Abstract
A 14-year-old Thoroughbred gelding with a history of acute onset of hematuria was presented for necropsy. Transitional cell carcinoma of the urinary bladder with intra-abdominal dissemination was diagnosed. Tumor masses were observed on the splenic capsule and surrounding the distal abdominal aorta. Tumor cells showed diffuse cytoplasmic reactivity for cytokeratin but were negative for epithelial membrane antigen, carcinoembryonic antigen, tumor-associated glycoprotein 72, and vimentin.
Primary neoplasms of the equine urinary bladder are uncommon. 2 10 Previous reports included descriptions of squamous cell carcinoma, 2 transitional cell carcinoma, 2 11 12 fibromatous polyps, 2 6 rhabdomyosarcoma, 12 and lymphosarcoma. 9 Squamous cell carcinoma is reported to be the most common primary urinary bladder neoplasm in horses. 2 10 Neoplasia of the urinary bladder is one differential diagnosis for hematuria in the horse, which occurs because of ulceration of overlying mucosal epithelium. 2 10 Prognosis is poor because of metastasis or, more commonly, direct extension into surrounding tissues and the abdominal cavity. 2 5 Metastasis occurs via lymphatic vessels. 5 Pyelonephritis and/or hydronephrosis are frequent secondary complications. 2
A 14-year-old Thoroughbred gelding was presented to the Livestock Disease Diagnostic Center, Department of Veterinary Science, University of Kentucky, for diagnostic examination. The horse had a history of acute onset of massive hematuria. Ultrasound had revealed a large mass in the urinary bladder. At necropsy, a 16-cm-diameter solid to loose mass of pale tan tissue was observed in the left ventral aspect of the mucosa of the urinary bladder (Fig. 1). The mass had an irregular surface. A pale tan, irregularly shaped mass 16 × 8 × 6 cm was observed surrounding the distal abdominal aorta and the origins of the left external and internal iliac arteries. A similar 4.5-cm-diameter mass was observed on the posterior surface of the base of the spleen. Samples of the tumor masses and of routine tissues were fixed in 10% neutral buffered formalin and embedded in paraffin. Sections were stained with hematoxylin and eosin (HE). Sections from urinary bladder and splenic masses were immunohistochemically analyzed. Antibodies to cytokeratin, epithelial membrane antigen (EMA), carcinoembryonic antigen (CEA), tumor-associated glycoprotein 72 (TAG-72), and vimentin were used to localize antigens in histopathologic sections from urinary bladder and splenic masses by an avidin–biotin complex immunoperoxidase method (ABC vector kit, Vector Laboratories, Burlingame, CA).
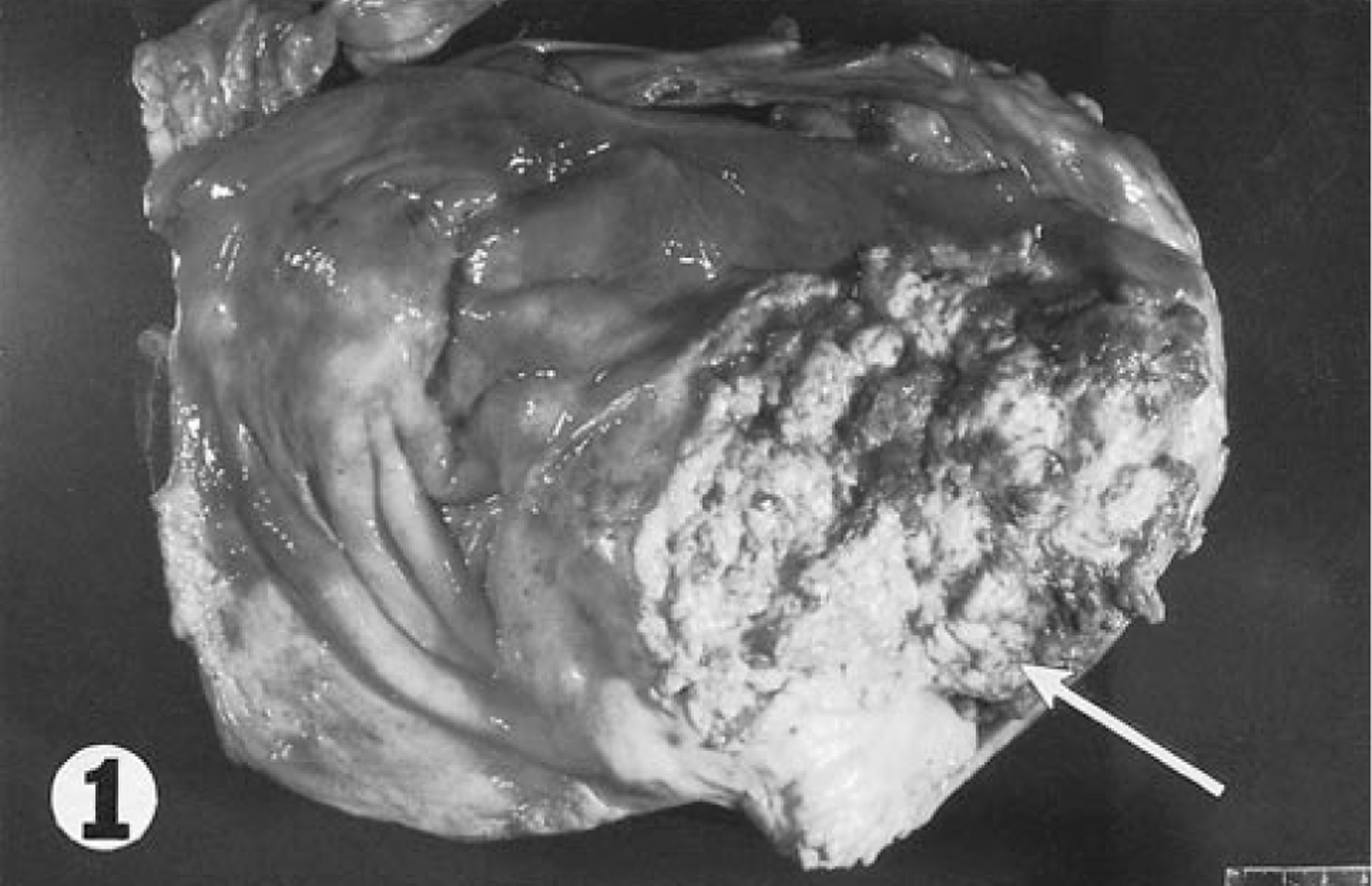
Urinary bladder; horse. Pale tumor tissue with an irregular surface is present in the ventral mucosa (arrow).
Histologically, the mucosa of the urinary bladder was markedly expanded by a moderately cellular, poorly demarcated exophytic neoplastic mass. Tumor cells were arranged in lobules and cords (Fig. 2) and in some regions were lining papillary projections of branching fibrovascular stroma. Broad bands of stromal tissue subdivided the mass into poorly defined lobules. Clusters of tumor cells were invading the papillary stromal projections in some foci. Tumor cells were cuboidal to polygonal, with small amounts of eosinophilic cytoplasm, and had moderately well-defined margins. Small clusters of tumor cells had finely fibrillar, more intensely eosinophilic cytoplasm, consistent with squamous metaplasia. Nuclei were central and round to ovoid, with finely granular, often marginated chromatin, and one to several prominent, central, deeply eosinophilic nucleoli. There was moderate anisokaryosis and two to six mitotic figures per 400× field. Small numbers of scattered binucleate and multinucleate tumor cells were observed. Tumor cells were invading deeply into the submucosa and multifocally into inner regions of the muscularis. In some foci, the submucosa and interstitium of the muscularis underlying the tumor mass were markedly expanded by collagenous fibrovascular tissue. Large clusters of tumor cells were observed distending scattered lymphatic vessels within this fibrovascular tissue (Fig. 3). Multifocal, mild hemorrhage was observed in superficial regions of the mass. There were multifocal, small to medium-size regions of necrosis of tumor cells, with infiltration of small numbers of neutrophils. A similar neoplastic mass was observed attached to the capsular surface in sections of spleen.
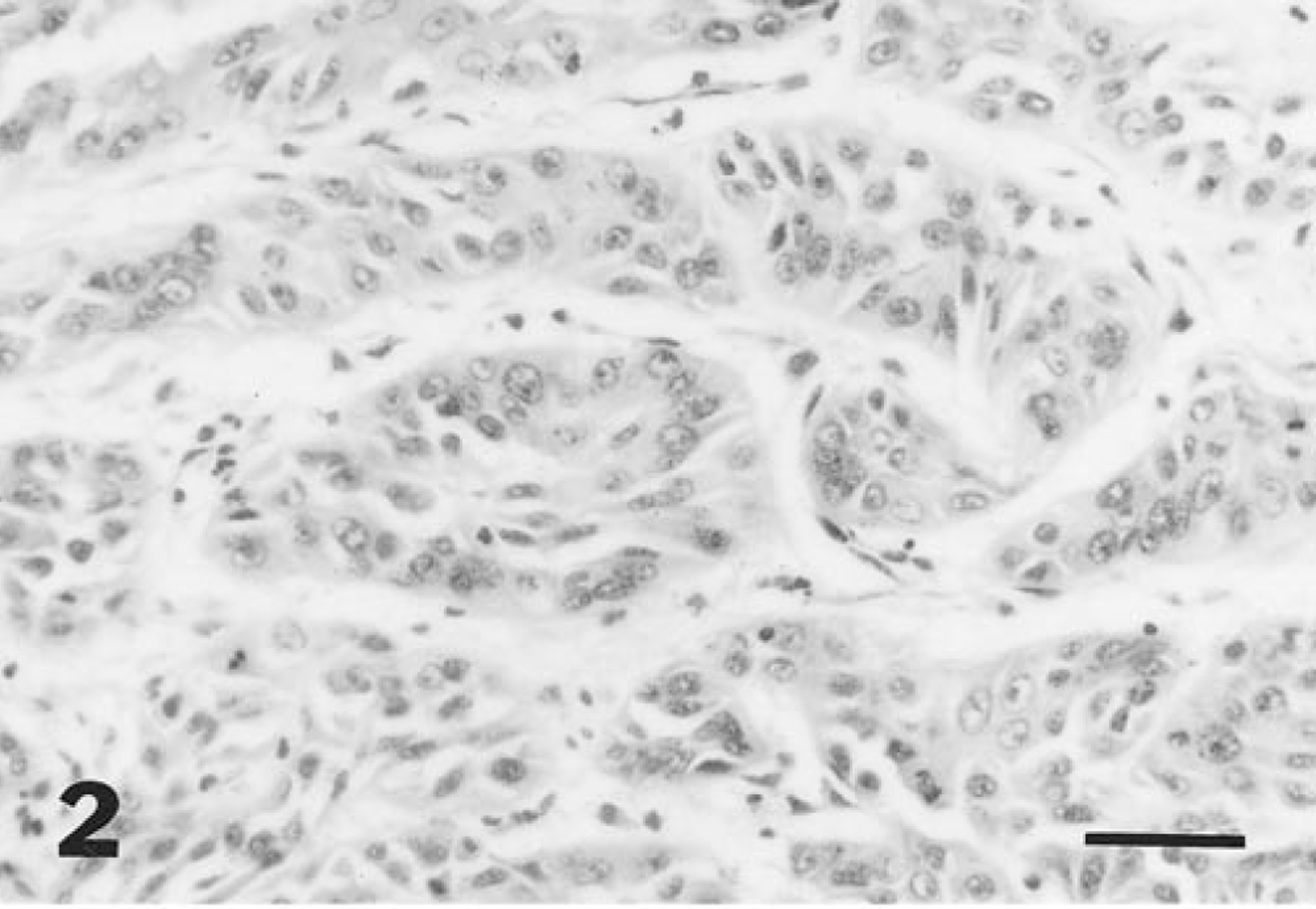
Urinary bladder tumor; horse. Neoplastic cells lining acini are cuboidal to polygonal. HE. Bar = 50 µm.
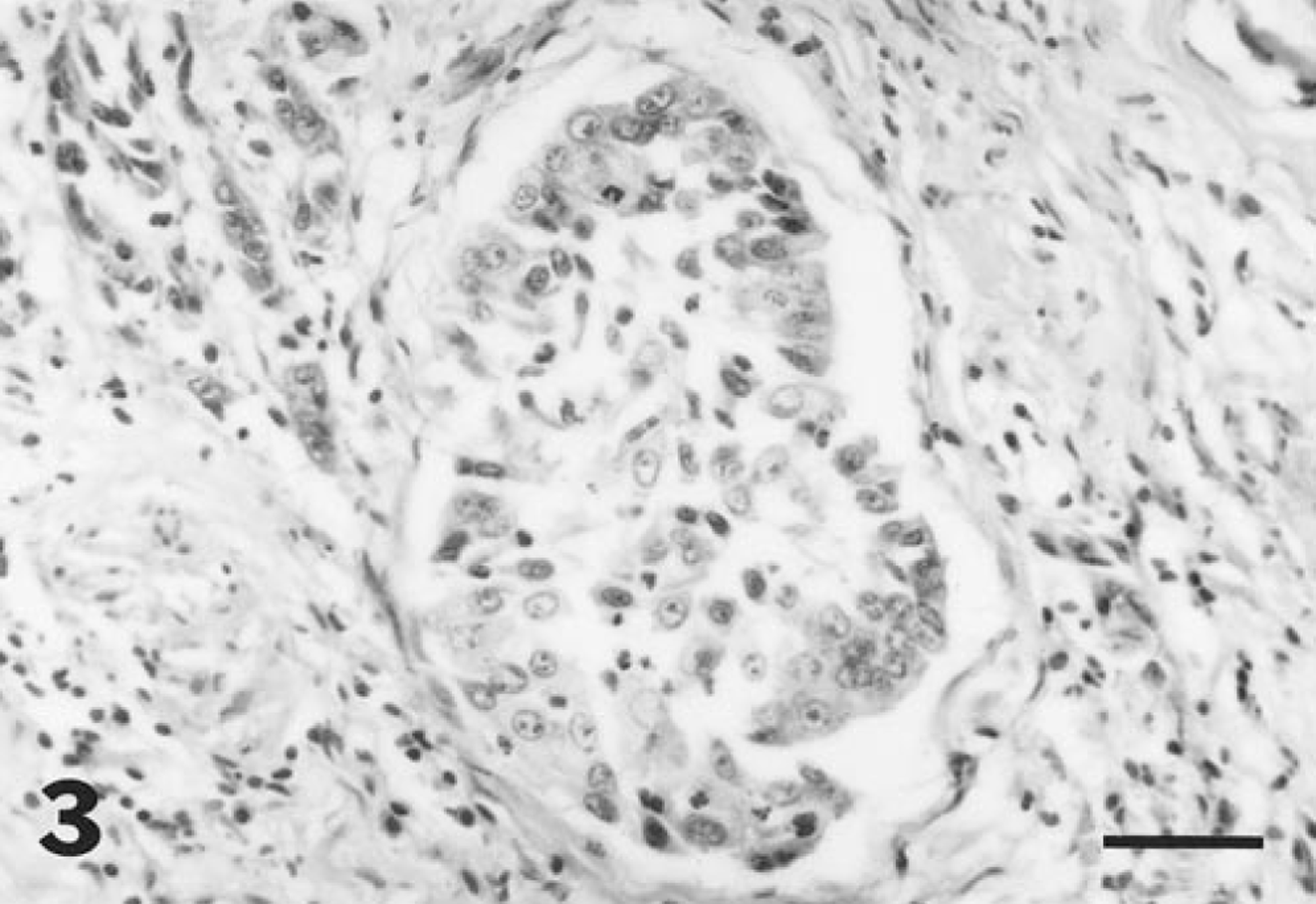
Urinary bladder; horse. A large cluster of neoplastic cells is distending a submucosal lymphatic vessel. HE. Bar = 50 µm.
Diffuse positive granular staining for cytokeratin antigen was present within the cytoplasm of tumor cells of urinary bladder (Fig. 4) and splenic masses, including tumor cells observed within lymphatic vessels of the urinary bladder. Immunohistochemical staining of tumor cells for EMA, CEA, TAG-72, and vimentin was negative.
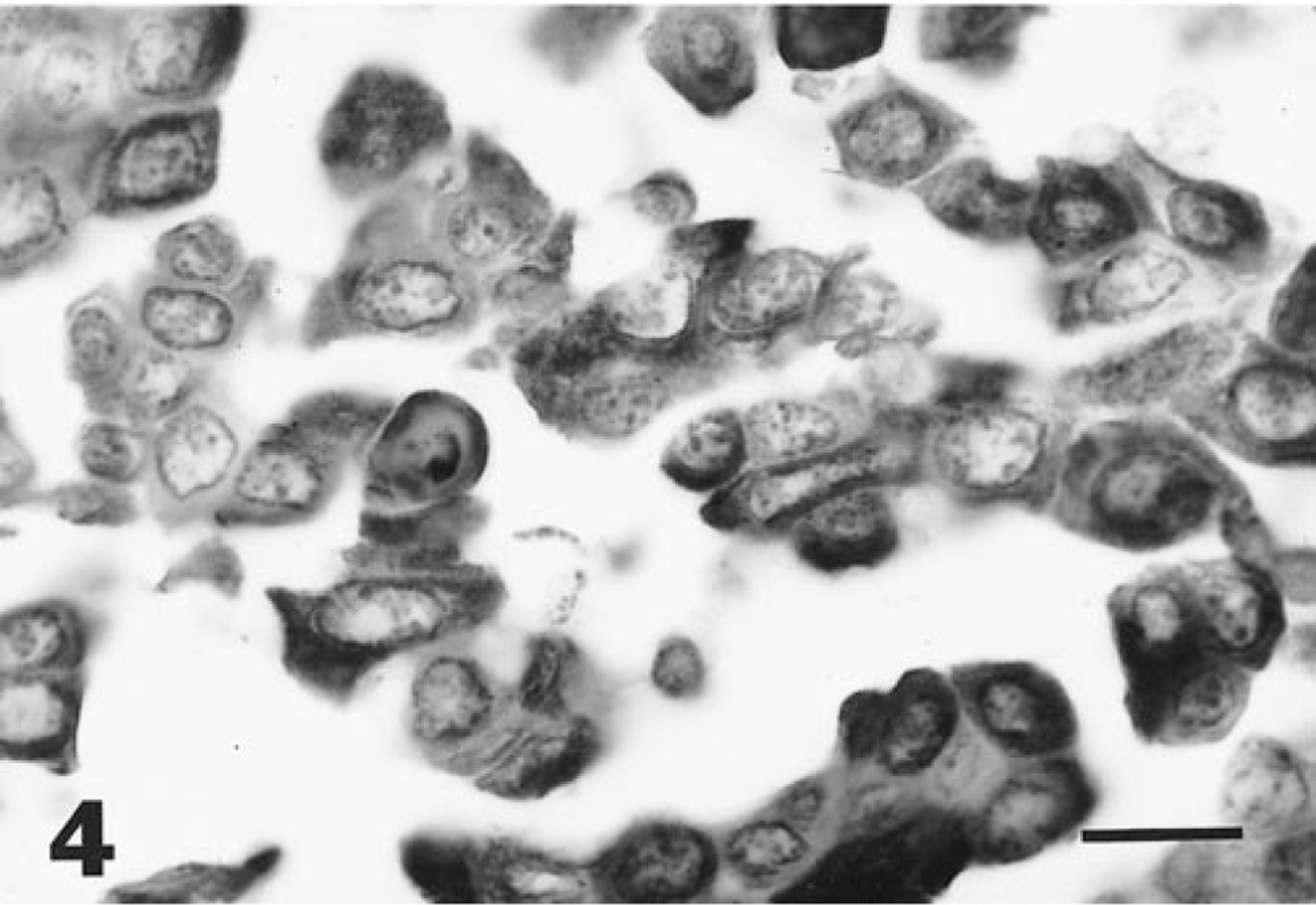
Urinary bladder tumor; horse. Neoplastic cells show diffuse, positive granular staining for cytokeratin protein. ABC immunoperoxidase method. Bar = 20 µm.
Carcinomas of the urinary bladder are classified as transitional cell carcinomas, squamous cell carcinomas, adenocarcinomas, or undifferentiated carcinomas. 5 In most species, the most common primary urinary bladder tumor is the transitional cell carcinoma. 5 In the horse, squamous cell carcinomas are more common. 10 Small clusters of squamous epithelial cells occur in the normal epithelium of the equine urinary bladder. It has been postulated that squamous cell carcinomas arise from these cells rather than from metaplastic transitional epithelial cells. 10
Transitional cell carcinomas may be classified as papillary or nonpapillary and as noninfiltrating or infiltrating. 5 Noninfiltrating, nonpapillary tumors do not extend beyond the normal mucosa. This tumor was papillary and infiltrating, with invasion of neoplastic epithelial cells into stroma of the papillary stalks and into subjacent submucosal and muscular layers. Urinary bladder carcinomas in humans are classified as stage A–D on the basis of invasion: A = into mucosa and submucosa; B = into muscularis; C = into perivesicular fat or peritoneum; D = distant metastasis has occurred. The gross appearance of this tumor was consistent with a stage C neoplasm, based on the presence of masses surrounding the aorta and attached to the capsular surface of the spleen. Clusters of tumor cells were observed within lymphatic vessels in sections of urinary bladder; however, distant metastatic masses were not detected. One prior report documented widespread intra-abdominal dissemination of a transitional cell carcinoma in a Percheron mare. 11 Another report of transitional cell carcinoma of the urinary bladder in a Anglo-Norman stallion documented tumor masses surrounding and compressing the aorta and iliac arteries, with other masses extending into the retroperitoneum and spinal canal. 12 Metastasis to the lung was also noted in that stallion. In this thoroughbred gelding, tumor masses surrounded the distal aorta and origins of the iliac arteries, but compression of the vessels was not noted.
Ingestion of bracken fern (Pteridium aquilinum) has been associated with the occurrence of urinary bladder neoplasms in cattle and sheep, with possible involvement of bovine papilloma virus type 2. 10 The relationship between bracken fern ingestion and occurrence of urinary bladder tumors in horses is less clear; however, involvement of an environmental carcinogen is less likely in sporadic cases such as that in this gelding. It is not known if this horse had a history of exposure to bracken fern.
Tumor cells in this horse showed diffuse cytoplasmic reactivity for antibody to cytokeratin. Detection of cytokeratin in transitional carcinomas of the human urinary bladder has been reported in >80% of cases. 8 In normal human urothelium and well-differentiated transitional cell carcinomas, cytokeratin reactivity is restricted to basal cells. 7 The pattern of basal cell staining for cytokeratin is lost in less well-differentiated tumors. 7
In one study of human transitional cell bladder carcinomas, EMA was detected in 62% of tumors, and CEA was detected in 70%. 4 The EMA and CEA content of tumor tissue has been correlated positively with grade and stage of transitional cell bladder carcinomas in humans. 3 4 Intra-abdominal dissemination of tumor cells in this horse was consistent with stage 3; however, tumor cells were diffusely negative for CEA and EMA. Carcinoembryonic and epithelial membrane antigens may not be correlated with stage or grade in transitional cell carcinomas of the equine urinary bladder. Another possibility is that the antibodies used do not cross-react with equine antigens.
TAG-72 is a mucinlike antigen that has been detected in numerous types of human carcinomas. In one study of canine transitional cell carcinomas of the urinary bladder, 53% of tumors were positive for TAG-72, using monoclonal antibody B72.3. 1 Tumor cells in this gelding were negative for TAG-72, which may not be a useful marker for transitional cell carcinoma in the horse.
Transitional cell carcinoma is an uncommon differential diagnosis for hematuria in the horse. Intra-abdominal dissemination of tumor cells has been reported on rare occasions, with one previous report of a tumor mass surrounding the abdominal aorta. Cytokeratin appears to be a useful marker for equine transitional cell carcinoma cells. Determination of the usefulness of EMA, CEA, and TAG-72 as markers for transitional cell carcinomas of the urinary bladder in horses will require analysis of further cases.
Footnotes
Acknowledgements
We thank P. Cross (Chandler Medical Center, University of Kentucky) for performing the immunohistochemical staining. This work is published as College of Agriculture, University of Kentucky Publication No. 99-14-177.
