Abstract
A 6–year-old, neutered male Labrador Retriever was diagnosed with congestive heart failure, and an echocardiogram revealed a large mass inside the pericardial sac associated with the left ventricle. At necropsy, the dog had marked ascites, mild hydrothorax, marked hydropericardium, and an 11.0 X 7.0 X 6.0 cm, tan and red, firm, well-demarcated mass attached to the left ventricular free wall. The mass was diagnosed as a fibrosarcoma based on the morphologic appearance and supportive immunohistochemical staining. To our knowledge, this is the first case report of a primary fibrosarcoma involving the left ventricular free wall myocardium, epicardium, and pericardium with a pulmonary metastasis in a dog.
Cardiac tumors are rare in both humans and domestic animals.9,12,21 The prevalence of primary heart tumors reported in dogs was 0.69% in a study of 1,383 dogs diagnosed with neoplasia.21 Similarly, the overall prevalence of primary cardiac neoplasia in humans is reported as 0.1–0.5% of all autopsies.12 In dogs, primary cardiac tumors are much more common than metastatic tumors to the heart at 84% and 16%, respectively.21 In humans, the reverse is true—metastatic tumors to the heart are reported to be 20–40 times more common than primary heart tumors.21 Over 80% of the canine primary heart tumors are malignant, of which 80% are hemangiosarcoma.21 Approximately 90% of the human primary cardiac tumors are benign, and 40–70% of these tumors are atrial myxomas.12,19,21 In both humans and domestic animals, primary cardiac fibrosarcomas are rare among primary cardiac malignant neoplasia.9,21 This case report describes a primary cardiac fibrosarcoma of the left ventricular free wall in a dog.
A 6-year-old, neutered male Labrador Retriever was referred to the Cardiology Service, University of Minnesota Veterinary Medical Center, with ascites due to suspected right-sided congestive heart failure. The dog had been treated with furosemide and abdominocentesis; 4.7 liters of fluid had been drained during the week prior to referral but the ascites had recurred. On physical examination, the dog was 8% dehydrated, had mild dyspnea manifested by increased abdominal effort, distended jugular veins, mild tachycardia (140 bpm), marked abdominal distension with a fluid wave, bilateral muffled lung sounds ventrally, and muffled heart sounds. Echocardiography demonstrated marked pericardial effusion and a large intrapericardial mass associated with the left ventricle and pericardium (Fig. 1). Because of the poor prognosis, the dog was euthanized and submitted to the University of Minnesota Veterinary Diagnostic Laboratory for necropsy.
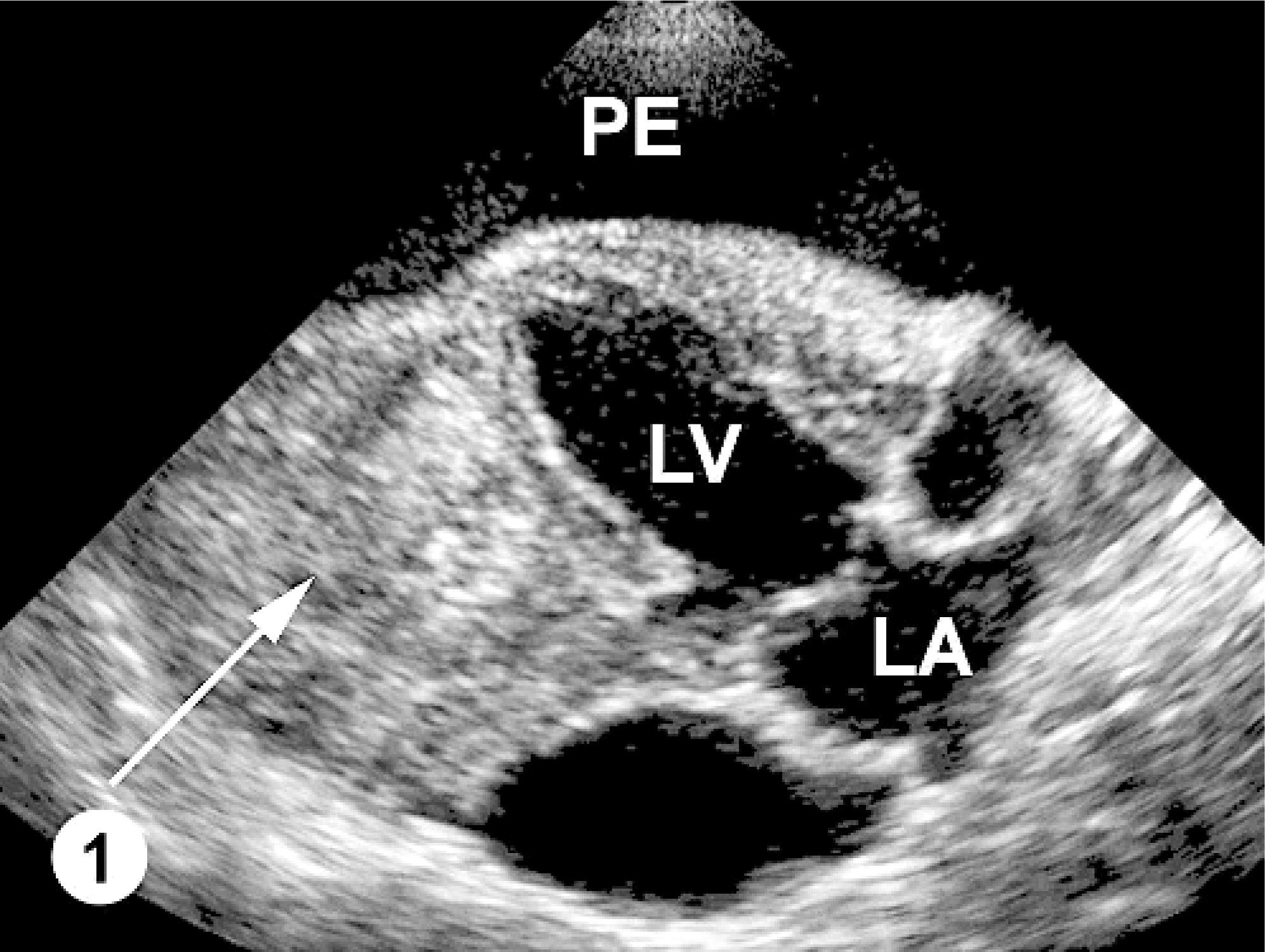
Echocardiogram; canine. Marked pericardial effusion (PE) forms an echolucent zone around the heart. The tumor (
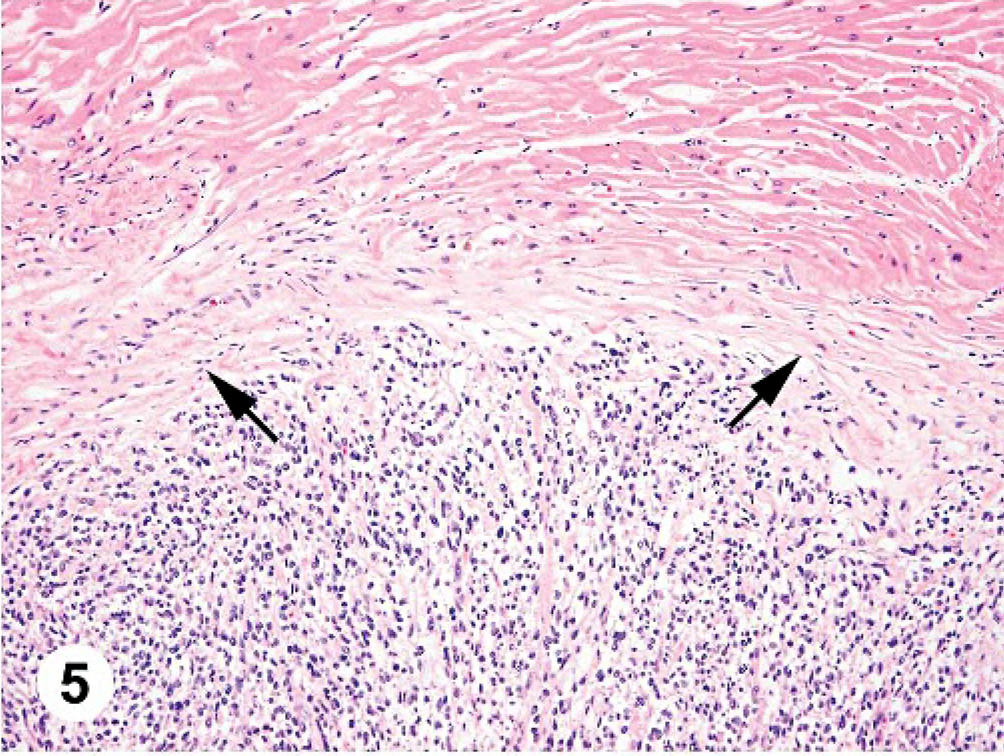
At necropsy, the abdominal cavity contained 3.8 liters of serosanguineous fluid; the thoracic cavity, 0.6 liters; and the pericardium, 0.5 liters. The pericardium was markedly and diffusely thickened (3 mm thick). Attached to the left ventricular free wall myocardium and expanding into the pericardial space was an 11.0 × 7.0 × 6.0 cm, multilobulated, soft, tan, well-demarcated mass. The pericardium had fibrous adhesions to the mass along 50% of the surface of the mass. Over the remaining epicardial surface of the heart, there was a fibrovascular sheet obscuring all normal architecture (Fig. 2). On cut surface, the majority of the mass was mottled tan to red and multilobulated (Fig. 3). The right ventricular free wall was markedly thickened (10 mm wide), and the left ventricular free wall was mildly thickened (20 mm wide); normal heart wall measurements for a dog of this body weight are considered to be 5 mm for the right ventricular free wall and 15 mm for the left ventricular free wall. There was a single, 1-cm-in-diameter, light-tan mass, similar in consistency to the heart mass, in the parenchyma of the right cranial lung lobe (Fig. 4). Tissues collected at necropsy and immersion-fixed in 10% neutral-buffered formalin included heart, pericardium, lung, liver, spleen, intestine, pancreas, kidney, urinary bladder, thyroid gland, adrenal gland, lymph nodes, and brain. Tissues were processed routinely, embedded in paraffin, and 5-μm-thick sections were stained with HE.
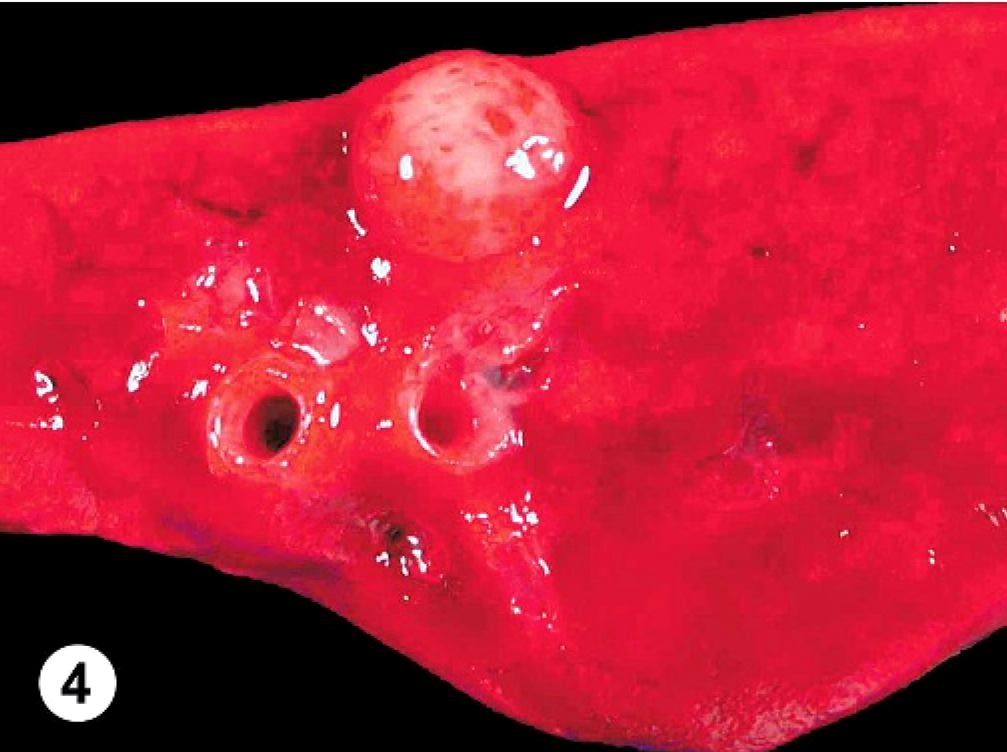
Lung; canine. There was a single, 1 cm-diameter, tan, firm, metastatic nodule within the lung parenchyma.
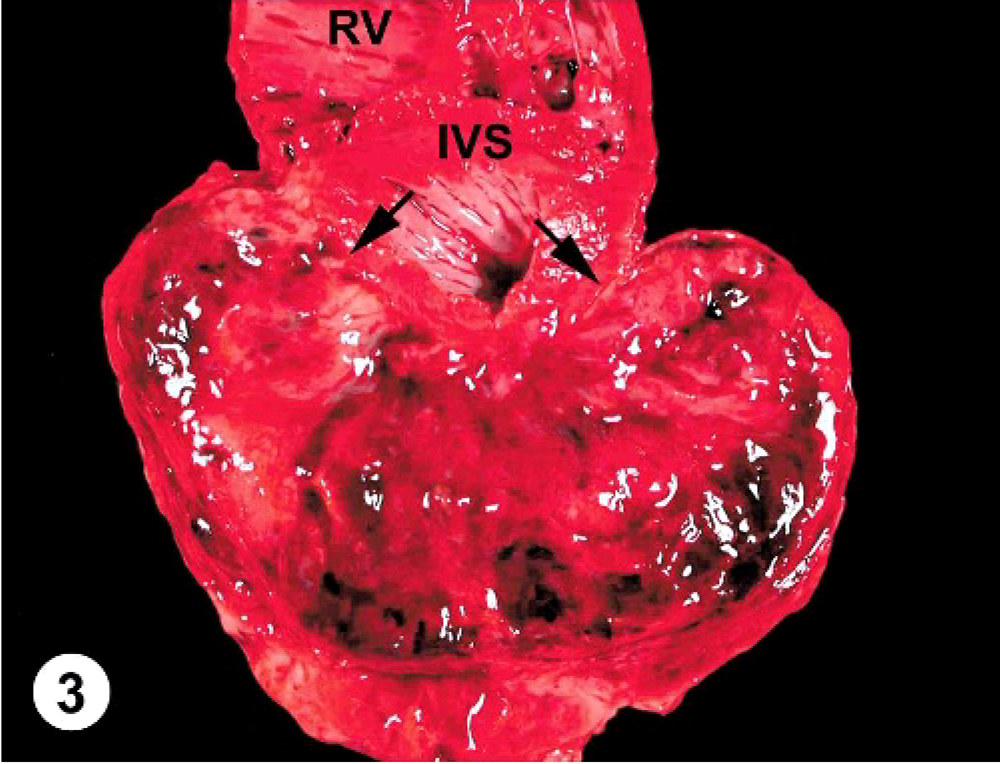
Heart; canine. The cut surface of the mass is mottled tan and red and multilobulated. The mass involves the left ventricular free wall with indistinct demarcation (
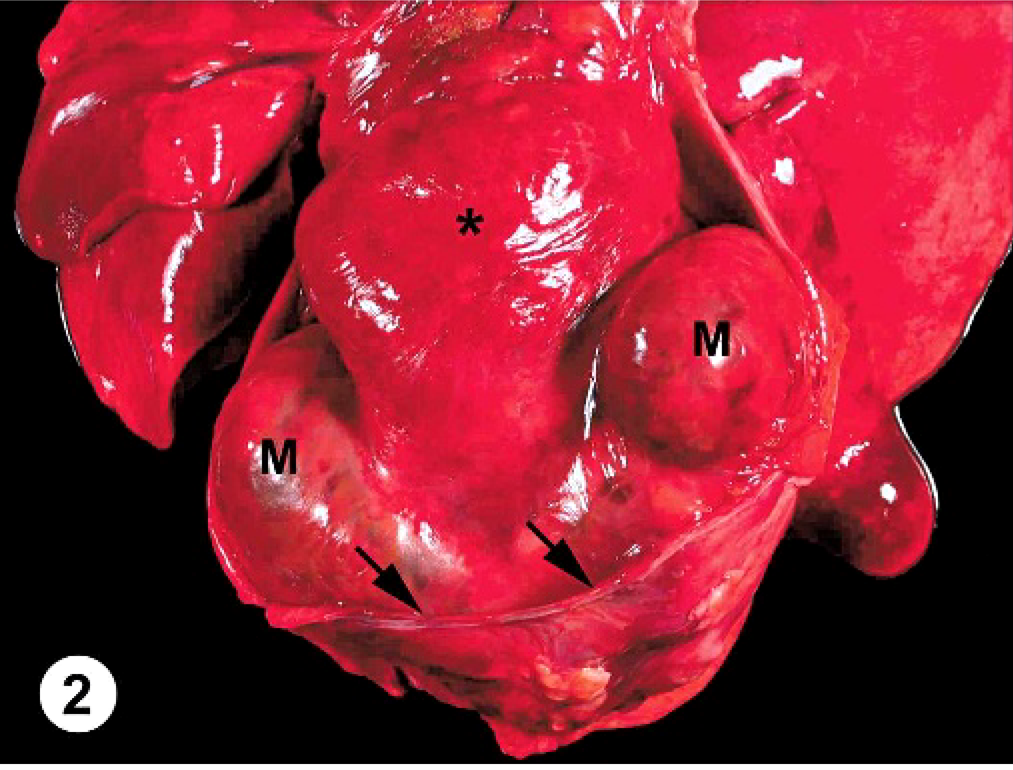
Heart and lungs, pericardium partially removed; canine. A large, multilobulated mass (M) is attached to the left ventricular free wall. The pericardium (
Histologically, the cardiac mass was a well-demarcated, highly cellular, unencapsulated neoplasm involving the subepicardial myocardium of the left ventricular free wall, epicardium, and adjacent pericardium. A compressed layer of collagen separated the neoplastic cells from the cardiac myocytes (Fig. 5). These cells were loosely arranged in sheets and interlacing bands with a scant amount of fine fibrovascular stroma and occasional, thick stromal trabeculae. The neoplastic cells were spindle-shaped with indistinct cell borders and small amounts of eosinophilic, finely fibrillary cytoplasm. The nuclei were ovoid to elongated and centrally located with finely stippled chromatin and single-to-multiple, basophilic nucleoli. The mitotic rate was 1–3 per 400× field (Fig. 6). There were occasional, variably sized, well-demarcated foci of necrosis scattered throughout the mass with slight palisading of the neoplastic cells admixed with infrequent neutrophils at the periphery of the necrosis. There were multifocal hemorrhages within the mass. The pericardium was markedly thickened with collagen, adhered to the mass and contained a small number of infiltrating neoplastic cells and hemosiderin-laden macrophages. Small clusters of neoplastic cells rarely infiltrated the coronary adipose tissue and coronary blood vessels. The entire remaining epicardium was covered by a thick layer of granulation tissue containing numerous hemosiderin-laden macrophages admixed with occasional neutrophils, plasma cells, and lymphocytes. The mass in the lung was unencapsulated and composed of spindle-shaped neoplastic cells similar to those described in the heart. The liver showed signs of chronic congestion with hepatic cord atrophy, mild centrilobular fibrosis, and slight, random hepatocellular necrosis.

Heart tumor; canine. The tumor consists of spindle cells with indistinct cell borders, ovoid nuclei, and finely fibrillary eosinophilic cytoplasm. Multiple mitotic figures are present (
Heart; canine. A compressed layer of collagen (
The tumor was histochemically stained with Masson trichrome, periodic acid–Schiff (PAS), and Alcian blue. The following immunohistochemical stains were prepared using the Dako EnVision/horse radish peroxidase (Dako, Carpinteria, CA) method at dilutions appropriate to the antibody: vimentin (Dako), S-100 protein (Dako), neuron-specific enolase (Dako), glial fibrillary acidic protein (Dako), myoglobin (Dako), pan-cytokeratin (LabVision Corp., Fremont, CA), α-smooth muscle actin (Dako), chromogranin (Dako), melan-A (Dako), Factor VIII-related antigen (Dako), neurofilament protein (Dako), synaptophysin (Dako), and desmin (Dako). Additional immunohistochemical stains using the avidin-biotin-complex detection system at appropriate dilutions included the following: tyrosinase (BioCare Medical, Walnut Creek, CA), MART-1 (Ventana, Tucson, AZ), CD68 (Dako), smooth muscle myosin (Ventana), muscle-specific actin (Ventana), CD117 (Dako), mast cell tryptase (Dako), lysozyme (Ventana), and collagen IV (Dako).
On the basis of the spindle cell morphology, the arrangement of tumor cells, positive staining of the neoplastic cells for vimentin (Fig. 7), and exclusion of other spindle cell tumors by negative results with special stains and a broad spectrum of immunohistochemical stains, a diagnosis of high-grade primary cardiac fibrosarcoma with pulmonary metastasis was made. Differential diagnoses for malignant cardiac spindle cell tumors include malignant peripheral nerve sheath tumor, leiomyosarcoma, rhabdomyosarcoma, malignant fibrous histiocytoma, hemangiosarcoma, myxosarcoma, granular cell tumor, mast cell tumor, malignant melanoma, perivascular epithelioid cell tumor (PEComa), and metastatic sarcomatoid carcinoma.
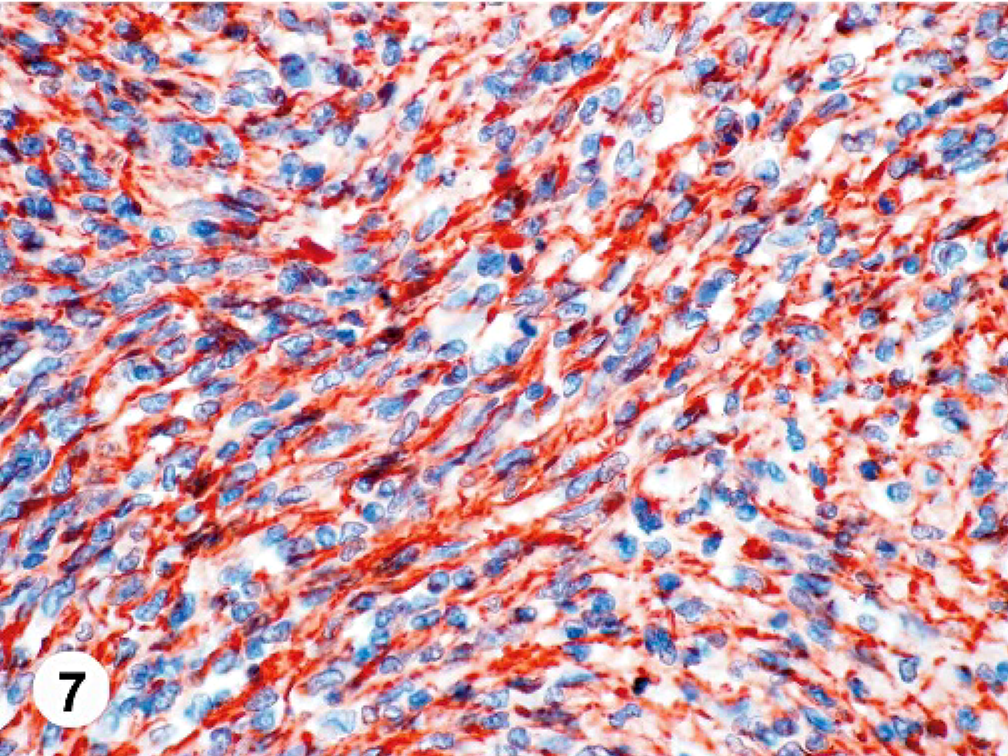
Heart tumor; canine. The cytoplasm of the neoplastic cells stain strongly for vimentin. EnVision/horseradish peroxidase method; Mayer's hematoxylin counterstain.
The origin of the tumor cells in this case was confirmed as mesenchymal by strong cytoplasmic staining with vimentin and no staining for pan-cytokeratin; this also minimized the possibility of a metastatic sarcomatoid carcinoma. Masson trichrome did not highlight significant collagen within the mass, which is typically associated with well-differentiated fibrosarcomas. Malignant peripheral nerve sheath tumors are consistently vimentin-positive,5,7,8,15–17 but stain variably for S-100. In one study, 73% of canine tumors diagnosed as malignant peripheral nerve sheath tumors on HE were positive for S-100.8 Collagen IV staining can be supportive of a diagnosis of a nerve sheath tumor since all tumor cells should have a basement membrane composed of collagen IV. Neither S-100 staining of the neoplastic cells nor collagen IV staining around the neoplastic cells was seen in this case. With the immunohistochemical results for this case and the neoplastic cell arrangement not classic for a nerve sheath tumor, this diagnosis was rejected. Muscle tumors were ruled out by negative immunohistochemistry for myoglobin, α-smooth muscle actin, smooth muscle myosin, muscle-specific actin, and desmin. Macrophage and histiocytic cell origin was evaluated using CD68 and lysozyme, which were negative. An unusual hemangiosarcoma was deemed unlikely since none of the neoplastic cells showed staining with Factor VIII–related antigen. No mucopolysaccharides were seen on an Alcian blue–stained section, which would indicate a myxosarcoma; and no PAS-positive granules were seen, which would be expected in a granular cell tumor. A neural origin of the tumor cells was considered and found improbable by the lack of staining of the neoplastic cells with glial fibrillary acidic protein, neuron-specific enolase, and neurofilament protein. The tumor was not of neuroendocrine origin since chromogranin and synaptophysin were negative. Mast cell markers were negative (mast cell tryptase) or showed only minimal, nonspecific, cytoplasmic staining (CD117). Malignant melanoma can have spindle-shaped cells but the neoplastic cells were negative for S-100, tyrosinase, and melan-A/MART-1, and no melanin granules were seen on HE slides. A PEComa has not been reported in domestic animals, but in humans, this tumor stains positively for smooth muscle actin and the previously listed melanocytic markers. Since all of these markers were negative on the tumor, PEComa was excluded.
Primary cardiac fibro sarcomas are rare in humans and dogs.9,21 In dogs, the fibro prevalence is reported as less than 1% of all primary heart tumors; in humans, the percentage drops to 0.01–0.02%.1,12,21 The reported sites for canine cardiac fibrosarcomas include the right ventricular free wall,1,20 endocardium of the right atrium,11 interatrial septum,20 and interventricular septum.20 None of these cases had metastases.1,11,20 In humans, the most common site of cardiac fibrosarcomas is within the right atrium, but fibrosarcomas can be found anywhere in the heart.2,3,9,10,12–14,19,22 These tumors are aggressive, infiltrate the myocardium and pericardium, and can metastasize to distant sites including the lungs, liver, bone, brain, and skin.19 Other animal species in which primary cardiac fibrosarcomas have been reported include a cat, on the wall of the left atrium;18 a sheep, in the right atrium;4 and a monkey, in the interventricular septum.6
To the authors' knowledge, this is the first report of a primary cardiac fibrosarcoma with a pulmonary metastasis in a dog. Other unique features of this case include the location of the tumor on the left ventricle and the large size of the mass. The mass in the dog in this report had an average diameter of 8 cm, which is more than twice that of other reported primary cardiac fibrosarcomas in dogs (mean, 3 cm; range, 0.5–4.5 cm).1,11,20 The mean diameter on human cases of cardiac fibrosarcoma for which a size was given is 4.5 cm (range, 2–6.6 cm).2,13,14 The exact origin of the tumor in this case cannot be determined but could be the pericardium, epicardium, or myocardium. The diagnosis of a fibrosarcoma is based primarily on excluding other spindle cell tumors because immunohistochemical markers specific for fibrosarcomas in either humans or domestic animals currently do not exist.
Footnotes
Acknowledgements
We thank Jan Shivers and the laboratory staff for their efforts on the immunohistochemistry and special stains for this case. We would also like to thank Dr. Erik Olson for his help with figures and editing.
