Abstract
Damaraland mole rats (Cryptomys damarensis) are among the longest-living rodents, with a maximum longevity of approximately 16 years. As one of the few mammals termed eusocial, these animals have been used in behavioral, genetic, metabolic, and physiologic research at the University of Connecticut since 1997. For individual identification at 3 to 4 months of age, mole rats were subcutaneously implanted with microchip transponders (11 mm in length) in the dorsal cervical region. In 2007, 2 of the 90 implanted adults, 10-year-old and 9-year-old females, developed subcutaneous masses at the site of the implant. Histopathological and immunohistochemical examinations revealed amelanotic melanoma and fibrosarcoma, respectively, with metastasis of the amelanotic melanoma. In 2008, a total of 3 adult males were castrated as part of a sex behavior study; 3 months later, all 3 castrated males developed subcutaneous masses around their implants, whereas none of the noncastrated males had masses. After an additional 9 months, these masses were found to be granulomas. To the authors' knowledge, this is the first report of neoplasia in this species. Both the tumors and the granulomas surrounded the microchip transponder.
Damaraland mole rats are indigenous to southwestern and central Africa. They spend nearly their entire lives underground, in a network of tunnels that connect nesting areas, defecation areas, food storage areas, and food source areas. They are one of the few mammalian species termed eusocial; that is, they live in a caste system, in which only one female, the queen, produces offspring. The queen mates with only one male in a stable relationship that lasts their entire lives. The other individuals in the colony assist in caring for the pups, foraging for and storing food, and maintaining and defending the tunnels. These workers are physiologically capable of reproduction but do not do so as long as they remain in the colony. Damaraland mole rats are among the longest-living rodents, with a maximum longevity of approximately 16 years. 2
At the University of Connecticut, microchip transponders are subcutaneously implanted in mole rats at 3 to 4 months of age for permanent individual identification. In 2007, 2 of 90 implanted adults, 10-year-old and 9-year-old females, developed masses at the site of the implant. Necropsy, histopathology, and immunohistochemistry were performed. Here, we report (to our knowledge) the first two cases of neoplasia in Damaraland mole rats diagnosed by histopathology and immunohistochemistry, and both tumors were associated with the microchip transponders. In 2008, 3 months after castration, 3 adult males developed granulomas associated with the microchip transponders that had been implanted at 3 to 4 months of age.
Materials and Methods
Colony Husbandry and Research
All procedures and studies involving Damaraland mole rats have been approved by the University of Connecticut Animal Care and Use Committee. The mole rats are housed individually or in pairs in single polycarbonate cages (48.0 × 26.6 × 20.2 cm) or as groups in 2 to 3 cages connected by acrylic tubes with a 7-cm internal diameter. Large groups have a polycarbonate nesting cage (70.7 × 44.1 × 38.4 cm). Filtered or perforated cage lids serve to reduce drafts. The standard diet is sweet potatoes, occasionally supplemented with vegetables, apples, squash, and pelleted laboratory rodent diet. Water is not provided. Aspen shredded or ground corncob bedding is provided, less than 3 cm deep. The light cycle is 16 hours of low-level light and 8 hours of dark, and the room humidity and temperature are maintained at 30 to 70% and 22 to 28°C.
At the University of Connecticut, 120 Damaraland mole rats are used in behavioral, genetic, metabolic, and physiologic research. 5 –7
Microchip Transponder
With a 12-ga hypodermic needle, microchip transponders (AVID Identification Systems, Inc, Norco, California) were subcutaneously implanted in the interscapular region of the mole rats. The microchips are sealed in bioglass capsules composed primarily of silicon and coated with a Parylene C polymer to encourage tissue encapsulation and reduce migration of the implant. The microchip transponder measures approximately 11.0 mm in length and 2.1 mm in diameter. When activated by a 125-kHz radiofrequency signal emitted by a handheld scanner, the microchip—a radio frequency identification device (RFID)—transmits a unique preprogrammed 9-digit identification number back to the scanner.
Histopathology
Representative samples of all internal organs—including brain, spinal cord, heart, lung, liver, kidneys, stomach, spleen, intestines, lymph nodes, and subcutaneous mass—of all 5 animals were fixed in 10% neutral buffered formalin. Following fixation, sections were routinely processed for histologic examination and stained with hematoxylin and eosin. Masson’s trichrome and periodic acid–Schiff hematoxylin was applied to the subcutaneous mass.
Immunohistochemistry
Immunohistochemistry was performed with formalin-fixed, paraffin-embedded 5- to 6-μm-thick sections. Briefly, microsections of tissues were placed on positively charged glass slides and deparaffinized in xylene (Allegiance Healthcare, McGaw Park, Illinois), rehydrated in a graded series of ethanol, and rinsed with distilled water. Antigen retrieval was performed by heating (98°C) in Target Retrieval Solution (Dako, Carpinteria, California) in a vegetable steamer (Fagor America, Cook’s Essentials Steamer, Lyndhurst, New Jersey). Following cooldown and washing in phosphate buffered saline (Dako wash buffer), endogenous peroxidase activity was blocked by incubation in 3% hydrogen peroxide for 5 minutes. Slides were incubated with the MAb (monoclonal) and PAb (polyclonal) primary antibodies (Tables 1 and 2) for 30 minutes at room temperature. After a short wash in Dako wash buffer, tissue sections were incubated for 30 minutes with labeled polymer (EnVision+ Dual Link System-HRP, Dako). Following a final wash in Dako wash buffer, sections were developed with 3,3′-diaminobenzidine chromogen (DAB+, Dako) or NovaRED (Vector Labs, Burlingame, California) for 5 minutes. After a washing in warm tap water, the slides were counterstained with hematoxylin and coverslipped. Appropriate controls were run: positive (human, rat, feline, and dog neoplasms) and negative (omitting primary antibody).
Primary Antibodies for Mole Rat No. 1 a

a Source for all antibodies: Dako. Chromogen for each antibody: 3,3′-diaminobenzidine (unless noted otherwise).
b Chromogen: NovaRED.
Primary Antibodies for Mole Rat No. 2 a

a Source for all antibodies: Dako. Chromogen for each antibody: 3,3′-diaminobenzidine.
Results
Clinical History and Gross Pathology
Mole rat No. 1 was an 8-year-old breeder female weighing 167 g. It had produced 22 litters in 9 years. On June 8, 2007, a soft subcutaneous mass (0.5 × 1.5 × 1.5 cm) was noted in the dorsal cervical midline. On August 10, 2007, the mass had increased in size to 2.50 × 2.50 × 3.35 cm (Fig. 1). The mass could not be completely surgically removed, and the mole rat was euthanized and submitted for necropsy. The surgically excised mass measured 1.8 × 2.0 × 2.8 cm and was nonencapsulated, soft, tan, and friable (Fig. 3). Multiple cystic areas (0.2 cm in diameter) were present in the mass, some of which contained yellow granular material and others, clear fluid. The lungs, liver (Fig. 4), and kidneys contained numerous soft gray–tan nodular foci (0.1 to 0.4 cm in diameter).
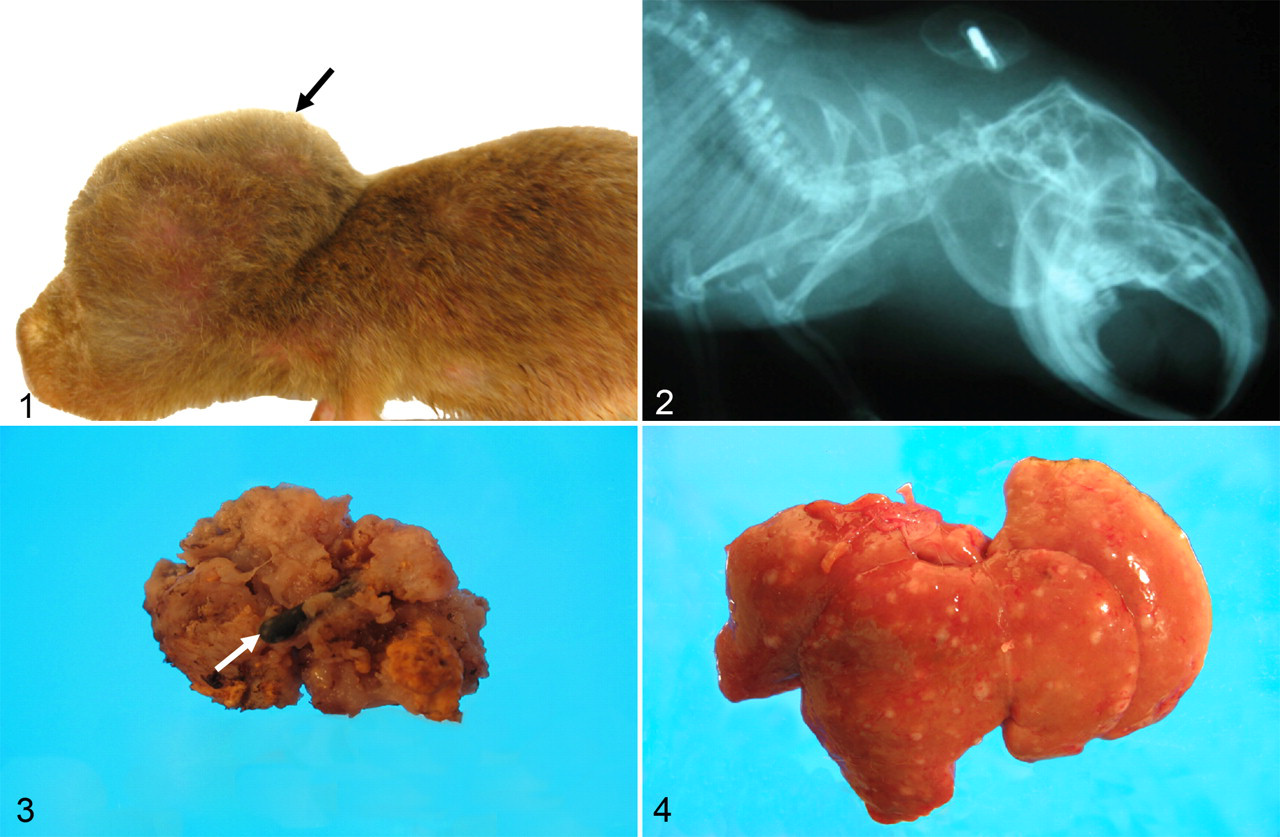
Mole rat No. 1. Subcutaneous mass (arrow) in the dorsal cervical midline.
Mole rat No. 2 was a 10-year-old breeder female weighing 144 g that had a soft gray–white subcutaneous mass (5.0 × 2.5 × 2.0 cm) in the dorsal cervical region, which extended to the right front limb. The mass affected locomotion, and on July 14, 2007, the mole rat was euthanized and submitted for necropsy. The mass infiltrated the surrounding skeletal muscles of the neck and extended to the proximal anterior right front limb.
Mole rat Nos. 3, 4, and 5 were males that were castrated at 26, 63, and 19 months of age, respectively, as part of a sex behavior study. Three months after castration, all 3 males developed subcutaneous masses around the implants that had been implanted at 3 to 4 months of age. Necropsies were done at the end of the study, approximately 1 year after castration and only on the castrated males. Mole rat No. 3 weighed 162 g and had a semifirm gray–brown subcutaneous mass (2.0 × 2.0 × 1.5 cm) in the dorsal cervical region. Mole rat No. 4 weighed 153 g and had a semifirm gray–brown subcutaneous mass (1.5 × 3.0 × 1.8 cm) in the dorsal cervical region. Mole rat No. 5 weighed 168 g that had a firm gray–tan subcutaneous mass (4.0 × 2.5 × 2.0 cm) in the dorsal cervical region. Lateral view radiographs were taken on mole rats Nos. 3, 4, and 5. In mole rat Nos. 3, 4, and 5, radiographs showed the microchip transponders embedded in the masses (Fig. 2), and in all males, the masses were firm and adhered to the skin and surrounding skeletal muscles.
Histopathology and Immunohistochemistry
In mole rat No. 1, the cervical mass was an nonencapsulated fairly well-demarcated infiltrative neoplasm composed of polygonal cells arranged in dense aggregates. Individual cells had pleomorphic nuclei with stippled chromatin and 2 or 3 large nucleoli, as well as a moderate amount of amphophilic cytoplasm. Anisocytosis and anisokaryosis were marked numerous karyomegalic and multinucleated tumor cells were present and the mitotic rate was high (5 or 6 mitotic figures per high-power field) (Figs. 5–8). Within and surrounding the mass were aggregates of small to moderate numbers of lymphocytes, macrophages, and rare plasma cells. Multifocal necrosis was present. Similar neoplastic cells were present in lung, liver, and kidneys (Figs. 9, 10). Immunohistochemical examination revealed diffuse and intense cytoplasmic staining of the majority of neoplastic cells for vimentin (Fig. 11), S-100 (Fig. 13), and the cytoplasm of approximately 50% of cells for Melan A (Fig. 14). Neoplastic cells were negative for CD68, CD3, and cytokeratin AE1/AE3 (Fig. 12). Based on histopathology and immunohistochemistry, the mass was consistent with amelanotic melanoma.
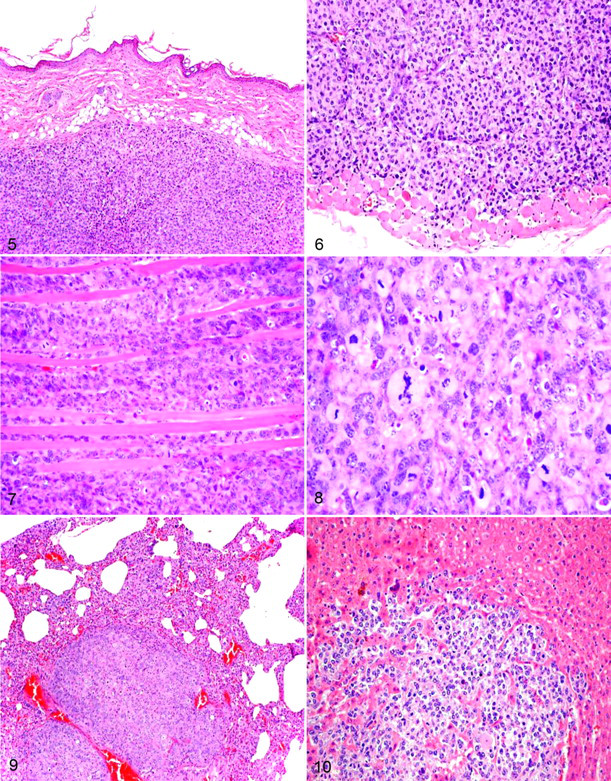
Mole rat No. 1. Nonencapsulated subcutaneous mass compressing the dermis. HE. 40×.
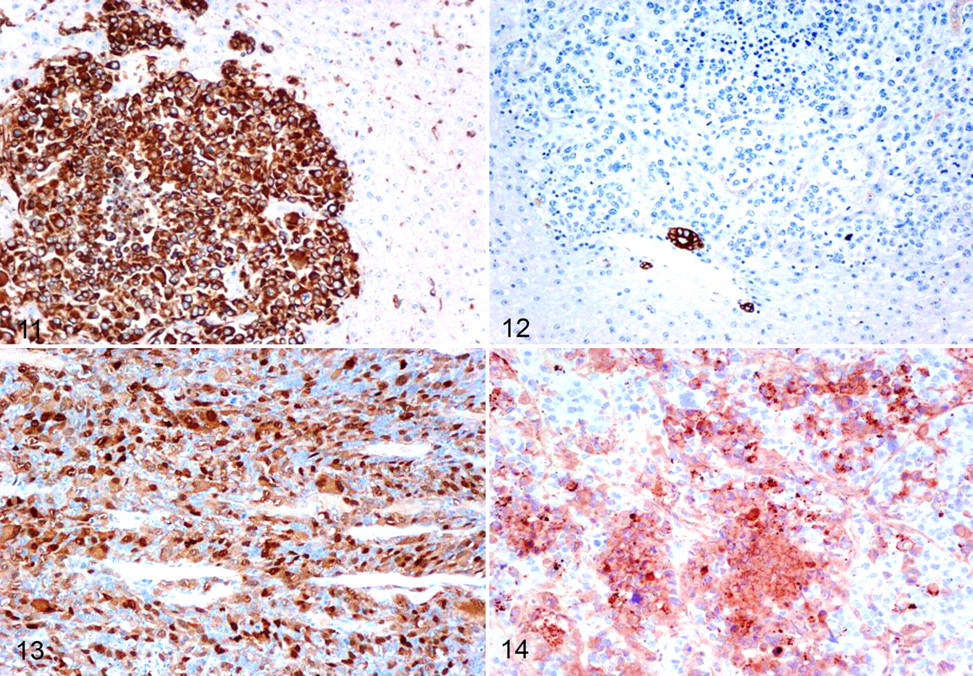
Mole rat No. 1. Immunoreactivity for vimentin in the neoplastic foci of liver. 100×.
In mole rat No. 2, the cervical mass was infiltrative, nonencapsulated, and composed of spindle-shaped cells arranged in whorls and fascicles (Fig. 15). Individual cells had oval nuclei with stippled chromatin and single nucleoli. There was mild anisocytosis and anisokaryosis, and the mitotic rate was 2 to 4 mitotic figures per 5 high-power fields. The neoplastic cells exhibited diffuse and strong immunoreactivity for vimentin (Fig. 16) and were negative for cytokeratin, S-100, desmin, and smooth muscle actin. These results are consistent with fibrosarcoma.
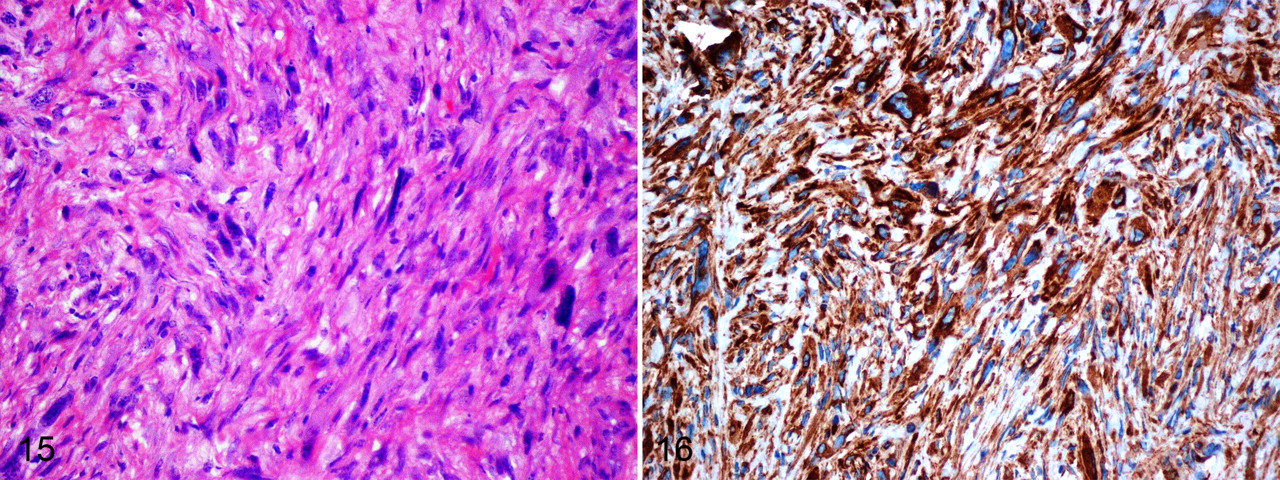
Mole rat No. 2. Haphazardly arranged neoplastic cells in the dermis. HE. 100×.
In mole rat Nos. 3, 4, and 5, the subcutaneous cervical masses were foreign-body granulomas. Individual granulomas often expand and compress the dermis and surrounding adnexa and are composed of aggregates of epithelioid macrophages, lymphocytes, plasma cells, reactive fibroblasts admixed with degenerate and necrotic neutrophils, and cellular debris.
Discussion
The use of implantable microchip transponders began in the late 1980s. They offer a means of permanent individual identification in long-term rat and mice toxicity and carcinogenicity studies, as well as in dogs, cats, horses, and livestock. 1,8 Microchip implantation provides a reliable and often less painful method of unalterable animal identification.
Although an initial inflammatory response typically follows implantation of the chip, the inflammation resolves within 3 months in dogs and laboratory mice, after which the microchip is surrounded by a connective tissue capsule. 9,11
Once encapsulation is complete, a microchip transponder is expected to function safely; however, transponder-associated neoplasia has been described in laboratory mice, laboratory rats, pet dogs, an Egyptian fruit bat, a degu, and a feathertail glider. 3,4,10,12 –15 The majority of transponder-associated tumors have been mesenchymal in origin, and the mechanism of carcinogenicity is thought to be foreign body–induced tumorigenesis. 4
In this report, we describe the findings of tumors associated with implanted microchip transponders in 2 adult female Damaraland mole rats. The additional findings of granulomas in 3 castrated male mole rats support the likelihood that the chronic inflammation may have induced the neoplasias in these long-lived rodents.
Footnotes
Acknowledgements
We thank Dr Richard Cartun at the Hartford Hospital and Mohammad Faizal at the University of Connecticut for timely assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
