Abstract
Tuberculosis in animals is caused by members of the Mycobacterium tuberculosis complex (MTC), with the tuberculous granuloma being the main characteristic lesion. The macrophage is the main cell type involved in the development of the granuloma and presents a wide plasticity ranging from polarization to classically activated or pro-inflammatory macrophages (M1) or to alternatively activated or anti-inflammatory macrophages (M2). Thus, this study aimed to analyze macrophage polarization in granulomas from cattle and pig lymph nodes naturally infected with MTC. Tuberculous granulomas were microscopically categorized into four stages and a panel of myeloid cells (CD172a/calprotectin), M1 macrophage polarization (iNOS/CD68/CD107a), and M2 macrophage polarization (Arg1/CD163) markers were analyzed by immunohistochemistry. CD172a and calprotectin followed the same kinetics, having greater expression in late-stage granulomas in pigs. iNOS and CD68 had higher expression in cattle compared with pigs, and the expression was higher in early-stage granulomas. CD107a immunolabeling was only observed in porcine granulomas, with a higher expression in stage I granulomas. Arg1+ cells were significantly higher in pigs than in cattle, particularly in late-stage granulomas. Quantitative analysis of CD163+ cells showed similar kinetics in both species with a consistent frequency of immunolabeled cells throughout the different stages of the granuloma. Our results indicate that M1 macrophage polarization prevails in cattle during early-stage granulomas (stages I and II), whereas M2 phenotype is observed in later stages. Contrary, and mainly due to the expression of Arg1, M2 macrophage polarization is predominant in pigs in all granuloma stages.
Tuberculosis (TB) in animals is a chronic progressive infectious disease that affects a wide range of domestic and wildlife animals, as well as humans. 33 The disease is caused by bacteria belonging to the Mycobacterium tuberculosis complex (MTC). 27 The characteristic lesion of the disease is the tuberculous granuloma,38,39 which is classified into 4 different stages of development in both cattle and pigs. Macrophages are the target cells for mycobacteria replication, and infected cells transition to epithelioid cells and multinucleated giant cells (MNGCs).5,29,39
The interaction between different cellular components reflects the crosstalk between the host’s immune response and the pathogen’s virulence factors. Despite the importance of the tuberculous granuloma, 29 the mechanisms triggered during the pathogenesis of this disease are not still well understood. Subsequent to the infection by MTC, the initial immunologic events include cytokine- and chemokine-mediated recruitment of monocytes, neutrophils, and macrophages. 19 Then, macrophages may act as a functional element to control or overcome the infection, cooperating with activated T cells to establish the granuloma. 26
The cytokine microenvironment in the granuloma can guide macrophage polarization, which adopt different functional responses according to the stimuli and signals.18,24,36 Thus, two main macrophage subpopulations may be identified, the classically activated (M1) pro-inflammatory macrophages and the alternatively activated (M2) anti-inflammatory macrophages. M1 macrophages are polarized by Th1 cytokines, such as interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α), and lipopolysaccharide.18,24 M1 macrophages have a high capacity for antigen presentation as well as secreting high levels of pro-inflammatory cytokines, nitric oxide (NO), reactive oxygen species, and antimicrobial peptides together with low levels of interleukin (IL)-10.28,36 In humans, M1 macrophage polarization is characterized by the upregulation of CD80/CD86, CD68, major histocompatibility complex class-II (MHC-II), IL-1 receptor, toll-like receptor (TLR)-2, TLR-4, inducible nitric oxide synthase (iNOS), and IL-12, and secretion of low levels of IL-10.25,36 Nevertheless, some of these markers, such as CD68 and MHC-II, have been also proposed as general markers for macrophages in mouse models, 23 thus the role of these markers may vary according to several factors, such as the species, age, or stimulus. Other markers, such as CD107a, also known as lysosome-associated membrane protein-1 (LAMP-1), have not been profoundly analyzed in the field of macrophage polarization; however, since CD107a is associated with a Th1 response following lipopolysaccharide stimulation, it may lead to M1 polarization in specific scenarios. By contrast, M2 macrophage polarization is mediated by Th2 cytokines, such as IL-4 and IL-13, through the IL-4 receptor alpha (IL-4Rα), IL-10, and glucocorticoids18,24,36 and can be further divided into at least four different subsets according to the activating stimulus (M2a to M2d). M2 macrophages are characterized in humans and/or mice by the upregulation of several surface molecules such as arginase 1 (Arg1), CD163, CD206, IL-1R, TLR1, and TLR8, and the downregulation of CD14, among others.25,36
Despite M1/M2 dichotomy, macrophage polarization is not fixed as their plasticity allows them to change during inflammation and disease. 28 In this line, macrophage polarization has been simplified in TB studies in different animal models, identifying M1 macrophages as those that express iNOS and M2 macrophages as those expressing Arg1;14,26,29 however, studies comparing the expression of different macrophage polarization markers and their location within the tuberculous granuloma in different animal species are scarce. Therefore, the aim of this study was to compare the expression and distribution of macrophage polarization related markers in granulomas from cattle and pig lymph nodes (LNs) from naturally MTC-infected animals.
Materials and Methods
Animals and Tissue Samples
This study is part of a large project from cattle subjected to the surveillance and monitoring for bovine TB in the framework of the Spanish national eradication program. Cattle included in this study were condemned at the slaughterhouse due to their positive result to the antemortem official test. 35 Samples from pigs belong to a study where animals were condemned at the slaughterhouse because of the presence of generalized TB-like lesions. 6 Ethical review and approval were not required for the animal study since no purpose killing of animals was performed for this study.
Samples for bacteriological, histopathological, immunohistochemical, and polymerase chain reaction (PCR) studies were collected along the slaughter-line after official veterinary inspection and consisted of retropharyngeal and tracheobronchial LNs from five cattle and mandibular LNs from four free-range pigs according to previous reports.9,30 All included animals yielded positive results to bacteriological culture and/or real-time PCR for MTC.6,35 The granuloma was considered as the experimental unit.
Histopathology
Samples from the LNs were fixed in 10% neutral-buffered formalin and embedded in paraffin. Four μm sections were stained with hematoxylin and eosin for histopathological examination and with the Ziehl-Neelsen (ZN) technique for the identification of acid-fast bacilli (AFB). Hematoxylin and eosin-stained sections were used to identify microscopic granulomas, which were further classified as stage I (initial), stage II (solid), stage III (minimal necrosis), and stage IV (necrosis and mineralization), as published elsewhere.5,17,30 A minimum of 10 granulomas per stage was required for the subsequent immunohistochemical study. Table 1 shows the breakdown of granulomas included in the study according to ZN technique for cattle and pigs, respectively. A sample was considered positive for AFB on ZN-stained sections when 1 or more AFB were detected in at least 1 high-power field (1000× magnification) of the granuloma. The total number of AFB present in each granuloma was recorded on a scale of 0 to 3 (0 = no bacilli, 1 = 1–10 bacilli, 2 = 11–50 bacilli, and 3 ≥ 51 bacilli).16,21
Number of tuberculous granulomas according to acid-fast bacilli (AFB) counts in each granuloma stage in lymph nodes from cattle and pig.
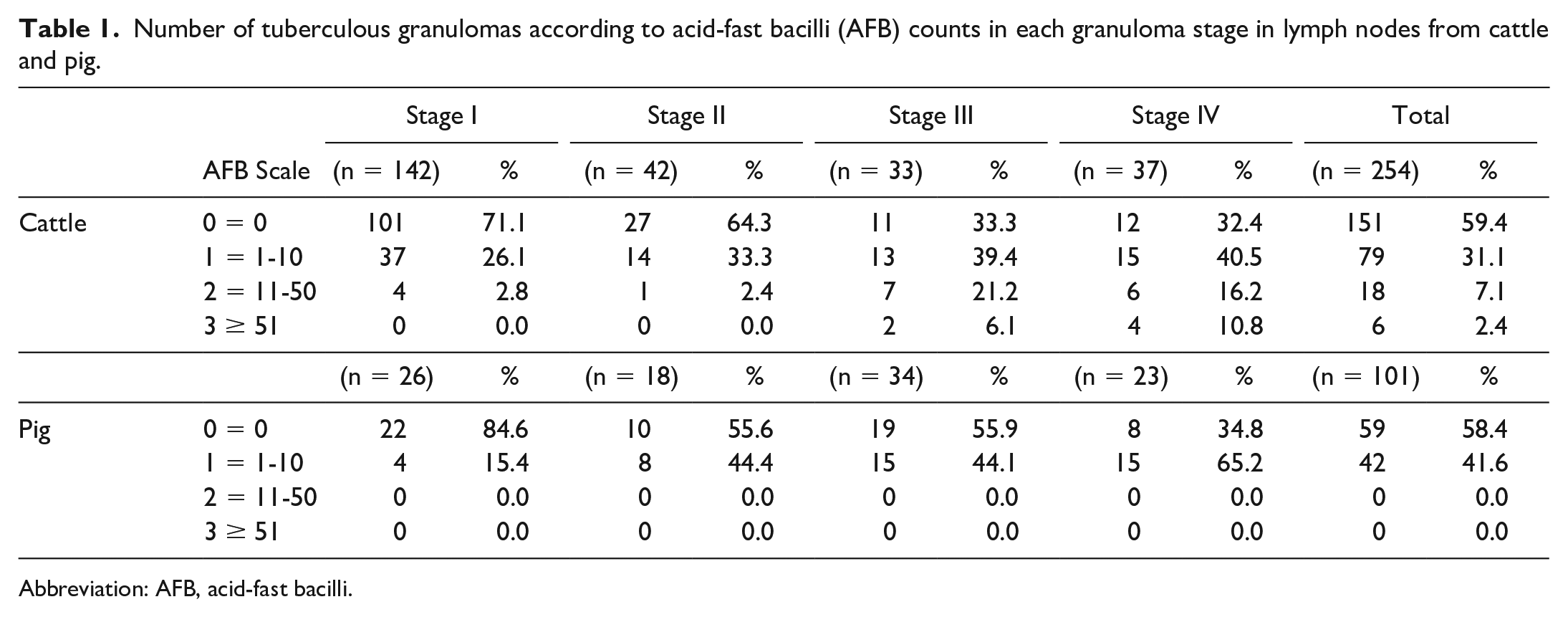
Abbreviation: AFB, acid-fast bacilli.
Immunohistochemistry
The avidin–biotin–peroxidase complex technique (ABC Vector Elite, Vector Laboratories, Burlingame, CA, USA) was carried out to identify and characterize macrophages (CD172a and calprotectin), M1 macrophages (iNOS, CD68, and CD107a), and M2 macrophages (Arg1 and CD163). Briefly, four-μm-thick tissue sections were deparaffinized and rehydrated through graded alcohols, followed by blocking of the endogenous peroxidase activity using 3% hydrogen peroxide in methanol for 30 minutes (min) in darkness. Table 2 summarizes antigen recovery method, blocking solution, and primary and secondary antibodies details. Primary antibody was applied either neat or diluted and incubated overnight at 4°C as described in Table 2. Additionally, a positive control, a negative control, and an isotype control were included for each antibody. For the positive control, a section from a tissue (bovine and porcine lymph node and lung) known to express the protein of interest was included; for the negative and isotype controls, the primary antibody was replaced by the corresponding blocking solution or the isotype-match antibody of irrelevant specificity, respectively, to confirm the lack of non-specific binding. The corresponding biotinylated secondary antibody was applied for 30 min followed by Avidin-Biotin-Peroxidase Complex (Vector Laboratories) for 1 hour at room temperature in the dark. Labeling was visualized by NovaRED substrate kit (Vector Laboratories).
Primary and secondary antibodies, suppliers, and conditions.
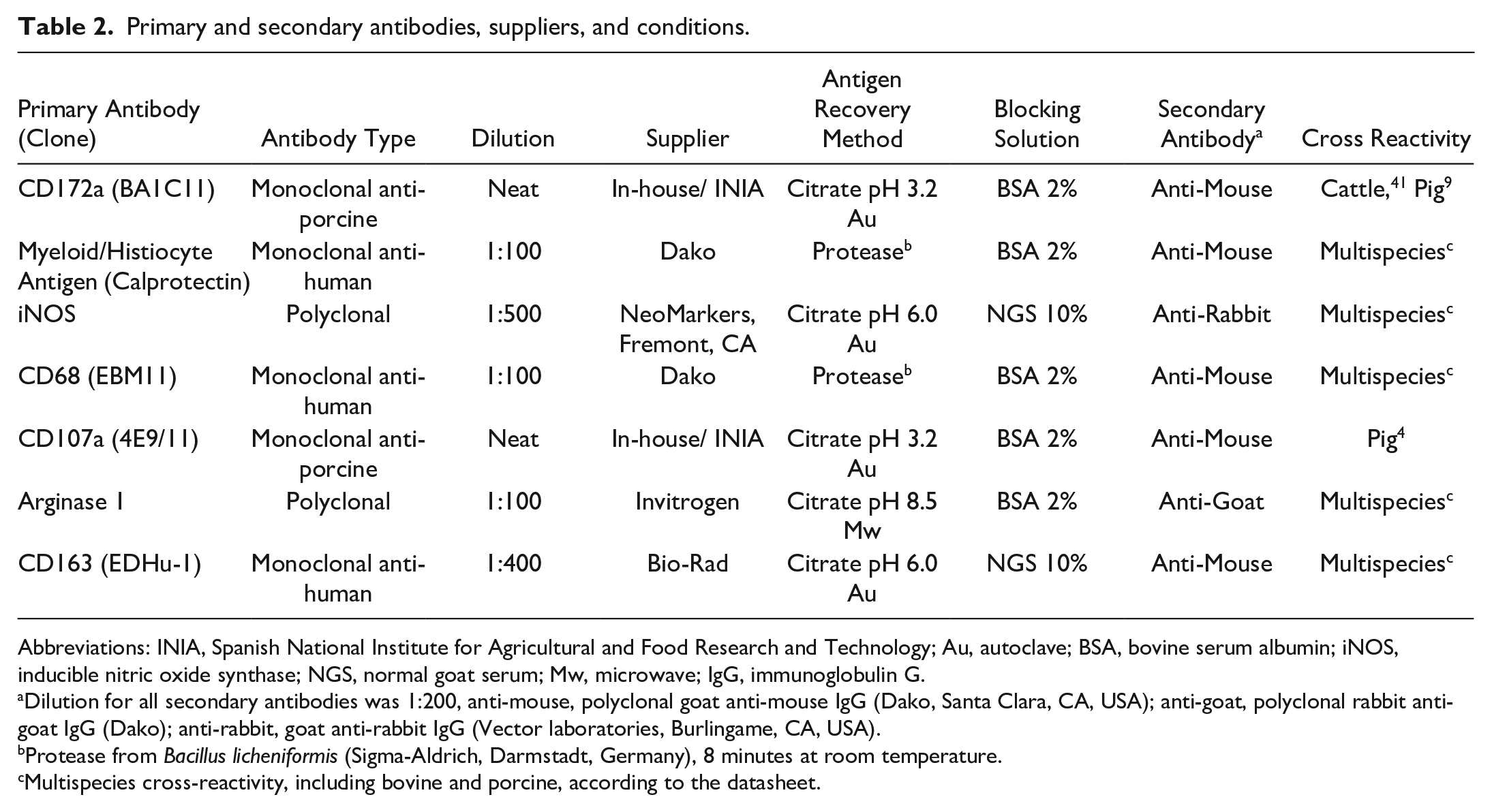
Abbreviations: INIA, Spanish National Institute for Agricultural and Food Research and Technology; Au, autoclave; BSA, bovine serum albumin; iNOS, inducible nitric oxide synthase; NGS, normal goat serum; Mw, microwave; IgG, immunoglobulin G.
Dilution for all secondary antibodies was 1:200, anti-mouse, polyclonal goat anti-mouse IgG (Dako, Santa Clara, CA, USA); anti-goat, polyclonal rabbit anti-goat IgG (Dako); anti-rabbit, goat anti-rabbit IgG (Vector laboratories, Burlingame, CA, USA).
Protease from Bacillus licheniformis (Sigma-Aldrich, Darmstadt, Germany), 8 minutes at room temperature.
Multispecies cross-reactivity, including bovine and porcine, according to the datasheet.
Digital Analysis
Immunolabeled slides were subjected to objective digital image analysis to determine the positive percentage (%) of CD172a+, calprotectin+, iNOS+, CD68+, CD107a+, Arg1+, and CD163+ immunolabeled cells. The slides were photographed at 100× magnification by digital and light microscopy (Olympus BX43, L’Hospitalet de Llobregat, Barcelona, Spain). Immunolabeled cells were registered for the different stages (I, II, III and IV) of tuberculous granulomas for each slide. All granulomas present in each slide were included in the study. The slides were digitalized and the whole area of the granuloma was selected as the region of interest (ROI), with the area of immunolabeled cells inside the ROI was calculated using QuPath software version 0.3.01 after setting thresholds for each marker and stage of granuloma. For stage III and stage IV granulomas, necrotic or mineralized centers were not included in the analysis, as previously described. 39 The results were expressed as the percentage of the area covered by immunolabeled cells within the whole area of the granuloma.
Statistical Analysis
Differences between granuloma stages and species were evaluated using D’Agostino & Pearson’s normality test, followed by Mann Whitney U nonparametric test or Student’s t-unpaired test for parametric data, as appropriate. Figures and data analyses were performed using GraphPad Prism 9.0 software (GraphPad Prism software 9.0, Inc., San Diego, CA, USA). For all data, P value lower than 0.05 was considered statistically significant. In the figures, (*) indicates P ≤ 0.05, (**) P ≤ 0.01, (***) P ≤ 0.001 and (****) P ≤ 0.0001.
Results
Distribution of Bovine and Porcine Granulomas According to Stage and ZN Technique
A total of 254 granulomas were evaluated from bovine LNs and 101 granulomas from porcine LNs. According to the granuloma stage classification, the distribution of the 254 bovine granulomas was: 142/254 (55.9%) were stage I, 42/254 (16.5%) were stage II, 33/254 (13.0%) were stage III, and 37/254 (14.6%) were stage IV. In the case of porcine granulomas, the distribution was as follows: 26/101 (25.7%) were stage I, 18/101 (17.8%) were stage II, 34/101 (33.7%) were stage III, and 23/101 (22.8%) were stage IV (Table 1).
ZN technique was performed to determine the presence of AFB in each tuberculous granuloma. Accordingly, no AFB were detected in most bovine granulomas (151/254; 59.4%). One to 10 AFB (grade 1) were observed in 79 out of 254 granulomas (31.1%). Grade 2 (11-50 AFB) and grade 3 (≥ 51 AFB) were present in 18 (7.1%) and in 6 (2.4%) of 254 granulomas, respectively. The number of AFB showed a trend toward increasing with the stage of the granuloma in bovine samples (Table 1). In porcine LNs, 59 of 101 granulomas (58.4%) did not contain any AFB whereas 1 to 10 AFB (grade 1) were observed in 42 of 101 granulomas (41.6%). No porcine granuloma presented more than 10 AFB (grades 2 and 3). In this species, no differences were observed between the number of AFB and the granuloma stage (Table 1).
CD172a and Calprotectin in Bovine and Porcine Granulomas as Markers of Myeloid Cells
CD172a expression showed a diffuse and cytoplasmic pattern with a stronger labeling in myeloid cells (including macrophages and epithelioid cells) in both species, while only scattered MNGCs from cattle were weakly positive for this marker (Fig. 1a, b). The expression was similar within the different granuloma stages in both species, but stage III and stage IV granulomas in pigs showed additional immunolabeling of neutrophils within the necrotic core and within the rim between the necrotic center and the fibrous capsule (Fig. 1b). A similar trend in the frequency of CD172a+ cells was observed in both species, although a significant increase was observed in stage II granulomas from pigs in comparison to cattle (P < .001) (Fig. 2a). Moreover, a slight decrease in the frequency of CD172a+ cells was observed in porcine granulomas when progressing from stage II granulomas to later stages (P < 0.05).
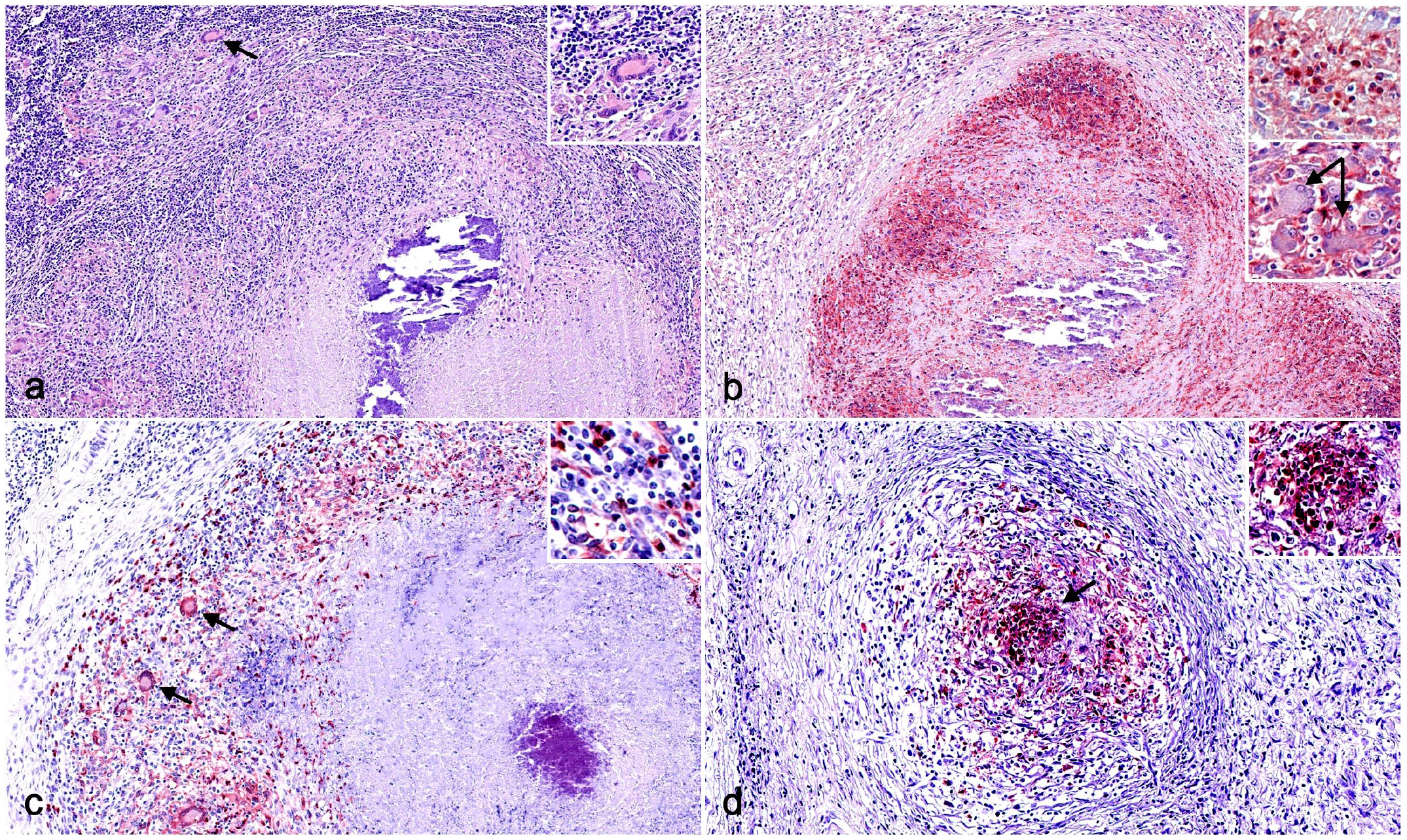
Immunohistochemistry (IHC) for myeloid cell markers in tuberculous granulomas in cattle and pig lymph nodes. (a) Cattle. CD172a+ macrophages, epithelioid cells, and multinucleated giant cells (MNGCs) (arrow) within the fibrous capsule of a stage IV granuloma. Inset: higher magnification. CD172a IHC. (b) Pig. Stage IV granuloma in a pig displaying a marked expression of CD172a in the cells within the surrounding capsule of connective tissue, as well as within the necrotic core. Upper inset: higher magnification of CD172a+ cells. Lower inset: higher magnification CD172a– MNGCs (arrows). CD172a IHC. (c) Cattle. Calprotectin+ macrophages, epithelioid cells, MNGCs (arrows), and neutrophils within the fibrous capsule of a stage IV granuloma. Inset: higher magnification. Calprotectin IHC. (d) Pig. Stage II granuloma displaying a marked expression of calprotectin in the center of a granuloma highlighting a strong neutrophilic infiltrate (arrow). Inset: higher magnification calprotectin+ neutrophils. Calprotectin IHC.
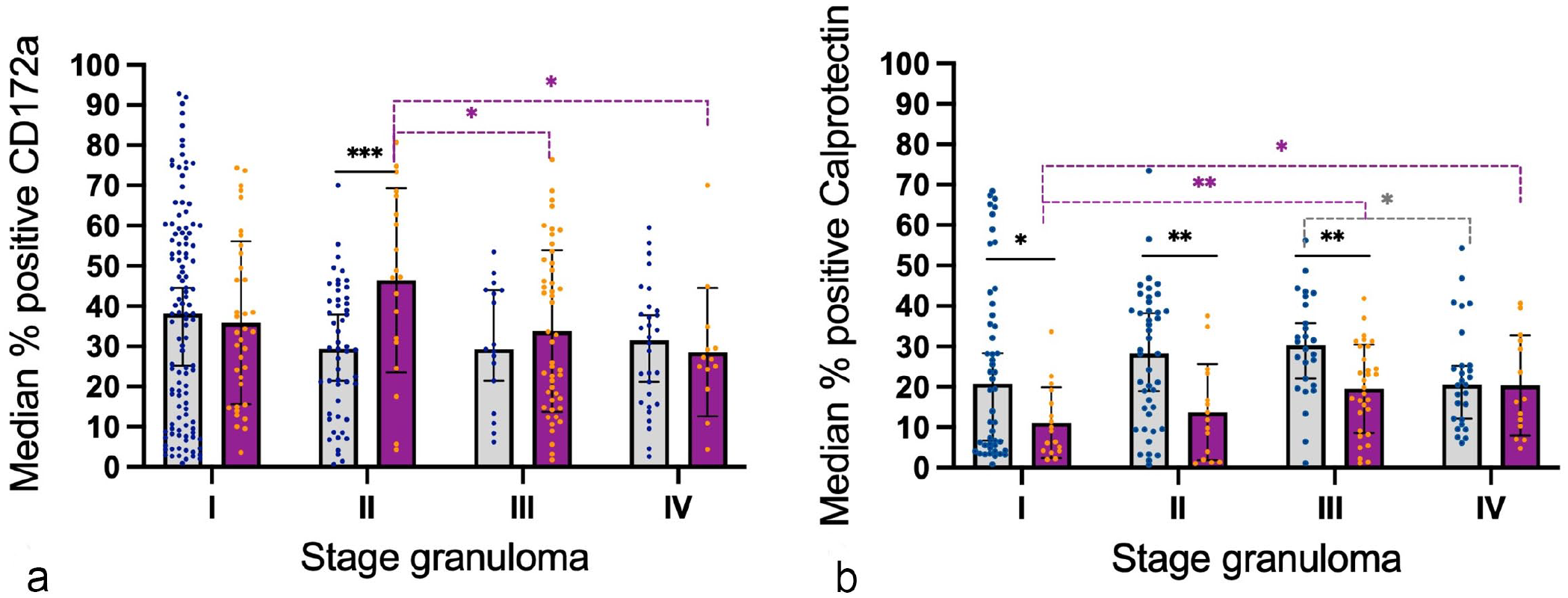
Graphs represent the median percentage of positively immunolabeled cells within each granuloma for (a) CD172a and (b) calprotectin in bovine and porcine lymph nodes. Gray columns and blue points represent bovine tuberculous granulomas and purple columns and orange points represent porcine tuberculous granulomas. Statistical differences are shown as * = P < 0.05, ** = P < 0.01 and, *** = P < 0.001.
Calprotectin expression showed a cytoplasmic labeling pattern within macrophages, epithelioid cells, MNGCs, and neutrophils in all stages of granulomas from both species (Fig. 1c, d). In cattle, immunolabeled cells were more numerous in the center of stage I and stage II granulomas in contrast to stages III and IV, where immunolabeled cells, including MNGCs, surrounded the necrotic center (Fig. 1c). However, in pigs, the frequency of calprotectin+ cells was lower than in cattle, with only few positive cells, mainly neutrophils, in the center of stage I and II granulomas (Fig. 1d). In stages III and IV granulomas in pigs, the distribution of immunolabeled cells was similar to the distribution observed in cattle. The quantitative analysis of calprotectin+ cells showed a higher frequency of immunolabeled cells for all stages of granuloma in cattle in comparison to pig and was statistically significant in stages I (P < 0.05), II, and III (P < 0.01). In general, there was a tendency to increase toward the most advanced granuloma stages, particularly in stages II and III in cattle and stages III and IV in pigs (Fig. 2b).
iNOS, CD68, and CD107a in Bovine and Porcine Tuberculous Granulomas as Markers of M1 Macrophages
iNOS expression showed a diffuse cytoplasmic labeling pattern, mainly in epithelioid cells and MNGCs in all stages of granulomas, with stronger labeling in granulomas from cattle when compared with pigs (Fig. 3a, b). In stage I and II granulomas, immunolabeled cells were distributed throughout the granuloma, whereas in stage III and stage IV granulomas iNOS+ cells consisted of epithelioid cells macrophages and MNGCs forming a rim surrounding the necrotic core and intertwining in the fibrous capsule of the granuloma. Although a similar kinetics was observed in both species, displaying a significant decrease from stage I granulomas toward more advanced stage granulomas (stage IV) (P < 0.0001), the median frequency of iNOS+ cells was higher in bovine than in porcine (Fig. 4a).
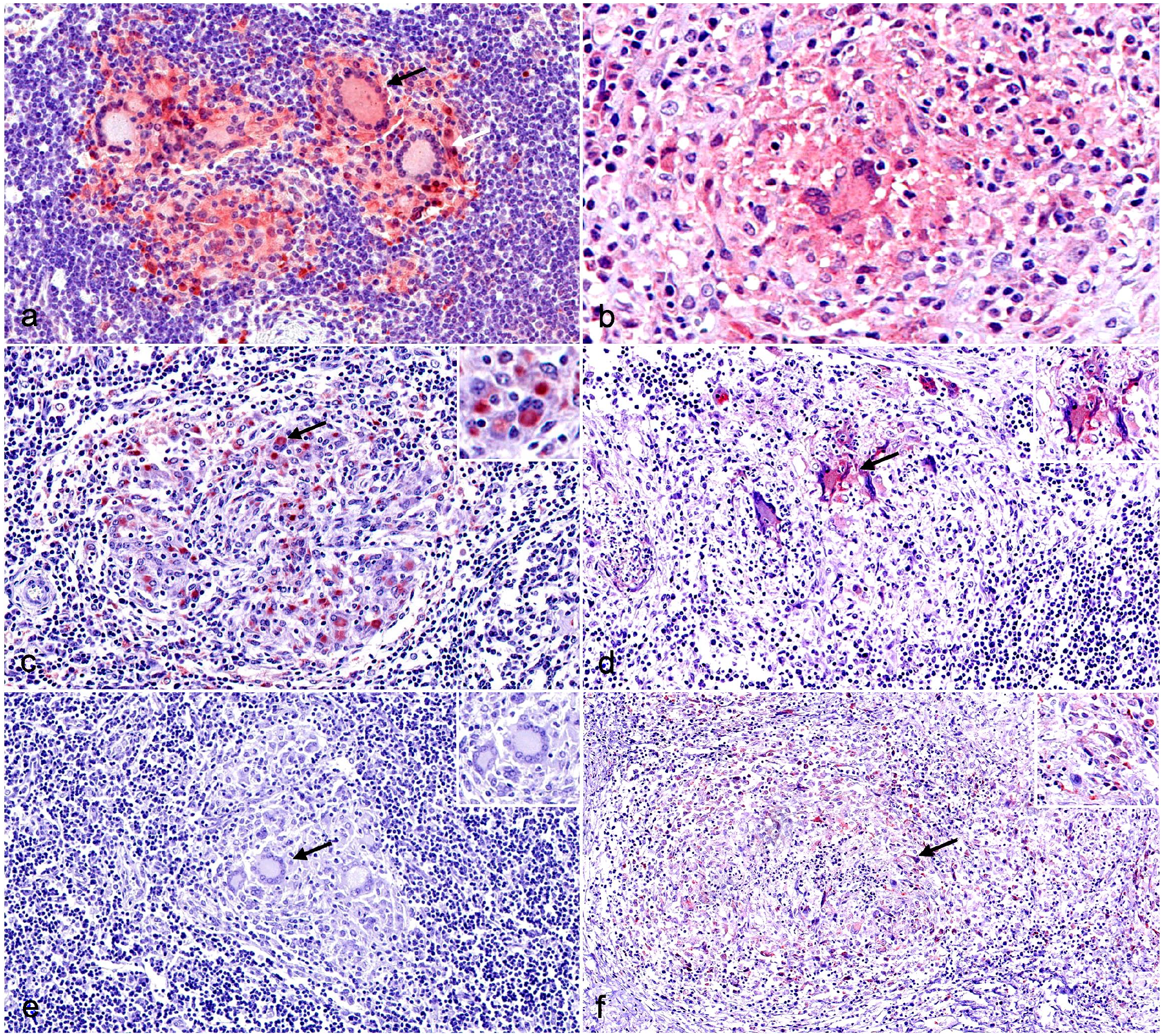
Immunohistochemistry (IHC) for M1 macrophage markers in tuberculous granulomas in cattle and pig lymph nodes. (a) Cattle. Strongly labeled inducible nitric oxide synthase (iNOS)+ macrophages, epithelioid cells, and multinucleated giant cells (MNGCs) (black arrow) and weakly labeled iNOS+ MNGCs (white arrow) of a stage I granuloma. iNOS IHC. (b) Pig. Stage I granuloma displaying a weak expression of iNOS in an MNGC (arrow). iNOS IHC. (c) Cattle. CD68+ macrophages, epithelioid cells, and MNGCs (arrow) of a stage II granuloma. Inset: higher magnification of an MNGC with an intense CD68 cytoplasmic immunolabeling. CD68 IHC. (d) Pig. Weak expression of CD68 in macrophages and epithelioid cells with a more evident labeling of MNGCs (arrow) in a stage II granuloma. Inset: higher magnification of a CD68+ MNGC. CD68 IHC. (e) Cattle. Stage I granuloma from cattle showing the lack of cross-reactivity against CD107a (arrow). Inset: higher magnification of MNGCs without CD107a labeling. CD107a IHC. (f) Pig. CD107a+ macrophages (arrow), epithelioid cells, and MNGCs of a stage II granuloma. Inset: higher magnification of CD107a+ macrophages within the granuloma. CD107a IHC.
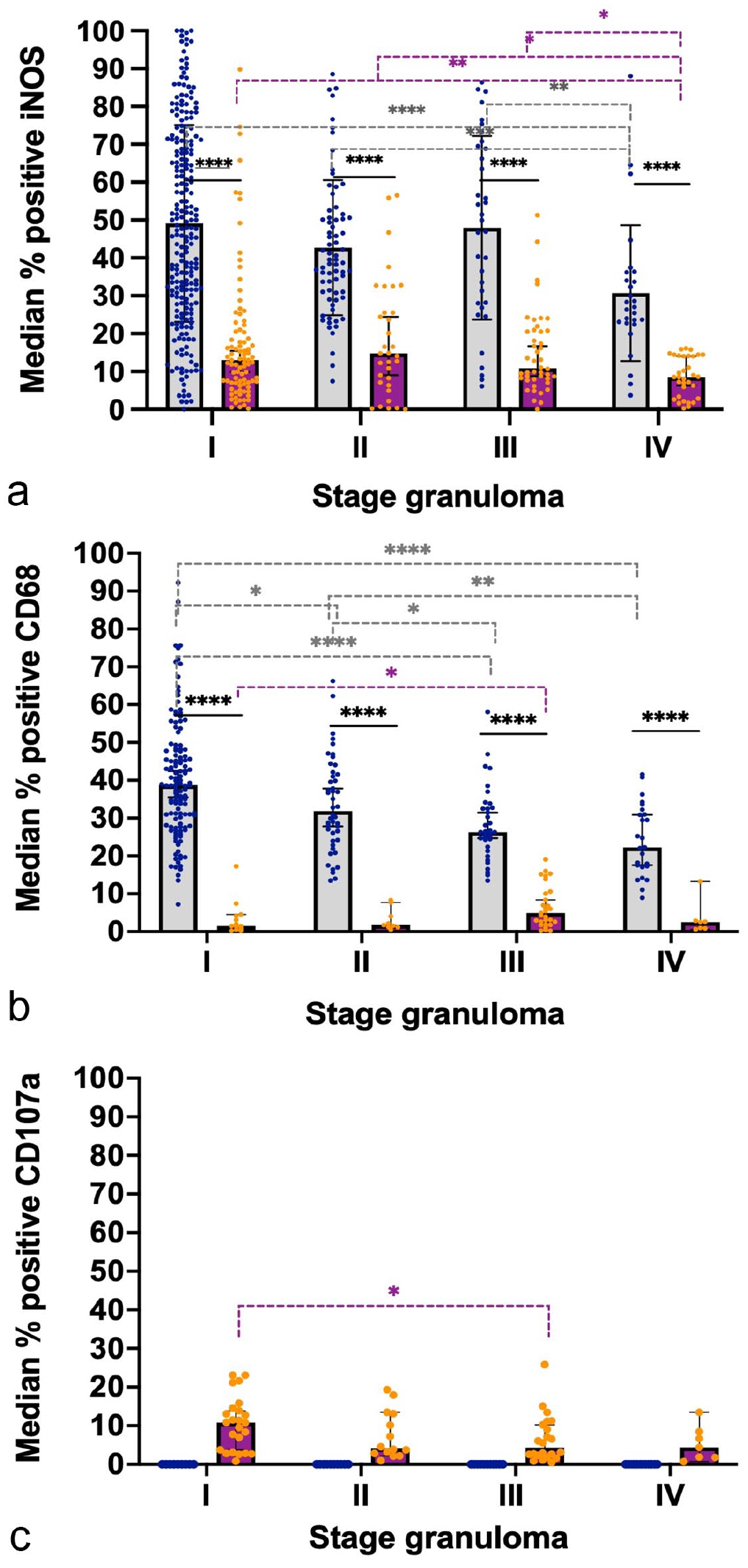
Graphs represent the median percentage of positively immunolabeled cells within each granuloma for (a) inducible nitric oxide synthase (iNOS), (b) CD68, and (c) CD107a in bovine and porcine lymph nodes. Gray columns and blue points represent bovine tuberculous granulomas and purple columns and orange points represent porcine tuberculous granulomas. Statistical differences are shown as * = P < 0.05, ** = P < 0.01, *** = P < 0.001 and, **** = P < 0.0001.
CD68 expression was observed in the cytoplasm of macrophages, MNGCs, and epithelioid cells from all granuloma stages (I-IV) in both species (Fig. 3c, d). However, the labeling was weaker in pigs compared with cattle, where MNGCs were strongly labeled. Scattered CD68+ macrophages and epithelioid cells were distributed throughout stage I and II granulomas in cattle and positive cells were within the fibrous capsule of stage III-IV granulomas, although in lower numbers. In contrast, porcine stage I and II granulomas contained only scattered CD68+ macrophages and MNGCs in the center of the granuloma. CD68+ macrophages and MNGCs were randomly distributed at the border of the necrotic center and within the fibrous capsule in stages III and IV of porcine granulomas, with a slight increase in the frequency of positivity as the granuloma stage progressed. The quantitative analysis of CD68+ cells (Fig. 4b) showed a significantly higher frequency of immunolabeled cells (P < 0.0001) in cattle compared with pigs with a progressive decrease from early stages to more advanced stage granulomas, as was observed for iNOS expression.
CD107a labeling was only observed in porcine granulomas; no cross-reactivity was observed in cattle tissue (Fig. 3e, f). In pigs, granulomas from the different stages (I-IV) contained scattered CD107a+ macrophages and MNGCs, which were mainly found within the granulomas’ fibrous capsule (Fig. 3f). The highest frequency of CD107a+ cells was observed in stage I granulomas, and there was a decreased frequency of CD107a+ in more advanced stage granulomas (P < 0.05, stage I vs stage III granulomas) (Fig. 4c).
Arg1 and CD163 in Bovine and Porcine Tuberculous Granulomas as Markers of M2 Macrophages
Arg1 was expressed in macrophages and MNGCs in both species, with a stronger expression in MNGCs (Fig. 5a, b). The frequency of Arg1+ cells was significantly higher in pigs than in cattle, notably in stage III granulomas (P < 0.001) (Fig. 6a). In the case of cattle, the frequency of Arg1+ cells was higher in stage I and II granulomas and was relatively decreased at higher stages, whereas a marked increase in the number of Arg1+ cells was observed in porcine stages III and IV granulomas, especially in the former. In late-stage granulomas, positive cells demarcated the necrotic core, particularly in pigs (Fig. 5b), and were less numerous toward the periphery.
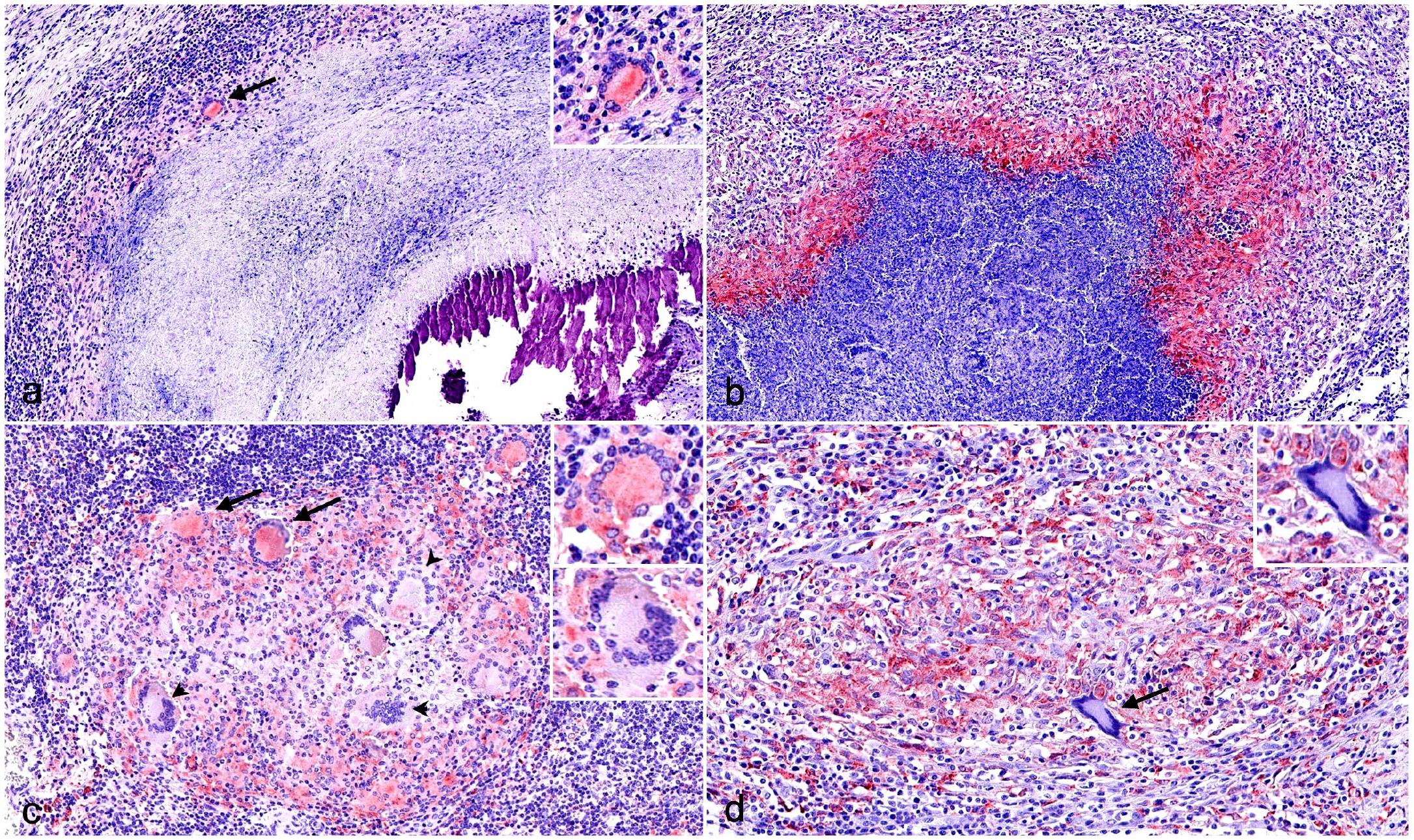
Immunohistochemistry (IHC) for M2 macrophage markers in tuberculous granulomas in cattle and pig lymph nodes. (a) Cattle. Weakly labeled arginase 1 (Arg1)+ macrophages, epithelioid cells, and multinucleated giant cells (MNGCs) (black arrow) within the fibrous capsule of a stage IV granuloma. Inset: higher magnification. Arg1 IHC. (b) Pig. Stage IV granuloma displaying a marked expression of Arg1 in the cells within the rim of the necrotic center of a stage III granuloma. Arg1 IHC. (c) Cattle. CD163+ macrophages, epithelioid cells, and MNGCs (arrows) and CD163– MNGCs (arrowheads) in a stage I granuloma. Upper inset: higher magnification CD163+ MNGC. Lower inset: higher magnification CD163– MNGC. CD163 IHC. (d) Pig. Stage II granuloma displaying a marked expression of CD163 in the cells within the surrounding capsule of connective tissue and lack of expression in an MNGC (arrow). Inset: higher magnification CD163– MNGC. CD163 IHC.
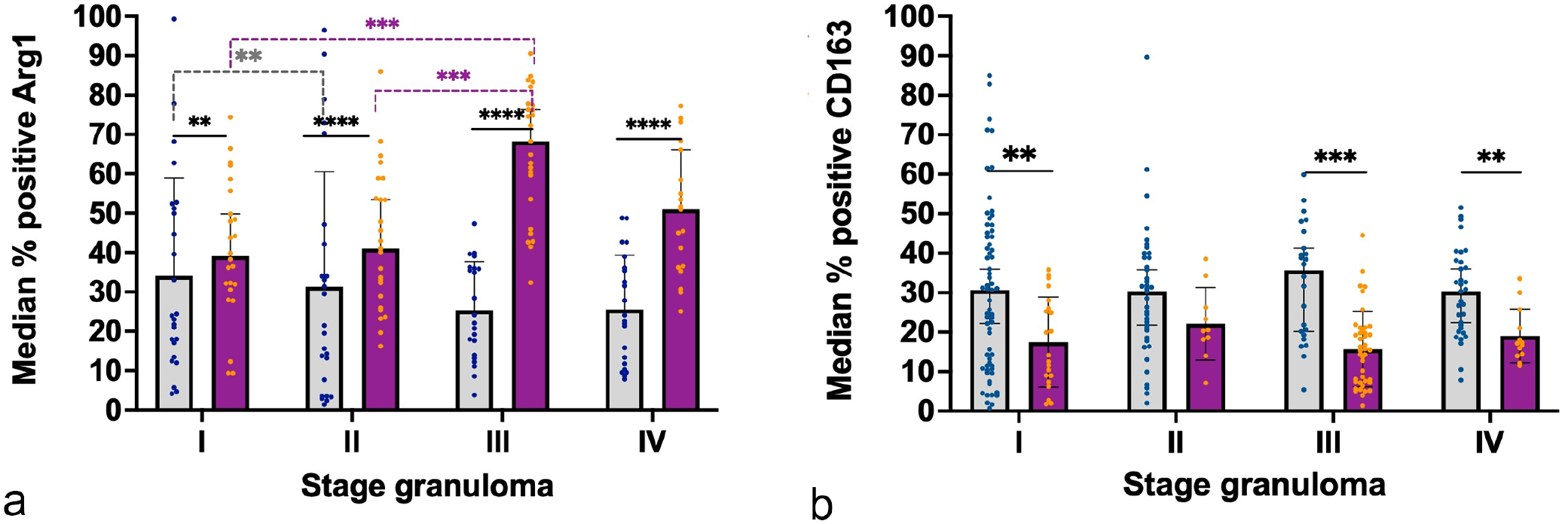
Graphs represent the median percentage of positively labeled cells within each granuloma for (a) arginase 1 (Arg1) and (b) CD163 in bovine and porcine lymph nodes. Gray columns and blue points represent bovine tuberculous granulomas and purple columns and orange points represent porcine tuberculous granulomas. Statistical differences are shown as * = P < 0.05, ** = P < 0.01, *** = P < 0.001 and, **** = P < 0.0001.
CD163 expression showed 2 immunolabeling patterns. A diffuse cytoplasmic pattern was seen in epithelioid cells and, inconsistently, in most of the MNGCs, while macrophages showed stronger labeling in all granuloma stages from cattle (Fig. 5c) and pigs (Fig. 5d). In stage I and II granulomas, immunolabeled cells were distributed throughout the granuloma (Fig. 5c, d). However, in bovine stage III and stage IV granulomas, CD163+ epithelioid cells and macrophages were observed surrounding the necrotic core together with CD163+ MNGCs. In pigs, CD163+ macrophages were mainly observed in the fibrous capsule and in few MNGCs in stages III and IV granulomas. The quantitative analysis of CD163+ cells (Fig. 6b) displayed similar kinetics in both species with a constant frequency of immunolabeled cells along the different granuloma stages; however, a significantly higher number of cells was observed in cattle compared with pigs in stage I, III, and IV granulomas (P < 0.01; P < 0.001).
Evaluation of M1/M2 Ratio Along Granuloma Stages in Bovine and Porcine Tuberculous Granulomas
To evaluate M1 and M2 polarization in bovine and porcine tuberculous granulomas, the ratio between iNOS/Arg1, as representatives of M1 and M2 polarization, respectively, as well as the ratio between the sum of the staining of each of the M1 (iNOS + CD68 + CD107a) or M2 (Arg1 + CD163) polarization markers used in the present study were calculated. Table 3 summarizes the results of both ratios for each granuloma stage. Our results showed a clear trend to M1 polarization in bovine tuberculous granulomas from stages I to II, which shifted to M2 polarization when progressing to stages III and IV (Table 3). In contrast, porcine tuberculous granulomas were governed by a clear M2 polarization along all granuloma stages (Table 3).
Immunohistochemistry ratios according to the analyzed iNOS/Arg1 and M1/M2 macrophage markers in lymph nodes from cattle and pigs.
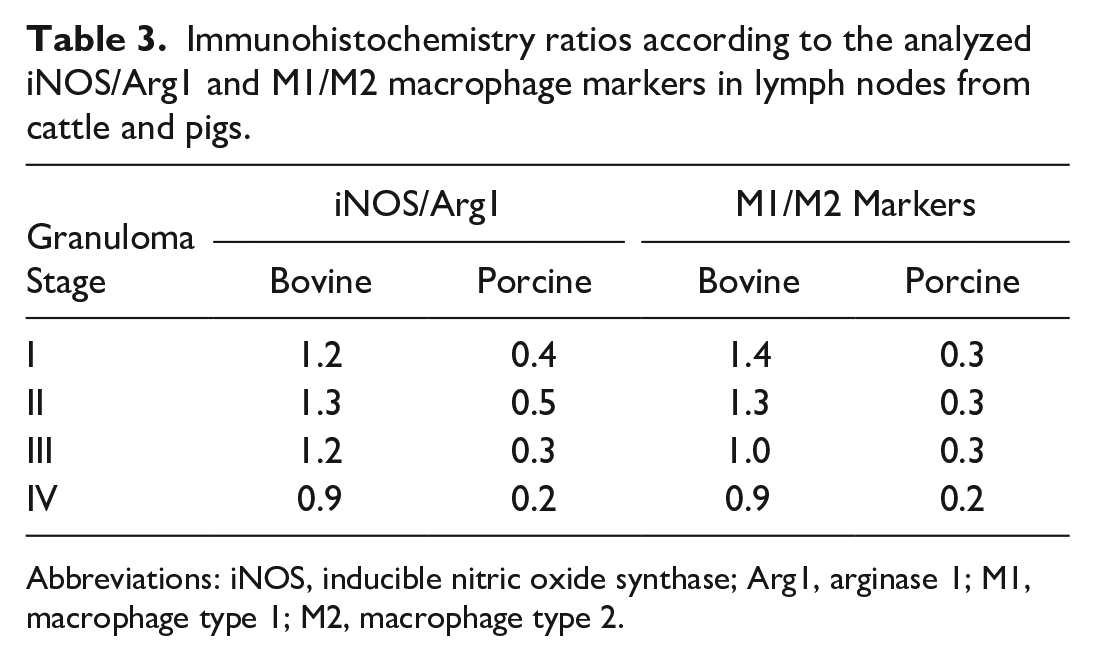
Abbreviations: iNOS, inducible nitric oxide synthase; Arg1, arginase 1; M1, macrophage type 1; M2, macrophage type 2.
Discussion
The complex interaction between mycobacteria and the host immune system makes it difficult to understand the pathogenesis of TB. Therefore, deepening our understanding of the mechanisms involved in the onset and progression of the tuberculous granuloma throughout the course of infection and its comparison among between different animal species may provide valuable information to advance the knowledge of this disease.
In this study, a high percentage of granulomas was negative for mycobacterium on ZN-stained section, with similar results in both cattle (59.4%) and pigs (58.4%), which is in agreement with prior studies.6,15,16 Pluribacillary lesions (grade 3) were only observed in cattle toward the more advanced stages of the granuloma (stages III and IV), highlighting an increase in the number of AFB and bacterial load during granuloma progression, as it has been previously proposed by others. 29
Specific markers for myeloid cells (macrophages and epithelioid cells)9,41 (CD172a and calprotectin) were used to characterize the macrophage population in the different granuloma stages. Thereafter, selected panels of specific markers for M1 macrophages (iNOS, CD68, and CD107a)4,26,30,34 and M2 macrophages (Arg1 and CD163)11,14,26 were evaluated. Results showed that CD172a was similarly expressed in bovine and porcine granulomas, was slightly decreased in advanced stage granulomas (III and IV). Considering its expression in MNGCs, they weakly expressed CD172a in cattle, but no labeling was found in the case of pigs. 40 However, from our results, the expression of CD172a does not seem to play a major role in the formation of porcine MNGCs.
Calprotectin behaved like CD172a in cattle, while in pigs calprotectin increased with advancing stages of the granuloma, differing from prior studies on fallow deer and wild boar, in which the percentage of calprotectin+ cells decreased toward more advanced stages of granuloma.16,17 Calprotectin was highly expressed in neutrophils of porcine tuberculous granulomas, highlighting another difference between cattle and pigs in our study, although this calprotectin+ neutrophils have been also observed in tuberculous granulomas from wild boars and different deer species.15,16,21 The role of neutrophils in the context of MTC infection is not clearly understood. 3 These cells have been reported to be able to phagocytose and eliminate Mycobacterium tuberculosis, but they could also supply nutrients to mycobacteria and cause tissue damage through their cellular storage of antimicrobials.3,12,31 However, when neutrophils fail to kill mycobacteria due to their virulence, the uncontrolled generation of reactive oxidative species within the neutrophils drive them toward necrosis. 8 Furthermore, the attraction of other inflammatory cells as well as persistent recruitment of cells to sites of chronic mycobacterial infection by neutrophils will promote the progression to more advanced granuloma stages.8,12 On the other side, the multi-etiological nature of lymphadenitis in pigs, 5 where it is common to find TB associated with other bacteria besides mycobacteria, may also explain the higher participation of neutrophils in pig granulomas. However, microbiological studies were not carried out in these cases to evaluate the participation of non-tuberculous microorganisms, thus, we cannot rule out the participation of other microorganisms. To better understand the function of these cells in the context of tuberculous granulomas, further research needs to be conducted.
Markers pointing toward M1 macrophage polarization were highly expressed in cattle, particularly in early-stage granulomas. Specifically for iNOS, our results agree with those previously reported in cattle,
30
deer,
16
and wild boar,
15
suggesting an important role for NO in the early stages of the disease as an attempt to eliminate mycobacteria.
20
Since iNOS and Arg1 compete directly for
CD68 expression followed the same trend as iNOS, but with a stronger expression in MNGCs, as reported elsewhere. 30 However, other authors have reported a lack of changes or even an increase in the number of CD68+ cells when lesions progressed in the same species.34,39 Interestingly, positive and negative correlations among iNOS and CD68 expression with respect to the count of AFB have been previously reported in bovine paratuberculosis, 13 which may suggest opposite roles for these molecules in cattle for this disease. However, in our study, focused on bovine TB, both parameters presented similar kinetics, displaying a higher expression in early-stage (paucibacillary) granulomas and decreasing onwards, where a higher number of AFB were observed within late-stage granulomas. Furthermore, the similar kinetics observed with respect to iNOS expression in cattle support a decrease of M1 polarization from initial stages to later stages of TB. In contrast, a lower percentage of positivity was observed in porcine, which could point to a lack of activation of macrophages by this pathway in pigs. 7
CD107a was weakly expressed in porcine granulomas, showing a slightly higher positivity in stage I granulomas and decreasing in late-stage granulomas, while no labeling was observed in cattle, probably due to a lack of cross-reactivity. The expression of iNOS, CD68, and CD107a support a higher polarization toward M1 macrophages in bovine than porcine granulomas. This is more evident during the early stages of TB and decreases with the evolution of the disease, which could inform the understanding of the persistence of the mycobacteria within the tuberculous granuloma.
M2 polarization was explored by the expression of Arg1 and CD163. Interestingly, Arg1 showed a higher expression in pigs compared with cattle with a higher frequency of Arg1+ cells toward more advanced stages of granuloma. There are few studies that analyzed this marker in TB in livestock, but similar results have been previously observed in mice, 22 macaques, 26 and human lungs. 32 The expression of Arg1 has been associated with necrosis in tuberculous granulomas, which is supported by the higher expression of this marker in porcine stages III and IV granulomas in our study, stages in which a necrotic core prevails. In this sense, Arg1 deletion has been associated with a lower mycobacterial count 22 and a higher expression of iNOS. 14 These results are in line with ours, which showed an increase in the number of Arg1+ cells together with a drop in the frequency of iNOS+ cells as well as a trend to pluribacillary granulomas. 11 According to the higher expression of Arg1 in porcine than in bovine granulomas in our study, further studies are warranted to determine the role of these molecules in the immunopathogenesis of TB in both species.
The expression of CD163+ did not display differences along the granuloma stages in both species; however, the expression was higher in cattle than in pigs and slightly higher in more advanced stages of granulomas.19,23 Others have observed that granulomas with a necrotic core have a high percentage of CD163+ cells close to the periphery of the granuloma in humans and macaques, as well as in the surrounding fibrous capsule, 26 similar to our findings in advanced stages of granulomas in cattle and pigs. These findings suggest a tendency toward an M2 profile in cattle and pigs, particularly in end-stage granulomas. In this line, although a pro-inflammatory status might be beneficial to control the development of the granuloma, once established, the anti-inflammatory response could act to control tissue damage generated by the granuloma itself.
Macrophage polarization may also be affected by previous exposure of the host to a given pathogen, such as mycobacteria, 37 adding more complexity to the dichotomy among classical and alternative macrophage activation. Furthermore, for an analysis of M1 and M2 markers, it is crucial to identify which markers are universally applicable across different species and which ones might exhibit variations depending on factors like the species or age of the organisms under study, or the specific stimuli being considered.
To summarize, this study shows the location and expression of several lineage and M1/M2 macrophage polarization markers in tuberculous granulomas from cattle and pigs naturally infected with MTC. Making a clear distinction between polarization specific markers is crucial to developing a more nuanced understanding of macrophage behavior across different contexts. Our results in cattle are in agreement with those previously reported in humans 2 and indicate that M1 macrophage polarization prevails during early granuloma stages (I and II), whereas an M2 phenotype is also observed in later stages. Contrary, and mainly due to the expression of Arg1, M2 macrophage polarization is predominant in pigs. These results are supported and represented in our study by the ratios iNOS/Arg1 and M1/M2 macrophage polarization markers.
Footnotes
Acknowledgements
We express our appreciation to Alberto Alcántara and Marta Ordóñez-Martínez for their technical assistance and the technical support offered by the Animal Health and Production Laboratory of Córdoba (Spain).
Author Contributions
JGL and LC conceived, designed, and performed the project. JSC, IRT, and FL-M helped in the sample collection. FL-M, MG-H, and JSC performed the laboratory experiments. JD and JGL contributed to the reagents, materials, and analysis tools. FL-M wrote the manuscript with contribution from the other authors. IR-T, IMRG, FJP, and JGL reviewed the manuscript. All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research project “New measures and techniques to control Bovine Tuberculosis in Andalusia” (Financial support for Operational Groups of the European Innovation Partnership for Agricultural productivity and Sustainability, EIP-AGRI) (GOP2I-CO-16-0010). FL-M was supported by a doctoral grant from ANID (National Research and Development Agency)/Doctoral grant Chile/2019/72200324.
