Abstract
Neurologic disease occurs sporadically in horses infected with the equine infectious anemia virus (EIAV). This report describes a case of clinically severe neurologic disease in a pony experimentally infected with EIAV. This pony did not have fever or anemia, which are the characteristic clinical signs of disease. The histopathologic changes were characterized as lymphohistiocytic periventricular leukoencephalitis. Polymerase chain reaction and in situ hybridization data showed that the brain lesions were directly associated with viral replication and that high-level viral replication occurred selectively within the lesion and not in other tissues. These findings suggest that EIAV-associated neurologic disease is the direct result of viral replication.
Keywords
Infection of horses with the equine infectious anemia virus (EIAV), a lentivirus in the family Retroviridae, typically causes clinical disease characterized by fever, anemia, and thrombocytopenia. 12 After initial disease and recovery, most horses experience recurrent episodes of disease that decrease in frequency and intensity over time, eventually resulting in lifelong subclinical carriers. 12 Neurologic disease is a rarely reported manifestation of EIAV infection but has been occasionally noted as causing clinical signs compatible with multifocal to diffuse encephalomyelitis. 7,9 Histologically, the lesions were described as granulomatous and lymphocytic ependymitis, meningitis, choroiditis, and subependymal encephalitis. 7,9,13
The primary host cell for EIAV replication is the tissue macrophage, and thus viral replication occurs in most tissues, 10 including brain. 5,8 Although the replicating virus can be detected at all times in infected horses, overt clinical disease is associated with high levels of viral replication. 5,10 In contrast, horses with subclinical infections have greatly reduced infectivity, viremias, and viral loads. 5,10 Interestingly, many of the previously described cases of EIAV-associated neurologic disease occurred in horses without the typical spectrum of clinical signs associated with acute EIAV infection. Conversely, the vast majority of horses with acute clinical EIAV infections do not have neurologic disease. The pathogenesis of EIAV-associated neurologic disease is unknown, and in the earlier reports the relationship between lesions and viral replication was not investigated. This report describes a case of fatal neurologic disease and periventricular leukoencephalitis in a horse that was chronically infected with EIAV and that did not have all the characteristic clinical signs of acute EIAV infection. It also demonstrates that the brain lesions were directly associated with high levels of EIAV replication and that high-level viral replication is occurring selectively within the lesion and not in other tissues.
Animal care and experiments were approved by the Washington State University Institutional Animal Care and Use Committee. An adult Shetland pony (pony No. 529) was infected intravenously with 1 × 108 50% tissue culture infectious doses (TCID50) of the WSU5 strain of EIAV (EI-AVWSU5). EIAVWSU5 is a weakly virulent tissue culture–adapted strain, 10 which rarely causes mortality and has not been previously reported to cause neurologic disease. An Arabian foal (foal No. 2079) was also infected intravenously with 1 × 106 TCID50 of EIAVWSU5. Another Arabian foal (foal No. 2084) was infected intravenously with 1 × 106 horse infectious doses of the highly virulent 10 Wyoming strain of EIAV (EIAVWYO). Monitoring for EIAV-associated disease included daily observation, daily recording of body temperatures, and periodic measurement of erythrocyte and platelet numbers.
Pony No. 529 was infected in September 1994. As is common with EIAVWSU5 infection, initial disease was very mild and consisted only of a low-grade fever (38.4–39 C) between days postinfection (dpi) 4–7. Between dpi 42 and 876, rectal temperature and platelet counts were not monitored routinely, although no overt clinical disease was observed. At dpi 877, temperature and platelet monitoring were resumed daily and weekly, respectively. No significant abnormalities were noted other than mild thrombocytopenia on dpi 1,116 and 1,130. At dpi 1,186, persistent mild thrombocytopenia was noted and was coincident with the onset of a 4-week course of progressive neurologic disease characterized initially by mild behavioral changes (difficult to catch and halter) followed by hyperesthesia and ataxia. Blindness was noted 3 days before death, and seizures occurred on the day of death. Fever and anemia were not present at any time during the course of neurologic disease. The pony was euthanatized and submitted to the Washington Animal Disease Diagnostic Laboratory for necropsy examination. Foal Nos. 2079 and 2084 were euthanatized at dpi 29 and 15, respectively, with acute clinical disease. Although the clinical signs in foal No. 2079 were mild and the clinical signs in foal No. 2084 were severe, both had all the classic disease signs of acute EIAV infection, including fever, anemia, thrombocytopenia, and viremia. 10 Neither foal No. 2079 nor foal No. 2084 had any evidence of neurologic disease.
After euthanasia of infected horses, complete necropsies were performed and samples of all major organ systems were collected. Tissues were fixed in 10% neutral buffered formalin, processed routinely for histopathology, and 3-µm sections were stained with hematoxylin and eosin (HE). Sections from all major organ systems were examined microscopically. In pony No. 529, sections of brain examined included cerebral cortex (frontal, temporal, and occipital lobes), midbrain (thalamus and rostral colliculi), brainstem (middle cerebellar peduncle and obex), and cerebellum.
Total cellular DNA and RNA were extracted from the brain and spleen of pony No. 529 using commercial DNA (Stratagene DNA extraction kit, La Jolla, CA) and RNA (Trizol Reagent, Life Technologies, Carlsbad, CA) extraction systems according to the manufacturer's instructions. Polymerase chain reaction for EIAV DNA was performed on 1–2 µg of total DNA with 35 cycles of 30 seconds at 94 C, 30 seconds at 58 C, and 30 seconds at 72 C using the EIAV capsid protein gene (gag)–specific primers (primer 897, 5′-ACTACTTGGGTGAATACCAT-3′; primer 1199, 5′-TCTGCCTAAACTGATCAAAA-3′). Reverse transcription (RT)–PCR for viral RNA was performed as stated above with the addition of a RT step using random hexamer primers and Moloney murine leukemia virus reverse transcriptase (SuperScript II, Life Technologies) to produce complementary DNA. Both the PCR and RT-PCR reactions produce EIAV amplicons of 322 base pairs. PCR and RT-PCR for the cellular glyceraldehyde-3-phosphate dehydrogenase gene was performed as described 5 to verify equal loading of template DNA and RNA and the lack of PCR inhibitors in the samples. In the RT-PCR assays, the absence of DNA was verified by PCR on extracted RNA without RT.
Sections of brain, spleen, lung, and liver from pony No. 529 and foal Nos. 2079 and 2084 were probed for EIAV RNA by in situ hybridization (ISH) as previously described. 10 Selected brain sections from pony No. 529, positive for viral RNA by ISH, were subsequently stained by immunohistochemistry as previously described 11 for glial fibrillary acid protein (GFAP) as a marker for astrocytes, 4 using as primary antibody rabbit anti-bovine GFAP (Dako Z0334, Carpinteria, CA; diluted 1 : 3,000). To rule out infection with equine herpesvirus type 1 (EHV1) in pony No. 529, brain sections were probed for the EHV1 antigen by immunohistochemistry as previously described, 2 using as primary antibody an anti-EHV1–specific monoclonal antibody (cell line F2, VMRD Inc., Pullman, WA). The positive control was spinal cord tissue from a horse with EHV1 infection confirmed by virus isolation. The negative control was an isotype-matched irrelevant monoclonal antibody used at a similar concentration on tissues from pony No. 529.
Grossly, pony No. 529 had widespread petechiae, which were most evident in the intestinal serosa, renal cortex, and subcutis. In the central nervous system (CNS), both cerebral cortices had random, nonsymmetrical, soft, yellow-brown areas of malacia in the white matter, sometimes progressing to malacia or cavitation (Fig. 1). Similar soft, yellow-brown foci were present in the periventricular white matter of the midbrain and brain stem. Histologically there was lymphohistiocytic leukoencephalitis with multinucleated giant cells. The lesions were random and primarily affected the white matter around the third and fourth ventricles and within the cerebral cortices (Fig. 2). Inflammatory aggregates consisted of perivascular to coalescing infiltrates of macrophages, lymphocytes, and occasional plasma cells and multinucleated giant cells (Fig. 3). The affected blood vessels were lined by plump, hypertrophied endothelial cells and sometimes had minimal inflammatory cell infiltration into the tunica adventitia and perivascular edema. The edema was severe in some areas and resulted in marked loosening of the neuropil, resulting in the formation of cavitations. In other areas, the neuropil was densely cellular with gliosis and loose to dense aggregations of macrophages and Gitter cells. The primary reactive glial cells were astrocytes that stained positively for GFAP (Fig. 4). The meninges had mild, multifocal infiltrates of lymphocytes and plasma cells. The lesions noted outside the CNS of pony No. 529 were compatible with EIAV infection, including random aggregates of lymphocytes and hemosiderin-filled macrophages in the portal regions of the liver, interstitium of the kidney, and lung. 8 Focal areas of acute hemorrhage were noted in the lung and lymph node but not in the brain. Foal Nos. 2079 and 2084 did not have gross or microscopic CNS lesions.
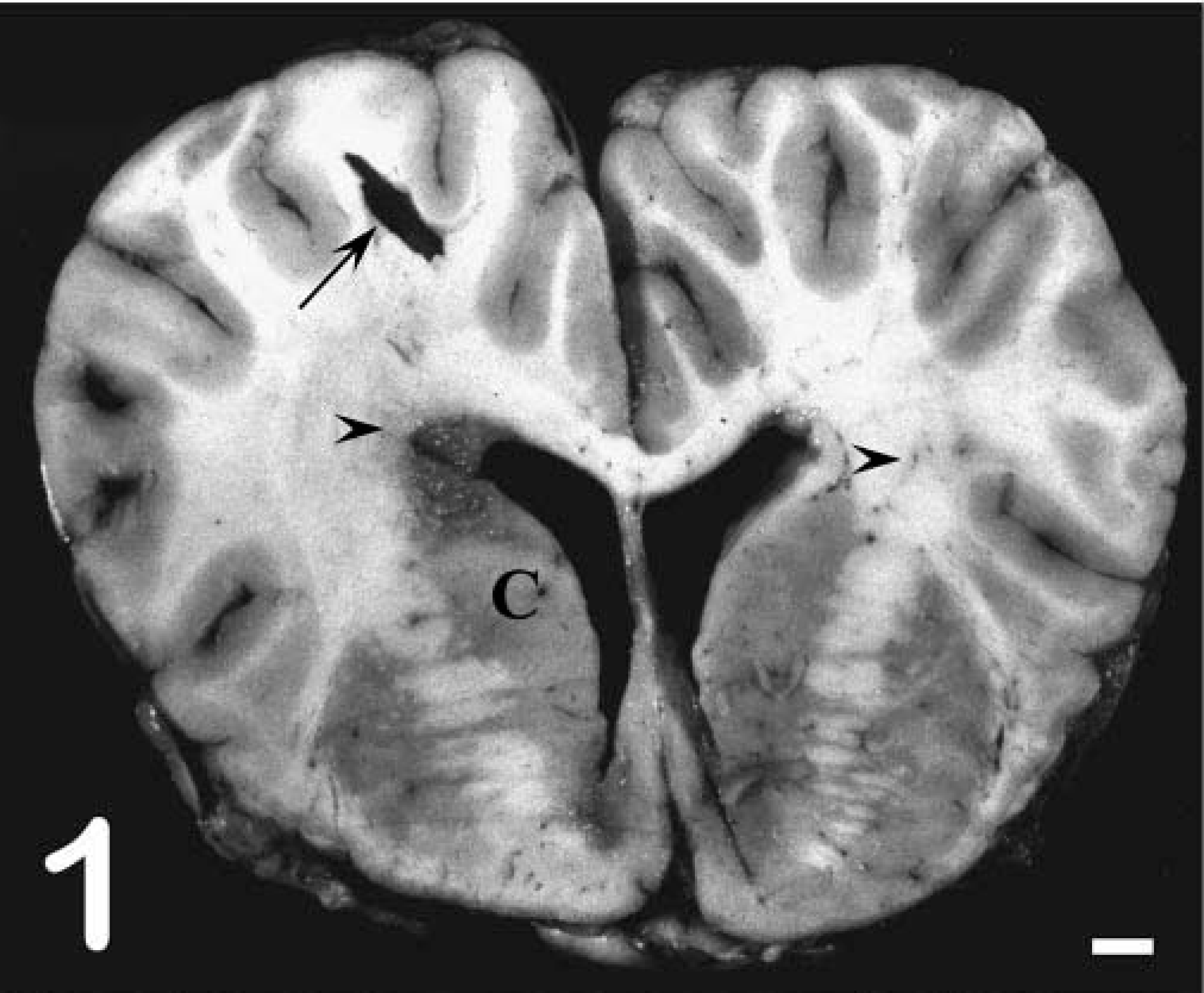
Cerebral cortex, coronal section of the frontal lobe; pony No. 529. Multifocal malacia in the white matter around the lateral ventricle and in the corona radiata (arrowheads) with focal cavitation (arrow). C = Caudate nucleus. Bar = 1 cm.
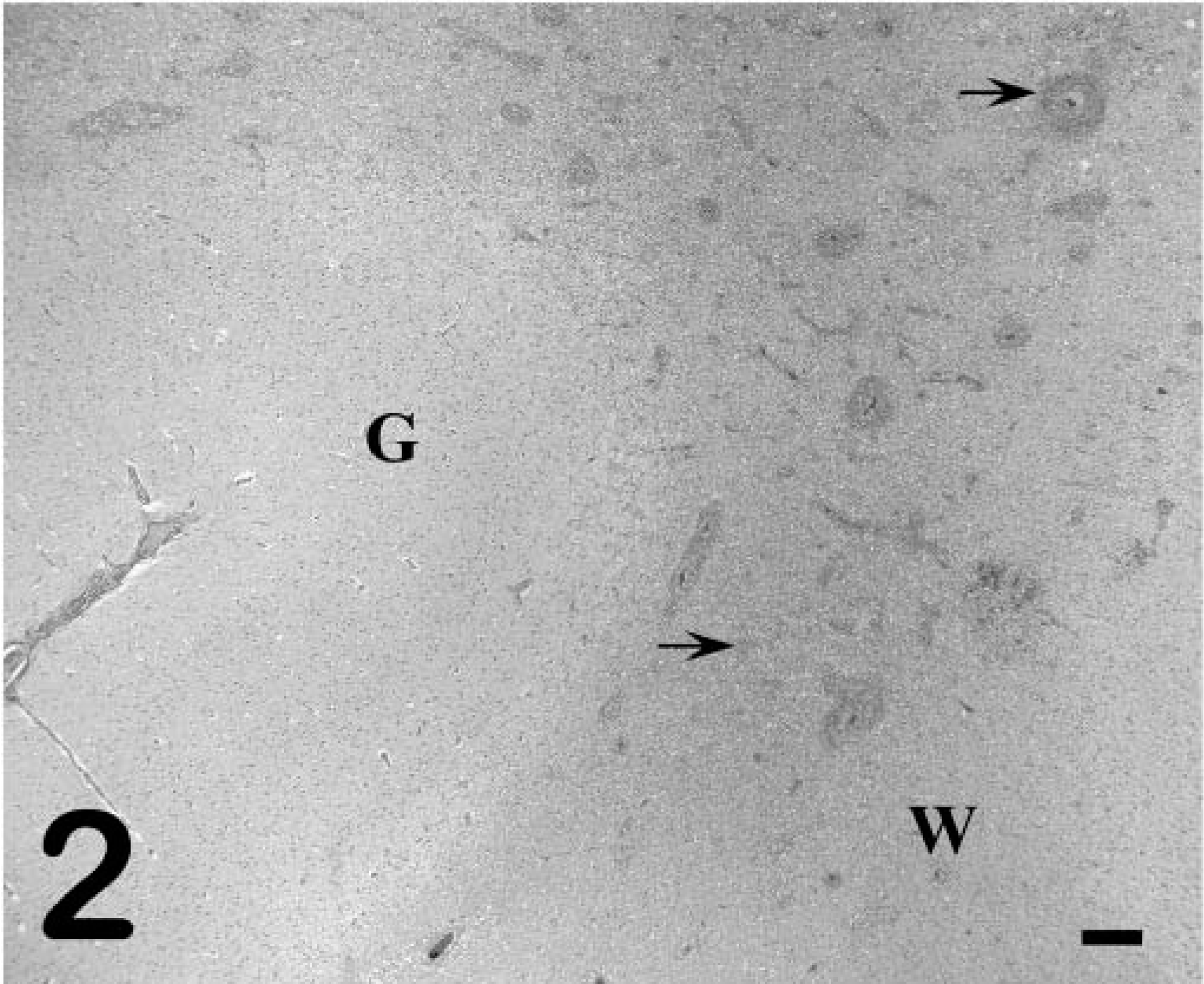
Cerebral cortex; pony No. 529. Multifocal leukocytic infiltrates around blood vessels and in the neuropil of the white matter (arrows) and rarefaction of the gray matter–white matter interface. G = gray matter, W = white matter. HE. Bar = 250 µm.
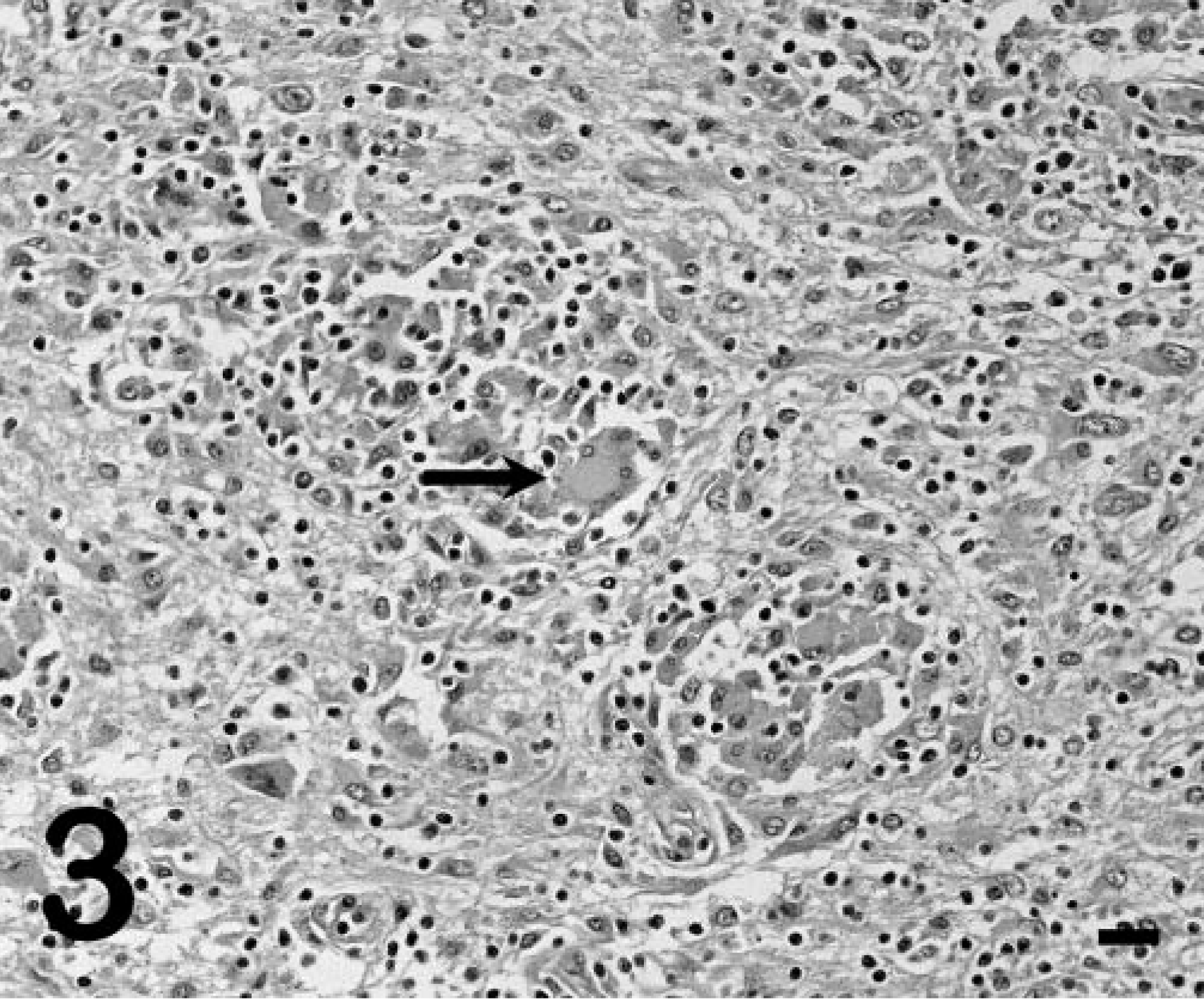
Cerebral cortex; pony No. 529. White-matter gliosis with aggregates of lymphocytes, macrophages, and multinucleated giant cells (arrow). HE. Bar = 30 µm.
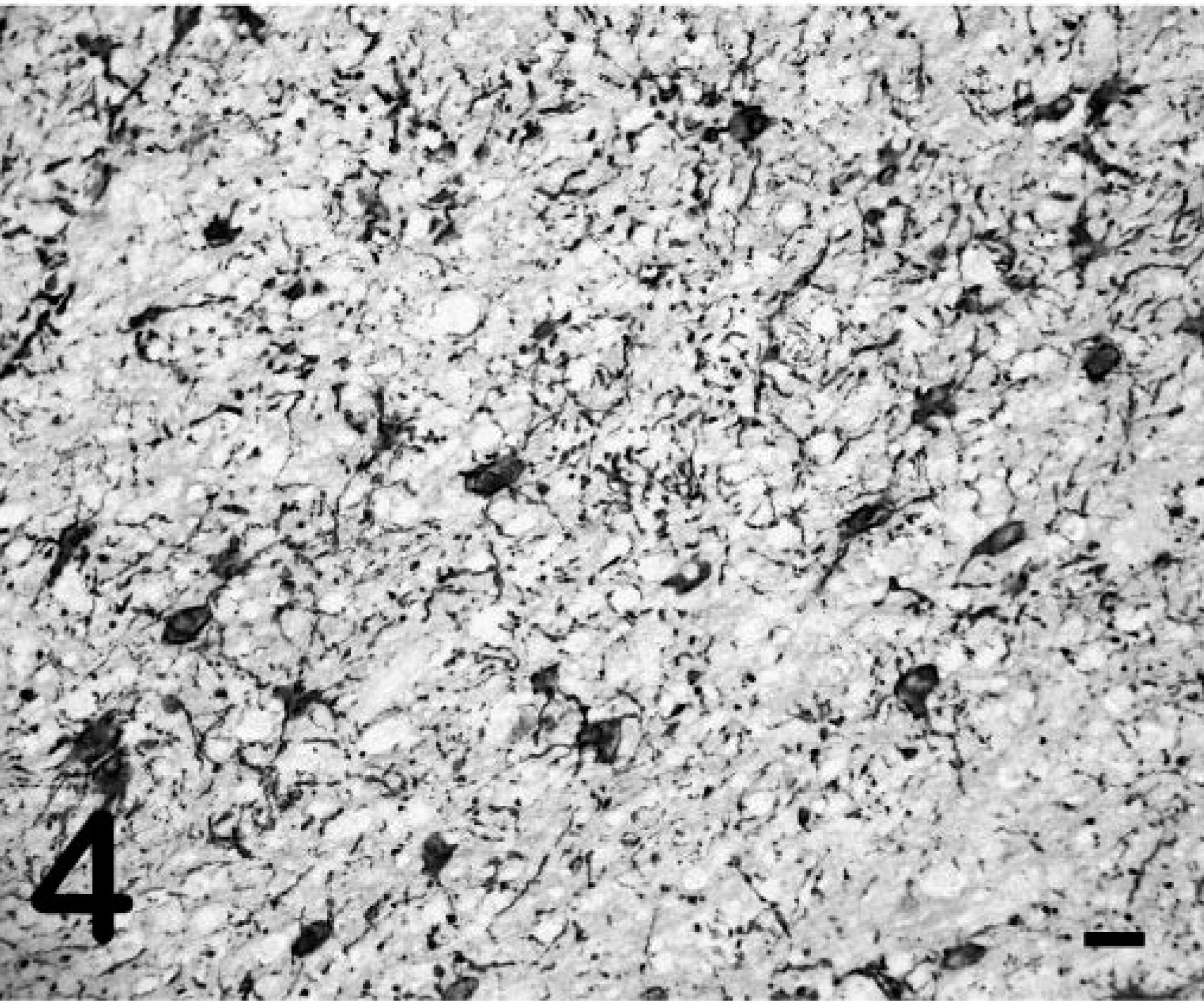
Cerebral cortex; pony No. 529. White-matter astrogliosis demonstrated by GFAP immunoreactivity showing abundant branching processes of hypertrophied astrocytes. Streptavidin–biotin complex immunoperoxidase with AEC chromogen and Mayer's hematoxylin counterstain. Bar = 60 µm.
The lesions in the CNS of pony No. 529 were atypical for nonneurologic acute or chronic EIAV infections, which do not typically have inflammatory changes, but were similar to the lesions previously described for EIAV-associated CNS disease. 7,9,13 The vasocentric pattern of inflammation, sometimes involving the vessel adventitia, and marked perivascular edema can be seen with equine herpesvirus encephalitis. 6 However, EHV1 antigen was not detected by immunohistochemistry. The cavitating lesion in the white matter of the cerebral cortices, sometimes with minimal inflammation, was reminiscent of leukoencephalomalacia caused by fumonisin toxicosis (moldy corn poisoning). 6 However, analysis of the feed by the National Veterinary Services Laboratory (Ames, IA) for fumonisins B1, B2, and B3 was negative. Sphingosine and sphinganine levels in serum and brain tissue of pony No. 529 were also within normal limits, with the findings not being consistent with fumonisin toxicosis. 15
PCR detected EIAV proviral DNA sequences for the capsid protein gene in both the spleen and brain of pony No. 529, indicating the presence of infected cells in these tissues as previously described in other studies involving subclinically infected horses. 5,10 Similarly, RT-PCR detected unspliced messenger RNA (mRNA) for the EIAV capsid protein gene, indicative of viral replication, in both the spleen and brain of pony No. 529. The presence of ongoing viral replication in subclinically infected horses has been previously described. 5,10 However, this case was unusual in that viral RNA could be readily detected in CNS tissues and at levels comparable with those in the spleen (Fig. 5). Because the levels of viral DNA and RNA in the spleen are typically much greater than those in the brain, 5,10 these findings suggested that there was extensive viral replication occurring in the brain of pony No. 529.
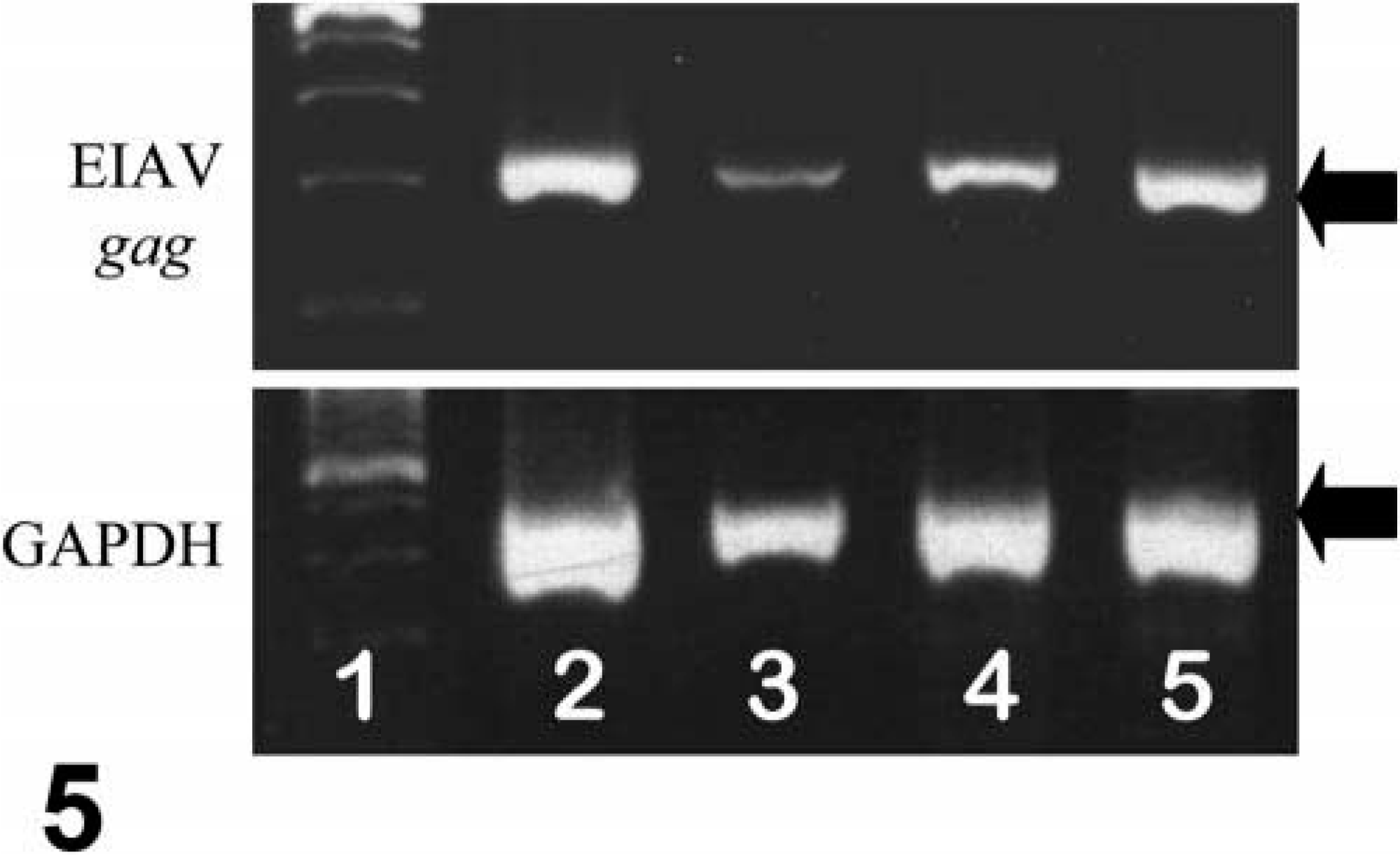
PCR/RT-PCR for EIAV DNA/RNA; pony No. 529. Lane 1, size markers. Lane 2, EIAV RNA in brain. Lane 3, EIAV RNA in spleen. Lane 4, EIAV DNA in brain. Lane 5, EIAV DNA in spleen. For all samples, equivalent loading of DNA or RNA is confirmed by PCR/RT-PCR for GAPDH DNA/RNA. Ethidium bromide–stained agarose gel. Arrow for EIAV PCR indicates 322 base pairs; arrow for GAPDH PCR indicates 452 base pairs.
ISHs for EIAV RNA performed on the liver, spleen, and lung of pony No. 529 were negative (Fig. 6), consistent with previous reports that fail to detect viral nucleic acids or proteins by in situ methods in horses with subclinical infections. 8,10 In contrast, tissues from the foals with acute clinical disease (foal Nos. 2079, 2084) had large numbers of cells with detectable viral RNA (Fig. 7). However, in pony No. 529, cells with detectable viral RNA were readily observed within the CNS white-matter lesions (Fig. 8), whereas sections from horses with acute clinical disease but no CNS lesions (foal Nos. 2079 and 2084) did not have positive cells in the CNS white-matter parenchyma (data not shown). ISHs on tissues from the infected horses with the sense polarity probe were negative, indicating that the hybridization was specific for EIAV RNA. ISHs on liver, spleen, and brain from an uninfected horse with the antisense probe were all negative. Dual-labeling experiments that combined ISH and immunohistochemistry for GFAP showed that the infected cells did not express GFAP (Fig. 9) and thus were not astrocytes.
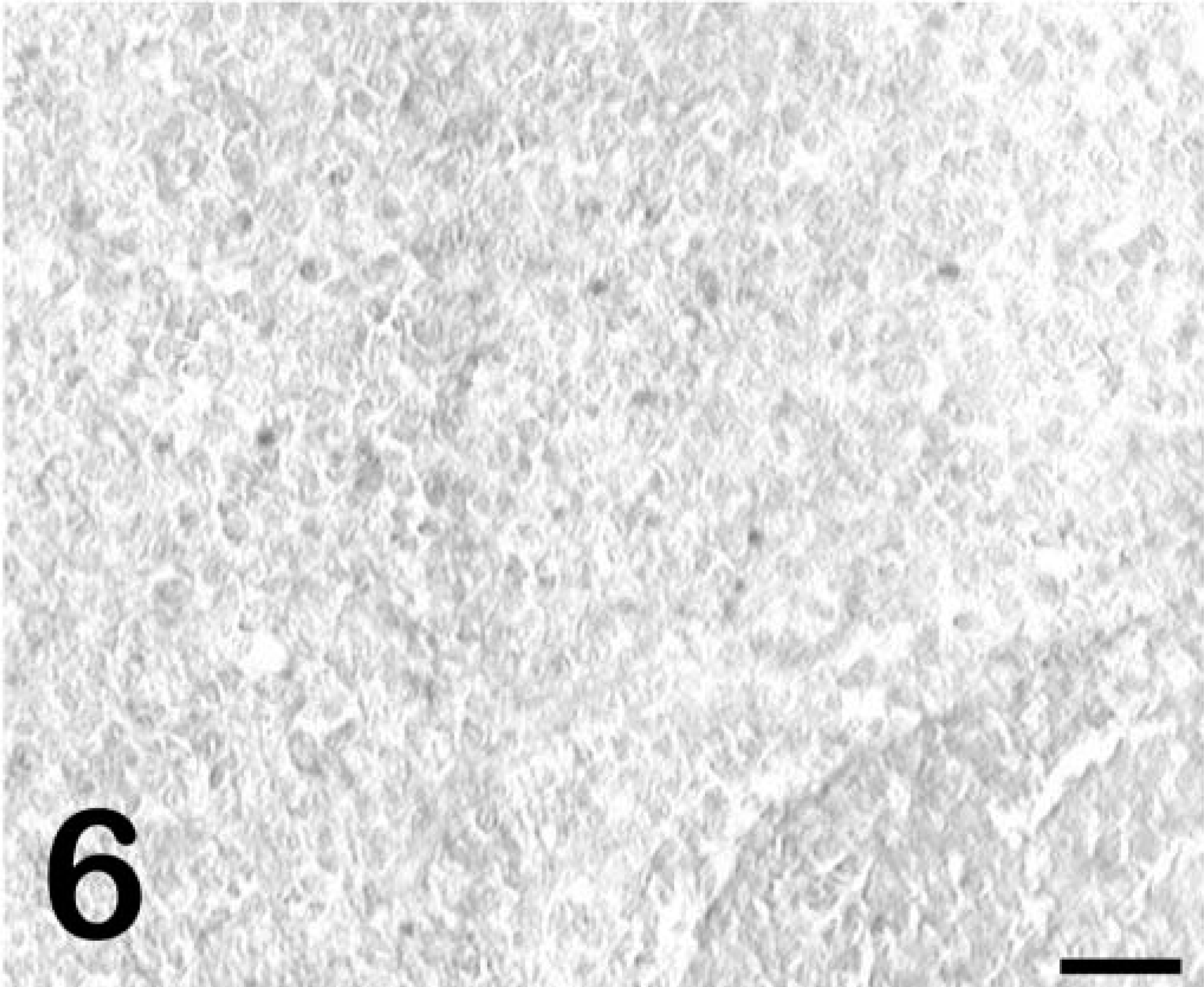
Spleen; pony No. 529. ISH for EIAV RNA demonstrating lack of detectable reactivity. Detection of digoxigenin-labeled probe by immunostaining with alkaline phosphatase and BCIP/NBT chromogen. Methyl green counterstain. Bar = 50 µm.
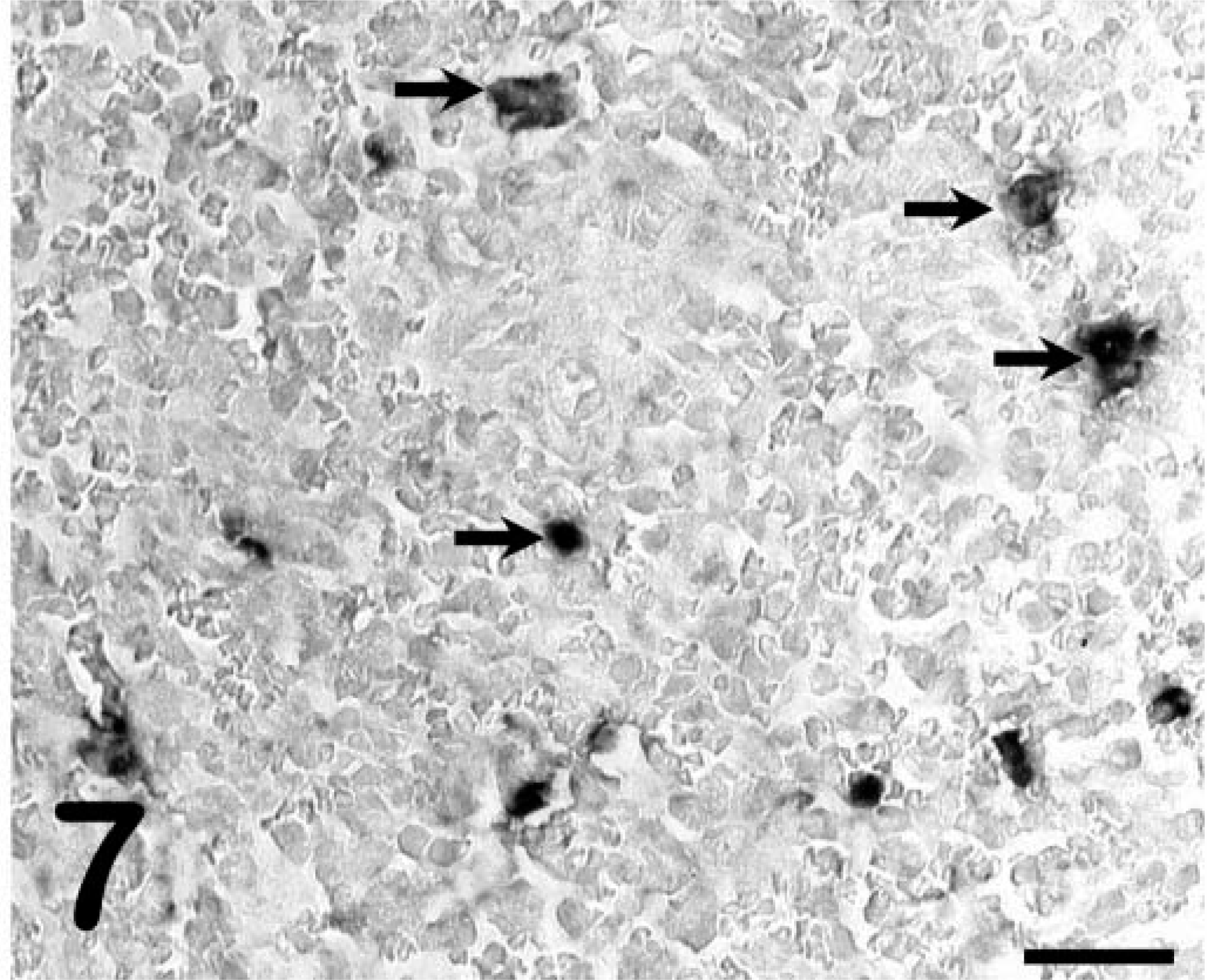
Spleen; foal No. 2079. ISH for EIAV demonstrating infected cells (arrows). Detection of digoxigenin-labeled probe by immunostaining with alkaline phosphatase and BCIP/NBT chromogen. Methyl green counterstain. Bar = 25 µm.
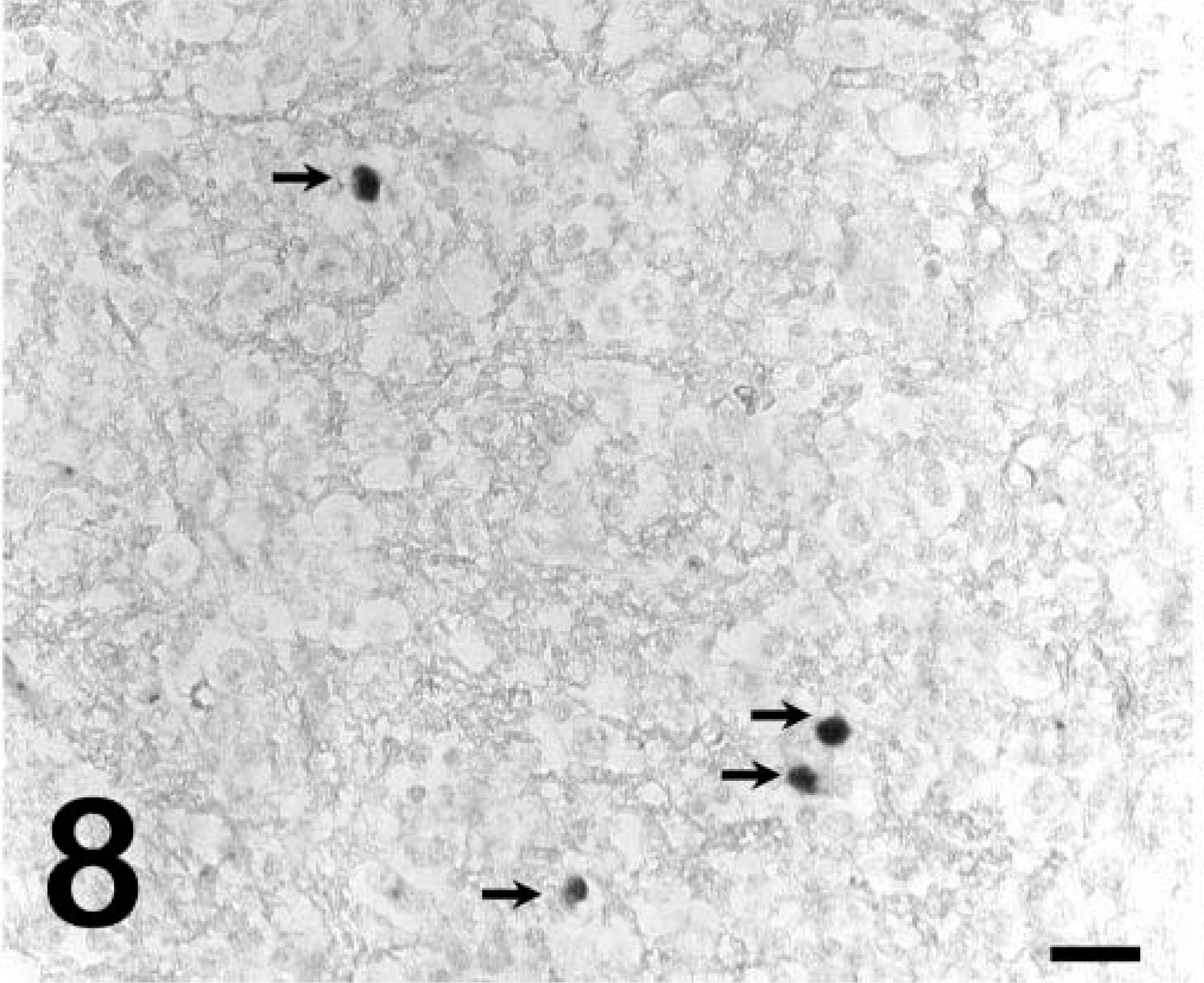
Brain; pony No. 529. ISH for EIAV RNA demonstrating infected cells (arrows) in the white-matter lesion. Detection of digoxigenin-labeled probe by immunostaining with alkaline phosphatase and BCIP/NBT chromogen. Methyl green counterstain. Bar = 25 µm.
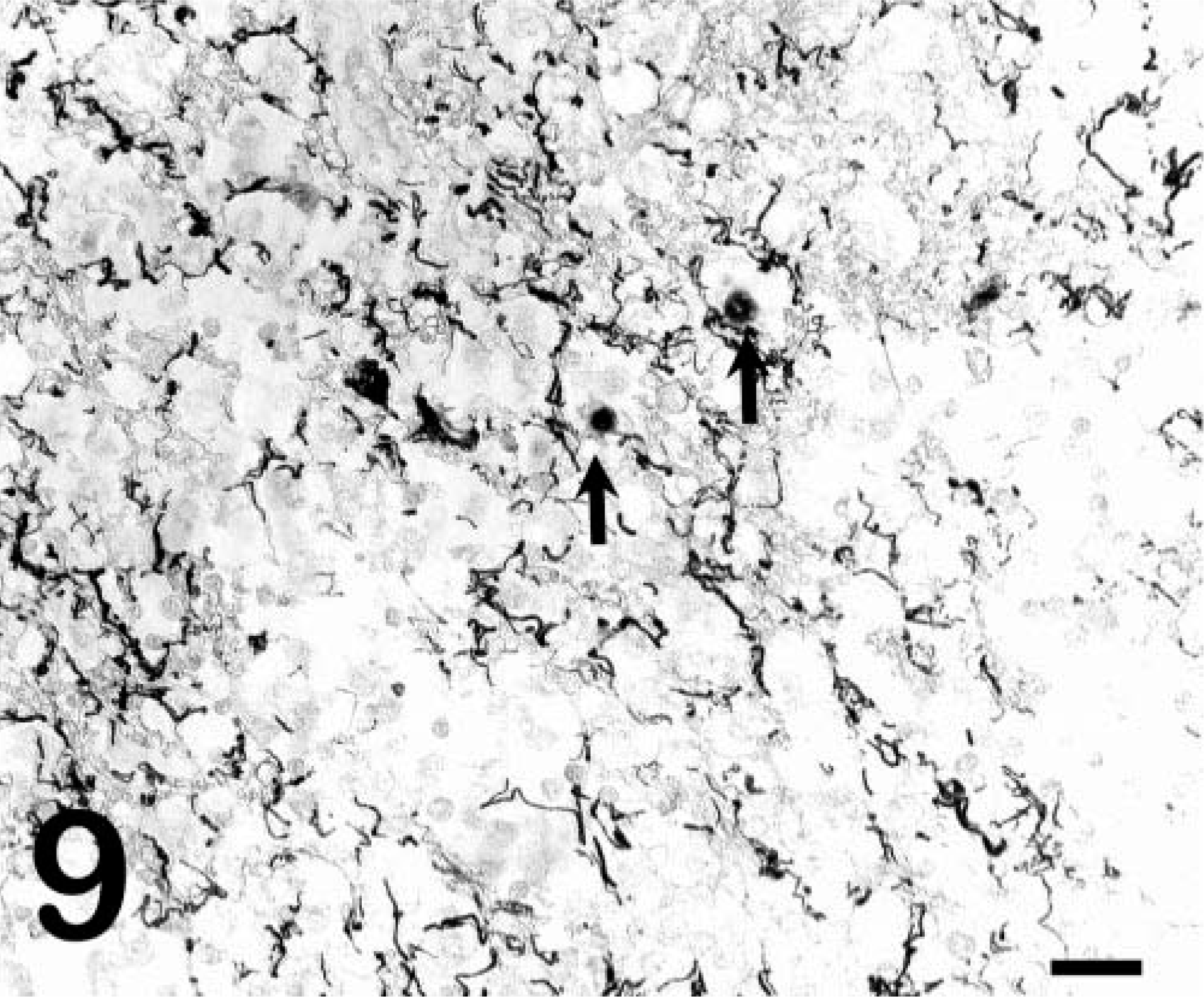
Brain, pony no. 529. ISH for EIAV RNA demonstrating that infected cells (arrows) do not colabel with GFAP and that infected cells are not astrocytes. Detection of digoxigenin-labeled EIAV probe by immunostaining with alkaline phosphatase and BCIP/NBT chromogen. Detection of GFAP by streptavidin–biotin complex immunoperoxidase with AEC chromogen. No counterstain used. Bar = 60 µm.
The clinical and pathologic abnormalities in pony No. 529 were very similar to those previously reported in horses with EIAV-associated neurologic disease. 7,9,13 The histologic lesions of necrosis (malacia) and nonsuppurative white matter and periventricular inflammation with multinucleated giant cells are also comparable with the lesions in other domestic animals infected with nonlymphotropic lentiviruses, including caprine arthritis encephalitis virus and ovine visna virus. 3 Although the lesions of EIAV-associated neurologic disease appear to be characteristic of lentivirus-related neuropathology, several features are unusual manifestations of EIAV infections in general. One is that neurologic disease is an inconsistent feature of either natural or experimental EIAV infections. Thus, neurotropism does not appear to be strain dependent as has been reported for some strains of simian immunodeficiency virus. 1 Another is that in this case, and in many of the previously described cases, neurologic disease occurs independently of some of the hallmark clinical signs of acute EIAV infection such as fever and anemia. And finally, typical EIAV lesions do not include overt tissue necrosis. The most characteristic lesions in EIAV infection appear to be primarily due to immune- and cytokine-mediated mechanisms, 12,14 and are described as mononuclear cell inflammatory infiltrates in the periportal regions of the liver, the interstitial areas of the kidney, spleen, lung, and other tissues. 6,8 Pony No. 529 had gross and microscopic evidence of hemorrhage, most likely related to the persistent thrombocytopenia. Although CNS disease may occur as a result of hemorrhage, this was unlikely in this case because the hemorrhages noted in the tissues of pony No. 529 were acute, whereas the neurologic disease and pathology were of weeks duration. Moreover, hemorrhage was not observed in the brain lesions of pony No. 529.
The mechanism of neurologic disease in EIAV infections is unknown, but this case provided an opportunity to examine the relationship between the CNS lesions and viral replication. In pony No. 529, the CNS lesions were directly associated with virally infected cells, and the high levels of mRNA for the viral structural proteins as detected by ISH indicated that these cells were also actively replicating virus. In contrast, ISH did not detect infected cells in unaffected areas of the brain or in other tissues, including tissues such as lung and spleen, which typically have large numbers of ISH-positive cells during acute disease. 10 In the tissues of subclinically infected horses, although the production of viral mRNA can be demonstrated by highly sensitive techniques such as RT-PCR, 5,10 viral antigens and RNA cannot typically be detected by the less sensitive in situ methods, 8,10 suggesting markedly restricted or latent infections at this time. This restricted EIAV replication is a feature in all tissues, and there is no evidence of tissue-specific replication during either acute or subclinical infections. 5,10 Thus, localization of ISH-positive cells to the brain lesions of pony No. 529 strongly suggested that viral replication was directly responsible for the neurologic disease. The neuroanatomic location of the infected cells in the white matter and the exclusion of astrocyte infection by the dual-labeling experiment make macrophages or microglial cells the probable infected cell type. The ability to selectively replicate in these cells in some EIAV cases may depend on host factors that either allow virus to cross the blood-brain barrier or an unusual biochemical or cytokine environment that allows these cells to become permissive.
Footnotes
Acknowledgements
This work was supported by NIH grant R01 AI44638 (to J. L. Oaks) and American Veterinary Medical Foundation grant 50-98 (to M. T. Long). We thank Sue Pritchard and Shirley Elias for excellent technical assistance.
