Abstract
Sixteen dogs (2–12 years of age) presented with one (
Collagen-rich non-neoplastic lesions and tumors are common in the skin of dogs. They include collagenous hamartomas, fibroadnexal hamartoma, nodular dermatofibrosis, fibromas, dermatofibromas, and fibrosarcomas. 21 Hyalinized collagen fibers are absent from these lesions, except in the keloidal type of dermal fibroma. 6 Here, we describe 17 subcutaneous lesions in 16 dogs. The lesions are characterized by deposition of thick hyalinized collagen fibers intermingled with fibrocytes and histiocytes and are histologically similar but not identical to keloids and hypertrophic scars of man. We propose the terms keloidal fibroma and keloidal fibrosarcoma for these lesions and suggest that keloidal fibromas may undergo malignant transformation into keloidal fibrosarcomas.
Skin tumors with hyalinized collagen fibers were collected from 12 February 2000 to 14 February 2001 from case material submitted to the West Sacramento division of Idexx Veterinary Services. Tissues were submitted to the laboratory fixed in 10% buffered formalin. Samples were trimmed, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). 12 All lesions were stained for canine CD18 (ready-to-use mouse monoclonal antibody to canine CD18; Leukocyte Antigen Biology Laboratory, University of California, Davis, CA), smooth muscle actin (ready-to-use monoclonal mouse antibody to synthetic NH2 terminal decapeptide of smooth muscle actin; Biogenex, San Ramon, CA), and vimentin (1:100 dilution monoclonal mouse anti-vimentin antibody, clone Vim3B4; Dako, Glostrup, Denmark) using a standard immunohistochemical method. 7 The antibody reaction products were visualized with 3,3′-diaminobenzidine tetrachloride and counterstained with Mayer's hematoxylin. The specificity of the immunoreactions was verified by staining negative and positive control tissue sections (pyogranulomatous inflammation for anti-CD18 antibody and dermal tissue for anti-vimentin and anti–smooth muscle actin antibodies). A section of each tumor was also stained under identical conditions, replacing specific antiserum with unrelated antiserum to detect nonspecific staining.
Seventeen skin lesions with hyalinized collagen fibers were diagnosed in 16 adult dogs (2–12 years of age; ○ = 7.6 years; Table 1). Intact males (
Keloidal fibromas and fibrosarcomas in dogs: affected breeds, age, gender, location, and size of tumors.
∗ M = male; MN = male neutered; F = female.
† ND = not determined.
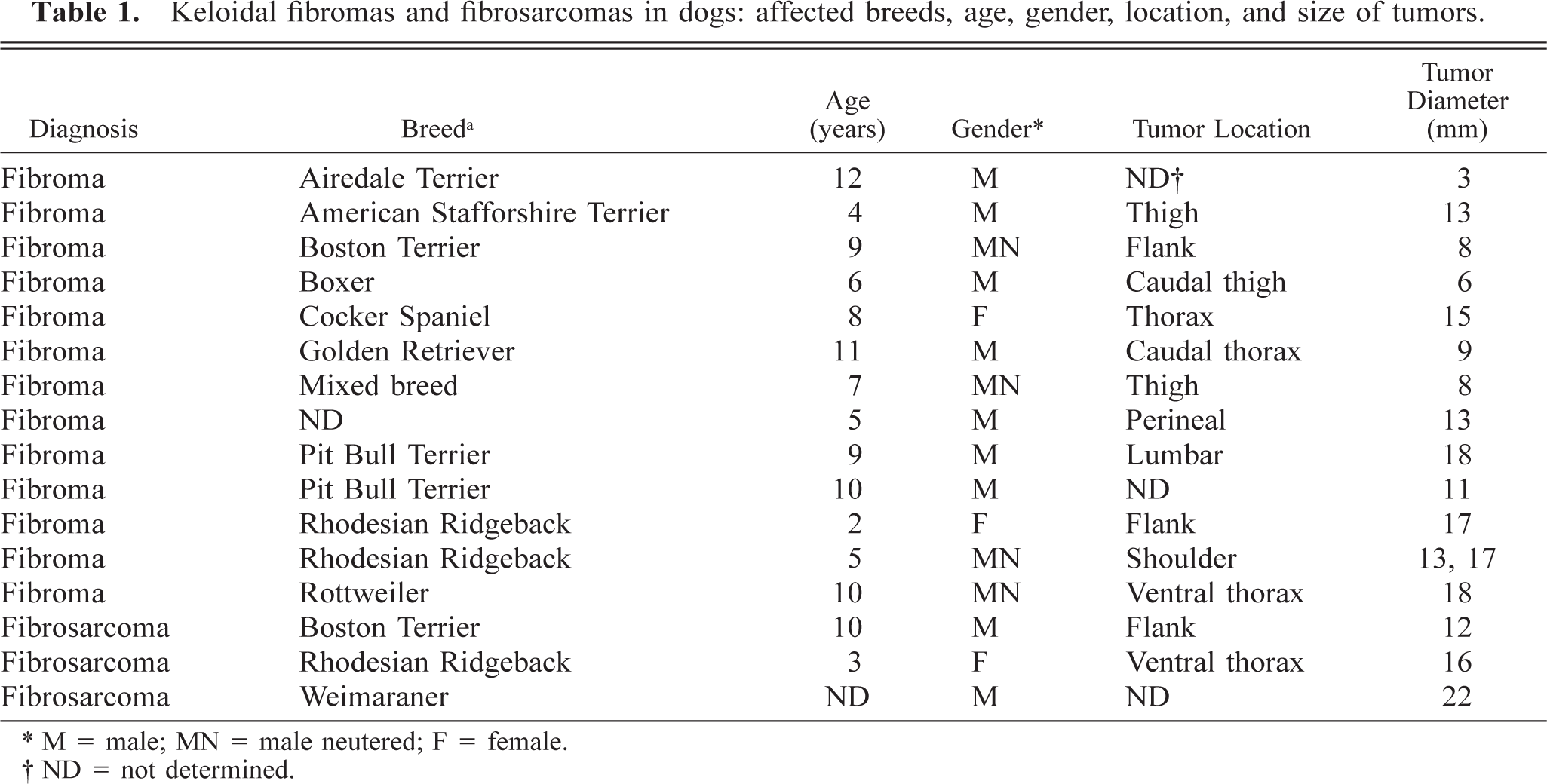
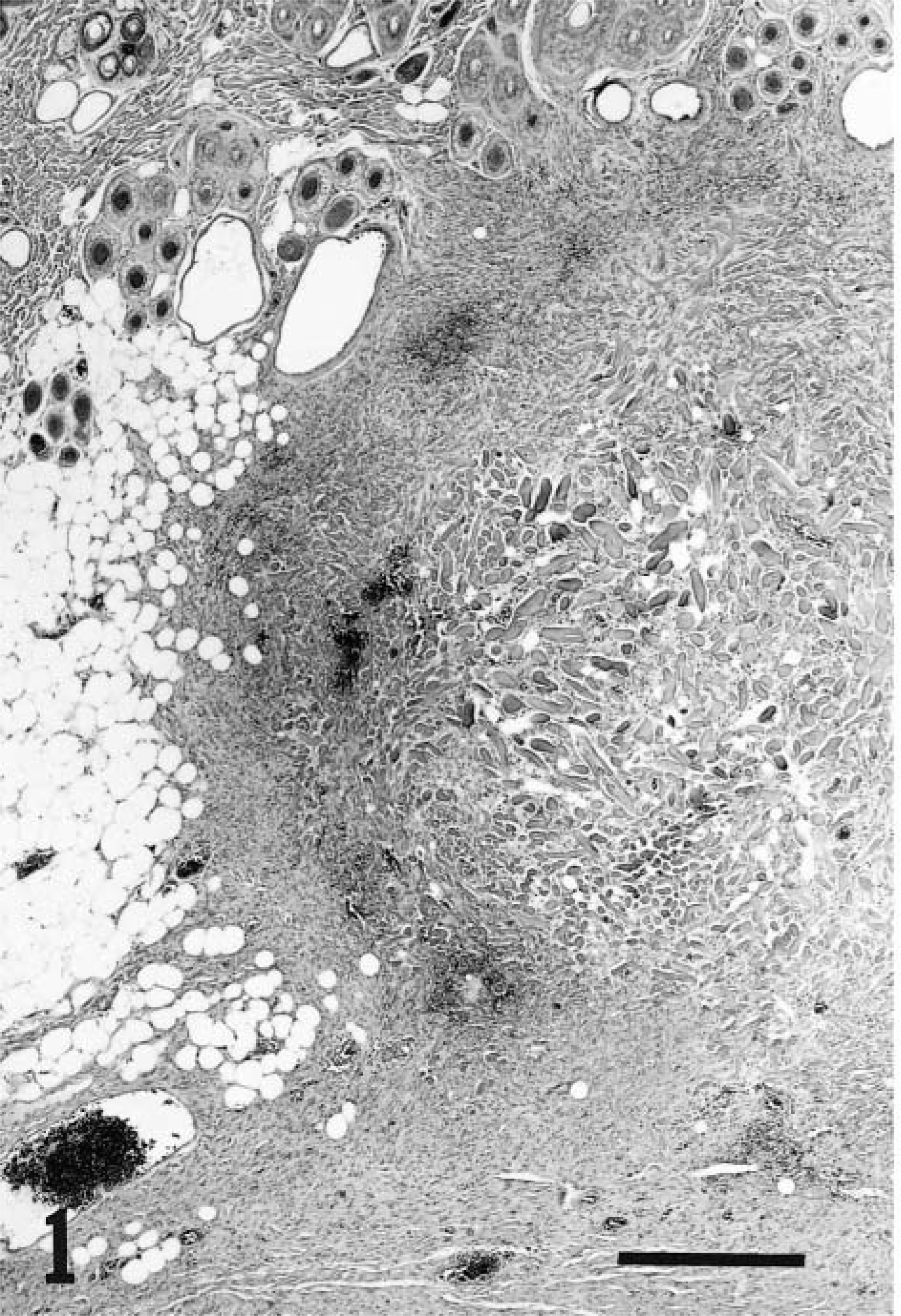
Keloidal fibromas were limited to the dermis (3), affected the dermis and extended into the subcutis (7), or were limited to the subcutis (3). The precise site could not be determined in one dog for which normal dermal and subcutaneous tissues were not submitted. Keloidal fibromas were nodular (13) or plaquelike (1) and 6–18 mm in diameter (○ = 12.3 mm). They gradually blended into the adjacent dermis and/or panniculus in 10 lesions (Fig. 1), but four lesions were discrete. The overlying epidermis was ulcerated in two lesions. Histologically, keloidal fibromas were comprised of streams of thick hyalinized collagen fibers surrounded by fascicles of closely packed plump cells. The lesions were more cellular at their periphery than at their center (Fig. 1). The cells had a moderate amount of pale gray and variably vacuolated cytoplasm. The nucleus was central, elongated, and normochromatic with a small basophilic nucleolus. Mitoses were rare. In all lesions, a variable number of thinner and more fibrillar collagen fibers were intermingled with and gradually replaced the thick hyalinized collagen fibers at the periphery of the tumor (Fig. 2). In three lesions, only a few hyalinized collagen fibers persisted and were predominantly located at the center of the lesion. Vascularization consisted of a small number of immature capillaries interspersed among the stromal cells or occasionally permeating hyalinized collagen fibers. Numerous minute hemorrhages were scattered throughout all keloidal fibromas.
Keloidal fibroma; dog. Ill-defined dermal and subcutaneous mass gradually blends into the adjacent dermis and subcutis. Cellularity is higher at the periphery of the mass than at the center, where there are thick collagen fibers. HE. Bar = 500 µm.
Keloidal fibroma; dog. The tumor is well delineated. Haphazardly distributed streams of thick hyalinized collagen fibers are surrounded by bundles of fusiform cells. HE. Bar = 100 µm.
Keloidal fibrosarcomas were invasive nodular subcutaneous tumors. Histologically, the superficial part of two of these tumors was composed of an ill-defined area that was indistinguishable from a keloidal fibroma. The deep portion of these two tumors and the entirety of the third tumor were composed of closely packed, interlacing fascicles of fusiform cells supported by a small amount of fibrovascular stroma in which a few adipocytes and a few thick hyalinized collagen fibers had been entrapped. Neoplastic cells had indistinct cell borders and a moderate amount of pale amphophilic cytoplasm. Their nucleus was central, elongated, and normochromatic, with a small basophilic nucleolus. Anisokaryosis and anisocytosis were mild. Mitotic index was low, with one or two mitoses per 10 high power fields in all three fibrosarcomas.
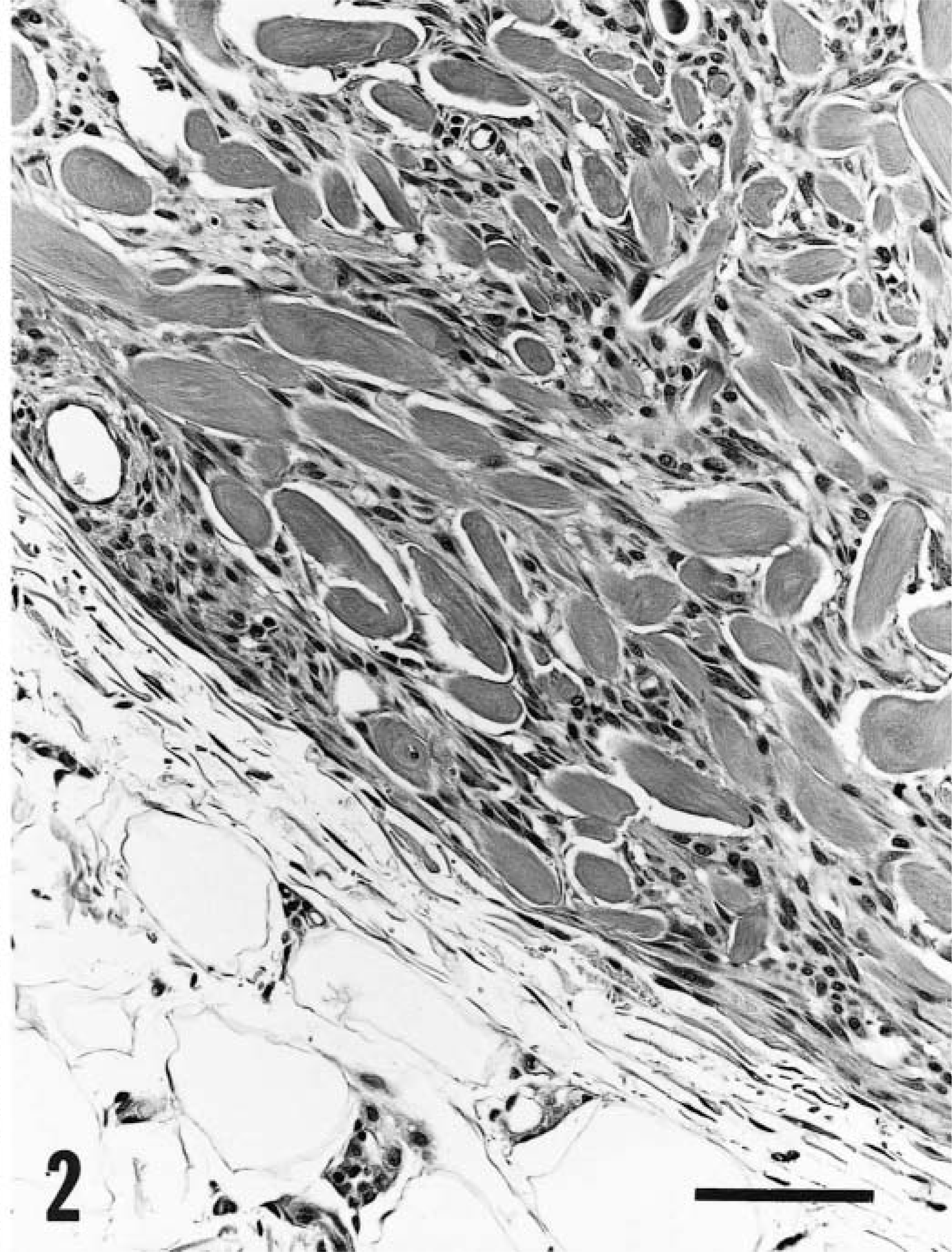
All stromal cells of keloidal fibromas and fibrosarcomas were strongly positive for vimentin. Immunohistochemistry for CD18 identified two populations among these cells: CD18-negative cells that were interpreted as fibroblasts and CD18-positive cells that were interpreted as macrophages. Fibroblasts predominated in all keloidal fibromas, whereas macrophages were present in 13 of 17 lesions and accounted for approximately 1–40% of cells (Figs. 3,4). Macrophages were present throughout keloidal fibromas, although they were more numerous in the vicinity of hyalinized collagen fibers. Macrophages accounted for about 10%, 20%, and 40% of cells of the keloidal fibrosarcomas. All stromal cells of keloidal fibromas and keloidal fibrosarcomas were smooth muscle actin negative.
Keloidal fibroma; dog. Cytoplasm of about 40% of cells is positive for CD18. These cells are interpreted as macrophages. Remaining cells are interpreted as fibroblasts. Avidin–biotin complex/diaminobenzidine–peroxidase, Mayer's hematoxylin counterstain. Bar = 50 µm.
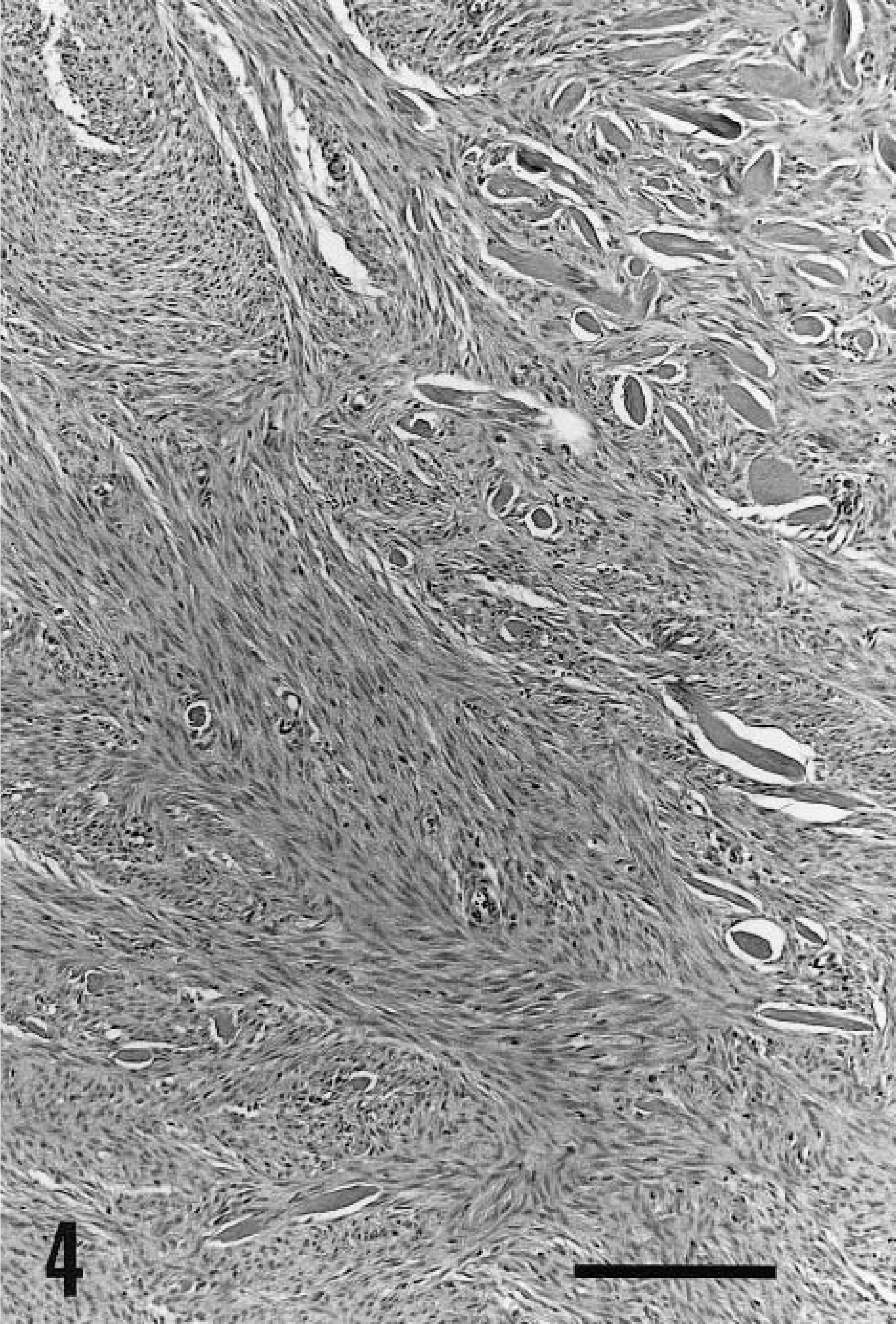
Keloidal fibrosarcoma; dog. Most of the tumor is made of interwoven fascicles of fibroblasts. A few thick collagen fibers persist within the tumor. HE. Bar = 200 µm.
Follow-up was available for five keloidal fibromas. There was no evidence of recurrence, additional keloidal fibroma, or metastasis 6–9 months (○ = 7.3 months) postexcision. The dogs with keloidal fibrosarcomas were lost to follow-up.
Keloidal fibromas have been briefly described in dogs by Goldschmidt and Shofer. 6 In our experience, these keloidal lesions are uncommon in dogs, and other animal species are not affected. The relative rarity of keloidal lesions in animals contrasts with the common occurrence of keloidal lesions (keloids and hyperplastic scars) in humans. 5,20 In humans, keloidal lesions preferentially develop at sites of trauma or high skin tension. 20 The presence of macrophages in most keloidal tumors suggests that in dogs, as in humans, keloidal fibromas and fibrosarcomas develop secondary to tissue injury and may represent a reactive inflammatory lesion rather than a true neoplasm.
The results of this study also suggest that fibrosarcomas may arise from keloidal fibromas, an observation that has also been reported in keloids of humans. 9 Malignant transformation of inflammatory lesions is relatively common in cats 4,11 and rodents 3 but is apparently rarer in dogs 13 and in humans. 2 Malignant transformation of inflammatory lesions associated with foreign bodies has been extensively studied in laboratory animals. 3 This transformation has been attributed to positive selection of proliferating mesenchymal cells in hypoxic chronic inflammation lesions. 3 Canine keloidal lesions are likely to provide the adequate hypoxic microenvironment for malignant transformation of fibrocytes because vascularization of these lesions is scanty.
Hyalinized collagen fibers are a distinctive feature of canine keloidal tumors and of keloids and hypertrophic scars in humans. However, keloidal fibromas in dogs differ from keloids in humans in that they do not extend above the surface of the skin. 5,16 Canine keloidal lesions also differ from hypertrophic scars in that they contain more hyalinized collagen fibers and are not linear lesions located at the site of previous surgery. 1,5 Thus, keloidal lesions in dogs histologically resemble but are distinct from keloids and hypertrophic scars of humans.
Other differential diagnoses for nodular dermal lesions with thick collagen fibers in humans include dermatofibromas 22 and sclerotic fibromas. 14,17,18 Dermatofibromas differ from keloidal fibromas in that collagen fibers are thickened but not hyalinized 22 and that the overlying epidermis is markedly hyperplastic. 8 Sclerotic fibromas are distinctive in that collagen fibers have a storiform pattern 14,17,18 that is not observed in canine keloidal tumors. Keloidal fibroma differs from all other collagen-rich nodular cutaneous lesions of dog, including nodular dermatofibrosis, collagenous hamartomas, dermatofibroma, fibroadnexal hamartoma, and dermal fibroma, in that it is the only one to have prominent thick hyalinized collagen fibers.
Histologic and immunohistochemical features of the predominant cell type of keloidal lesions are consistent with those of fibroblasts. These cells did not stain for smooth muscle actin, suggesting that they are not myofibroblasts, which is the major cell type in keloids 10 and in hypertrophic scars. 15 However, not all myofibroblasts express smooth muscle actin. 19 Electron microscopic examination of keloidal tumors may help to categorize the cellular component of these lesions.
The epidemiologies of keloidal tumors in dogs and keloidal lesions in humans differ. Male dogs were more often affected by keloidal tumors than were females, whereas keloidal lesions in humans predominantly affect females. 1 There is no age predominance in dogs with keloidal tumors, whereas keloidal lesions predominantly develop in young adult humans. 1 These epidemiologic differences further suggest that keloidal tumors in dogs are not the exact counterpart of keloidal lesions in humans.
Canine keloidal lesions have distinctive histologic features. Possible malignant transformation of canine keloidal fibromas into fibrosarcomas warrants specific diagnosis and wide excision of these lesions. Monitoring of more cases over a longer period of time is needed to better characterize the clinical behavior of canine keloidal tumors.
Footnotes
Acknowledgements
We express our sincere thanks to Ms. T. Cabral, Ms. S. Puerner, M. R. Havens, and M. B. Shibata for outstanding technical assistance. We thank Dr. Barnes, Dr. Griffith, Dr. D. L. Mabley, Dr. W. E. Lipman, and Dr. I. K. Saikashi for follow-up on these cases.
