Abstract
A 2-week-old crossbred male piglet with numerous congenital, variably sized macules, plaques, and papules distributed all over the body was submitted for necropsy. Significant gross and histological lesions were restricted to the skin. On light microscopic examination, these cutaneous lesions corresponded to dermal and/or subcutaneous masses composed of spindle-shaped to round cells that multifocally contained hemosiderin; epidermotropism was not observed. Immunohistochemically, the neoplastic cells were strongly positive for CD204; moderately positive for CD163, lysozyme, and vimentin; and negative for Mac 387, α-1-antitrypsin, S-100 protein and E-cadherin; frozen tissues were not available for CD1a and CD11c. Transmission electron microscopic examination of sections from formalin-fixed tissues did not reveal Birbeck’s granules. The clinical, morphological, and immunohistochemical results were consistent with a congenital cutaneous histiocytosis of non-Langerhans cell origin. The condition most resembled juvenile xanthogranuloma in humans, a generally skin-limited non-Langerhans histiocytic disorder that can be congenital. Cutaneous and/or systemic histiocytic disorders are well characterized in dogs and have been described in cats, and a case with some similarities to ours has been reported in a neonatal piglet, but this is to our knowledge the first immunohistochemically supported report of histiocytosis in the pig and congenital histiocytosis in animals.
Histiocytoses are a clinically and pathologically heterogeneous group of proliferative diseases of macrophages and dendritic cells (DCs) described in dogs, cats, and humans; most involve the skin, alone or along with other organs. 1,7,11 Cutaneous DCs consist of the epidermis-associated Langerhans cell (LC) and the interstitial (IDC) or dermal dendritic cell (DDC), derived from a CD34+ bone marrow precursor (myeloid DCs). 1,12 These histiocytic disorders range from benign, often self-limiting proliferations to malignant neoplasms; 1,7,11,12 their etiopathogenesis is poorly to incompletely understood. In veterinary medicine, they are common and well characterized in dogs and uncommon in cats. 1,7 In dogs, there are 3 major entities: (1) canine cutaneous histiocytoma (CCH), a generally benign neoplasm of LCs; (2) reactive histiocytosis (RH), a cutaneous or systemic nonneoplastic proliferation of IDCs and T lymphocytes; and (3) histiocytic sarcoma (HS), a malignant neoplasm of IDCs or, in its rare hemophagocytic form, of macrophages. 7 In cats, feline progressive histiocytosis, a cutaneous and eventually metastatic HS, is the best described condition; pulmonary LC histiocytosis has been reported in 3 cats. 1 In dogs and cats, DC lineage is established mainly by a CD1+/CD11c+ and CD3–/CD79 or CD20– immunophenotype; LCs, but not other DCs, also express E-cadherin. 1,3,7 In humans, cutaneous and/or systemic histiocytic disorders are classified as DC-related and macrophage-related disorders of varied biologic behavior and malignant disorders; they are rare to uncommon conditions that can occur at any age but mostly involve children and include neonatal cases. 11 Most human histiocytic disorders involving the skin fall into 1 of 2 subgroups: LC histiocytosis (LCH) and non-LC histiocytosis (NLCH). 11,12 The diagnosis of LCH relies on the demonstration of CD1a+/S100+/langerin+ expression, with clinical criteria defining different entities; the ultrastructural demonstration of Birbeck’s granule, a LC-specific organelle, is no longer essential as langerin (CD207) is intimately associated with them. 12 The diagnosis of NLCHs is based on clinical, morphological, and immunophenotypic criteria, but there is considerable overlap between the different entities, many of which are now considered part of a spectrum and referred to as the “juvenile xanthogranuloma (JXG) family.” 4,12 The cell of origin in JXG and related entities is generally considered the DDC or “dermal dendrocyte,” based on factor XIIIa+ and CD1a–/S100–/langerin– immunophenotype, most being also CD14+/CD68+/CD163+/fascin+. 12 It has, however, been shown that there are 2 major distinct populations of dermal histiocytes with dendritic morphology: true CD1b+/CD1c+/CD11c+ (myeloid) DCs and CD1–/CD11c– and FXIIIa+/CD163+ cells that are functionally macrophages (ie, mostly phagocytic and not antigen-presenting cells); these dermal DC-like macrophages can phagocytose hemosiderin, among others. 8 There is currently no single specific histiocytic marker, and some markers used in veterinary and human medicine, such as CD1s and CD11s, can only be used on frozen tissues, which are often not available. 3 In recent years, 2 monocyte/macrophage scavenger receptors, CD163 and CD204, have been used as histiocytic markers in some canine and human histiocytic disorders. CD163 and CD204 have been shown to be expressed in canine HS but not in normal canine LCs or in CCH. 3 CD163 is an excellent histiocytic marker for some NLCHs in humans, including JXG; 9 in pigs, it is strongly expressed in macrophages and a subtype of dermal DCs but not in LCs. 5,6
A male crossbred piglet was born with numerous, widely disseminated cutaneous lesions that had not resolved by 2 weeks of age; it was reported to be otherwise healthy. No other pig on the facility was affected. It was euthanized on the farm and brought to the University of Montreal’s Faculty of Veterinary Medicine Diagnostic Laboratory. The piglet was in good body condition. Numerous variably sized tan to brown macules, plaques, and papules were distributed all over the body. Most lesions were less than 1 cm in diameter, with a few larger ulcerated plaques up to 4 cm in diameter (Fig. 1). On cut surface, they were mottled gray to tan, and their location was dermal to subcutaneous (Fig. 2). Superficial lymph nodes appeared normal except for the superficial inguinal nodes that were mildly increased in size and brownish (iron injection). Internal examination did not reveal any significant changes; the stomach was full of milk. Several cutaneous lesions, brain, heart, thymus, lungs, liver, spleen, kidneys, palatine tonsil, and superficial and mesenteric lymph nodes were sampled. They were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin-eosin-phloxine-saffron (HEPS). Selected slides with skin lesions were also stained with Gram, periodic acid Schiff, Ziehl-Neelsen, Giemsa, Perl’s Prussian blue, and Fontana. For transmission electron microscopy, small samples of formalin-fixed samples of 2 skin lesions were taken 2 weeks later; they were postfixed in 5% glutaraldehyde with 0.05 M cacodylate buffer, followed by 1% osmium tetraoxide fixation and stained with 0.05% uranyl acetate. Immunohistochemistry (IHC) was performed for CD163, CD204, vimentin, lysozyme, α-1-antitrypsin, Mac-387, S-100 protein, E-cadherin, and smooth-muscle actin (DCPAH, MI); unfortunately, no frozen tissues were kept for markers such as CD1a, CD11c, and CD4. Routine bacteriology was done on skin, a superficial lymph node, lung, liver, and spleen. Viral culture of cutaneous lesions was also performed.
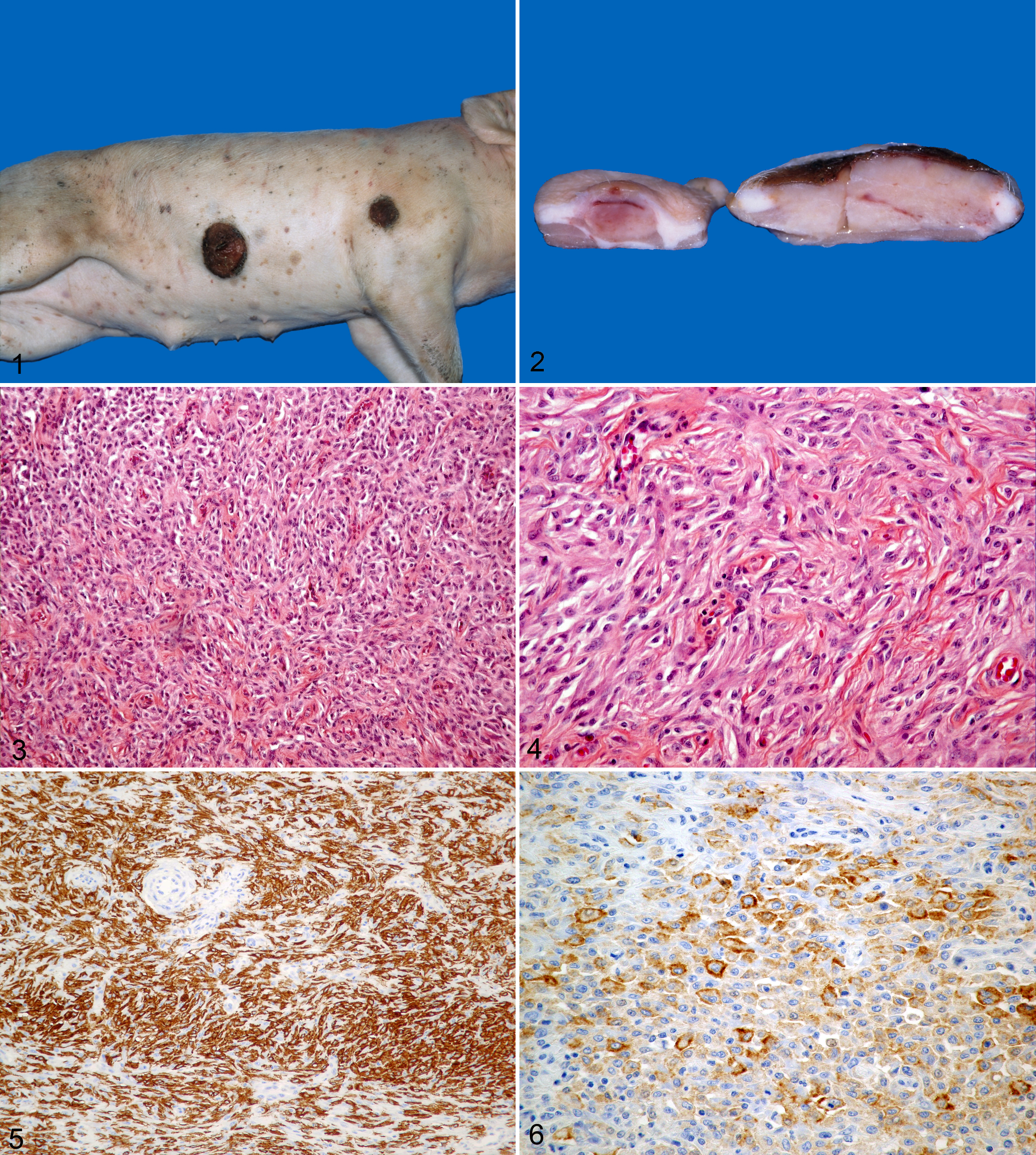
Pig with cutaneous histiocytosis. Numerous brown/tan macules, plaques, and papules are widely distributed over the body. A larger plaque on the abdomen is extensively ulcerated.
On light microscopic examination, cutaneous masses ranged from moderately well to poorly demarcated, were densely cellular, and located in the dermis and/or panniculus, and some were extensively ulcerated; in one section, the mass infiltrated the cutaneous muscle. They were composed of medium-sized spindle-shaped cells with fewer round to polygonal histiocytoid cells that were located mostly at the periphery of the mass. Dermal masses multifocally extended to the dermoepidermal junction, but epidermotropism was not observed. Spindle-shaped cells were arranged in short bundles, sometimes with a storiform pattern (Figs. 3, 4). Stroma was scant except in areas with a storiform pattern, where small to sometimes moderate amounts of collagen were often present. Most cells had moderate amounts of pale eosinophilic cytoplasm with ill-defined limits. Spindle-shaped cells had elongated nuclei while round cells had oval and occasionally reniform nuclei; nucleoli, when present, were small and unique. Rare bi- or trinucleated cells were seen. Cytonuclear atypia was mild, and mitoses were rare (0–1/10 high-powered field), with no abnormal mitotic figures. Melanin granules were not seen, even with Fontana’s stain. Multifocally in some masses, numerous cells contained moderate amounts of hemosiderin (Perl’s Prussian blue); hemorrhage was not present in most of these foci. A few eosinophils, lymphocytes, and plasma cells were multifocally interspersed throughout the spindle and histiocytoid cells. In ulcerated dermal masses, inflamed granulation tissue blended in with the mass. The other organs examined did not have any significant changes. Sections for transmission electron microscopy were of passable quality; they did not reveal any Birbeck granules. Immunoreactivity (IHC) was diffuse and strong for CD204 (Fig. 5); moderate and less extensive for CD163 (Fig. 6), lysozyme, and vimentin; and negative for the other markers. Special stains did not reveal any micro-organisms. Routine aerobic bacteriology was negative, as were cell cultures for viral isolation.
Discussion
Based on the clinical, morphological, and immunohistochemical results, a diagnosis of congenital cutaneous histiocytosis of non-Langerhans cell origin was made. The histiocytic nature of the lesion was strongly supported by CD204, CD163, and lysozyme immunoreactivity. 3,5,6,11 In human and, to a lesser degree, veterinary medicine, CD68 is often used as a histiocytic marker, but it is less specific than CD163 and CD204, since it is a lysosomal marker. 9 In veterinary medicine, CD18, a pan-leucocytic marker, is often used in cases where frozen tissues are not available, and the CD18+/CD3–/CD79 or CD20– immunophenotype is used to support histiocytic lineage; however, other round cell tumors such as non–B-cell/non–T-cell lymphomas cannot be excluded. 3 The exact phenotype (DC or macrophage) could not be determined, since CD1a and CD11c IHC was not possible; however, the CD163+/CD204+ and S-100–/E-cadherin– immunophenotype, lack of epidermotropism, and absence of Birbeck’s granules are not consistent with an LC phenotype. CD204 and CD163 immunoreactivity have been shown in the non-LCH dendritic disorders of canine HS (both) and human JXG (CD163). 3,9 CD163 is expressed by macrophages in several species, including pigs, in which it is also expressed in subsets of dermal DCs. 5,6 From a comparative pathology standpoint, the condition in this piglet most resembled JXG, the most frequent NLCH in humans. This disease is mostly seen in infants and children, generally limited to the skin, and self-limiting, with multiple lesions reported in about 20% of cases, mostly in very young children, and 35% of cases present at birth; the latter must be differentiated from congenital cutaneous forms of LCH. 2,4,11,12 The NLCHs are a diverse group of clinopathologic entities with considerable overlap; the literature can sometimes be confusing, with conflicting IHC results. Most have a similar immunophenotype, including factor XIIIa+/ CD163+ and CD1–/S100–, and are considered parts of a spectrum sometimes referred to as the “JXG family”; of this group, only JXG and benign cephalic histiocytosis (BCH) have been reported in neonates. 4,12 In JXG, cells are also immunoreactive for vimentin but not for Mac 387. 2,11 The histopathology of JXG, as its name implies, typically involves foam (xanthomized) cells and multinucleated Touton giant cells, but other morphologic forms, including spindle cells, are present. However, early lesions are often monomorphous with rare to absent foam cells and Touton cells; some predominantly consist of spindle cells often arranged in a storiform pattern (spindle cell variant). 2,4 Hemosiderin has been observed in some cases; 11 in our case, since hemosiderin was not associated with hemorrhages, it is possible that it was due to the neonatal iron injection and subsequent phagocytosis by the histiocytic cells. A single porcine case with some similarities to ours has been reported, but the cell morphology was different (foam cells) and the condition likened to Letterer-Siwe disease, a form of LCH; the diagnosis was not supported by immunohistochemistry or ultrastructural studies. 10
Footnotes
Acknowledgements
We thank Dr René Sauvageau for initial necropsy work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
