Abstract
The expression of immunohistochemical markers that have been used in diagnosis and/or prognostication of urothelial tumors in humans (uroplakin III [UPIII], cytokeratin 7 [CK7], cyclooxygenase-2 [COX-2], and activated caspase 3) was evaluated in a series of 99 canine proliferative urothelial lesions of the urinary bladder and compared to the lesion classification and grade as defined by the World Health Organization / International Society of Urologic Pathology consensus system. There were significant associations between tumor classification and overall UPIII pattern (P = 1.49 × 10–18), loss of UPIII (P = 1.27 × 10–4), overall CK7 pattern (P = 4.34 × 10–18), and COX-2 pattern (P = 8.12 × 10–25). In addition, there were significant associations between depth of neoplastic cell infiltration into the urinary bladder wall and overall UPIII pattern (P = 1.54 × 10–14), loss of UPIII (P = 2.07 × 10–4), overall CK7 pattern (P = 1.17 × 10–13), loss of CK7 expression (P = .0485), and COX-2 pattern (P = 8.23 × 10–21). There were no significant associations between tumor classification or infiltration and caspase 3 expression pattern.
Keywords
Proliferative lesions of the urothelium of the canine urinary bladder range from benign polyps and papillomas to carcinomas with varying metastatic potential. Also, inflammatory conditions such as polypoid cystitis can form tumor-like masses within the urinary bladder that can be confused with urothelial neoplasms. Classification of urothelial proliferative lesions and histologic grading of urothelial carcinomas is key to accurate prognostication and treatment selection. A classification and grading scheme based on the 1986 World Health Organization (WHO) classification scheme for human urinary bladder and urethral cancer was proposed in 1995 for use in evaluating urothelial neoplasms in dogs based on pattern of growth, nuclear atypia, and degree of infiltration into the urinary bladder wall. 50 In the initial description of this system’s use in dogs, significant correlations were found between tumor grade and depth of infiltration, the presence of metastases, and survival time. However, a subsequent smaller study failed to find a significant correlation between grade and prognosis. 41 To our knowledge, this scheme is not widely used in dogs and has been replaced in human medicine by more modern classification schemes. There remains a need for a clear, reproducible, and well-accepted classification and grading scheme for urothelial proliferative lesions in dogs.
Currently, the most widely accepted scheme for classification and grading of proliferative urothelial lesions in humans is the WHO / International Society of Urologic Pathology (ISUP) consensus classification system, published in 1998 and updated in 2004. 6,7 In multiple studies, the WHO/ISUP consensus classification system has been demonstrated to be significantly associated with clinical outcome. 2,32 –34,37,42,43,51,59 It has long been known that canine urothelial neoplasms are similar to urothelial neoplasms in humans in terms of morphology, biological behavior, and response to chemotherapy. 14,18,19,27,28,59 Patrick et al recently examined the potential use of the WHO/ISUP consensus classification system in classifying canine proliferative urothelial lesions. 36 In that study, the authors demonstrated that the histomorphology of proliferative urothelial lesions was homologous between dogs and humans and that canine lesions could easily be classified according to the system; however, data regarding the prognostic relevance of classification system remain lacking. Additional study is needed of the biological differences between classifications and grades of proliferative urothelial lesions in dogs.
Uroplakins compose a group of membrane-associated proteins expressed by urothelial cells that are important for cell-to-cell adhesion and maintenance of water impermeability. 57,58 These proteins form a plaque-like complex along the apical membrane of the umbrella cells that form the most superficial layer of the urothelium. 57 Cytokeratin 7 (CK7) is a cytokeratin expressed by simple epithelium as well as differentiated urothelial cells and a variety of carcinomas in humans, including those of urothelial, pancreatic, cholangiolar, and ovarian origin. 49,52 Uroplakin III (UPIII) and CK7 are used in dogs and humans as diagnostic markers of urothelial differentiation in primary tumors and metastases. 8,13,31,35,39,47 In humans, loss of UPIII has been associated with several prognostic features. 13,25
The inducible enzyme cyclooxygenase-2 (COX-2) and the resulting production of prostaglandin E2 have been ascribed significant roles in carcinogenesis, including immunosuppression, inhibition of apoptosis, increased metastatic potential of neoplastic epithelial cells, promotion of drug resistance, and stimulation of angiogenesis. 9,11,23,54 –56 Numerous studies have shown significant correlations between COX-2 expression and tumor grade, infiltration, metastasis, and survival. 21,24,30,40,44 –46,53 Concordantly, a decreased risk for urinary bladder cancer development has been seen in humans undergoing long-term nonsteroidal anti-inflammatory therapy, and in vitro and in vivo studies have suggested potential use of COX-2 inhibitors in treatment. 3,5,10,29,53 In humans and dogs, COX-2 is not expressed by normal urothelium of the urinary bladder. 17,24 Substantial expression of COX-2, however, has been observed in transitional cell carcinomas. 17,20,22
Caspase 3 is an effector or executioner caspase that is activated by intrinsic and extrinsic apoptosis signaling pathways to cleave multiple cellular structural and repair proteins. 4,12,38 Due to the fact that this protein is activated late in the apoptotic pathway, immunohistochemical detection of activated caspase 3 has been used to evaluate apoptotic rate. 1,48 In human urinary bladder cancers, expression of caspase 3 has been suggested to have prognostic significance. 15,16,26
The goals of the current study were twofold: first, to evaluate the expression of UPIII, CK7, COX-2, and caspase 3 in nonneoplastic and neoplastic proliferative lesions of the canine urothelium of the urinary bladder; second, to correlate the observed patterns of expression of each of these markers with specific lesion classification and grade as defined by the WHO/ISUP consensus classification system.
Materials and Methods
Selection of Cases and Histologic Classification
A series of 99 formalin-fixed, paraffin-embedded proliferative urothelial lesions from 99 dogs that had been submitted as diagnostic cases were selected from the tissue archives of the Diagnostic Center for Population and Animal Health of Michigan State University. Of these, 93 had previously been classified and, where applicable, graded with the WHO/ISUP consensus classification system. 36 Six additional diagnostic cases that presented to the center were included in the study set, including additional low-grade neoplasms. For grading of these additional samples, 5-μm sections of all samples were routinely processed and stained with hematoxylin and eosin for microscopic examination. Proliferative urothelial lesions were categorized according to the WHO/ISUP consensus classification system as previously described and summarized in Table 1. 36 For urothelial carcinomas, the degree of infiltration into the urinary bladder wall was scored as no infiltration, infiltration into the substantia propria, or infiltration into the tunica muscularis.
Histologic Features of Proliferative Urothelial Lesions According to the World Health Organization / International Society of Urologic Pathology Consensus Classification System.a

aAdapted from descriptions made by Patrick et al. 36
bPapillary urothelial neoplasm of low malignant potential.
Of the 99 proliferative urothelial lesions examined, 44 were nonneoplastic and categorized as either urothelial polyps or polypoid cystitis. Of the 55 neoplasms, there were 2 urothelial papillomas and 1 papillary urothelial neoplasm of low malignant potential (PUNLMP). The remainders of the urothelial neoplasms were papillary urothelial carcinomas of varying grade. Low-grade (grade 1) papillary urothelial carcinomas were rare, with only 2 being included in the set. Both these had some degree of infiltration into the urinary bladder wall, with one sample having infiltrative clusters of neoplastic urothelial cells within the muscularis. Papillary carcinomas infiltrated at least into the substantia propria in 49 of 52 cases (94%) and into the muscularis in 21 of 52 (40%).
Immunohistochemistry
Sections (5 μm) of all samples were processed for immunohistochemistry and labeled with a mouse monoclonal anti-UPIII antibody (1:5, RDI, Fitzgerald Industries Intl, Concord, MA), a mouse monoclonal anti-CK7 antibody (1:75, Dako Cytomation, Carpentaria, CA), a rabbit polyclonal anti-COX-2 antibody (1:100, Cayman Chemical Company, Ann Arbor, MI), or rabbit polyclonal anti-activated caspase 3 antibody (1:5000, RDI). Deparaffinization, antigen retrieval, immunohistochemical labeling with 3,3′-diaminobenzidine chromogen, and counterstaining with hematoxylin were performed on the Bond Max Automated Staining System (Vision BioSystems, Leica, Bannockburn, IL) with the Bond Polymer Detection System (Vision BioSystems). Sections of normal canine urothelium were similarly labeled as positive controls for UPIII and CK7. A canine squamous cell carcinoma known to express COX-2 and a lymph node with large numbers of cells positive for activated caspase 3 were respectively used as positive controls for these antibodies. For negative controls, homologous nonimmune sera or buffer replaced primary antibodies.
Immunoreactivity for UPIII and CK7 was scored according to overall pattern within the urothelium and partial loss of immunoreactivity (Figs. 1 –6). Immunoreactivity for UPIII and CK7 in positively labeled cells was variably perimembranous, predominately cytoplasmic without distinct perimembrane labeling, or associated with both the membrane the cell membrane and the cytoplasm. Specifically, for overall pattern of UPIII and CK7 immunoreactivity within the urothelium, pattern 1 was defined by labeling limited to the most superficial layer of cells (Figs. 1, 4); pattern 2 was defined by labeling extending to the middle layer of the urothelium (Fig. 2); pattern 3 was defined by cell labeling throughout the full thickness of the urothelium (Fig. 5); and pattern 4 was defined by immunoreactivity that was patchy and randomly distributed (Figs. 3, 6). Partial loss of immunoreactivity was noted when greater than 50% of epithelial cells were immunonegative and there were areas within the proliferative urothelium with no immunopositive cells in at least 2 contiguous high-power (400×) fields.
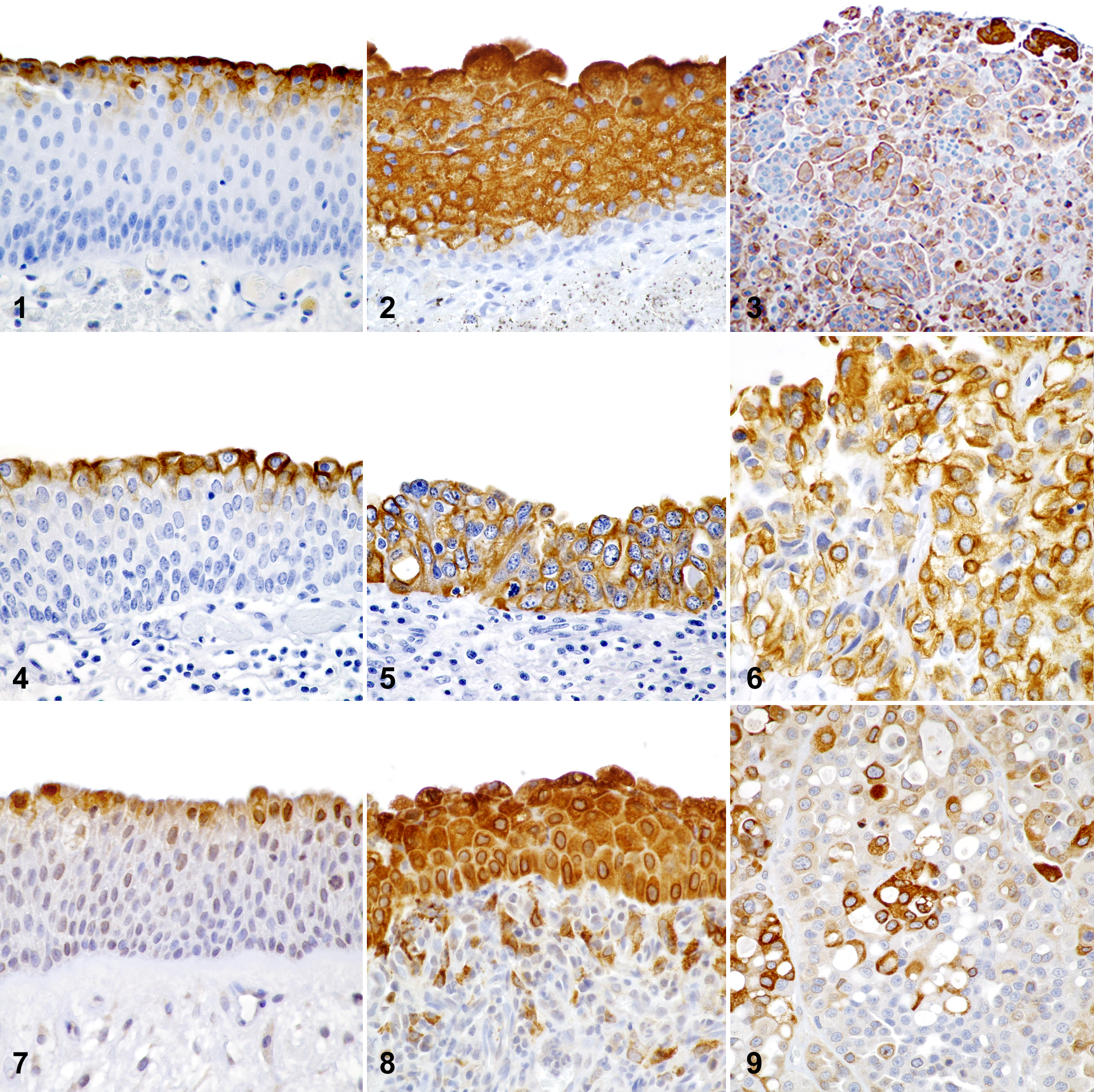
Immunoreactivity for COX-2 and activated caspase 3 was classified according to overall pattern within the urothelium. Cells immunoreactive for COX-2 had diffuse cytoplasmic to perinuclear labeling. Cells immunoreactive for activated caspase 3 had diffuse or, more often, finely granular labeling within the cytoplasm and/or nucleus. In pattern 1 for these markers, immunoreactivity was limited to the superficial (1–3) cell layers (Fig. 7). In pattern 2, cells were labeled throughout the full thickness of the urothelium (Fig. 8). In pattern 3, immunoreactivity was patchy and randomly distributed throughout the proliferative urothelium, but greater than 15% of neoplastic cells were positively labeled (Fig. 9).
For each marker, any section that completely lacked immunoreactivity was excluded from analysis, as it could not be determined whether this was true loss of expression or was artifactual.
Statistical Analysis
Statistical Analysis Software version 9.1.3 (2002, SAS Institute Inc, Cary, NC) was used for the data analysis. Fisher exact test was used to test the association between grade or degree of infiltration and the pattern of immunoreactivity for all evaluated markers and loss of immunoreactivity for UPIII and CK7. For all statistical analyses, lesions were categorized as follows: nonneoplastic lesions (urothelial polyps and polypoid cystitis), low-grade neoplasms (urothelial papillomas, PUNLMPs, and grade 1 urothelial carcinomas), grade 2 urothelial carcinomas, and grade 3 urothelial carcinomas. Significance was set at P = .05.
Results
Immunohistochemistry
UPIII and CK7. The urothelium of control urinary bladders and areas of normal urothelium in tumor samples demonstrated strong expression of UPIII and CK7 diffusely throughout the superficial layers (umbrella cells). This distribution of immunolabeling typified UPIII and CK7 pattern 1.
In the hyperplastic urothelium of polyps and polypoid cystitis, expression of UPIII and CK7 was generally limited to the superficial (umbrella) cell layers (UPIII pattern 1; CK7 pattern 1) or extended to only the midportion of the urothelium (UPIII pattern 2). Only 1 urothelial polyp had UPIII pattern 4, and none had CK7 pattern 4. The PUNLMP and urothelial papillomas demonstrated UPIII pattern 3 and CK7 pattern 2 or 3. With few exceptions, papillary carcinomas of all grades demonstrated patchy randomly distributed UPIII expression consistent with pattern 4. Partial loss of UPIII expression was not detected in any grade 1 papillary carcinoma and in only 1 grade 2 papillary carcinoma. In contrast, 14 of 33 (42%) grade 3 papillary carcinomas had partial loss of expression of UPIII. More grade 2 and 3 carcinomas had a CK7 pattern 3 than had a UPIII pattern 3 (11 of 50 CK7 pattern 3 cases compared to 3 of 50 UPIII pattern 3 cases). The majority (71%) of grade 2 and 3 urothelial carcinomas, however, had a CK7 pattern 4 similar to that observed for UPIII. Only grade 2 and 3 urothelial carcinomas had significant loss of CK7 expression (Tables 2 and 3).
Immunohistochemical Scoring of Uroplakin III Expression in Canine Proliferative Urothelial Lesions by Overall Pattern, n (%).
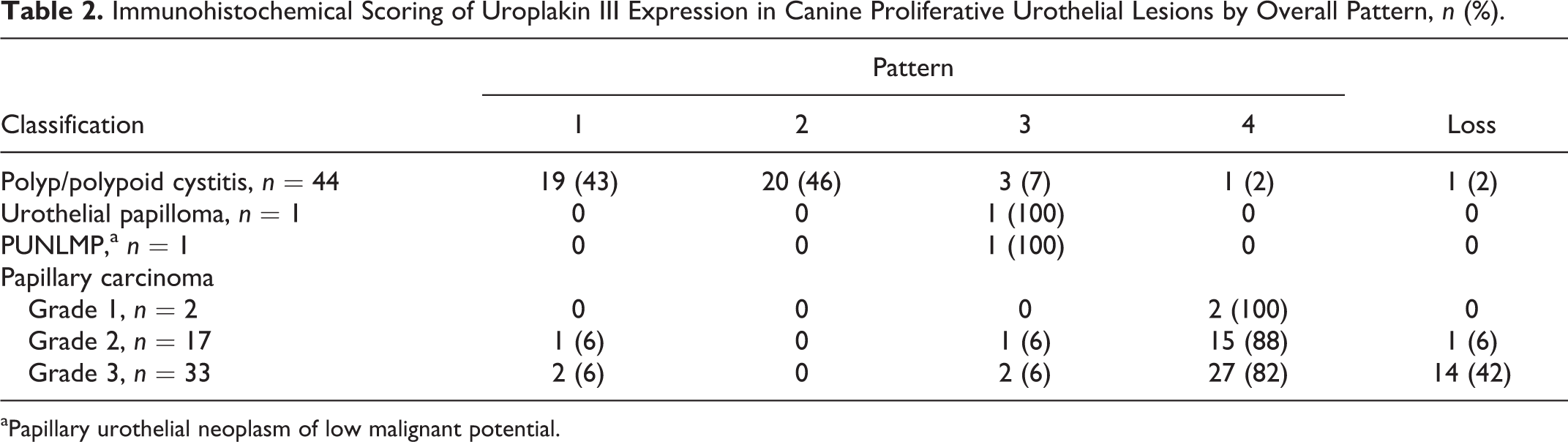
aPapillary urothelial neoplasm of low malignant potential.
Immunohistochemical Scoring of Cytokeratin 7 Expression in Canine Proliferative Urothelial Lesions by Overall Pattern, n (%).
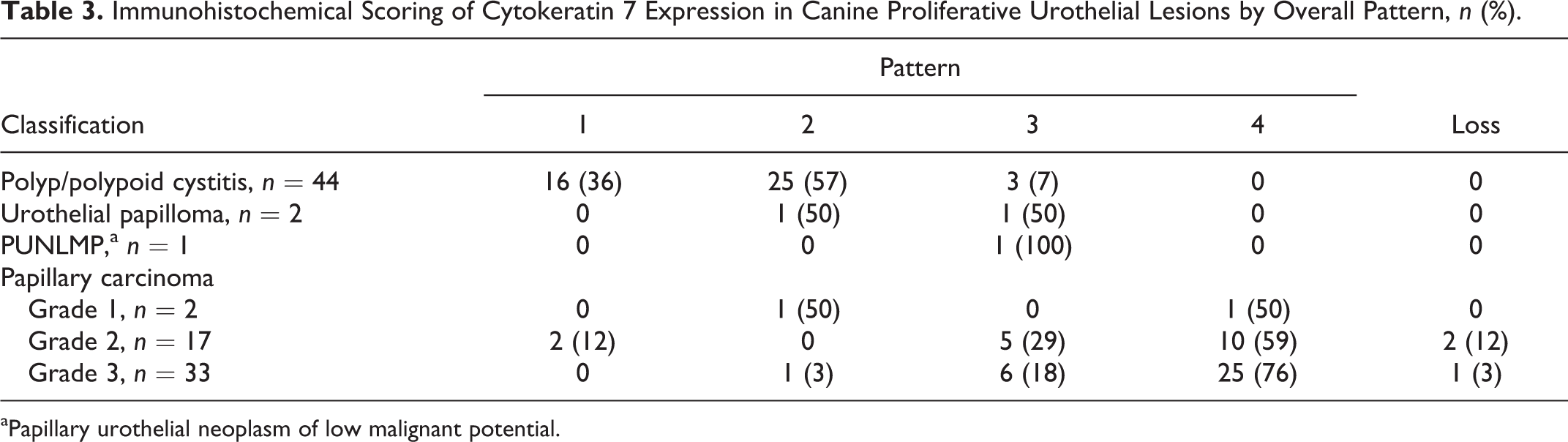
aPapillary urothelial neoplasm of low malignant potential.
COX-2. The squamous cell carcinoma from the digit of a dog used as a positive control demonstrated strong positive cytoplasmic and mainly perinuclear immunoreactivity for COX-2 in 30% of neoplastic cells. There was no positive immunoreactivity for COX-2 in any part of the normal urothelium from control urinary bladders; however, in the superficial layers of nonproliferative urothelium adjacent to proliferative lesions, there were often a small percentage (< 10%) of COX-2 positive cells.
In all proliferative lesions, at least 10% of proliferative urothelial cells exhibited positive COX-2 immunoreactivity. In urothelial polyps and polypoid cystitis, COX-2 expression in the proliferative urothelium was usually restricted to the superficial layers (COX-2 pattern 1) or was less commonly full thickness (COX-2 pattern 2). Only 1 polyp had randomly distributed and patchy expression of COX-2 (COX-2 pattern 3). The PUNLMP and the 2 papillomas demonstrated COX-2 pattern 1 while the grade 1 papillary carcinomas exhibited either pattern 2 or 3. All high-grade (grades 2 and 3) papillary urothelial carcinomas in which COX-2 expression was detected exhibited COX-2 pattern 3 (Table 4).
Immunohistochemical Scoring of Cyclooxygenase-2 Expression in Canine Proliferative Urothelial Lesions by Overall Pattern, n (%).
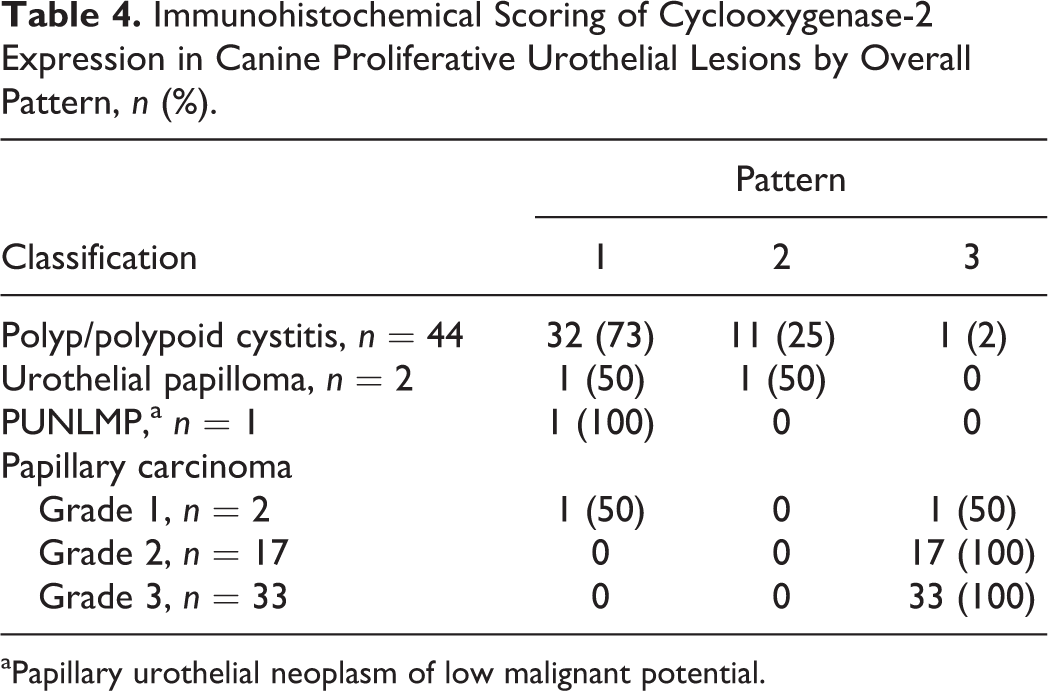
aPapillary urothelial neoplasm of low malignant potential.
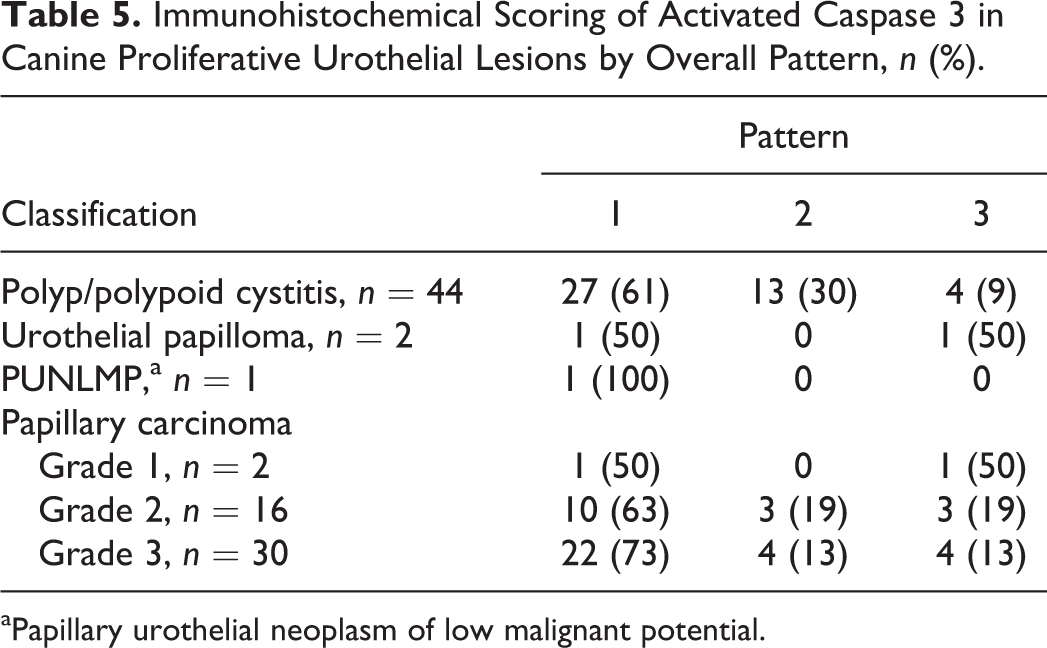
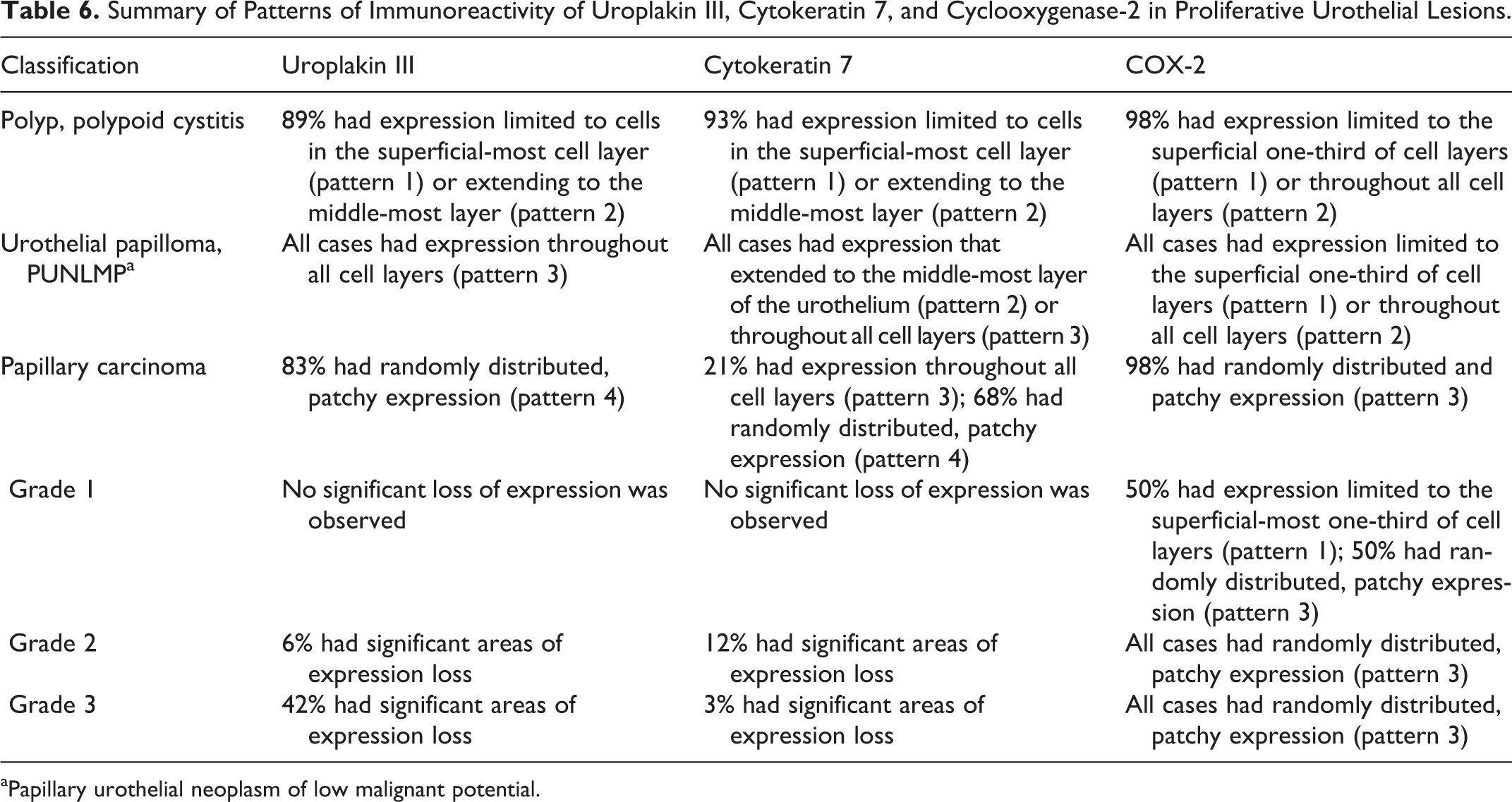
Activated caspase 3. Approximately 10% of cells in the lymph node used as a positive control for activated caspase 3 had positive finely granular labeling of the cytoplasm and/or nucleus. In the normal urothelium of control urinary bladders and adjacent to proliferative lesions, immunoreactive cells composed less than 10% of the total urothelium and were largely limited to the superficial (1–2) cell layers. The total percentage of caspase 3–immunoreactive cells ranged from 10% to 40% in all other proliferative lesions; however, there was no appreciable variation in the percentage of positive cells among tumor types. Immunoreactivity for activated caspase 3 was most commonly noted in the superficial-most cell layers of all proliferative lesions (caspase 3 pattern 1) but was also seen diffusely throughout the urothelium (caspase 3 pattern 2) and in a randomly distributed patchy distribution (caspase 3 pattern 3) (Table 5). A summary of the predominant patterns of immunoreactivity for UPIII, CK7, and COX-2 observed in each proliferative urothelial lesion classification is presented in Table 6.
Immunohistochemical Scoring of Activated Caspase 3 in Canine Proliferative Urothelial Lesions by Overall Pattern, n (%).
aPapillary urothelial neoplasm of low malignant potential.
Summary of Patterns of Immunoreactivity of Uroplakin III, Cytokeratin 7, and Cyclooxygenase-2 in Proliferative Urothelial Lesions.
aPapillary urothelial neoplasm of low malignant potential.
Statistical Analysis
Based on Fisher exact test, there were significant associations between tumor classification and overall UPIII pattern (P = 1.49 × 10–18), loss of UPIII (P = 1.27 × 10–4), overall CK7 pattern (P = 4.34 × 10–18), and COX-2 pattern (P = 8.12 × 10–25). Also by Fisher exact test, there were significant associations between depth of neoplastic cell infiltration and overall UPIII pattern (P = 1.54 × 10–14), loss of UPIII (P = 2.07 × 10–4), overall CK7 pattern (P = 1.17 × 10–13), loss of CK7 expression (P = .0485), and COX-2 pattern (P = 8.23 × 10–21). There were no significant associations between tumor classification and loss of CK7 or caspase 3 pattern. There were also no significant associations between depth of infiltration and caspase 3 expression.
Discussion
In the current study, strong differences in expression of UPIII, CK7, and COX-2 were observed between urothelial polyps and cases of polypoid cystitis, urothelial papillomas and PUNLMPs, and papillary carcinomas. However, no association was observed between the expression of activated caspase 3 and tumor classification, grade, or depth of infiltration into the urinary bladder wall.
Overall, urothelial polyps and cases of polypoid cystitis predominately had expression of UPIII and CK7 that either was limited to the superficial-most cell layers (UPIII/CK7 pattern 1) or extended to the middle-most cell layer (UPIII/CK7 pattern 2). Urothelial papillomas and a PUNLMP had UPIII and CK7 expression throughout the full thickness of the urothelium or extending at least up to the middle-most cell layer. The majority of papillary carcinomas, in contrast, had randomly distributed patchy immunoreactivity for UPIII and CK7 (UPIII/CK7 pattern 4). In addition, there was often partial loss of UPIII and CK7 expression in high-grade carcinomas that invaded into the urinary bladder wall.
Based on the association with infiltration into the urinary bladder wall, loss of UPIII and CK7 in urothelial carcinomas may suggest a lack of differentiation or epithelial-mesenchymal transition in that favors infiltration. In humans, loss of UPIII expression is frequently seen in metastases of high-grade tumors and has been associated with lymphovascular infiltration, stage, and grade. 13,25 Loss of cell membrane adhesion molecules may also result in an increased propensity for metastasis in dogs; however, information on clinical outcome including presence of metastasis and survival was not available for the examined canine tumor set. The observed significant loss of UPIII and CK7 in many high-grade carcinomas suggests that when used as diagnostic markers on small specimens, some urothelial carcinomas might not be positively labeled.
There was a clear difference in the location of COX-2-positive cells within the urothelium between nonneoplastic and neoplastic lesions as well as among papillary urothelial carcinomas with different degrees of infiltration into the urinary bladder wall. Urothelial polyps, cases of polypoid cystitis, urothelial papillomas, and the one PUNLMP exhibited expression of COX-2 in the superficial layers of the urothelium (COX-2 pattern 1) or, rarely, diffusely throughout the urothelium (COX-2 pattern 2). In contrast, papillary carcinomas had a randomly distributed, patchy pattern of COX-2 expression (COX-2 pattern 3).
Previous studies of COX-2 in canine urinary bladder urothelium have focused on the difference in COX-2 expression between normal urinary bladder and urothelial carcinomas and reported differences as a percentage of COX-2 positive cells. In the current study, COX-2 immunoreactivity was observed in the majority of nonneoplastic and neoplastic lesions, often in a relatively high percentage of cells, precluding the use of percentage of positive cells as a differentiating criterion. In one previous immunohistochemical study, only 30 of 52 (58%) canine transitional cell carcinomas demonstrated COX-2 expression by IHC. 22 In a study of 18 canine transitional cell carcinomas, all tumors expressed COX-2, and 3 showed more than 30% of cells to be positive for COX-2. 20 The reasons for discrepancy between these studies in terms of the numbers of cases and the number of cells within a given case that are positively labeled for COX-2 may be due to the low number of cases examined in each study, differing methods of fixation and processing of the tissues, differences in immunohistochemical methods including the anti-COX-2 antibody used, or interobserver variation.
To our knowledge, COX-2 expression has not been previously evaluated in nonneoplastic proliferative urothelial lesions of the urinary bladder in dogs. In contrast to normal urothelium from urinary bladders used as controls, all urothelial polyps and cases of polypoid cystitis had expression of COX-2. Urothelial polyps and polypoid cystitis are often associated with mucosal irritation such as what might occur with cystoliths. Expression of COX-2—often throughout the superficial portion or the full thickness of the proliferative urothelium of nonneoplastic lesions, as observed in the current study—might represent a consequence of such surface irritation.
Overall, there were distinct differences in the patterns of UPIII, CK7, and COX-2 expression in canine urothelial proliferative lesions of the urinary bladder as defined and categorized by the WHO/ISUP consensus classification system. As the cases included in the study set comprised diagnostic samples, reliable follow-up data regarding clinical outcome were not available. Definitive prospective studies of the clinical outcome based on these markers and the WHO/ISUP consensus classification system in dogs remain lacking, but this study encourages the continued prognostic evaluation of the WHO/ISUP consensus classification system and these immunohistochemical markers in canine proliferative urothelial lesions.
Footnotes
Acknowledgements
The work described in this article composed a portion of Dr Sledge’s PhD graduate program, for which a fellowship was provided from Bristol-Meyers Squibb through the American College of Veterinary Pathologists / Society of Toxicological Pathologists Coalition.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
