Abstract
Fatal meningoencephalitis caused by equine herpesvirus-1 (EHV-1) was diagnosed in a reticulated giraffe (Giraffa camelopardalis reticulate). The giraffe died following a history of stumbling, incoordination, and abdominal pain. Gross examination of the brain revealed asymmetric edema and red-brown discoloration, predominantly within the telencephalon. Microscopically, there was perivascular lymphohistiocytic cuffing, multifocal gliosis, and neuronal necrosis in the cerebrum. Necrotic neurons contained acidophilic intranuclear inclusions. EHV-1 was isolated from the brain of the giraffe, and polymerase chain reaction was positive on sections of the brain. Immunohistochemistry using an EHV-1-specific antibody identified positive staining in neurons, astrocytes, and endothelial cells. The giraffe had been housed with a group of zebras that were serologically positive for EHV-1 and suspected as the source of infection. This raises concerns for cross-species transmission of EHV-1 when housing equids together with other species in zoologic collections.
Keywords
Equine herpesvirus (EHV)-1 and EHV-4 are widely distributed within horse populations, and respiratory disease, abortion, and myeloencephalitis caused by either virus has been described worldwide. 6, 8, 11 However, EHV-1 accounts for the vast majority of cases with neurologic lesions. 6 Although neurologic disease tends to occur sporadically, in recent years outbreaks of clinical disease with high morbidity have been observed, similar to abortion storms caused by EHV-1. 6 Neurologic disease commonly follows an outbreak of rhinopneumonitis or abortion; however, the central nervous system (CNS) disease may occur alone, and circumstantial evidence suggests this disease is caused by particularly neurovirulent strains of EHV-1. 2, 6, 8, 9, 11 Horses of all ages are susceptible to an infection with EHV-1, but CNS disease predominantly affects adult horses, unlike other herpesvirus infections. 11 Although vaccination significantly reduces the incidence of respiratory disease and abortion in equine populations, there is no current vaccine that provides protection against the neurologic disease caused by EHV-1. 6, 8, 11
This report describes the clinical, gross pathologic, and histopathologic findings of herpesviral meningoencephalitis in a reticulated giraffe caused by a herpesvirus with sequence similarity to EHV-1. CNS disease caused by EHV-1 has been reported in other equid as well as ruminant and camelid species. 1, 3, 9, 11 EHV-1 is known to affect zebras 1 and can establish silent infections in donkeys, which serve as reservoir hosts for infection in horses. 11 It appears as though zebras act primarily as silent carriers; however, overt clinical disease has occurred in this species. 1, 3, 5 There are a few reports of EHV-1 affecting nonequid species such as llamas and alpacas, 5, 9 as well as a Thompson's gazelle. 3 Other important herpesviruses that cause clinical disease in zoo species include the recently described endotheliotropic betaherpesvirus of Asian and African elephants. 4, 7 Distribution and severity of herpesviral encephalitis often differs between natural and accidental hosts in terms of enhanced neurovirulence; herpes simplex virus in rabbits causes a severe and fulminating encephalitis as opposed to the disease in humans, and herpes B virus in humans is rapidly fatal and widespread in contrast to other nonhuman primates. 10 Such cross-species differences in disease distribution and severity may explain the lesions observed in this giraffe as contrasted with the disease in horses. Alternatively, a new novel giraffe herpesvirus with sequence similarity to EHV-1 may be responsible for the disease in this animal. To date, alphaherpesvirus infections have not been described in any giraffe species. The uncommon distribution of microscopic lesions is reviewed and contrasted to the lesion in horses with EHV-1 meningomyeloencephalitis.
An 18-month-old male reticulated giraffe housed in a zoo presented with a one and a half day history of incoordination, stumbling, and abdominal pain. The giraffe was housed in a pen with several zebras that were reportedly healthy; however, the vaccination status of these animals was unknown. Approximately 7 days before the onset of clinical signs, the giraffe was bitten by a zebra stallion in the enclosure. Because of a worsening clinical condition and poor response to treatment, the giraffe was euthanized and submitted for necropsy examination.
On necropsy examination, the giraffe was in good body condition and weighed 1,050 pounds. There was a traumatic wound on the dorsal aspect of the tail base that was 10 cm in diameter and 2-cm deep, covered by a thick crust. There was a focal 3-cm area of malacia within the cerebrum on cut section (Fig. 1). There were no other lesions noted on gross necropsy examination.
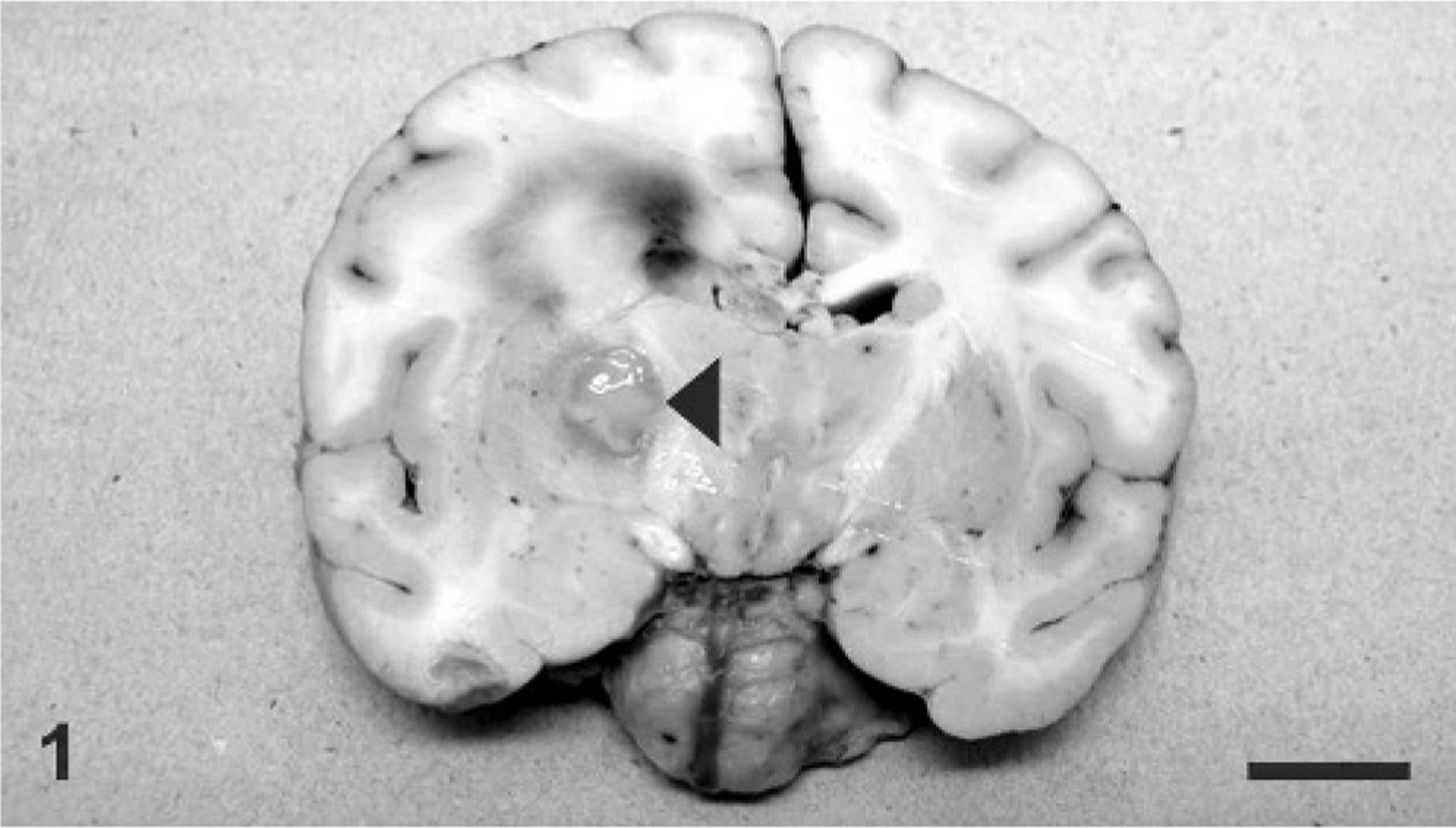
Brain, coronal section; giraffe. A fairly well demarcated focus of malacia (arrowhead) is within the putamen with extensive edema and multifocal hemorrhage of the entire right hemisphere, leading to a midline shift.
Sections of brain; cervical, thoracic, and lumbar spinal cord; heart; lungs; liver; spleen; kidneys; adrenal glands; rumen; abomasum; jejunum; ileum; and colon were examined microscopically. There was severe multifocal nonsuppurative meningoencephalitis within the white and gray matter of the cerebral cortex, hippocampus, thalamus, midbrain, and medulla oblongata, and was most severe in the thalamus and ventrolateral portions of the cerebral cortex. Lesions were characterized by multifocal perivascular cuffs composed of moderate numbers of lymphocytes and histiocytes (Fig. 2). The endothelium of affected blood vessels was swollen, and there were numerous microglial cell foci distributed diffusely in the gray and white matter. Multifocal neuronal necrosis was most evident in the ventral portion of the cerebral hemispheres, and areas of neuronal necrosis were occasionally associated with characteristic herpetic eosinophilic intranuclear inclusions in neurons and astrocytes (Fig. 3).
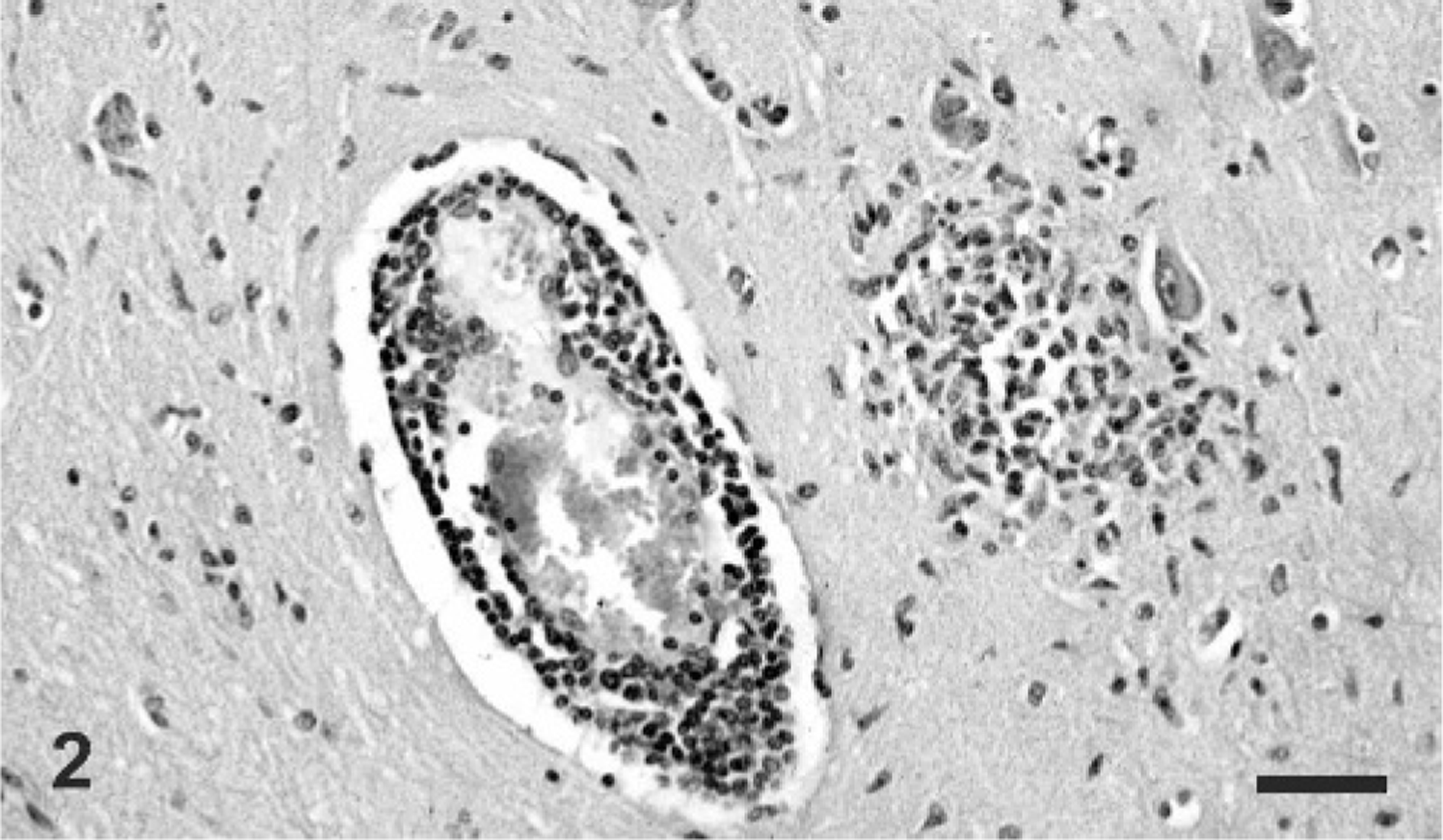
Brain, cerebrum; giraffe. Prominent lymphocytic perivascular cuffing and a focal glial nodule is present within the neuropil. HE. Bar = 100 μm.
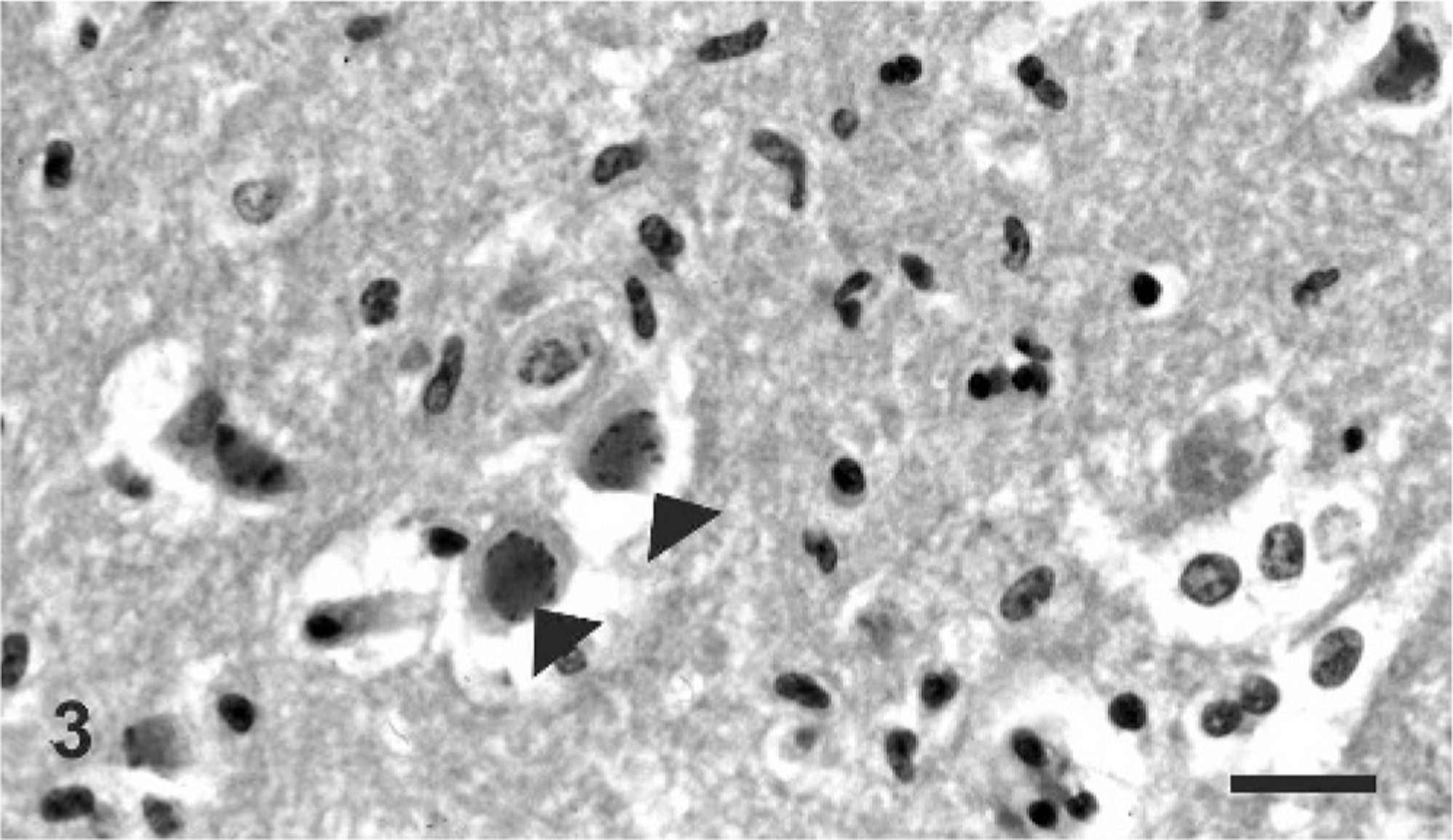
Brain, cerebrum; giraffe. Distinct Cowdry type A intranuclear inclusion bodies are present within neurons (arrowheads). HE. Bar = 40 μm.
Using an antibody against EHV-1 (Ken Smith, Animal Health Trust, Newmarket, UK), immunohistochemistry examination revealed positive labeling within astrocytes, neurons, and endothelial cells (Fig. 4). A herpesvirus was isolated from sections of brain and confirmed as EHV-1 by fluorescent antibody testing. Polymerase chain reaction (PCR) was performed on brain samples from the giraffe for the herpesvirus terminase gene that encodes for a packaging protein and the DNA polymerase gene. Agarose gel electrophoresis revealed a 500-base pair (bp) fragment of the terminase gene, and a 150-bp fragment of the DNA polymerase gene (Figure 5) Following the sequencing of portions of the PCR products and a basic local alignment search tool (BLAST) search, there was 100% (118/118) sequence homology to the terminase gene region of EHV-1 and 96% (59/61) sequence homology to the DNA polymerase gene region of EHV-1. When compared to EHV-4, there was only 95% (113/118) sequence homology to the terminase gene region and 90% (55/61) sequence homology to the DNA polymerase gene region.
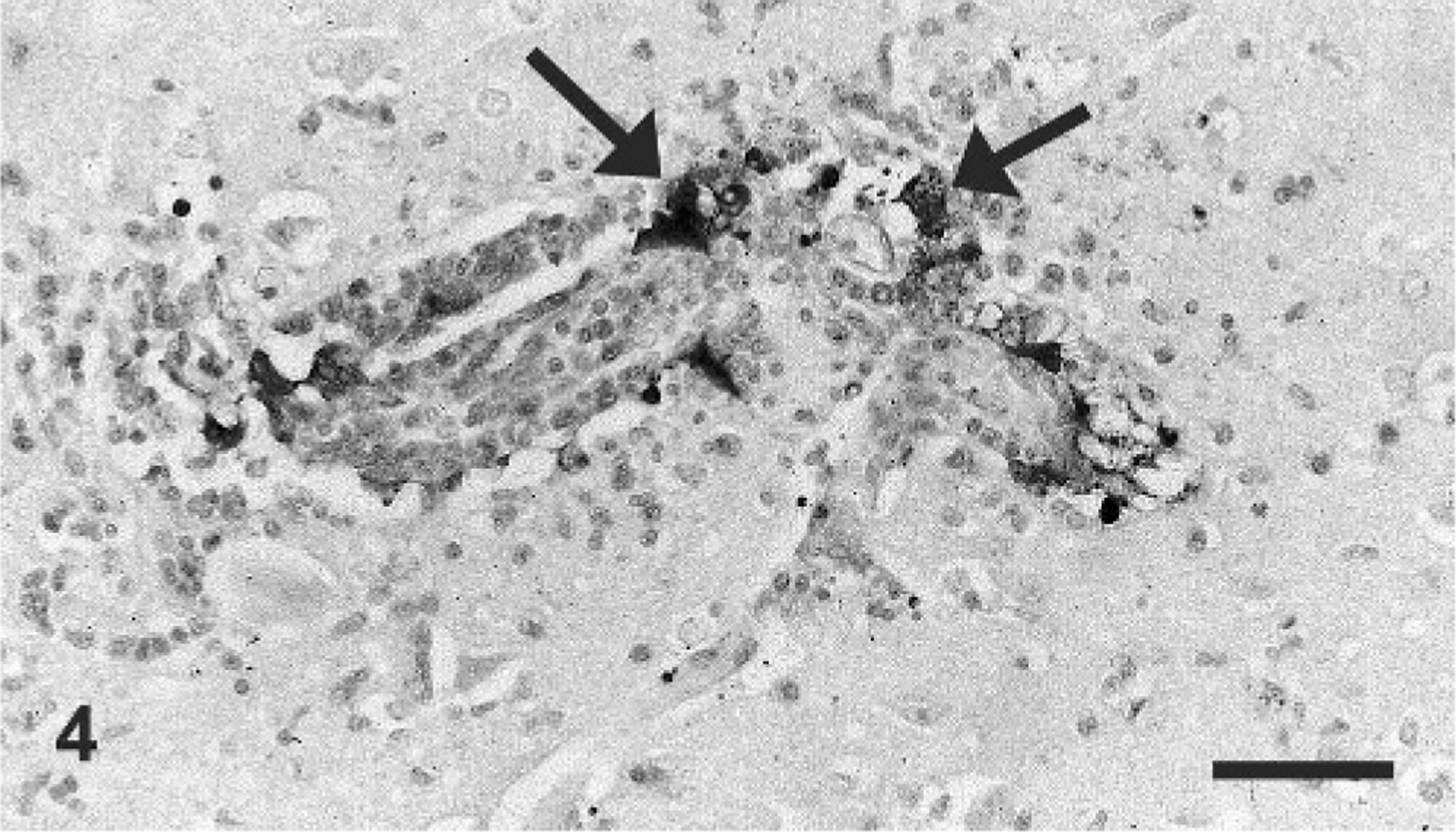
Brain, cerebrum; giraffe. Positive immunoreactivity to anti–EHV-1 antibody is present within endothelial cells (arrows). Anti–EHV-1 antibody, strepavidin–biotin method, hematoxylin counterstain. Bar = 100 μm.
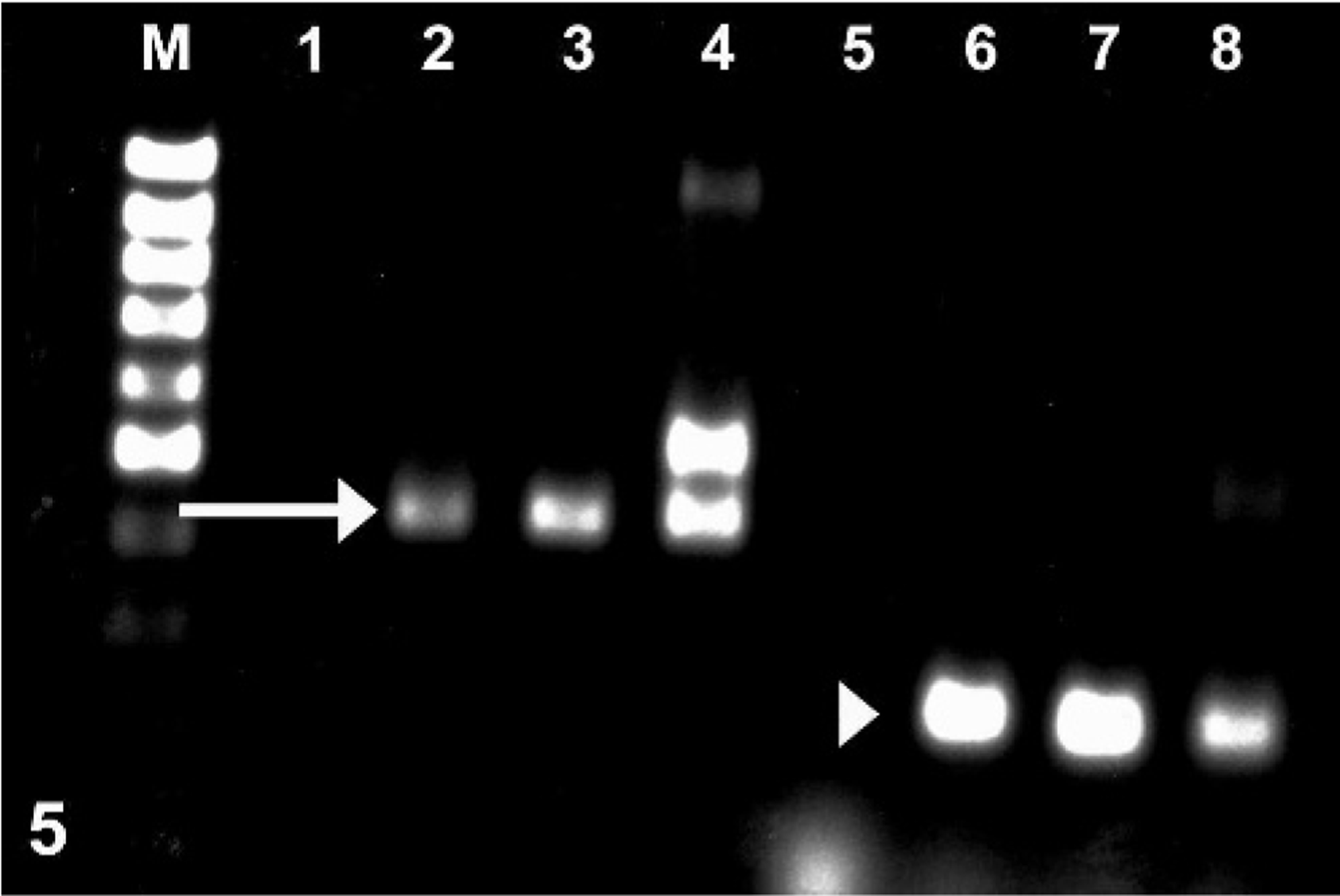
Gel electrophoresis of DNA obtained from giraffe brain: 500-bp fragment corresponding to the EHV1 terminase gene (arrow) and 150-bp fragment corresponding to the EHV1 DNA polymerase gene (arrowhead). M = 100-bp DNA ladder, 1–4 = EHV-1 terminase; 1 = negative control, 2 = positive control, 3 = giraffe brain tissue, 4 = culture material from giraffe brain, 5–8 = EHV-1 DNA polymerase; 5 = negative control, 6 = positive control, 7 = giraffe brain tissue, 8 = culture material from giraffe brain.
Serologic testing was performed on blood samples from the male zebra, which had attacked the giraffe, and the 2 female zebras, which were also housed in the same enclosure. Samples revealed serum titers in all zebras against a control EHV-1 as well as against the herpesvirus isolated from the brain of the giraffe. The 3 zebras had positive titers that ranged from 8 to 64 against EHV-1 and against the virus isolated from brain homogenate of the affected giraffe. Titers of the control serum of EHV-1–infected horses were 128 and 64, respectively. Brain homogenate (approximately 10%) was originally inoculated onto equine dermal cells (monolayer at 1 day on 4 coverslips in Leighton tubes, inoculum 0.25 ml per tube). At day 1 of incubation cytotoxicity was observed. At day 2 the inoculum was diluted approximately 1°2 and reinoculated onto equine dermal cells for 1 day, using the same L-tube system. Less cytotoxicity was observed, and the homogenate was incubated on monolayers for 7 days. Tissue culture fluid (TCF) was harvested and passed onto fresh ED4 cells, bovine kidney, and embryonic bovine lung cells using the L-tube system. At 8 days, the TCF was harvested and passed onto new monolayers. Cytopathic effect was evident on day 2 in all 3 cell lines. A fluorescent antibody test done on cells for EHV-1 and pseudorabies virus was negative. The TCF was examined by electron microscopy and structures observed were consistent with herpesvirus-like particles.
To the authors' knowledge, this is the first report of an EHV-1 infection in a giraffe species. The histopathologic changes observed in the brain of nonsuppurative encephalitis with prominent perivascular cuffing and neuronal necrosis with intranuclear inclusions are consistent with lesions of herpesviral encephalitis in a wide variety of species. Immunohistochemistry using an antibody against EHV-1 confirmed infection of neurons, endothelium, and astrocytes in this case. An infection with EHV-1 was also identified by PCR on brain tissue using specific primers for herpesviral DNA polymerase and terminase regions and subsequential sequencing of PCR products confirming 100% and 96% sequence homology to the DNA polymerase and terminase regions of EHV-1, respectively. Lastly, serologic examination confirmed exposure of the giraffe and the 3 zebras in the enclosure to EHV-1; suggesting a subclinical infection in the zebras, a likely source for infection of the giraffe.
The diagnostic and pathologic workup in this case confirmed infection with a herpesvirus with a high degree of sequence similarity to EHV-1 as the cause of meningoencephalitis in this giraffe. Although the disease presented clinically similar to that in horses, the pathology of neurologic diseases caused by EHV-1 in horses and in this giraffe differs significantly. The lesion distribution exclusive to the brain with sparing of the spinal cord in this case is unusual for the neurologic form of EHV-1 in horses. The typical disease in horses is a myeloencephalopathy, with lesions consistently affecting the spinal cord, with or without lesions in the brain. Microscopically, the lesion in horses is reported to be predominantly endotheliotropic, with resulting necrotizing vasculitis and secondary parenchymal infarction. 2, 8, 11 Neither of these lesions was a feature in this case. Although there was evidence of endothelial cells expressing EHV-1 immunoreactivity, there was no evidence of primary vasculitis or vasculopathy in sections of brain or spinal cord from this giraffe. In addition, prominent intranuclear viral inclusions are not consistently noted in neurons of horses with the neurologic form of EHV-1. 3 Certain strains of EHV-1 are thought to be particularly neurovirulent, which may explain the difference in lesion distribution and susceptibility of this animal to the virus. 3 The case presented here exhibited true neurotropism, consistent with other alphaherpesviral infections caused by pseudorabies virus in swine, infectious bovine rhinotracheitis in cattle, herpes simplex virus in a number of species including rabbits, and varicella–zoster virus in humans. 10 As an alternative explanation, cross-species infection may have resulted in altered cellular tropism similar to lesions seen in rabbits with herpes simplex virus infection.
Encephalitis caused by a variant of EHV-1 reported in a Thompson's gazelle had a similar distribution of lesions, with sparing of the spinal cord as in this case. 3 In addition, the histopathologic changes were very similar in that the characteristic necrotizing vasculitis was not present, and inflammatory changes consisted of perivascular accumulations of lymphocytes; however, intranuclear neuronal inclusions were not observed. 3 The report in the gazelle was the first incidence of EHV-1 in a ruminant species 3 ; given that giraffes are ruminants, this distribution may speculatively represent a particular species difference from the disease in equids.
Transmission of EHV-1 occurs by horizontal spread through direct or indirect contact with infective aerosols, fetal membranes or tissues, or uterine fluids, or from fomites harboring the virus. 6, 11 The virus initially replicates within the nasal epithelium, leading to infection of predominantly T lymphocytes, and a cell-associated viremia with spread to distant organs such as the lungs, reproductive tract, and central nervous system. 6 It is known that alphaherpesviruses have the ability to evade the immune system and become latent within lymphoid tissues, peripheral leukocytes, and trigeminal ganglia following infection, with potential for reactivation and shedding following immune suppression or stress. 6, 11 The 3 zebras housed with this giraffe were seropositive for EHV-1. Although the vaccination status of these zebras was not known, reactivation and subsequent shedding of infective virus from any one of the zebras in the same enclosure could have resulted in systemic infection and disease in this animal. This cross-species transmission of EHV-1 raises important concerns for latent infection and transmission of the disease from zebras to other zoologic species, with potentially devastating results. Zebras are an important carrier of EHV-1 in zoologic collections and should be regularly serologically tested for evidence of EHV-1. Cross-species transmission to previously unreported susceptible animal species must be considered in both the screening of susceptible animals for subclinical infection, as well as husbandry and housing issues for various irreplaceable species.
Footnotes
Acknowledgements
The authors would like to thank Mary Woodruff, Animal Disease Diagnostic Laboratory, Purdue University, for her technical expertise.
