Abstract
A 9-year-old, male French Bulldog was examined for a subcutaneous mass located at the site of a microchip implant. Cytologic examination of the mass was suggestive of a malignant mesenchymal neoplasm. Histologically, the mass was confirmed as a high-grade infiltrative fibrosarcoma, with multifocal necrosis and peripheral lymphoid aggregates. By immunohistochemistry, the sample was investigated for vimentin, smooth-muscle actin (SMA), CD3, CD79α, and CD18. All the neoplastic cells were positive for vimentin. Scattered cells at the periphery of the lesion were also positive for SMA, highlighting a myofibroblastic phenotype. The lymphoid cells were positive for CD18 and CD3. No aluminum deposits were detected by the aurintricarboxylic acid method. A diagnosis of fibrosarcoma morphologically similar to feline postinjection sarcomas was made. Fibrosarcomas at the site of injections have been reported in dogs and ferrets. Furthermore, neoplastic growth at the site of microchip implant in dog and laboratory rodents has been described.
On April 2004, Leon, a 9-year-old male French Bulldog, was examined by the referring veterinarian, based in Guelph, Ontario (Canada), for the sudden growth of a subcutaneous 3 × 3-cm mass located on the dorsal midline of the neck, just cranial to the shoulders. The dog was regularly vaccinated against the most common canine infectious diseases and rabies, and was microchipped (Indexel, Merial, Lyon, France) in September 2003. A complete physical examination excluded any further alteration. Fine needle aspiration of the mass highlighted a single population of large, bipolar streaming spindle cells in swirling bundles. Cells had moderate nuclear/cytoplasm ratios and oval nuclei, with fine chromatin, multiple dark nucleoli, and prominent anisocytosis and anisokaryosis. Mitotic figures were rare, and no extracellular matrix was present. No evidence of inflammation or sepsis was observed. The cytologic diagnosis was fibrosarcoma.
On the veterinarian's advice, the owner decided to have the mass removed. The mass was surgically excised with 2 cm of margins. The microchip, which was detected attached to the mass, was also removed. The tissue was immediately fixed in 10% neutral buffered formalin, routinely processed, and paraffin embedded for histologic examination.
Histologically, a not well-demarcated, not encapsulated nodular mass was evident in the subcutis. Neoplastic cells were characterized by elongated pleomorphic nuclei, with prominent nucleoli and high mitotic rate (2–4 mitotic figures per high-power field; Fig. 1). Multifocal random necrosis and peripheral lymphoid aggregates were also present (Fig. 2). Further serial transverse sections were made to find the microchip. The microchip was found, not embedded within the tumor, but immediately adjacent to it, surrounded by a very thin fibrous wall (approximately 1 mm thick) and some fresh hemorrhage. The mass was confirmed as a high-grade infiltrative fibrosarcoma. Because the histologic features of this tumor were remarkably similar to feline postinjection sarcoma, further investigations were strongly recommended. On this advice, the dog's owner submitted paraffin blocks of the tumor to the Histopathology Department of the Istituto Zooprofilattico Sperimentale delle Venezie, Legnaro (PD), Italy. Further 3-µm sections were evaluated for the presence of aluminum deposits by the aurintricarboxylic acid method 21 and were immunohistochemically stained for vimentin (V9, Dako, Carpinteria, CA, USA, M0725, 1 ° 25), which reacts with the 57 kDa intermediate filament protein present in cells of mesenchymal origin, and smooth-muscle actin (SMA; 1A4, Dako, M851, 1 ° 50), which labels the smooth-muscle cells of vessels and different parenchyma, including myofibroblast in benign and reactive fibroblastic lesions. Each primary antibody was incubated for 30 minutes at room temperature. Antigen retrieval for SMA was obtained by trypsinization for 30 minutes at 37°C. The EnVision Peroxidase Dual Link System detection system (Dako, K4063) and DAB (3,3 diaminobenzidine tetrahydrochloride; Sigma, St. Louis, MO) as chromogen were applied. The sections were counterstained with Mayer's hematoxylin.
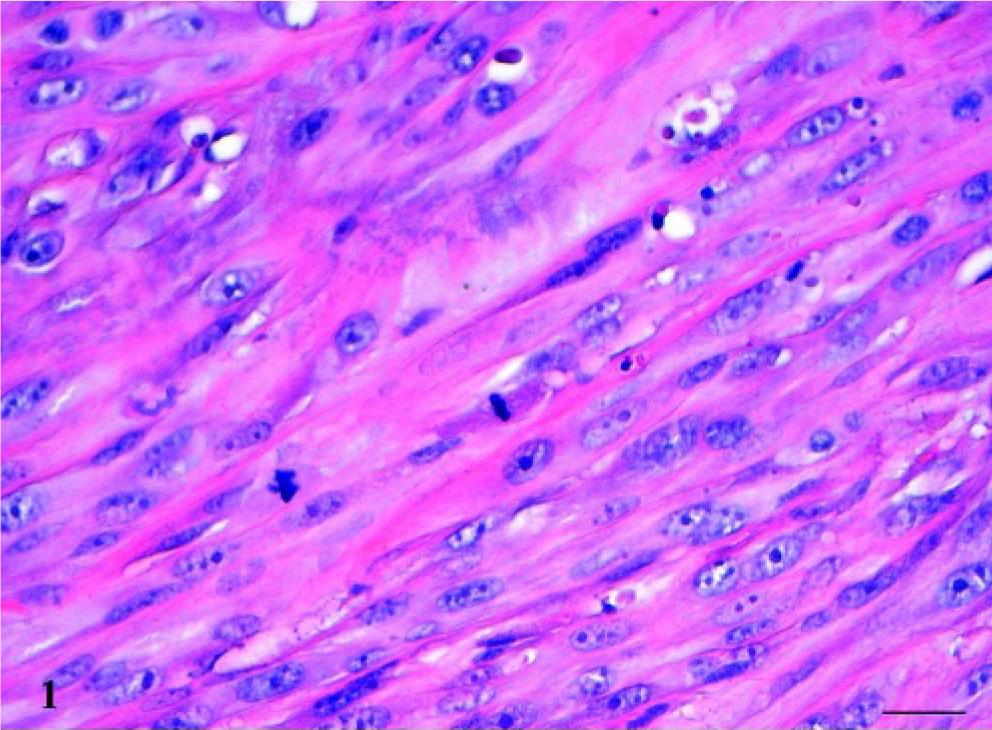
Subcutis; dog. Histologic aspect of neoplastic cells, characterized by evident nuclear pleomorphism, multiple and prominent nucleoli, and frequent mitotic figures. HE. Bar = 25 µm.
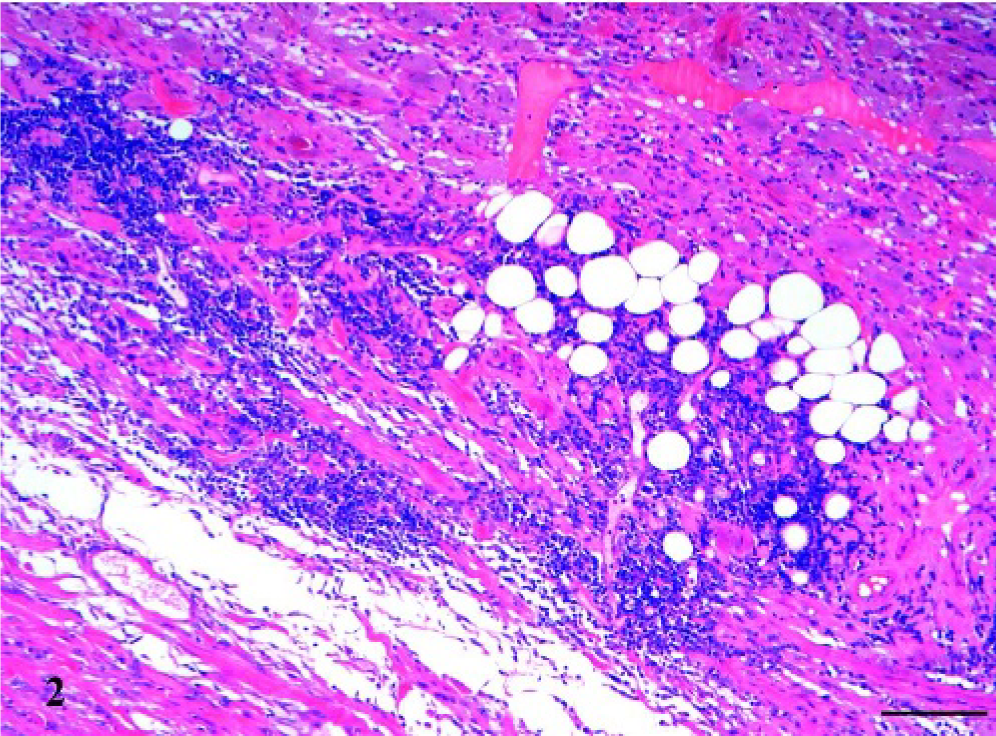
Subcutis; dog. Multifocal lymphoid aggregates were present at the tumor periphery, infiltrating the subcutaneous adipose tissue. HE. Bar = 100 µm.
The immunophenotype of lymphoid aggregates was also investigated, by immunohistochemistry using monoclonal antibodies for CD18 (canine leukocytes, University of California-Davis), CD3 (canine T-cells, University of California-Davis), and CD79α (human B-cells, Dako, M7051, 1 ° 10). Antigen retrieval was conducted by steaming chamber at 98°C for 30 minutes in citrate buffer pH 6.1 (Dako; S1699) for CD3 and CD18, and by pressure cooking for 25 minutes (Dako, S2031) for CD79α. The EnVision Peroxidase Dual Link System detection system (Dako, K4063) and DAB, as chromogen, were applied. Sections were counterstained with methyl green (Dako, S1962). Appropriate positive and negative controls were included in each immunohistochemical run.
No aluminum deposits were detected in the sample. All the neoplastic cells were positive for vimentin. Furthermore, some scattered cells located at the periphery of the tumor were also positive for SMA, highlighting a myofibroblastic phenotype. CD18 immunohistochemistry highlighted numerous cells diffusely infiltrating the tumor mass, which were negative for CD3 and CD79α. Lymphoid aggregates at the tumor periphery exhibited a T-cell phenotype, being positive for both CD18 and CD3. No CD79α positivity was observed. Based on histologic and immunohistochemical results, a diagnosis of high-grade fibrosarcoma with typical features of postinjection sarcoma was made. The inflammatory cells exhibited histiocytic and T-cell phenotype, indicative of a type IV hypersensitivity reaction.
After surgery, the dog was not vaccinated or microchipped again. Up to now, the dog is well, and no recurrence has been observed.
Postinjection fibrosarcoma is a well-known pathologic entity, first described in cats and recently observed in dogs and ferrets. 13, 21 Histologically, feline postinjection fibrosarcomas are characterized by inflammatory peritumoral infiltration, multinucleated giant cells, and myofibroblastic cells. 6 Data suggest that local inflammation caused by aluminum or other potentially irritant inoculated substances may predispose tissues to tumor development. It is accepted that substances other than aluminum can be involved in the pathogenesis of these fibrosarcomas. For close to 100 years, investigators have observed that irritation, inflammation, and/or wounds are promoters of tumor development. 12 Virtually anything that causes a local inflammatory reaction may potentially be responsible for neoplastic initiation. 23 Sarcomas developing at sites of subcutaneous administration of long-acting drugs and at sites with deep nonabsorbable sutures, as well as ocular posttraumatic sarcomas are clinical examples that support these findings. 3– 5, 8
In November 1996, in response to the increased incidence of soft-tissue sarcomas occurring at vaccination sites, the American Veterinary Medical Associations, the American Animal Hospital Association, the American Associations of Feline Practitioner, and the Veterinary Cancer Society formed the Vaccine-Associated Feline Sarcoma Task Force (VAFSTF). The goals of the VAFSTF are to facilitate investigation of the epidemiology, etiopathogenesis, treatment, and prevention of these malignancies, as well as to disseminate information to veterinarians and the cat-owning public. 1, 20 In Europe, the British Small Animal Veterinary Association, in conjunction with the Federation of European Companion Animal Veterinary Associations, launched a scheme to record information on adverse reactions to vaccination 18 and to microchips. 15
Microchipping is the most widely diffuse method of identification for domestic animals, based on its indelibility, and safety. The histologic effects of microchip implantation have been evaluated in dogs up to 6 years after implantation. 14 A foreign-body reaction to the subcutaneously implanted microchips was observed in the form of infiltration of inflammatory cells, fibroblast proliferation, and granulation-tissue formation. The inflammatory reaction disappeared 3 months after implantation, and the enclosure of the microchip by a capsule consisting of fibroblasts, collagen fibers, and elastic fibers was complete after 12 months. No marked difference was observed in the histologic findings 36 and 72 months after implantation, compared with those 12 months after implantation. 14
However, despite clear advantages over other identification techniques, some adverse reactions to microchips have been recorded. 16, 17 Some cases of soft-tissue tumors surrounding a microchip have been described in laboratory mice and rats. 2, 7, 19 All the tumors described in these papers were mesenchymal in origin and contained embedded microchips. The mechanism of carcinogenicity was ascribed to a foreign-body–induced tumorigenesis. 7 Recently, a case of liposarcoma embedding the microchip (Indexel, Merial) was described in a dog. 22 In the present case, the microchip was not embedded within the tumor but was found intimately adjacent to it. The Indexel microchip is equipped with an antimigrational capsule, located in the anterior part of the microchip, to prevent migration after implantation. The capsule is made from bioglass, the main components of which are silicon, sodium, calcium, potassium, magnesium, iron, and aluminum, which has been classified in the silicon sodium group. 10 One of the properties of such bioglasses is their insolubility, and they tend to become encapsulated with fibrous tissue when implanted subcutaneously. 9, 11 The apparent biocompatibility of the bioglass with the tissues formed the basis for its final application on a wide scale.
Many observational and epidemiologic studies have been performed to better define the temporal interval between exposure and the neoplastic outcome. In this case, the microchip was applied 8 months before tumor development. In the previous report of a liposarcoma development at the site of the microchip implant, the interval time was about 18 months. 22 In laboratory rodents, neoplasm occurred between 15 weeks to 2 years after microchip implant. 2, 7 In cats, the interval between vaccination and tumor development has been observed to be highly variable, ranging from 1 month to 3.5 years (E. K. Meyer, personal communication).
In this case, it is difficult to establish which was the primary cause of the neoplastic growth, because the dog had received several rabies vaccines and the microchip was detected close to but not included in the mass. A concause could then be hypothesized. Notwithstanding, reports on adverse reactions to vaccination and microchips are strongly encouraged to deepen the current knowledge on their possible role in tumorigenesis. In fact, the cause-and-effect relation between exposure (injection) and the outcome (sarcoma) is still to be defined and is a matter of discussion for experts. 20
Footnotes
Acknowledgements
The authors wish to thank Dr. Barb Deter for providing clinical and surgical information, and Dr. Brian Wilcock for the histologic evaluation and for making further investigations possible by providing the paraffin blocks. Jeanne, Leon's owner, is also thanked for her support and deep interest so that this case could be documented.
