Abstract
In murine models the inducible nitric oxide synthase (iNOS) and the natural resistance associated macrophage protein (NRAMP1) play major roles in host defense against mycobacteria. iNOS regulates nitric oxide (NO) production, which is noxious for ingested mycobacteria, and NRAMP1 displays pleiotropic antimicrobial effects, including upregulation of iNOS expression. Little is known about the role of these molecules in bovine tuberculosis (TB). In this work we demonstrate by Western blot a high expression of NRAMP1 in peripheral blood mononuclear cells (PBMCs), alveolar macrophages (obtained by bronchioalveolar lavage), and lymph node granulomas from 8 Holstein-Freisian cattle with autopsy-proven bovine TB. Immunohistochemistry revealed the abundant expression of NRAMP1 and iNOS in lymph node and lung granulomas. Immunoreactivity was abundant in the cytoplasm of many epithelioid macrophages and multinucleated giant cells of the Langhans type. A striking accumulation of nitrotyrosine (NT), an indicator of iNOS activity and local NO production, was observed in granuloma cells, particularly in multinucleated Langhans cells. This study shows that the expression of NRAMP1 and iNOS is costimulated in granulomas, which are protective T-cell reactions against mycobacteria.
Keywords
Mycobacterium bovis (M. bovis) is an intracellular pathogen with a wide range of hosts, including domestic and wild animals. M. bovis is the etiologic agent of cattle tuberculosis (TB), a disease that results in economic loss and represents a human health risk. 13 World Health Organization programs to eradicate bovine TB include tuberculin testing with purified protein derivative (PPD) and slaughtering of reacting animals to verify lesions and to carry out microbiologic cultures to identify mycobacteria. 14 Cattle are usually infected by the respiratory route. 26 The control of mycobacteria initially depends largely upon activated macrophages (MØs), which display an array of innate antimicrobial mechanisms. In vitro studies in cattle have shown that M. bovis activates MØs to produce tumor necrosis factor alpha (TNF-α), interleukin 12, and interleukin 10. 22
Over the last decade the roles of inducible nitric oxide synthase (iNOS) and natural resistance associated macrophage protein (NRAMP1) in mycobacterial infections have been the focus of much attention. 7, 11, 25, 36 iNOS catalyzes nitric oxide (NO) production, converting arginine into citrulline and NO, an MØ-derived radical highly toxic to intracellular mycobacteria whose production is regulated by pro-inflammatory cytokines such as interferon gamma (IFN-γ), TNF-α, and interleukin 1β. 11, 25, 33 It has been demonstrated that iNOS inhibitors aggravate the course of murine TB and that NOS2(-/-) mice are highly susceptible to mycobacterial infection. 11, 25 In human TB, iNOS expression by monocytes and alveolar MØs has been documented 16, 27 ; however, its role in protection has been questioned. 34 The production of iNOS by bovine MØs in response to M. bovis has not been documented, although production of iNOS and NO has been shown after in vitro stimulation with LPS and Salmonella dublin, and in vivo in listeric encephalitis. 1, 30
NRAMP1 has attracted much attention for its role in innate immunity. 5, 36 In the mouse, NRAMP1 is linked with genetic resistance to Mycobacterium bovis/BCG infection, which is abolished by a single non-conservative Gly to Asp substitution. 38 In humans, several NRAMP1 polymorphisms seem to be associated with genetic susceptibility to TB. 5, 10 NRAMP1 is primarily expressed in MØs and tissues of the recticuloendothelial system, but its role in the resistance of or susceptibility to mycobacterial infection remains undetermined. 3, 4, 35 In a recent study we demonstrated high expression of NRAMP1 in granulomas of M. bovis–infected cattle; 15 since granulomas are protective inflammatory reactions, 17, 20, 21 it was thought that NRAMP1 plays a defensive role against mycobacteria locally. This finding, in addition to recent studies showing a regulatory role for NRAMP1 in iNOS production, 2, 18 prompted this study, which aims to analyze the expression of NRAMP1 and iNOS in M. bovis–infected cattle. In addition, we analyzed nitrotyrosine (NT) expression as an indication of iNOS enzymatic activity and NO production. 23, 32
Materials and Methods
Study population
Eight head of milk-producing Holstein-Friesian cattle with autopsy-proven TB were included in this study. Before slaughtering, all animals were shown to be M. bovis infected by the single intradermal comparative tuberculin PPD test (SICTT) and the IFN-γ assay. 31, 39 All cattle showed lesions grossly consistent with TB granulomas 12 at autopsy; in all cases acid-fast bacilli were demonstrated by Zhiel-Nielsen staining of tissue sections, and M. bovis was isolated by bacteriologic cultures of granulomatous lesions. Bacteriologic cultures and identification of M. bovis were carried out by conventional techniques. 12 Animals came from a herd with a TB history previously confirmed by M. bovis culture of slaughtered animals. A 30% TB prevalence was established by PPD and IFN-γ assays. All cattle were born within the herd and, with the exception of one animal, were 3 years or older (range, 49–91 months). Three cattle of the same lineage coming from a TB-free herd were included in the study as controls. At autopsy, the 3 controls were free of grossly evident TB lesions, and Zhiel-Nielsen staining and bacteriologic cultures of retroperitoneal lymph nodes samples were negative. Two controls were PPD- and IFN-γ–negative, and the third control was positive for both tests; this control was included to assess the effects of exposure and sensitization to mycobacterial antigens without ongoing disease in the expression of iNOS and NRAMP1. It is known that about 5% of cattle free of TB lesions as shown by postmortem examination can give positive PPD and IFN-γ. 31, 39
PPD test and IFN-γ assay
Despite the small number of false-positive and false-negative results, these tests are important tools in the assessment of the TB status of cattle. 26, 31, 39 Both tests indicate T-cell–mediated immunity to mycobacteria. 17, 31, 39 The single intradermal comparative tuberculin test (PPD) was performed by the intradermal injection of 0.1 ml of PPD-B (1 mg protein/ml; M. bovis AN5 strain) and 0.1 ml of PPD-A (0.5 mg protein/ml; M. avium D4 strain) (reagents obtained from Productora Nacional de Biológicos Veterinarios, Mexico City, Mexico) in the neck at sites 12 cm apart. Reactions were measured after 72 hours. A test was considered positive when the thickness elicited by PPD-B exceeded that of PPD by 12 mm. For the IFN-γ assay, blood samples with heparin were obtained (20 units heparin/ml) before the PPD test and were processed within a few hours of collection. Plasma was obtained after stimulation of the blood samples with 100 μl of avian PPD (PPD-A, 300 mg/ml), bovine PPD (PPD-B, 300 mg/ml), or phosphate-buffered saline (Nil). The assay for IFN-γ then was carried out following the manufacturers' instructions using a commercial kit (Bovigam, CSL, Parkville, Australia). An IFN-γ test was considered positive when the OD450 of samples stimulated with PPD-B exceeded 100 OD450 units in cases of unstimulated samples, and by 50 OD450 units in cases of PPD-avian–stimulated samples.
Postmortem examination and obtention of samples for study
Postmortem examination was carried out in all animals included in this study. At autopsy, blood, bronchioalveolar lavage (BAL) cells, and tissue samples from lungs and retropharyngeal, tracheobronchial, or mediastinal lymph nodes suspicious of harboring tuberculous lesions were obtained. TB was suspected when lymph nodes were enlarged or fused and, on sectioning, exhibited focal areas of whitish necrosis, calcification, or fibrosis. 12 Blood samples, BAL cells, and retroperitoneal normal-looking lymph nodes were obtained from the 3 control animals. Peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll-Hypaque density gradients (Sigma Chemical Co., St. Louis, MO). BAL cells were collected immediately after slaughtering in 500 ml of Hank's solution (5% Hank's solution, 35 mg NaCO3) that was perfused into the lungs through the main bronchi. The lavage fluid was recovered by suction and pelleted, and, after rinsing, BAL cells were cytospinned on poly-L-lysine–treated glass slides (Sigma Chemical Co.). Tissue samples were frozen in liquid nitrogen or fixed in 10% paraformaldehyde. Fixed tissues were embedded in paraffin, and microtome sections were taken for hematoxilin and eosin staining and for immunohistochemistry. Frozen samples of tuberculous granulomas obtained at autopsy were thawed and cultured by conventional techniques. 12 Zhiel-Nielsen staining of histologic sections of tuberculous lesions also was performed.
Antibodies
The anti-NRAMP1 antiserum was elevated in rabbits immunized with the carboxyl end of murine NRAMP1 (kindly donated by Dr.Vianney Ortiz, CINVESTAV, Mexico City, Mexico). By Western blot, this antibody was shown to react with both the characteristic 65-kDa NRAMP1 band of J774 murine MØ activated with PMA and with a similar band in cattle PBMCs. 15 A monoclonal antiactin immunoglobulin G (IgG) antibody was purchased from Pierce (Rockford, IL). Polyclonal rabbit antibodies to iNOS and NT were obtained from Transduction Laboratories (Lexington, KY). A polyclonal goat antibody to rabbit IgG labeled with peroxidase was obtained from Zymed Laboratories (San Francisco, CA). The anti-MØ mAbMCA874G was from Serotec (Raleigh, NC). A monoclonal antiactin IgG antibody was obtained from Pierce Biotechnology (Rockford, IL).
Western blot
The expression of NRAMP1 in PBMCs, BAL cells, and lymph node and lung granulomas was analyzed by Western blot. Proteins were extracted from cells and frozen tissues in RIPA buffer (50 mM Tris, 150 mM NaCl, 1% NP40, 0.5% sodium deoxycholate, and 0.1% sodium dodecyl sulfate [SDS]), containing a cocktail of protease inhibitors (pepstatin, leupeptin, aprotinin, chemostatin, antipain, and phenylmethylsulfonyl fluoride) and phosphatase inhibitors (Na3VO4 and NaF), and clarified by centrifugation at 4°C for 20 minutes. Protein concentration was determined with the Lowry protein assay (Bio-Rad, Hercules, CA). Fifty micrograms of total protein were mixed with loading buffer, electrophoresed on 7.5% SDS-polyacrylamide gels, and transferred to polyvinylidene difluoride membrane (Bio-Rad). After blocking with 5% milk and 1% bovine serum albumin, membranes were incubated overnight with the anti-NRAMP1 antiserum (1 ° 250). A second anti-rabbit IgG antibody coupled with peroxidase was used (1 ° 5000). Blots were developed using a chemiluminescence system (Amersham Pharmacia Biotech, Piscataway, NJ). To control the amount of protein loaded in each lane, a monoclonal antiactin IgG antibody (1 ° 1000) was used.
Immunohistochemistry
The expression of NRAMP1, iNOS, and NT was analyzed by immunolabeling of cytospin slides of BAL cells and 5-μm microtome sections from formalin-fixed, paraffin-embedded tissues. Cells and tissue sections were placed on poly-L-lysine–coated slides (Sigma Chemical Co.). After deparaffinization and rehydration, sections were immersed in 10 mM of sodium citrate (pH 6.0) and heated for 5 minutes at 95°C. Endogenous peroxidase was eliminated with H2O2 for 10 minutes. After rinsing, slides were incubated with the anti-NRAMP1 antiserum (1 ° 50) for 3 hours at room temperature. For iNOS detection, the polyclonal rabbit was diluted 1 ° 30. To detect NT, a similar procedure was followed using a polyclonal rabbit antibody (1 ° 150). Bound antibodies were labeled with a goat anti-rabbit IgG antibody labeled with peroxidase and diluted 1 ° 200 for 90 minutes. Immunolabeling with the anti-MØ mAb (MCA874G) was also performed (Serotec). Peroxidase was developed with 3,3′-diaminobenzidine. Negative controls consisted of slides in which the first antibody was omitted, as well as BAL cells and lymph node sections from TB-free cattle as shown by postmortem examination. Positive controls for the anti-NRAMP1 antibody were PMA-activated MØs of the J774 murine line. 15 Positive controls for the anti-iNOS and anti-NT antibodies were provided in the kits. Slides were counterstained with Mayer's hematoxylin. The percentage of immunolabeled cells for the 3 antibodies was estimated according to the following scale: +-, 5% or less; +, 6%–20%; ++, 21%–50%, +++, more than 50% labeled cells (Table 1).
Immunologic, pathologic, and immunohistochemical findings in granulomas ofM. bovis naturally infected cattle.

Percentage of immunolabeled cells: −, no labeling; +/−, 5% or less; +,6%–20%; ++, 21%–50%; +++, more than 50%.
PPD = purified protein derivative; INF-γ = interferon gamma; NRAMP1 = natural resistance associated macrophage protein; iNOS = inducible nitric oxide synthase; NT = nitrotyrosine; ✓ = positive results; Neg = negative; TB = animals with autopsy–proven tuberculosis; Controls = animals free of tuberculous lesions at autopsy; T = tracheobronchial; RP = retropharyngeal; M = mediastinal; L = lung.
Results
Immunologic and pathologic findings (Table 1)
Postmortem examination revealed tuberculous granulomas in all cases, which involved mediastinal lymph nodes in 4 animals, tracheobronchial in 3, and retropharyngeal in 2. Lung granulomas were observed in 2 animals.
In hematoxilin and eosin–stained slides (not shown) the characteristic granulomatous tissue reactions of bovine TB were observed. Lymphocytes, epithelioid MØs, and giant cells of the Langhans type were observed throughout the granuloma and organized around fibrocaseous or calcified foci. Zhiel-Nielsen staining revealed few acid-fast bacilli in granulomas (not shown), and bacteriologic cultures successfully demonstrated M. bovis infection in samples taken from lesions of all animals. Postmortem examination failed to identify granulomatous lesions in control cattle, including the one with PPD- and INF-γ–positive test results. Zhiel-Nielsen staining and bacteriologic cultures of retroperitoneal lymph nodes were negative.
Western blot to demonstrate high-level expression of NRAMP1 in PBMCs, BAL cells, and tuberculous granulomas of cattle with TB
Results are shown in Figure 1. Findings were similar with PBMCs, BAL cells, and retroperitoneal lymph node samples of all 8 TB-positive cattle. Two representative examples are shown (TB-1, TB-2). As previously reported, 15 in blots of PBMCs and lymph node granulomas the anti-NRAMP1 antibody revealed a 65-kDa band. A similar band was demonstrated in lung granulomas from 2 cattle and in BAL cells from cattle without grossly apparent TB lung lesions but with lymph node granulomas. Western blots with PBMCs, BAL cells, and samples of normal-looking retroperitoneal lymph nodes of the 3 TB-free controls were negative. A representative example is shown (Contr).
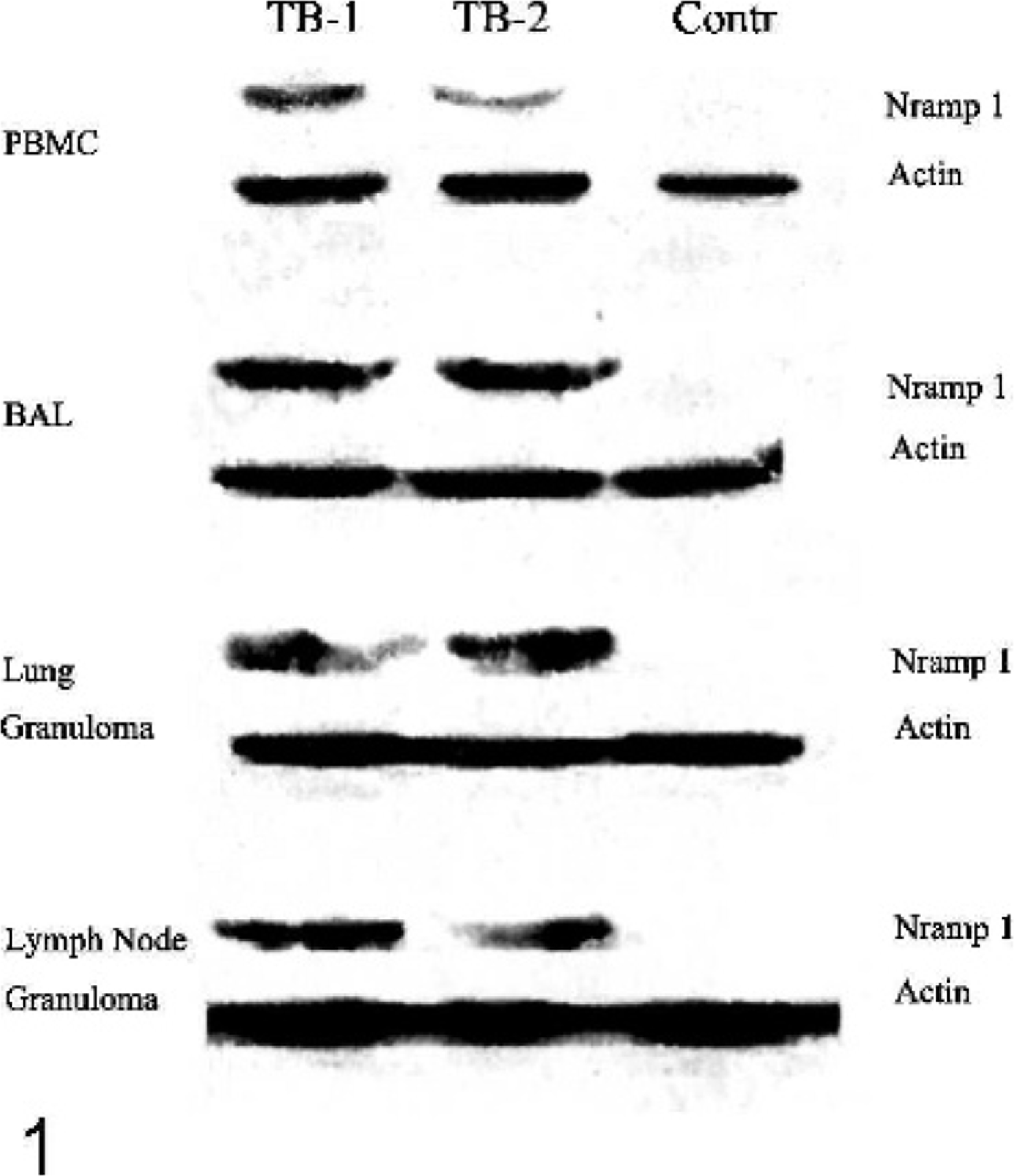
Western blot showing NRAMP1 expression in bovine tuberculosis. Ox No. 8 (TB-1) and cattle No. 212 (TB-2) are shown. Results were obtained with PBMCs, BAL cells, and lung and lymph node granulomas; results also were obtained with cells and tissues from healthy control cattle (Contr; cattle No. 395). Quantitative actin control lanes were included. A second peroxidase-labeled antibody and a chemiluminescence kit were used.
Immunohistochemical demonstration of NRAMP1 expression by granuloma cells
Table 1 presents a semiquantitative estimation of the immunolabeling studies of granulomas with the anti-NRAMP1 antibody. The intensity of labeling (subjectively estimated) and the number of NRAMP1-labeled cells were very high in granulomas from TB cattle (++ to +++). Representative immunohistochemical findings are shown. In well-organized granulomas, numerous epithelioid MØs with abundant intracytoplasmic NRAMP1 granules were observed throughout the inflammatory reaction (Fig. 2a–b). Multinucleated giant cells of the Langhans type with immunoreactive cytoplasmic granules also were frequent (Fig. 2c). In lymph node sections of the TB-free PPD/IFN-γ–positive cattle, few NRAMP1 cytoplasmic granules were observed in occasional cells (not shown). In the 2 PPD/IFN-γ–negative controls, immunostaining was negative (not shown). Cytospin slides of BAL cells (>95% with MØ morphology) immunostained with the anti-NRAMP1 antibody showed cells with abundant clear cytoplasm displaying numerous NRAMP1-positive cytoplasmic granules (Fig. 2d). BAL cells of control animals were negative.
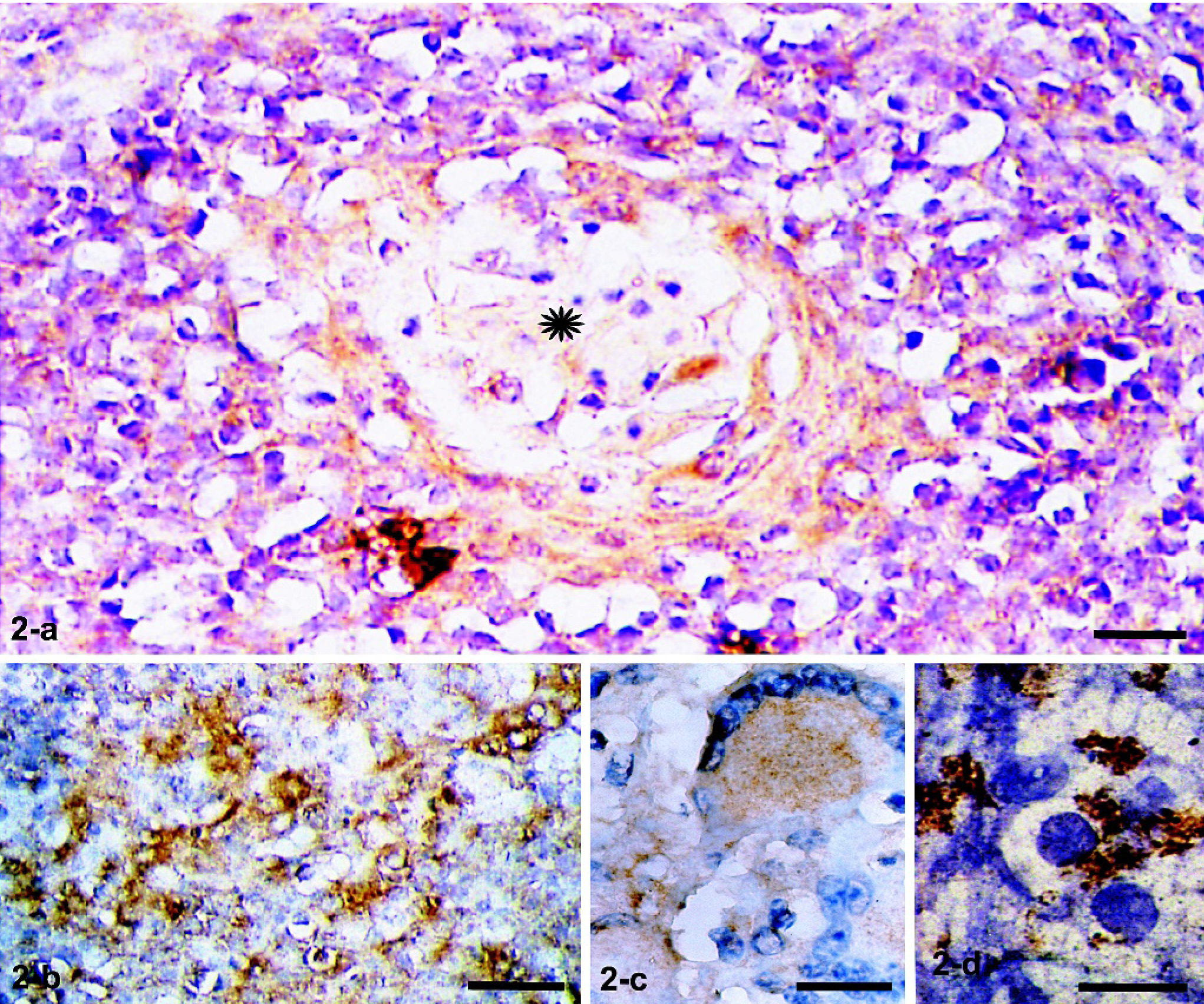
Mediastinal lymph nodes. Ox No. 212. Immunohistochemistry with a polyclonal anti-NRAMP1 antibody and a second peroxidase-labeled antibody is shown. Many cells have abundant intracytoplasmic immunoreactive granules in a well-organized granuloma (
Coexpression of iNOS and nitrotyrosine in tuberculous granulomas of M. bovis–infected cattle
Table 1 presents a semiquantitative estimation of cells expressing iNOS and NT. In TB granulomas from 5 cattle, iNOS expression was very high in cell numbers (++ to +++) and in labeling intensity. In 2 cattle, iNOS expression was low, similar to that observed in control cattle. The immunohistochemical findings in a high iNOS-expressing, well-developed granuloma are presented. The pattern was similar to that observed with the anti-NRAMP1 antibody. Many cells displayed strongly positive cytoplasmic granules, particularly in the vicinity of necrotic areas (Fig. 3a–b). Multinucleated giant cells with numerous cytoplasmic fine granules were frequent (Fig. 3c). In lymph node sections from the TB-free controls, a few iNOS-expressing cells with an elongated appearance were observed (Fig. 3d). No differences were found between PPD- and IFN-γ–positive and –negative controls. Immunostaining of BAL cells obtained from TB cattle showed occasional cells with granular immunoreactivity (Fig. 3e). BAL cells from control animals were negative for iNOS (not shown).
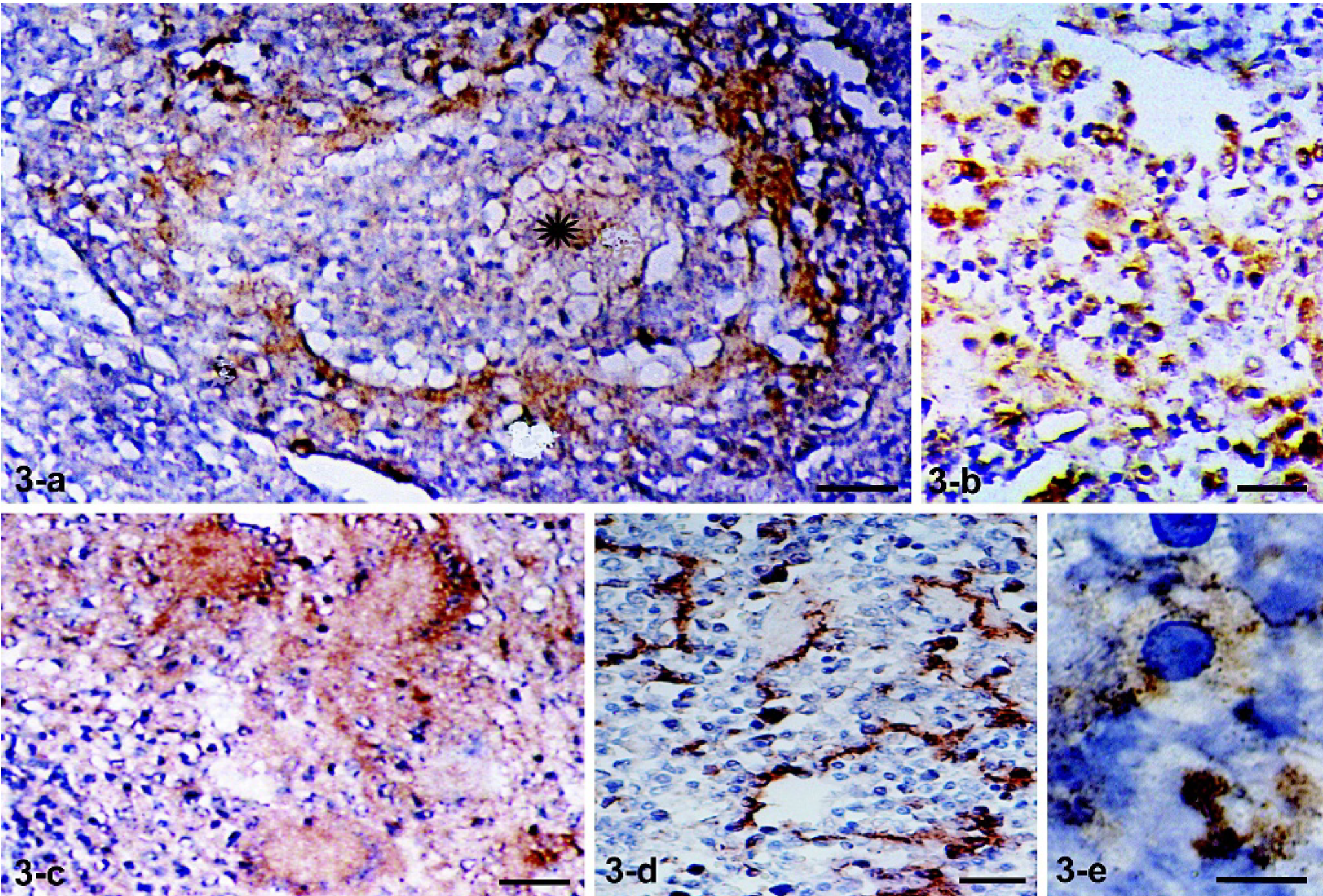
Mediastinal lymph node. Ox No. 529. Immunohistochemistry with a polyclonal anti-iNOS antibody and a second peroxidase-labeled antibody is shown. Around a fibrocaseous central area (∗) of a well-developed granuloma, marked iNOS immunoreactivity was observed in macrophages (
With the exception of 1 cattle, granulomas of all TB animals contained numerous NT-labeled cells (Table 1). Representative findings with the anti-NT antibody are presented. An old granuloma with a calcified center is shown (Fig. 4a, arrow). The great majority of epithelioid MØs and multinucleated giant cells in the granulomatous reaction were strongly NT positive (Fig. 4a). In higher magnifications, the cytoplasmic labeling of MØs and multinucleated giant cells was seen to be diffuse and intense (Fig. 4b). In lymph node sections of healthy controls, a few labeled cells showed a pattern similar to that observed with the anti-iNOS antibody (not shown). Immunostaining of cytospin slides of BAL cells obtained from TB-positive cattle showed a few cells with scantily labeled cytoplasmic granules (Fig. 4c). BAL cells of control animals were negative (not shown). That the cells expressing NRAMP1, iNOS, and NT in the granulomas or in cytospin slides of BAL cells were MØs was demonstrated by parallel slides stained with the anti-MØ mAb MCA874G (not shown). Immunostainings of control slides in which primary antibodies to NRAMP1, iNOS, NT, or MØs were omitted were consistently negative (not shown).
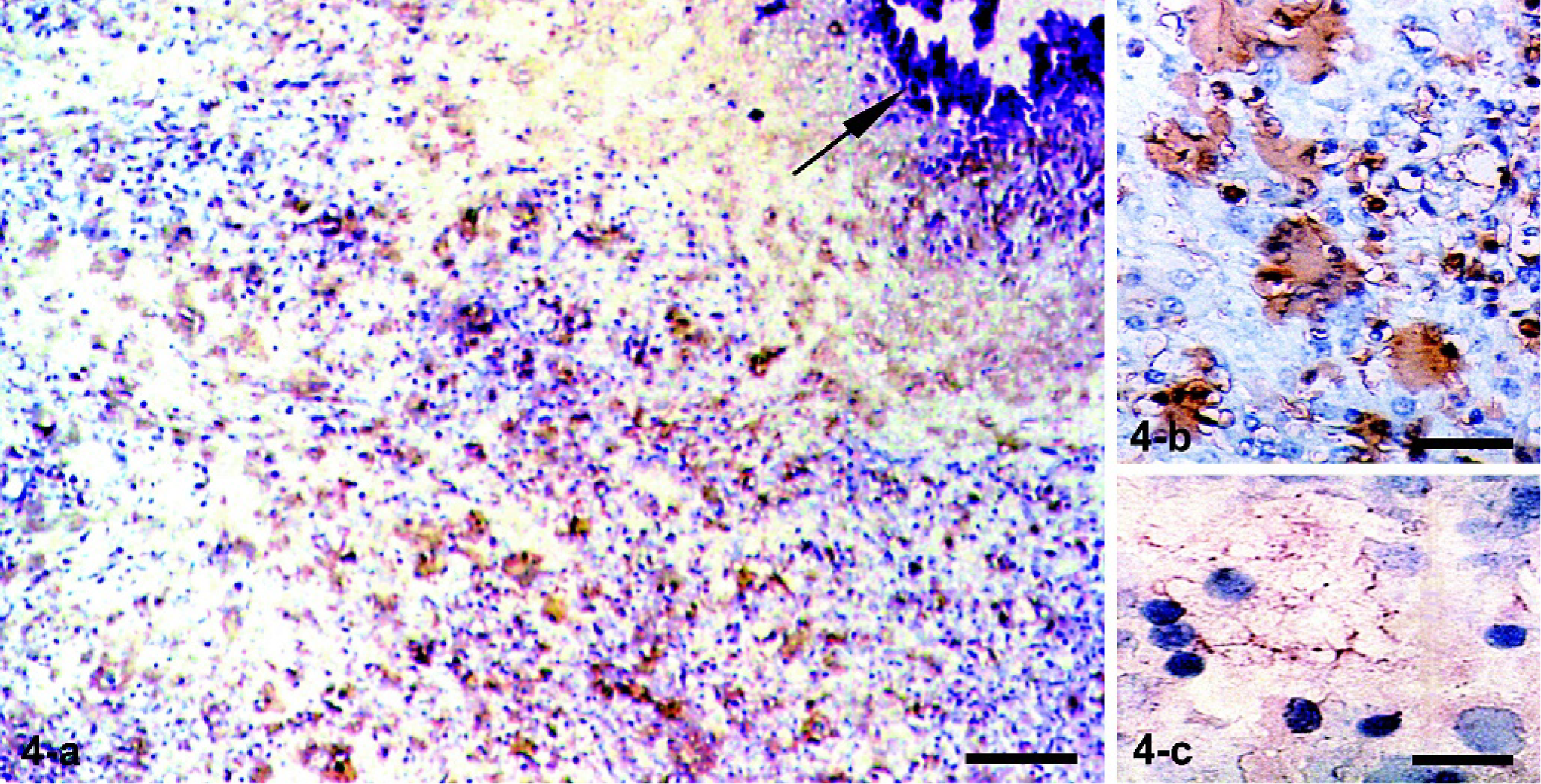
Fig. 4. Tracheobronchial lymph nodes. Ox No. 220. Immunohistochemistry with a polyclonal antinitrotyrosine antibody and a second peroxidase-labeled antibody. Numerous strongly immunolabeled epithelioid macrophages and multinucleated giant cells were observed around a fibrocalcified area (
Discussion
In this work we describe the high expression of NRAMP1 and iNOS in granulomas of M. bovis–infected bovines. It seems that the full-blown expression of these MØ effector molecules requires ongoing active TB, since in healthy control cattle iNOS-producing cells were rare, and NRAMP1-labeled MØs were rare in the PPD- and IFN-γ–positive control. These observations can help us to better understand the pathogenesis of cattle TB. In murine models the central role of NRAMP1 and iNOS has been well documented. 7, 11, 25, 36 In man, these molecules seem to play similar roles, although the evidence is less conclusive. 5, 10, 16, 27, 34 Current findings are potentially significant, since granulomas are protective; they are T-cell–regulated inflammatory reactions in which infecting mycobacteria are confined and eventually eliminated. 17, 20, 21 When T-cell–mediated immunity fails, as occurs in severe combined immunodeficiency (SCID) mice and in AIDS/TB coinfection, the granulomatous response to mycobacteria is defective, and proliferation and dissemination of bacilli go unchecked. 28, 37
To the best of our knowledge, the participation of iNOS in the response to M. bovis infection has not been previously documented, although in vitro studies have shown that M. bovis/BCG–infected bovine MØs produce NO after stimulation with recombinant IFN-γ. This effect, however, was not associated with increased bactericide or bacteriostatic capacity. 9 How bovine MØs regulate iNOS is unknown. In mice, iNOS synthesis is regulated by IFN-γ and TNF-α, 2 cytokines which are produced within the mycobacterial granuloma. 6, 20, 21, 33 These cytokines, however, do not increase iNOS synthesis by bovine MØs, thus suggesting regulatory mechanisms different from those of the murine protein. 1 iNOS is an important effector molecule that catalyzes NO, an agent that is highly toxic for mycobacteria. In vitro, NO kills mycobacteria with a molar potency exceeding that of most antituberculosis drugs, and human alveolar MØs infected with M. bovis/BCG produce NO and peroxynitrite that kill mycobacteria. 24, 29 In this regard, the abundant expression of NT by MØs and Langhans cells that might indicate iNOS activity and NO production by granuloma cells is of particular interest. NO reacts with hydroxyl radicals to generate peroxynitrite. These species are not only powerful oxidants but also strong nitrating agents. 23, 32 Protein tyrosine residues are highly susceptible to peroxynitrite-dependent nitration reactions forming 3-nitrotyrosine, which, as in this work, can be detected by immunolabeling. 23, 32 It is possible that nitration of mycobacteria with NT production and damage to proteins and DNA may affect mycobacterial survival within the phagosome. In vitro studies are needed to determine whether bovine MØs are capable of killing mycobacteria via NO production.
In this study we confirmed previous observations showing the high expression of NRAMP1 in PBMC and MØs of granulomas of M. bovis–infected bovines. 15 Herein, the alveolar MØs are shown to express the protein even in the absence of evident lung TB lesions. NRAMP1 has been the focus of much attention because of its role in genetic susceptibility to mycobacterial infection in mice, 7, 8, 36 a role thus far undetermined for bovine NRAMP1 3, 4 Based on our current observations and on published data, it is possible that NRAMP1 enhances the capacity of granuloma MØs to deal with mycobacteria. 7, 8, 19, 38 This protein upregulates MHCII, IL-1β, TNF-α, and CXC chemokine 7 expression by murine MØs, which are important in the host response to mycobacteria. Another interesting NRAMP1 function is the transport of divalent metals across the phagosomal membrane, which may deprive bacteria of essential micronutrients. 8 Moreover, Fe2+ transported from the cytosol to phagolysosomes may catalyze the Fenton and Haber-Weiss reaction to generate hydroxyl radicals with high bacteriostatic and bactericidal activity. 7 In view of these findings, NRAMP1 must be considered within the arsenal of molecules with antimycobacterial capacity that are expressed in the tuberculous granuloma, and which include IL-1β, IFN-γ, TNF-α, granulocyte-macrophage colony-stimulating factor, IL-12 (p40), and lymphotoxin-β. 6, 20, 21 The similar topographical and cellular expressions of NRAMP1 and iNOS in the granuloma should be noted. These could be related to upregulation of both molecules by similar cytokines produced within the granuloma, such as TNF-α and IFN-γ. 33 It could also be that iNOS is upregulated by NRAMP1. It has been reported that murine MØs carrying functional NRAMP1 display increased NO synthesis that is abrogated by iNOS inhibitors; in contrast, in mice with mutated NRAMP1, iNOS production is diminished drastically. 2 In this regard, it is interesting to mention a recent study describing upregulation of iNOS by NRAMP1 that is associated with increased expression of interferon regulatory factor 1 and augmented binding affinity to the iNOS promoter. 18
In conclusion, our current observations suggest that M. bovis infection promotes the expression of NRAMP1 and iNOS by bovine MØs. Evidence that this expression is particularly strong in the granuloma suggests that these molecules are involved in the host defense against mycobacteria in cattle TB.
Footnotes
Acknowledgements
We thank Ismael Ramirez for skillful technical assistance. This project was supported by grant PROMEP UAEHGO-PTC-301289.302. Isabel Pérez Montfort corrected the English version of the manuscript.
