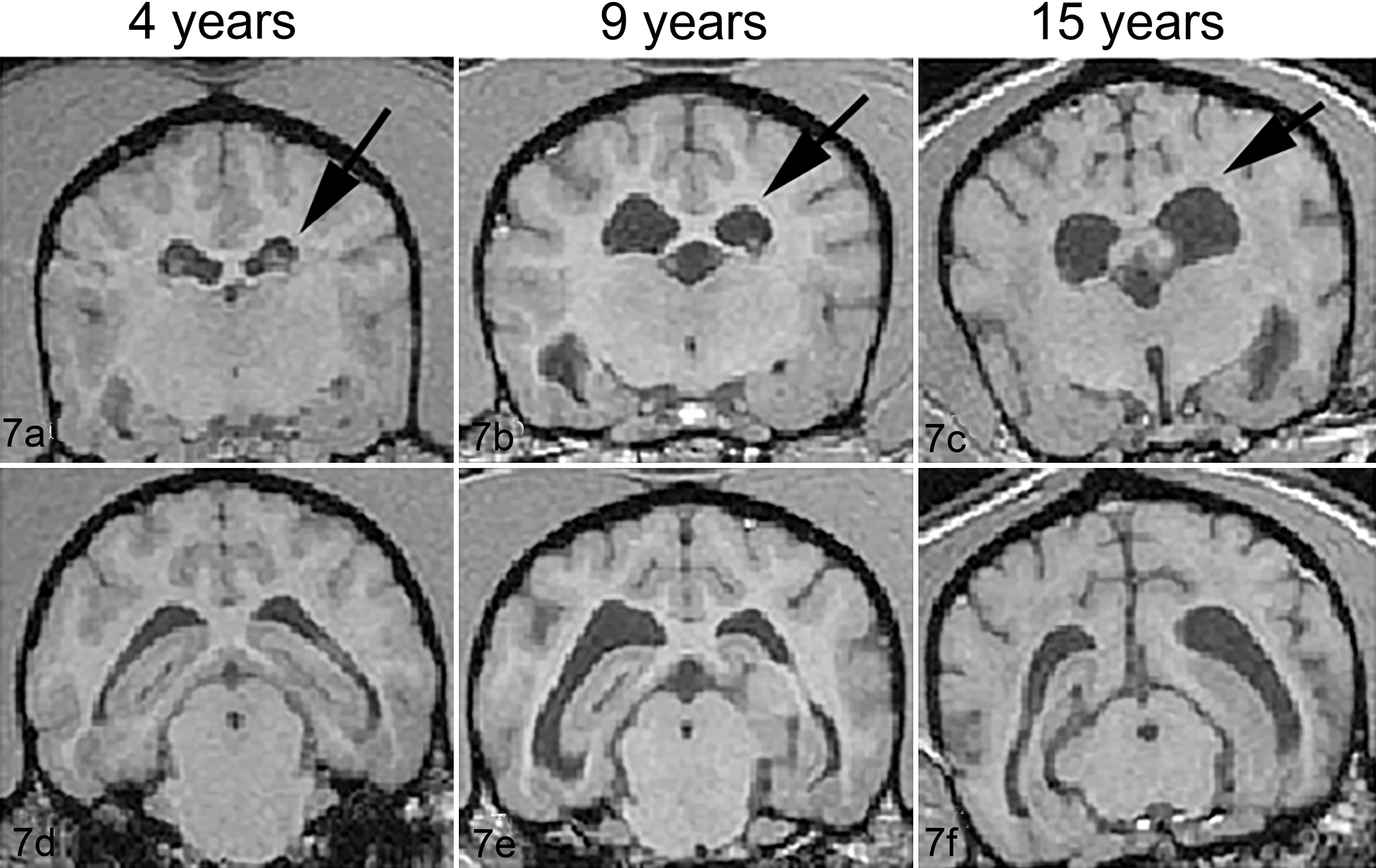
Cortical atrophy and ventricular dilatation. Magnetic resonance (MR) images (1.5 T) taken from locations through thalamus (upper panel) and hippocampus (lower panel) from a 4-year-old (a, d), 9-year-old (b, e), and 15-year-old dog (c, f). The old dogs show moderate increases in ventricular volume and cortical atrophy (with deep gyri and widened sulci) compared to the 4-year-old dog. Lateral ventricle is indicated with arrows. Adopted with permission. Courtesy of Neurobiology and Aging (Su et al 175 ).
Neuronal loss and reduced neurogenesis
Several mechanisms may contribute to brain atrophy. The neurobiological basis for brain atrophy may be due to a loss of neurons or potentially shrinking of neurons (ie, changes in density) as observed in normal brain aging and more extensively in AD in people. 13,167,206 Scoring or evaluating neuronal loss in sections stained with hematoxylin and eosin is challenging particularly if the loss is mild. It can be more readily demonstrated by Nissl staining or a neuronal specific marker such as NeuN. In aging beagles, counting neurons using unbiased stereological approaches shows neuron loss. Indeed, aging dogs can show up to a 30% loss of neurons in the hilus of the dentate gyrus in the hippocampus. 171 The loss of neurons in the hilus is linked to losses in cognitive ability in correlation studies and dogs with more intact numbers of neurons tend to be those animals with better learning ability. 171 In addition to loss of neurons in the hippocampus, aging dogs show Purkinje cell loss, which in a study by Pugliese et al was correlated with cognitive impairments. 145 Neurons in locus coeruleus nuclei that provide noradrenergic input to the brain, which are also lost in AD in people 44,58 are significantly reduced in dogs with cognitive impairment. 77 In parallel, a study by Kiatipattanasakul et al found the age-dependent decline of cognitive function in dogs correlated with the number of apoptotic (TUNEL-positive) neuronal cells, but not the number of amyloid plaques, they possess. 88
We recently detected significant neuronal cell loss in the hippocampi of aged cats, which occurred after formation of NFT consisting of highly phosphorylated tau protein (HN, JKC, KU, unpublished data). We also investigated the roles of autophagy in several canine degenerative neuronal diseases and neuronal loss using several autophagy markers, such as microtubule-associated protein 1 light chain 3 and P62. 133 These molecules colocalize with lipofuscin granules, and their expression tends to increase with aging. Taken together with the significant increase in the number of ubiquitin-positive granules observed in the neuropils of aged dogs, degradation processes that break down aberrantly metabolized proteins or damaged organelles, such as autophagy or the ubiquitin/proteasome system, might be important for neuron survival during the various physiological stresses caused by aging. The disturbance of these processes might induce cell death, resulting in neuronal cell loss.
Although losses in cortical thickness observed by MRI may be due to several factors, hippocampal cell loss may be exacerbated by reduced neurogenesis and replacement of lost neurons. Beagles show a dramatic age-related loss of ability to generate new neurons in the hippocampus (90-95% decline), which was linked to losses in cognition. 168,169 Studies in other breeds of dogs, have reported similar results of reduced neurogenesis. 75,140 Thus, declining neurogenesis, replacement of dying or dead neurons are factors that may contribute to cognitive aging. However, these hypotheses have yet to be tested systematically using intervention approaches.
Neurofibrillary tangles
NFTs are formed due to the intraneuronal deposition of hyperphosphorylated and misfolded insoluble tau protein in the neuronal cytoplasm and processes (Figs. 1 and 6), leading to degeneration. 79,196 After the tangle-laden neurons die, the remains of NFT reside in the neuropil and becomes extraneuronal (“ghost” tangles). NFT are argyrophilic and Thioflavin-S positive and can be detected using immunostaining with antitau antibodies and also by Gallyas-Braak silver staining. Tau proteins belong to the family of microtubule-associated proteins that mediate axonal transport. 196 In AD, tau proteins undergo hyperphosphorylation, ubiquitination, misfolding, and aggregation into paired helical filaments (PHFs). 119 Tau proteins in PHFs are different than phosphorylated native Tau and are considered to be highly neurotoxic and synaptotoxic due to their interference with cellular homeostasis and micortubular axonal transport system.8,107 There are no mutations associated with tau that leads to AD as there are for Aβ. One hallmark of AD pathology that is not found in dogs is NFT. 158,162 Although no research to date confirmed the presence of typical NFT in the canine brain, increased phosphorylation of tau is observed at some amino acid sites as observed in AD cases. 71,93,137,205,219 This lack of NFT pathology could possibly be due to significant differences in the tau protein sequence between dogs and humans (http://www.ensembl.org/Canis_familiaris/). Although the lack of NFT formation in dogs might be considered a weakness of the model, this does allow researchers to focus on Aβ pathology and provides opportunities to test interventions that focus on targeting this critical pathology for AD.
Interestingly, there is evidence of increased phosphorylated tau in other species although typical NFTs per se have not been identified including the cat, 60,61,71 sheep and goats, 22,128 cheetahs, 166 2 bison, a baboon, bears, cynomolgus monkeys, and a chimpanzee. 35,63,135,152,160,161 Typically these tau changes tend to be sporadic and do not adopt the full blown features of intracellular NFTs and are usually not associated with neuronal loss. Interestingly, even in nonhuman primates, which display substantial plaques formation and tau proteins with highly conserved amino acid sequences, NFT are rarely observed. 57 On the basis of the “amyloid hypothesis” of AD, it is generally considered that Aβ deposition precedes the development of NFT and the subsequent neuronal loss and cognitive deterioration; however, recent findings show NFT formation may precede Aβ pathology. 21,62,82
Recently, we detected Gallyas-Braak stain-positive NFT in cheetahs, leopards, and domestic cats (JKC, unpublished data), all of which belong to the subfamily Felinae (Fig. 6). 28,166 In these animals, abundant NFT were found together with Aβ deposition and neuronal loss. As is the case in humans, the NFT found in cats were composed of hyperphosphorylated tau.
In horses, an unusual observation by Capucchio et al was the positive staining of sporadic hippocampal neurons with total tau protein antibody, in most of the horses (14/18) aged 9Y to over than 15Y. 23 Same neurons stained negative for hyperphosphorylated Tau AT8 and no NFT detected by the Gallyas silver stain. The authors hypothesized that an age-dependent axonal transport deficiencies in the hippocampal neurons might led to accumulation of non phosphoryled-S202 tau without the formation of typical NFT. 23
Information about NFTs and tauopathies in other species is summarized in the third section and Table 1 of this review.
Occurrence of Amyloid Plaques (AP) and Neurofibrillary Tangles (NFT) in Various Animal Species.
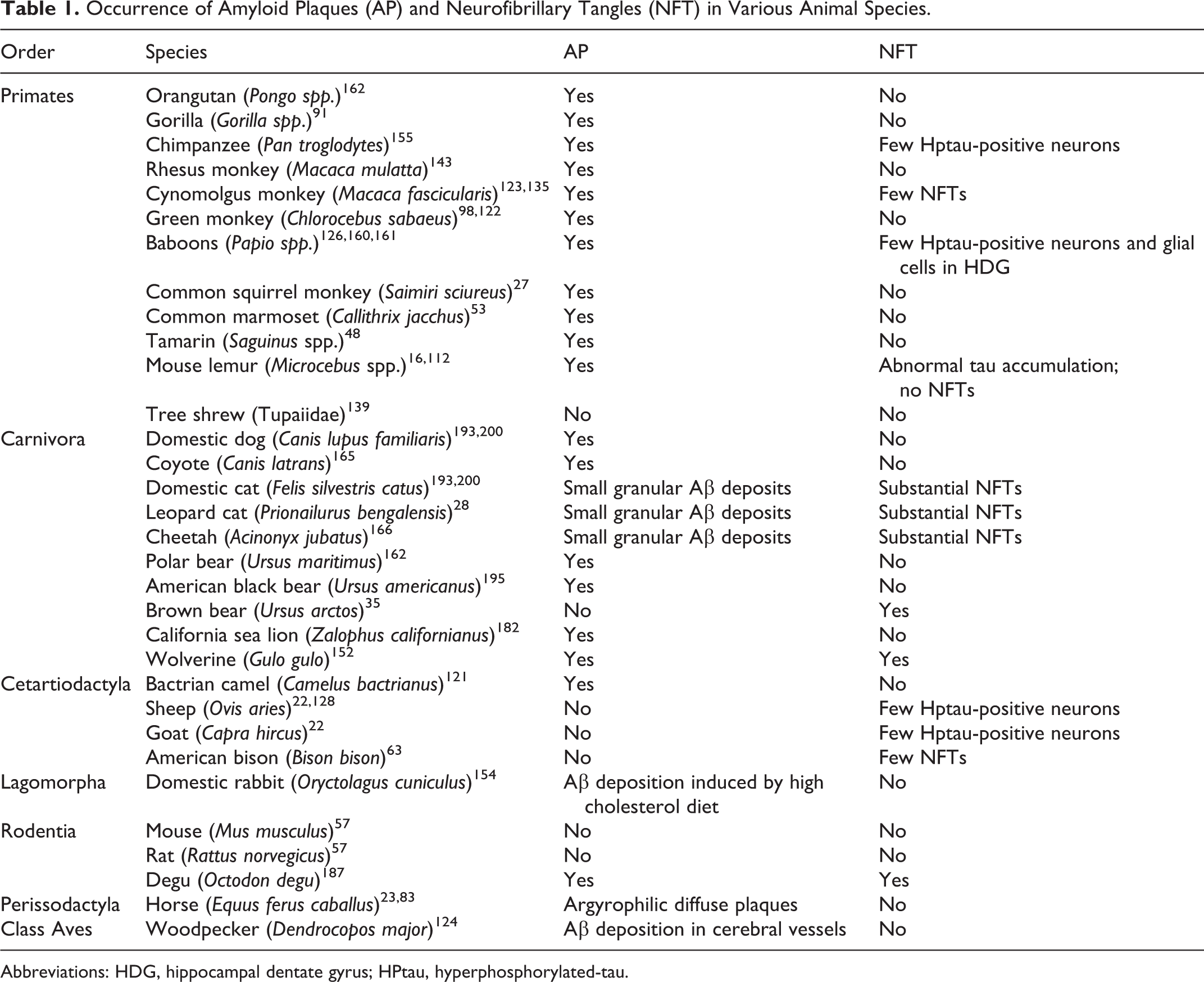
Abbreviations: HDG, hippocampal dentate gyrus; HPtau, hyperphosphorylated-tau.
Pigment accumulation
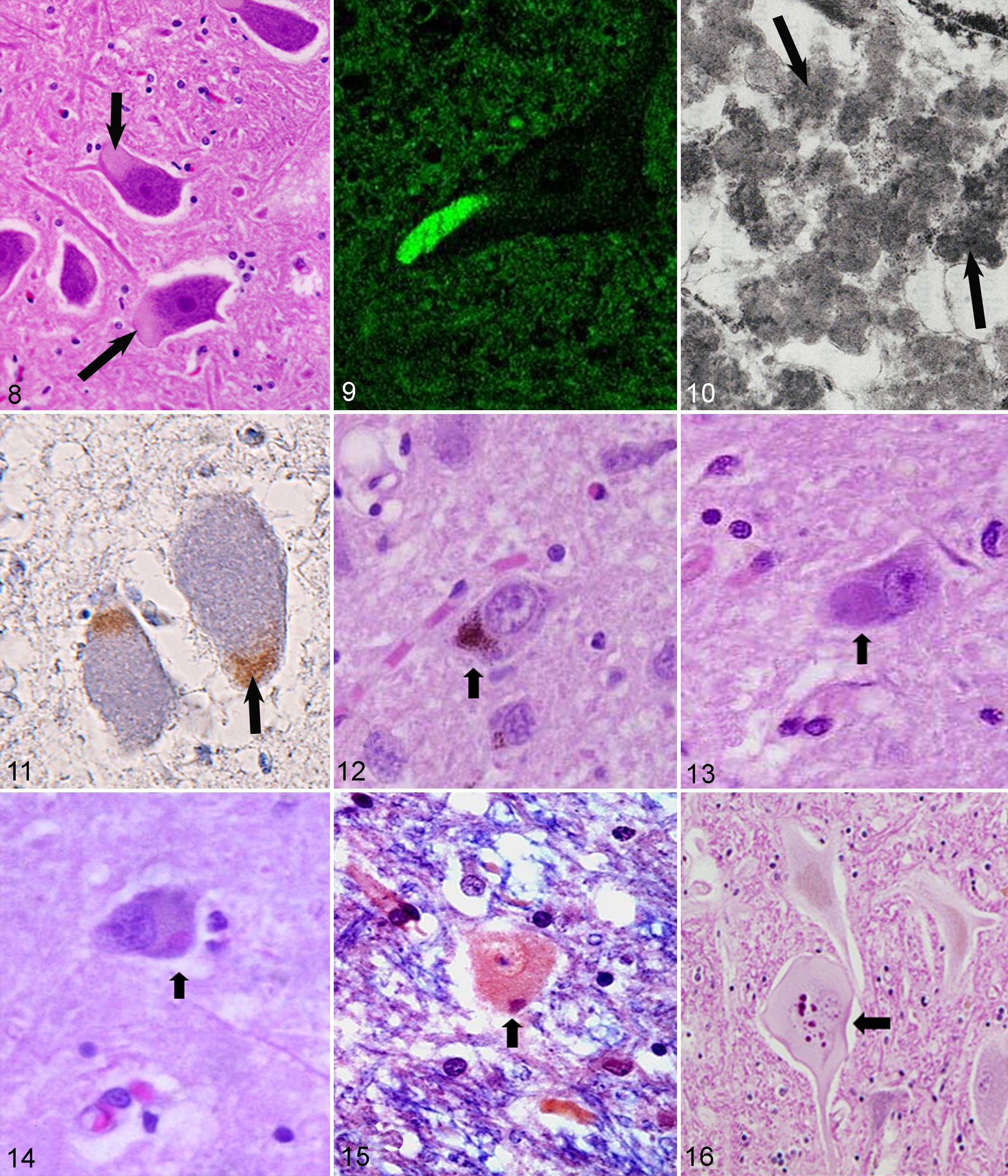
Lipofuscin is a yellowish pigment found in postmitotic cells, such as neurons and cardiomyocytes (Fig. 8). Ceroid is a similar pigment that is mainly found in phagocytes, and these 2 pigments are often collectively referred to as ceroid-lipofuscin. These pigments are autofluorescent (Fig. 9) and can be stained with periodic acid–Schiff (PAS), Luxol fast blue, Sudan black B, and Ziehl-Neelsen acid fast stain. 176 Under electron microscopy, these pigments are observed as dense granular bodies (Fig. 10). Lipofuscin is known as an age-related pigment, and its appearance is considered to be a phylogenetically independent intracellular morphological change in normal aging. 144,176 Intraneuronal deposition of lipofuscin can be seen in very young animals, however, the deposition increases dramatically with aging. Lipofuscin appears in neurons in humans by the age of 9 years and in rat by age 2-3 months particularly in large neurons. 104,132,176 The role of lipofuscin accumulation in neuronal cell death remains unclear, while the extensive accumulation of ceroid-lipofuscin causes fatal progressive neurodegenerative diseases known collectively as neuronal ceroid-lipofuscinosis (NCL), which is the most common inherited lysosomal storage disease affecting humans and domestic animals. 84,177 Molecular biological studies have revealed the genetic features of these inherited diseases, and various mutations have been confirmed in 9 human NCL-associated genes. 84 Among animals, some gene NCL-associated mutations are found in sheep (CLN5, CLN6, and CL10), cattle (CLN5), and dogs (CLN2, CLN5, CLN8, and CL10). 130 Biochemically, subunit c of mitochondrial adenosine triphosphate synthase is the main material that accumulates in NCL (Fig. 11), but saposins A and D also accumulate in some subtypes of the condition. Research into the processes involved in neuronal cell death in NCL, such as oxidative stress, the autophagy-lysosome pathway, or apoptosis, might provide new insights into the biological significance of neuronal lipofuscin accumulation in aged animals. 84 Borras et al demonstrated significant neuronal lipofuscinosis in all 20 aged dogs that were examined (8-18 years old), however, lipofuscin was also present in young dogs (1-5 years old) although at a lower level. 17 In young dogs, lipofuscin exhibited as small perinuclear and granular deposits affecting large neurons such as Betz’s giant motor cells and neurons of some cranial nerve nuclei neurons. In aged dogs, the lipofuscin deposits were more diffuse and appeared as granular deposits affecting the perikaryon and proximal dendritic tree in the neurons of cerebral cortex, basal nuclei, thalamus, hippocampal pyramidal neurons, cerebellar dentate nuclei, and some midbrain nuclei. Lipofuscinosis is a common histologic finding in the brains of aged horses. 23 Neuronal lipofuscinosis was found in the brain of 57 horses aged between 11 and 23 years, but was not found in 3 horses aged 8-10 years old or in 5 young horses age 3 months to 5 years. 23 The pigment was present mainly in the large cortical neurons and numerous neurons of the brainstem nuclei (nucleus vestibularis, formatio reticularis of the pons, nucleus tractus spinalis nervus trigemini, nucleus parasympathicus nervus vagi, and rubber nucleus). Lipofuscin was present mainly in the perikaryon, between the nucleus and the axon hillock; the lipofuscin deposits increased significantly with age. No degenerative changes were noted in the affected neurons. Similar results were reported by Jahns et al, who found an age-dependent increase in the amount and the intensity of neuronal, glial, and neuropilar lipofuscinosis in 100 horses aged between 2 and 25 years. 83 Age-related neuronal lipofuscinosis is also reported in nonhuman primates, 14,117 cat, 199 and cattle. 14
Neuromelanin, a dark brown pigment that is not autofluorescent, is an organic polymer produced by dopamine metabolism. It is primarily found in the neurons of the substantia nigra
Other neuronal changes
Neuronal granulovacuolar degeneration (GVD) is one of the histopathological hallmarks of AD, together with NFT, Aβ plaques, and CAA. 163,190 In humans, hippocampal GVD is characterized by the presence of dense granules (0.5 to 1.5 μm in diameter) in each vacuole together with a doubling of the cytoplasmic diameter in pyramidal cells. 190 The granules are composed of several components including neurofilament proteins (NF), ubiquitin, phosphorylated tau, and other microtubule-associated proteins. 213 Although there have been a few studies of hippocampal GVD in aged animals including rats, wolverines, and dogs, its morphological and immunohistochemical features have not been described in detail. 80,152,219
Neuronal vacuolization was reported in the brain of aged cats, 200 cattle, 52 and horses, 23 however, the correlation of this lesion to aging was not completely confirmed.
Various intracytoplasmic inclusions have been found in the brains of aged animals, especially dogs, and animals suffering from neurodegenerative diseases. 177,179 Negri body-like eosinophilic inclusions (ie, the accumulation of intracisternal microtubules in the rough endoplasmic reticulum) were observed in the pontine nuclei and SN of aged dogs, although they might not have pathological significance. 179 Uchida et al previously detected 2 types of cytoplasmic inclusions in the SN of aged dogs, amorphous basophilic inclusions (9 of 34 cases, Fig. 13) and Negri body-like inclusions (2 of 34 cases, Fig. 14). 192 The former were strongly positive for PAS, and some of the latter inclusions were positive with phosphotungstic acid hematoxylin stain (Fig. 15). Both types of inclusions were negative for α-synuclein and NF, and weakly positive for ubiquitin using immunohistochmesitry. 192 The morphological characteristics of these canine inclusions are quite different from those of the Lewy bodies seen in PD patients.
Several types of intracytoplasmic inclusions have been identified in the motor neurons of patients with familial or sporadic ALS. Bunina bodies are characterized as round to oval eosinophilic intracytoplasmic inclusions. 134 They are immunopositive for cystatin C and negative for ubiquitin and TAR (transactive response) DNA-binding protein of 43 kDa (TDP-43). In addition, intracytoplasmic inclusions that are positive for ubiquitin and TDP-43, and called skein-like inclusions or Lewy body-like/round inclusions are seen in patients with sporadic ALS. 12 The aberrant distribution and accumulation of TDP-43 also occurs in another age-related human disease called frontotemporal dementia or Pick’s disease. 129 In the latter disease, intracytoplasmic inclusions called Pick bodies, which contain highly phosphorylated tau together with TDP-43, are observed in the affected regions. 92 Among these inclusions, Bunina body-like inclusions (Fig. 16) have been detected in large spinal cord neurons in cases of equine motor neuron disease (EMND). 108,177 In addition, the aberrant accumulation of TDP-43 can occur in EMND, 46 however, the pathological roles of TDP-43 in neuronal diseases affecting animals including EMND and canine degenerative myelopathy remain to be clarified.
Neuropilar Lesions
Amyloid plaques
Amyloid plaques (APs) are formed from the accumulation of Aβ, a 36 to 43 amino acid peptide derived from APP. APP is an integral membrane protein, made by neurons and other brain cells and is also expressed in extraneural tissues such as pancreas, kidney, and spleen and is abundant in blood platelets. 25 Its function is unknown. Neurotoxic APs are composed predominantly of amyloid-β (Aβ) peptides of 40 (Aβx-40) and 42 (Aβx-42) amino acids in length, formed by sequential cleavage of APP by β-, and γ-secretases. Aβx-42 is more abundant in plaques than Aβx-40 because of its higher rate of fibrillization and insolubility, and its ability to form insoluble clumps of oligomers. 4 APs accumulate mainly in the neocortex and to a lesser extent in the allocortex (including entorhinal cortex and hippocampal formation), the basal ganglia, relevant nuclei of the brainstem, and the cerebellum. 127,163 APs have been classified histologically into 2 types: the mature or neuritic plaque (NP) and the diffuse or amorphous plaque (DP). DPs are spherical extracellular deposits of Aβ that are up to 100 microns diameter and are negative for both Congo red and thioflavin S. DPs may not be specific for AD, as they commonly found in cognitively intact elderly people. In contrast to NP, DPs are typically not associated with a glial reaction, or neuronal and synaptic loss. NPs are spherical structures composed of a compact central core of fibrillar amyloid deposits that are positive for both Congo red and thioflavin S. NPs are surrounded by degenerating or dystrophic neurons and nerve endings (neurites) containing abnormal PHFs containing hyperphosphorylated tau. NPs are accompanied by reactive astrocytes and microgliosis, and are associated with synaptic loss. The degree of cognitive impairment in AD patients is correlated with the severity of NPs. 127
Age-related Aβ deposits have been detected in the brains of birds, primates, carnivores, bears, and some other mammalian species. 123,154,162,195,200 The amino acid sequence of Aβ is relatively conserved among mammalian species, except in cats and rodents including mice, rats and degus (Table 2). 85 Most AP that form in the canine brain are diffuse plaques. Mature and NPs, which are common in humans, are very rare in dogs. In dogs, AP first form as early as 8 years of age and are observed in half of dogs over the age of 15 years (see Figs. 2 and 3). A strength of the canine model is that the plaques observed to increase with age in the brain contain Aβ that have an identical amino acid sequence (Table 2). 85,162 This feature of canine brains was observed several decades ago and led to the initial suggestion that dogs may a useful model system in which to understand human brain aging and age associated neurodegenerative diseases. 209 As with human brain aging and AD, aging dogs deposit Aβ in areas such as the prefontal cortex and entorhinal cortex, consistent with reports in the aged human brain. 20,69,81,162,188,209 Specifically, if Aβ deposition is quantified in different regions of the brain involved with cognition a different age of onset is observed, which can provide some interesting research opportunities. 69 Aβ deposition occurs earliest in the prefrontal cortex and at later ages in temporal and occipital cortex, similar to previous reports in humans. 188 Thus, one could, based on the age of the dog, test hypotheses regarding the ability of an Aβ modifying intervention to prevent or reverse preexisting pathology and determine the impact on cognition. Aβ is a worthwhile target for these studies as the amount of Aβ and its location is correlated with the type and severity of cognitive decline. 34,38,67,153 More Aβ pathology in the prefrontal cortex of beagles is associated with poorer executive function. 39,67 Entorhinal Aβ is correlated with poor visual learning ability. 67 Another very useful noninvasive and indirect measure of brain Aβ can be by measuring the ratio of Aβ42/40 in the cerebrospinal fluid (CSF), which decreases in a linear fashion with increasing amounts of Aβ measured biochemically in the brain. 72
Amyloid β (Aβ) Amino Acid Sequences of Animals.

Over the last decade, there is an increasing recognition that not all Aβ behaves similarly and it can adopts different conformations and assembly states that have varying levels of toxicity to neurons. Small soluble forms of Aβ called oligomers can dramatically interfere with synaptic function and cognition and have been considered as a highly toxic form of Aβ. 87,203 As with measures of total Aβ in CSF, the amount of Aβ oligomers in CSF is inversely related to the amount of total Aβ measured in the brain, suggesting that oligomers are may be increasingly deposited into plaques. 72
Aβ deposition has also been observed in the brains of aged domestic cats (Felis silvestris catus), 61,71,200 Tsushima leopard cats (Prionailurus bengalensis euptilurus), 28 and cheetahs (Acinonyx jubatus), 173 but there are some interesting differences relative to Aβ accumulation in human brain. For example, feline Aβ appears to deposit as a truncated form of Aβ into diffuse plaques and is not detectable using an Aβ 1-16 antibody (6E10) but can be visualized with an Aβ 17-24 antibody (4G8), both of which can detect human and canine Aβ. 71 The amino acid sequences of the Aβ molecules found in domestic cats and Tsushima leopard cats are one residue different from the human Aβ sequence (at the 7th residue of the N-terminal sequence), which might result in weak Aβ aggregation in Felidae cats. 28 Methenamine silver positive–diffuse plaques were observed in the neocortex of 4/18 horses 9-23 years old, however, these plaques were negative with both Aβ (bA4) immunostaining and Congo red. 23
Information about Aβ deposits and Aβ plaques in other species are summarized in section III and Table 1 of this review.
Polyglucosan bodies
Inclusion bodies composed of glycoproteins are collectively referred to as polyglucosan bodies (PGBs) and are found in normal brain aging as well as neurodegenerative diseases. PGBs are PAS-positive, binds to lectin concanavalin A, and spherical to radial or conglomerate in morphology. 5 In general, PGBs that develop in glial cells, especially astrocytes, are called corpora amylacea (CA), which are often seen in the central nervous systems of aged individuals without any apparent functional impairments (Fig. 17). On the other hand, PGBs that develop in neurons in association with Lafora disease, a genetic disease that causes progressive myoclonic epilepsy, are called Lafora bodies (Fig. 18). However, in the veterinary field the terminology used to describe age-related PGBs is inconsistent (ie, Lafora-like bodies or CA) due to the uncertainty about their functional significance and cellular location (see Fig. 17).
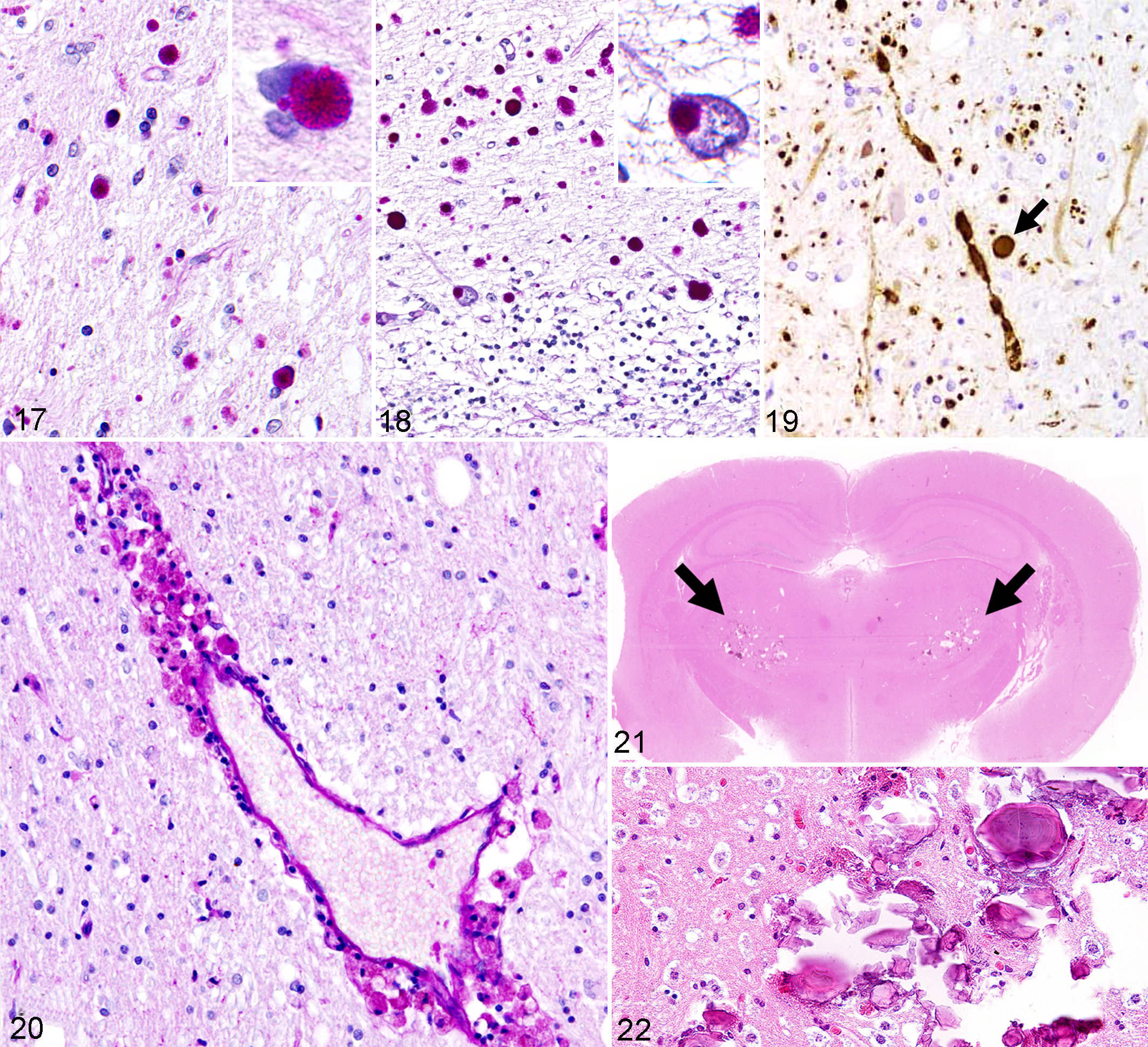
Age-related PGBs have been detected in various animal species, 26,180,195 however, their frequency and distribution have been studied most extensively in dogs. In a study by Suzuki et al, PGBs were found in the brains of 56% of 3- to 6-year-old dogs and 100% of 8- to 16-year-old dogs. 178 In aged dogs, PGBs are frequently distributed in the diencephalon, particularly in the neuropil and neuronal cell bodies, based on light microscopic observation. Ultrastructurally, PGBs are detected in neuronal perikarya and axons, although no neurological signs were observed in these dogs. In the authors’ experience, the number of PGBs found in the canine brain varies among individuals and the pathological significance of their number, distribution, and size remain unknown.
Lafora disease has been described in several animal species. Expanded repeats in the Epm2b gene have been identified as a cause of progressive myoclonic epilepsy in miniature wirehaired dachshunds (MWD). 100 Mutations in the Epm2b gene are also known to cause Lafora disease in humans. 30 Unlike human Lafora disease, in which signs are first seen in juveniles (teenagers), in MWD neurological signs in miniature wirehaired dachshunds appear in adulthood (6 to 9 years old), and some affected dogs live out their natural life span (over 15 years) (Fig. 18).
Neuroaxonal dystrophy
Axonal swelling (spheroid formation) is a characteristic lesion of neuroaxonal dystrophy, which can be congenital (not related to aging) or acquired (age-related), and the distribution and diameter of swollen axons differs between each type of the condition. 191 Spheroids are also seen in animals without any neurological signs, and their frequency increases with age. 181 In aged dogs, spheroids are often observed in the brain stem, especially in the dorsal column nuclei of the medulla oblongata as well as in the spinal cord (Fig. 19). In addition to the changes that affect the cerebral white matter (discussed below), ubiquitin-positive granules are often observed together with swollen axons in aged dogs. The functional significance of these changes remains unknown. Similarly, ubiquitin-labeled axonal spheroids and neuronal swellings were identified, mainly in the midbrain and striatum of a 17 horses out of 18 aged horses (9-23 years old) using the Gallyas silver stain and immunostaining for ubiquitin., indicating a proteolytic phenomenon occurring in these structures. 23
Other neuropilar lesions
In aged dogs, the cerebral white matter atrophy, apparently due to significant myelin loss has been observed. 29 In addition, macrophages laden with ceroid-lipofuscin pigment accumulate in the perivascular space in the white matter (Fig. 20), and thus these oxidized lipids are likely to be originated from degenerated myelin lipids. Recently, the role of cerebral white matter degeneration in cognitive impairment in elderly humans has been thought to play a larger role in dementia than originally appreciated, especially when it occurs in association with vascular lesions. 76 Capillary Aβ deposition is related to apolipoprotein E (ApoE) genotype in humans and is considered to be an important factor for white matter degeneration. 6 The colocalization of capillary Aβ deposits with ApoE protein is commonly seen in the brains of aged dogs. 29 The affected capillaries are often occluded, which might subject the white matter to oxidative stress, leading to cognitive decline. 68
In aging mice and other small rodents, focal mineralization occurs in the thalamus. 49,218 Although the etiology and functional significance of such lesions remain unknown, they might involve a substantial amount of mineral deposition, and hence, space occupying (Figs. 21 and 22).
Vascular Lesions
Vasculopathy such as CAA and atherosclerosis are important causes of cerebral hemorrhages, infarctions, and leukoencephalopathy in AD patients. The aged canine is considered to be a unique model to study the cerebrovascular features of aging and AD due to the spontaneous nature of these same neuropathological features in older animals.
Cerebrovascular amyloid angiopathy
CAA is a common lesion observed in both normal human aging brain and particularly in AD. 6,163 In people, CAA is thought to be more extensive in the posterior occipital and parietal areas and affects mainly cortical capillaries, small arterioles and middle-size arteries as well as leptomeningeal arteries, however, venules, veins, and white-matter arteries are rarely involved. 163 CAA is common in aged dogs and is seen in almost 100% of dogs over the age of 13.2 years (see Figs. 4 and 5), however, the density may vary dramatically between affected dogs. 202,204 In dogs, Aβ is mainly deposited in the vascular walls of meningeal arterioles and/or capillaries in the deep cortex. Vascular walls containing Aβ deposits become fragile, which may lead to micro and macro hemorrhages, breakdown of the blood brain barrier, and be related to the loss of myelin in the affected region. 29,193,194,199 The occipital cortex also appears to be more vulnerable to CAA in aging dogs as with human brain aging and AD. 6 Vascular Aβ typically includes the shorter 1-40 species, which is identical in dogs and humans. 208 As with human brain aging and AD, there is increasing recognition that CAA can be a contributor to clinical signs of cognitive dysfunction in pet dogs. 34 Furthermore, beagles have been used as a model for ischemic stroke and both FLAIR and T2-weighted imaging shows significant white matter hyperintensities suggesting pathology (eg, hemorrhage and edema) resulting from vascular lesions can be visualized with the appropriate imaging approaches. 86 Despite significant CAA at necropsy in canine brain, there are currently few reports of losses in white matter integrity in aging dogs, 29,120 or increased neuroinflammation. 145,146,164 Chambers et al, studied 51 dogs ranging in age from 1-20 years including multiple breeds and found an age-dependent loss of myelin basic protein by immunohistochemistry associated with increasing amounts of CAA. 29 Interestingly, there is little data regarding changes in the neuroinflammatory milieu in aging dog brain despite reports of higher levels of activated microglia and astrocytosis. 145,146,172 Thus, the prevalence of cerebrovascular disease (CVD) and how it relates to white matter integrity and inflammation has yet to be determined. Overall, dogs are thought to be a good model for cerebrovascular pathology and CAA. In terms of designing future clinical trials, characterizing CVD in dogs will be critical given that CVD is mediated to large extent by lifestyle factors that are amenable to intervention. Individuals with hypertension, high cholesterol, obesity, and type 2 diabetes show a higher risk of CVD. 150 Furthermore, neurobiological studies may help identify molecular pathways that could be manipulated in future canine aging studies. Several environmental manipulations and pharmacological studies that modify lifestyle factors have been successfully implemented in the canine model with some showing significant benefits to cognition. 36 Age-dependent CAA is also reported in the cerebral blood vessels of cats, 200 and nonhuman primates. 117,123 In contrast, CAA was not detected in the brain of 18 horses aged from 9 to 23 years using immunostaining for Aβ and Congo red staining. 23
Vascular degeneration
Spontaneous vascular degeneration such as atherosclerosis is extremely rare in animals with the exception of aged pigs and birds, and also it occurs in dogs in association with hypothyroidism. Chronic arterial changes (arteriosclerosis) consist of luminal narrowing due to proliferative and degenerative changes in the media and the intima are more common than atherosclerosis in domestic animals even if they normally do not cause disturbance of blood flow. Chronic arterial degenerative changes were described in equine meningeal blood vessels in 58% of aged horses. 23 Similar degenerative changes were observed in the brain/meningeal arteries of old/aged pigs and cattle (MTC, unpublished data). Atheromas and fibrofatty plaque or cholesterol clefts (lesions observed in advanced human atherosclerosis) were not detected. The significance of these vascular changes remains unknown.
Vascular mineralization
Mineralization (calcification, siderocalcinosis) of brain vasculature not pertaining to generalized vascular diseases is well documented in humans, 103 monkeys, 217 cows, 214 mice, 218 rats, 216 cats, 105 dogs, 47 and horses. 23,118 The clinical significance and the pathogenesis of these brain changes remains undetermined. They have been considered an age-dependent incidental finding that has no clinical significance, 83,215 although neurological signs 118 and areas of infarction with neovascularization were described in association with severe siderocalcinosis and vascular occlusion. 23,108,177 Mineralization involves venules, arterioles, and/or capillaries. The largest deposits usually are present in and around the arterioles, occasionally showing a concentric lamellar structure, with occlusion of the lumen. Small deposits are occasionally found in the neuropil, with no relation to the blood vessels. X-ray microanalysis of the deposits confirmed the presence of both calcium and phosphorus in small and large deposits in mice and horses. 116 In the horse, particularly calcium, phosphorus and magnesium were observed most frequently, with small quantities of other minerals including aluminum, zinc, potassium and sodium. 23,214 This mineralization increases with age in horses. 23 The deposits generally stain positively with PAS, von Kossa, and Perl’s stains, indicating the presence of mucopolysaccharides, calcium, and iron salts. In horses and cows, vascular mineralization involves particularly the corpus striatum (globus pallidus) and thalamus and the white matter, peduncles, and roof nuclei of the cerebellum. 23,118
Meningeal Lesions
Meningeal thickening as result of fibrosis with collagen deposition may be found in old animals particularly in dogs, 17,177 cats, 136 and horses. 177 This can be diffuse or focal in distribution, but more prevalent in the dorsal aspect of the cerebral sulci. Macroscopically meningeal fibrosis is observed as a white prominence of cerebral sulci. Focal mineralizations are sometimes organized in concentric structures resembling psammomatous bodies. Meningeal fibrosis and mineralization are considered age-related changes without clinical significance.
Plaques of osseous metaplasia (also named spinal dural ossification, osseous dural metaplasia, or ossifying pachymeningitis) are occasionally observed in the spinal dura mater of old dogs particularly in the cervical and lumbar tracts, of old large breed dogs. 157,201 These islands of bone, located ventral or dorsal to the spinal cord, may contain adipocytes and myeloid elements. They are usually considered as an incidental age related finding, however, in rare cases they were thought to have caused functional effects such as hyperesthesia of 1 or both thoracic limbs, progressive paresis, ataxia, atrophy of the limb musculature, urine retention, and incontinence. 101,105 In 1 report a 6-year-old Ragdoll cat with neurological signs had ventrally located dural plaques of osseous metaplasia on the C3-C5 segments and caudal lumbar areas. 3 The etiology of dural ossification remains unknown. It was suggested that mechanical factors such as rubbing of the dura matter against adjacent vertebral structures (eg, articular facets) or against other spinal disorders, such as disk protrusion may cause the lesion. 7 To our knowledge, no studies have confirmed that ossification results from inflammation, suggesting that the term “ossifying pachymeningitis” is not appropriate. 40
Cholesterol granulomas (Cholesteatoma) are typically intracranial pathologic lesions in animals and also occur rarely in extraneural sites. Intracranial granulomas are described in the meninges, choroid plexuses and rarely in the brain parenchyma. They are mostly reported in aged horses and only sporadically in other species, including sheep, cat, dog, meerkats, reptiles, and amphibians. 23,108,177 Microscopically meningeal granulomas are characterized by an accumulation of needle-like or fusiform empty structures (cholesterol clefts) inducing a severe granulomatous reaction composed of epithelioid macrophages, multinucleated giant cells, plasma cells, and glial cells. 108 Although neurologic deficits associated with this lesion have been reported in domestic species, most are found incidentally at necropsy particularly in aged animals. 102,189
Glial Lesions
Reactive astrocytosis and microgliosis are nonspecific lesions associated with multiple inflammatory, neurodegenerative, and neoplastic neurologic diseases. Age-related astrocytosis or microgliosis not related to these conditions are poorly documented in humans and animals. Age-related astrocytosis and/or microgliosis have been reported in cattle, 52 and dogs. 200 Borràs et al studied age-related lesions including glial changes in the brain of 20 dogs (8 to 18 years) and found diffuse bilateral astrocytosis in 7 dogs (35%) and increased staining intensity of glial fibrillary acidic protein (GFAP) in 17 dogs (85%). 17 Lesions were more prominent in the white matter, specifically in the corticomedullary junction, corpus callosum, internal capsule, hippocampus, and cerebellar white matter. These lesions were not present in the brains of 10 young dogs (1-5 years old). Multifocal or disseminated astrocytosis was reported in the brains of 18/60 (30%) horses from 7 to 23 years old, and occurred mainly in the white matter of brainstem and occasionally in the globus pallidus, where increased GFAP staining intensity was also observed. 23 There was no significant relationship with age of the horses. No alteration in microglia or oligodendroglia cells was observed. 23 The same article reported small foci of satellitosis in 55/60 (92%) horses, especially in the frontal cerebral cortex, thalamus and midbrain. This lesion was significantly more common in the older animals (15-23 years).
Animal Models of Age-Related Neurodegenerative Human Diseases
We describe here spontaneous and transgenic animal models of human age-related neurodegenerative diseases including AD, PD, and progressive supranuclear palsy. Other neurodegenerative diseases such as DLB, Huntington’s disease, multiple system atrophy, and spinocerebellar degeneration are only reported in humans.
Alzheimer’s Disease
Evolution of Alzheimer’s disease pathology
Figure 23 shows the life spans of representative species and the relative timing of the processes that lead to AD pathology. 125 The production and deposition of Aβ occur at the beginning of the aged period. This process results in a zone of NFT formation in humans, but not cynomolgus monkeys or dogs. In the latter species, the process stops after the subsequent formation of the Aβ zone due to disorders affecting other organs. However, transgenic mice with genetic mutations in the human APP develop Aβ deposits due to their increased Aβ production. 51,74 The speed of brain aging might be fixed in each species, and species differences in brain aging are presumably smaller than differences in whole body aging. Therefore, the short life span of mice means that they do not develop Aβ zones; the life spans of dogs and cynomolgus monkeys allow them to develop Aβ zones, but not NFT zones; and the extended life spans of humans results in them developing both Aβ and NFT zones. It has been suggested that if the life spans of animals could be extended considerably, NFT would also form in their brains (the brain aging speed hypothesis).
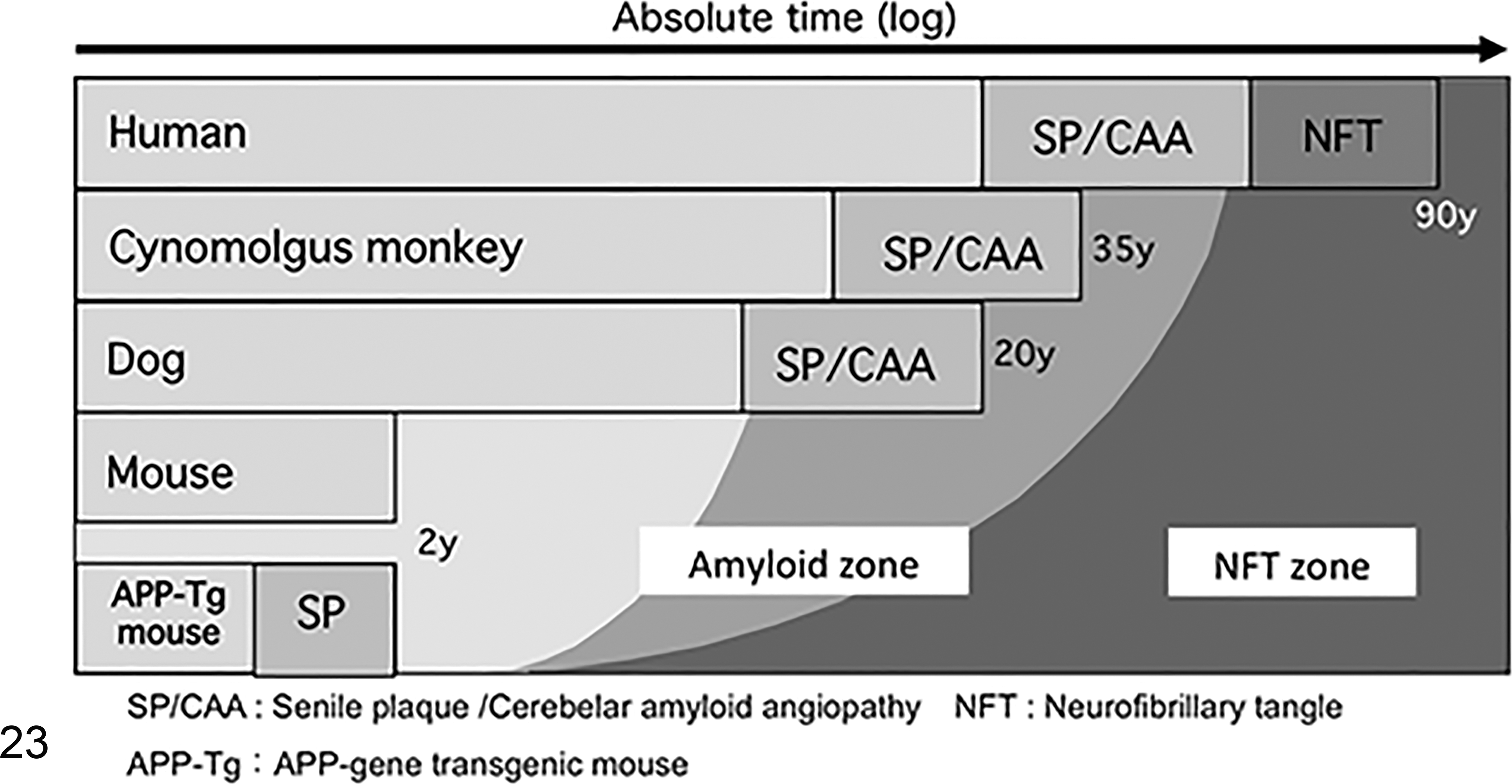
The process of age-related brain lesions in animals (the brain aging speed hypothesis). Each animal species shows amyloid-β plaque (Aβ) deposition in the brain in their old age. The Aβ lesion induces NFT over a long period of time in humans, but does not in cynomolgus monkeys or dogs. No Aβ lesions are formed in mice. On the contrary, APP-transgenic mice, which have artificially enhanced Aβ production, can develop Aβ deposition in the brain.
Recently, it has been revealed that NFT composed of hyperphosphorylated tau form in the brains of domestic cats, Tsushima leopard cats, and degus, 24,28,187 which cannot be explained by the brain aging speed hypothesis. Table 2 summaries the Aβ amino acid sequences of various animals. The species differences in the amino acid sequences of the N-terminal region of the Aβ molecule might play a role in Aβ aggregation and/or tau phosphorylation.
Spontaneous animals models of Alzheimer’s disease
Models of AD should possess 2 histopathological hallmarks, namely Aβ deposition and NFT formation. However, no natural animal models that fulfilling these conditions have been reported. Therefore, most research into AD has been performed using transgenic rodents. Various age-related lesions are observed in the brains of animals (Table 1), and some of them are identical to those seen in AD patients. The brains of nonhuman primates and of humans brains have multiple structural, functional, and molecular similarities that make nonhuman primates an excellent model to study human neurodegenerative diseases including AD. In this section we describe the AD-like lesions that were reported in the different species and families of nonhuman primates. Aβ deposition has been reported in the brains of aged apes, old world monkeys, new world monkeys, and strepsirrhines. Apes (Superfamily Hominoidea). Apes are the most closely related monkeys to humans and are classified into great apes, including orangutans (Pongo spp.), gorillas (Gorilla spp.), and chimpanzees (Pan troglodytes), and small apes (Hylobatidae). AP have been observed in the brains of aged orangutans,
162
gorillas,
91
and chimpanzees.
162
Most of these AP were diffuse plaques, but a small number were mature plaques. PHFs were observed in the brain of a 41-year-old chimpanzee. However, the case involved a large ischemic infarct, which might have been responsible for the formation of PHFs. Old world monkeys (Superfamily Cercopithecoidea). Rhesus monkeys (Macaca mulatta),
143
cynomolgus monkeys (Macaca fascicularis),
123
green monkeys (Chlorocebus sabaeus),
98,122
and baboons (Papio spp.),
126
all of which are old world monkeys, exhibit a higher frequency of classical plaques than other animals. The AP found in the brains of cynomolgus and rhesus monkeys display a similar distribution to those seen in humans. In addition, as these monkeys are established laboratory animals, they have been used as models for studying AP formation. The deposition of hyperphosphorylated tau protein in neurons, which were positive with Gallyas-Braak stain, was observed in the cerebral cortices of aged cynomolgus monkeys. However, few NFT were seen in these animals, and neuronal loss has never been detected in old world monkeys.
135
Hyperphosphorylated tau-positive fibrillary structures are observed in the cerebral neurons, astrocytes, and oligodendrocytes of 90% of baboons over the age of 26. These hyperphosphorylated tau-positive cells are mainly distributed in the hippocampal dentate gyrus, which is different from the distribution seen in the brains of AD patients.
160
Schultz et al examined 50 aged baboon brains and reported that argyrophilic NFT were only observed in one 30-year-old animal, and on ultrastructural examination demonstrated that the NFT were composed of single filaments.
160,161
In addition, a considerable number of individuals demonstrated hyperphosphorylated tau deposition without Aβ deposition. Therefore, the relationship between tau and Aβ deposition in baboons remains unclear. New world monkeys (Parvorder Platyrrhini). Age-dependent Aβ deposition has been reported in new world monkeys, such as common squirrel monkeys (Saimiri sciureus),
27
common marmosets (Callithrix jacchus),
55
and tamarins (Saguinus spp.).
48
New world monkeys are smaller than old world monkeys, have shorter life spans, and develop Aβ deposits earlier. For example, rhesus monkeys develop such deposits in their twenties whereas squirrel monkeys develop these deposits in their early teens. Squirrel monkeys are therefore more often used for research on brain aging. Mature AP are rarely observed in the brains of aged squirrel monkeys, but marked CAA, is seen. Therefore, squirrel monkeys have received much attention as a model of CAA. Primitive monkeys (Suborder Strepsirrhini). Aβ deposition has been reported in the brains of the smallest primate species, mouse lemurs (Microcebus spp.).
15,16
Some mouse lemurs develop diffuse AP and CAA lesions at less than 5 years of age, which makes them the youngest animal species known to develop spontaneous AD-related brain lesions earliest. Tree shrews (Family Tupaiidae). Tree shrews (Tupaia spp.) were previously classified into the order Primates, but are now considered to belong to the order Scandentia. The amino acid sequence of tree shrew Aβ is identical to that of human Aβ, but Aβ deposition has never been observed in the brains of aged (7 to 8 years old) tree shrews.
139
Transgenic animal models of Alzheimer’s disease
Spontaneous aging lesions in rodent brains are poorly documented. Wild-type rodents do not develop spontaneous AD-like lesions (AP and/or NFT) as they age but they develop cognitive decline and several behavioral changes that are correlated with those seen in AD. Therefore, wild-type rodents are not used to study pathologic AD phenotypes. The sequences of the Aβ proteins in mice and rats differ by 3 amino acids from that of the human Aβ protein. 85 The only known nontransgenic mouse strain that naturally develops an accelerated brain aging phenotype is the senescence-accelerated mouse 8 (SAM8) which was generated through aging phenotypic selection of AKR/J mice. The genes responsible for the accelerated aging in SAM8 mice are yet to be determined. SAM8 mice develops age related increase in hippocampal Aβ, gliosis, status spongiosis from 4 to 12 months of age but with no plaques or NFT formation. 212 They also exhibits age-associated cognitive and behavioral deficits. Several transgenic mice were developed to mimic the Aβ pathology (eg, deposition of Aβ plaques) and Tau pathology (eg, formation of NFT) characteristic of AD. These models have single, double or multiple mutations in the genes responsible for production of Aβ and/or tau protein. Examples of these transgenic models are discussed below and summarized in Table 3.
Selected Transgenic Animal Models of Alzheimer’s Disease (AD), Parkinson’s Disease (PD), and Progressive Supranuclear Palsy (PSP).

Abbreviations: AP, amyloid plaques; CAA, cerebrovascular amyloid angiopathy; DP, diffuse plaques; MP, mature plaques; NFT, neurofibrillary tangles; TH, tyrosine hydroxylase.
The most common mouse models of amyloidosis affecting the brain include APP23, PDAPP, and Tg2576 models. All of these 3 models overexpress mutant human APP and support the amyloid cascade hypothesis of AD. They lack NFT transformation, but develop severe amyloidosis seen as diffuse and NPs, CAA, and synaptotoxicity, in the hippocampus, corpus callosum, and cerebral cortex, in addition to behavioral, and cognitive abnormalities. 51,74,174 The first Aβ deposits are usually observed at 6 months in APP23 and PDAPP mice and at 9 months in Tg2576 mice. 174 However, these mouse models lack the tau protein abnormalities, therefore, the mutated human tau mice (THY-Tau22) were developed to mimic the tau pathology (ie, misfolding of tau protein and the formation of NFT). THY-Tau22 mice develop learning and behavioral deficits and tau pathology at age of 3 months which includes hyperphosphorylation of tau on many epitopes (eg, AT8, tau-pSer396, and AP422) with development of NFT-like structures, PHF-like structures, and rare extraneuronal (ghost) tangles. 159 Several double and multiple mutant mice models were developed to combine both the Aβ and tau pathology in 1 animal. For instance, the Tg2576/Tau(P301 L) (APPSwe-Tau) mouse which is generated by crossing Tg2576 mice that overexpress a mutant form of APP with the JNLP3 mouse that carries mutant tau protein. The resultant bigenic mouse model develops NFT and gliosis as early as 3 months and plaques at 7-9 months of age. 99
Brain Lesions Resembling Parkinson’s Disease in Animals
PD, which generally develops after middle age, is a neurodegenerative disorder whose characteristic signs include tremors, akinesia, and rigidity. The signs of PD are caused by damage to the SN pars compacta (pc) and the subsequent effects on the midbrain and striatum, which receive projections from the SNpc. Intracytoplasmic inclusions called Lewy bodies, which are composed of phosphorylated α-synuclein, appear in the dopaminergic neurons in the SN in patients with PD. The sporadic type of PD is the most common form of the condition and the cause remains unknown, whereas several mutations have been identified in the genetic type of PD. No spontaneous diseases that are identical to PD have ever been reported in animals.
Examination of dogs for PD-like brain lesions
Dopaminergic neurons disappear from the SN in PD patients. As these neurons possess tyrosine hydroxylase (TH) in their cytoplasm, TH is widely used as a specific marker of dopaminergic neurons. The number of TH-positive cells, as estimated by immunostaining with an anti-TH antibody, is decreased in the SN of PD patients. In a canine study, the brains of 34 dogs aged from 2-months-old to 18 years were immunostained for TH, glial fibrillary acidic protein (GFAP), and ubiquitin. 192 It was found that the number of GFAP-positive and/or ubiquitin-positive regions increased with age, while the number of TH-positive cells in the SN remained constant regardless of age. Lewy bodies have never been detected in the canine brain. In humans, the number of dopaminergic cells in the SN decreases with age. Accordingly, the amount of dopamine produced declines, and PD symptoms appear when the amount of dopamine falls to 20% of the normal level. In contrast, no reduction in the number of dopaminergic cells has been reported in the midbrains of dogs.
PD-like brain lesions in aged cats
In cats, it was found that the numbers of ubiquitin-positive cells and TH-positive cells remained constant regardless of age. In addition, the number of α-synuclein-positive cells increased with age, but not significantly. Furthermore, alpha-synuclein accumulates in the cytoplasm of dopaminergic cells in the SN of cats in the form of coarse granules and later spreads to the whole cell body by the age of 10 years. However, no Lewy bodies have been detected in the brains of aged cats. The above-mentioned descriptions are unpublished personal observations (HN, KU, and JKC). According to the above findings, α-synuclein is deposited in the SN of cats during aging, but cats die before Lewy bodies have time to form.
Drug-induced Parkinson’s disease models
The administration of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), which is a by-product of opioid production can induce PD-like signs in young men, mice and several monkey species. 64 MPTP reaches the brain parenchyma through the blood brain barrier and is biotransformed to 1-methyl-4-phenylpyridium (MPP+) by monoamine oxidase-B in glial cells. MPP+ damages dopaminergic cells in the SN, and the subsequent reduction in the amount of dopamine causes PD. Lewy bodies, which are the most typical histopathological hallmark of PD, were not detected in MPTP-induced models of the condition.
When rotenone, which is used as an insecticide or pesticide, was subcutaneously administered to rats for a long period, the animals displayed PD-like signs, such as tremors, akinesia, and rigidity. 11 Histopathologically, Lewy body-like α-synuclein aggregations were observed in dopaminergic cells in the rats’ SN, and the dopaminergic cells disappeared as the condition progressed. PD-like signs have also been reported in mice and rats administered 6-hydroxydopamine or paraquat/maneb. 10
All the drug-induced conditions mentioned above could be used as models of PD, but none would elucidate the cause of the condition.
Transgenic models of PD
Strategies to develop transgenic animals to mimic PD are based on deletion and or inducing mutation of alpha-synuclein, PINK1, Parkin, and/or LRRK2 genes. Mutations in these genes are linked to autosomal dominant PD (alpha-synuclein and LRRK2) and autosomal recessive PD (Parkin and PINK1). 39 For example, LRRK2 mutant mice develop axonopathy of nigrostriatal dopaminergic neurons, associated with hyperphosphorylated tau. 211 Other transgenic models such as the Park2 knockout mouse and Pink1 knockout rat develop different pathological (Table 3), neurochemical and behavioral phenotypes that are more or less comparable to human PD. 39
Progressive Supranuclear Palsy-Like Disease in a Cynomolgus Monkey
Progressive supranuclear palsy (PSP) is one of the tauopathies. The pathologic phenotypes include neuronal loss, gliosis, and NFT. No APs are seen in PSP. A previous report described a case of a PSP-like condition in an albino cynomolgus monkey over 35 years of age. 89 The animal displayed neurological signs such as gait disturbance, trembling, a drowsy tendency, and reduced activity. Histopathological examinations of the animal’s brain revealed fibrillary tangles in its oligodendroglia and neurons. The cytoplasmic tangles were argyrophilic and tau-positive, and some of them were also positive for α-synuclein. The abovementioned clinical and histopathological findings demonstrate that the monkey had been suffering from a PSP-like disease. Finally, transgenic mouse models such as THY-Tau22 and MAPT develop taupathies that resemble PSP and can be used as an animal model for PSP. 42
Summary and Conclusions
We have reviewed spontaneous age-associated diseases in animals that parallel changes observed in human brain. In some animals, the extent of neuropathology can reach levels where cognitive decline is observed, and may serve as animal models for AD. In addition, we have discussed the most common transgenic or induced animal models of AD, PD, and PSP. The use of several different models providing converging evidence of a role for specific pathologies on cognition should be encouraged as each provides unique information. Animals that spontaneously develop diseases resembling those of humans are particularly useful to evaluate treatments that modify disease and improve cognition as these models may be more easily translated to human clinical trials. Given the current lack of effective treatment for AD, it is critical to continue to refine and develop natural models of human brain aging and disease.
Footnotes
Acknowledgements
We are grateful to Ms Paula Thomason from the College of Medicine of the University of Kentucky, who did extensive English editing to this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
