Abstract
The epidemiologic role of passerine birds in the spread of highly pathogenic avian influenza virus (HPAIV) remains controversial. However, confirmed natural infections with HPAIV in Passeriformes, their close contact to poultry and humans, and their role as a human food source indicate a need for increased research on passerines. To date, there are only a few studies on viral shedding and pathomorphologic changes in songbirds infected with HPAIV. To investigate susceptibility, clinical outcome, virus spread, and pathomorphology, the authors inoculated oculo-oronasally 22 red-billed queleas (Quelea quelea) and 11 blackcaps (Sylvia atricapilla) with A/Cygnus cygnus/Germany/R65/2006 (H5N1) using 2 different doses of either 104 EID50 (50% egg infective dose) or 106 EID50 per animal. They monitored all birds for clinical signs and oropharyngeal and cloacal virus shedding. They also performed immunohistochemistry and obtained molecular virologic data by real-time reverse transcription polymerase chain reaction in tissue samples. In contrast to blackcaps, where 100% of the infected individuals died, queleas were much less susceptible, with a mortality of 82% and 18%, depending on the doses applied. In both species, the virus was shed within 3 to 6 days postinfection, mainly via the respiratory tract. Viral antigen was detected in 100% of the succumbed birds, particularly in the central nervous system. In blackcaps, the heart, lungs, and pancreas were mainly infected. In contrast, the pancreas was predominantly affected in queleas, whereas the heart and the lower respiratory tract were of minor relevance. The authors hypothesize that neurotropism should be considered a main factor for the fatal course of disease in Passeriformes after infection with HPAIV.
Keywords
Highly pathogenic avian influenza virus (HPAIV) is a striking disease that has led to severe economic losses in susceptible poultry. 26 Although aquatic birds represent the major reservoir of influenza A viruses, several bird species are in principle susceptible to the infection. Influenza A viruses are classified into subtypes on the basis of their hemagglutinin and neuraminidase antigens. 31 To date, 16 hemagglutinin subtypes and 9 neuraminidase subtypes are confirmed. Viruses of H5 or H7 subtype have been defined as notifiable avian influenza viruses (AIVs). 32 This derives from the fact that H5 or H7 virus of low virulence can become virulent during multiple passages in poultry species by mutation. 32 The panzootic HPAIV H5N1 virus spread rapidly through Asia and into Europe and Africa, and wild migratory birds were suspected to play a role in the geographic distribution of HPAIV H5N1, at least in Europe and Africa.15,24
However, the epidemiologic role of passerines regarding the spread of AIV is controversial. Although surveillance studies suggest that the prevalence of influenza A virus in passerines in Europe, North America, Africa, and Asia is rather low,4,6,7,18,19,20,25 several publications report natural infections with AIV, including HPAIV, in passerine species.8,9,17,21,28 Varying morbidity, mortality, and pathomorphologic findings are described in experimental infection studies of house sparrows, stonechats, zebra finches, house finches, and European starlings.3,12,21,23 Brown et al recently provided the first study on infecting passerines using different dosages. 3
Here we investigate whether 2 passerine species—blackcaps and red-billed queleas—are susceptible to the established HPAIV H5N1 isolate A/Cygnus cygnus/Germany/R65/2006.12–14,30 Blackcaps are resident or migratory insectivorous birds in Europe. 2 Amin et al and Gronesova et al already isolated low pathogenic AIV (LPAIV) from birds belonging to the genus Sylvia. 1,8 Red-billed queleas are one of the most abundant wild bird species of the world. They often travel long distances as they migrate in response to seasonal patterns of rainfall to track the availability of their principal food: the seeds of annual grasses. 29 Queleas are a major agricultural pest on grain crops throughout sub-Saharan Africa; they also serve as a food source for humans. 5
The objective of this study was to investigate whether susceptibility, clinical outcome, virus spread, and pathomorphology vary between blackcaps and queleas owing to high- or low-dose infection with HPAIV H5N1.
Material and Methods
Virus Propagation
The third passage of the reference strain A/Cygnus cygnus/Germany/R65/2006 (H5N1) originating from a dead whooper swan found in early February 2006 on the island of Ruegen was used for this study.12–14,30 Allantoic fluid from inoculated embryonated eggs was collected and stored at –70°C until usage. All experiments using HPAIV H5N1 virus were conducted using biosafety level 3+ containment facilities at the Friedrich-Loeffler-Institut (FLI), Insel Riems, Germany.
Virus Isolation and Real-Time Reverse Transcription Polymerase Chain Reaction
All swabs were stored at –70°C until virus isolation was performed. All individual samples were tested with real-time reverse transcription polymerase chain reaction (rRT-PCR) specific for H5, and the genomic load was semiquantified. 10 Virus titers of the oropharyngeal and cloacal/fecal swab samples were calculated as 50% tissue culture infectious dose (TCID50) per milliliter swab sample on Madin-Darby canine kidney cells (collection of cell lines in veterinary medicine, RIE1061, FLI) as described, without supplemental trypsin. 11 The minimal detectable titer was 101.625 TCID50 per milliliter.
Serology
Preexperimental sera were collected from all birds. Sera from birds that survived the experimental setting were also collected. The serum samples were heat inactivated at 56°C for 30 minutes and examined for the presence of antibodies against the nucleoprotein of AIV type A. A commercial epitope-blocking ELISA was used according to the manufacturer’s manual (POURQUIER ELISA Avian Influenza Type A, Pourquier Montpellier, France).
Animals and Experimental Design
Fifteen adult blackcaps (wild caught) were obtained from the Institute of Avian Research, Wilhelmshaven, Germany. Twenty-four adult female red-billed queleas (wild caught) were provided by the Max Planck Institute for Ornithology, Seewiesen, Germany. Animals were housed in individual cages within one stable of the high-containment facility of the FLI, with a 12-hour lighting regimen. Feed and water were provided ad libitum. For sampling procedures, the birds were captured by hand.
The blackcaps were divided into 2 groups and infected with 2 different virus doses. HPAIV H5N1 was inoculated oculo-oronasally with 106EID50 (50% egg infective dose) per animal (3 male and 3 female birds: blackcap high-dose group, BHIGH) or 104 EID50 per animal (3 male and 2 female birds: blackcap low-dose group, BLOW) in 0.25-ml cell culture medium. The infection was performed with an Eppendorf pipette (maximum volume, 0.2 ml) to supply a drop in each eye and nostril. The remaining volume was given into the beak. Two noninoculated animals were used as controls. Similarly, the queleas were divided into 2 groups of 11 birds each and inoculated with 106 EID50 per animal (quelea high-dose group, QHIGH) or 104 EID50 per animal (quelea low-dose group, QLOW). One additional bird of each quelea group served as noninoculated control. The trial gained governmental approval under the registration number LVL M-V/TSD/7221.3-1.1-003/07.
All birds were monitored daily for clinical signs. Oropharyngeal swabs were collected every 3 to 4 days, and samples from individual feces/cloaca were collected almost daily for 21 days in Dulbecco’s modified Eagle medium, supplemented with 5% fetal bovine serum, and antimicrobial drugs (enrofloxacin, 1 mg/ml; gentamicin, 0.05 mg/ml; lincomycin, 1 mg/ml). If an individual bird exhibited neurological symptoms, it was killed humanely.
Gross Pathology, Histopathology, and Immunohistochemistry
Tissues of adrenal gland, bone, bone marrow, cerebrum, cerebellum, esophagus, eye, gizzard, heart, intestine, kidney, liver, lung, skeletal muscles, nasal cavity, gonads, pancreas, proventriculus, skin, spinal cord (thoracic and lumbar spine), spleen, and trachea were sampled from all birds in 4% phosphate buffered neutral formaldehyde and processed for paraffin embedding. Paraffin wax sections (3 to 4 μm) were dewaxed and stained with hematoxylin and eosin. To determine distribution of influenza A antigen, sections were mounted on positively charged SuperFrost Plus microscope slides (Menzel, Braunschweig, Germany) and dewaxed, and inactivation of endogenous peroxidase was performed by using hydrogen peroxide (3%) in methanol (90%) during rehydration procedure. Sections were microwave irradiated for antigen retrieval (2 × 5 min, 600 W, 10 mM citrate buffer, pH 6.0). Briefly, after blocking unspecific background staining with normal goat serum (30 minutes, room temperature), slides were incubated with a rabbit anti-NP serum. This antibody was applied at a dilution of 1:750 in Tris-buffered saline (0.1 M Tris base, 0.9% NaCl, pH 7.6). A biotinylated goat anti-rabbit immunoglobulin G1 (Vector, Burlingame, CA; diluted 1:200 in Tris-buffered saline) was applied for the avidin–biotin complex method. A bright red intracytoplasmatic and nuclear signal was produced with an immunoperoxidase kit (Vectastain Elite ABC Kit, Vector), and the substrate 3-amino-9-ethylcarbazole (DAKO AEC substrate-chromogen system, Dako, Carpinteria, CA). The sections were counterstained with Mayer’s hematoxylin and sealed with aqueous medium (Aquatex, Merck, Darmstadt, Germany). Positive control tissues of chickens experimentally infected with HPAIV H5N1 were included, and a primary rabbit serum against bovine papillomavirus (1:2000) was used as nonrelated control antibody. Autolytic and diminutive tissues were not considered in the analysis.
Results
Mortality and Clinical Signs
Clinical signs were mostly absent in both species and for both infectious doses. Few birds showed ruffled feathers and lethargy or neurologic disorders. The incubation period for the BHIGH group was at least 3 days, and all birds of this group died or had to be humanely killed because of exhibited neurologic disorders (mainly, ataxia) through 6 days postinoculation (DPI; Fig. 1). The minimum incubation period of the BLOW group was 6 days, with 100% mortality by 9 DPI. In strong contrast, 2 birds from the QHIGH group survived the infection, with 9 of 11 birds succumbing to the disease between 6 and 9 DPI (Fig. 1). In the QLOW group, however, only 2 birds died (between 5 and 9 DPI), giving a relatively low mortality rate of 18% (Fig. 1) through the experiment (21 days).
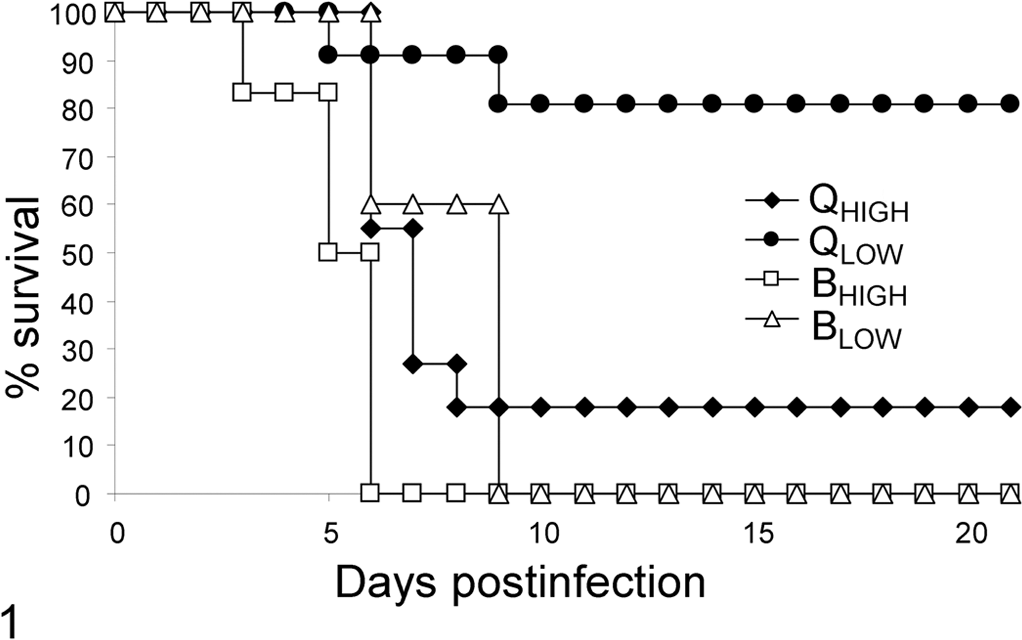
Mortality of blackcaps and red-billed quelea after inoculation with A/Cygnus cygnus/Germany/R65/2006 H5N1 virus; percentage survival of 2 groups of blackcaps and red-billed quelea infected with different dose rates, expressed as mean value of individuals pergroup. The animals were observed daily for a period of 21 days. BHIGH, blackcap high-dose group; BLOW, blackcap low-dose group; QHIGH, quelea high-dose group; QLOW, quelea low-dose group.
Serology
All preexperimental sera from the individual birds tested negative for antibodies against the nucleoprotein of influenza A.
Sera from surviving queleas were retested. Both surviving individuals of the QHIGH group reacted positive, whereas all 9 surviving birds of the QLOW group did not seroconvert.
Virus Isolation and rRT-PCR
HPAIV H5N1 shedding in oropharyngeal and fecal swabs was monitored daily by virus titration on Madin-Darby canine kidney cells. Shedding in fecal samples could be demonstrated from 3 DPI through 4 DPI for the BHIGH and BLOW groups (Table 1). Viral excretion from fecal samples was verified on 3, 4, and 6 DPI for the QHIGH group, whereas fecal shedding from the QLOW group was restricted to 3 DPI. Viral excretion from oropharyngeal samples was detected in both species and both dose groups on 3 DPI. Furthermore, in all populations, rRT-PCR revealed positive viral genome for the same period, whereas all swab samples taken from control birds remained negative (data not shown). Generally, viral titers in the oropharyngeal swabs were slightly higher than titers detected in fecal samples (Table 1). The maximum of infectivity was detected from swab samples taken from deceased birds (Table 1).
Viral Shedding Detected on Cell Culture From Oropharyngeal and Fecal Swab Samples of Blackcaps and Queleas After Experimental Infection With Highly Pathogenic Avian Influenza Virus H5N1 a
a Queleas and blackcaps inoculated oculo-oronasally with 106 EID50 (50% egg infective dose) per animal (QHIGH and BHIGH) and 104 EID50 per animal (QLOW and BLOW). DPI, days postinoculation.
b Oropharyngeal shedding (OS): sampling on 3, 7, 14 DPI; the number indicates the DPI on which virus was isolated in each group. Fecal shedding (FS): sampling every day.
c Number of individuals alive shedding virus per infected birds.
d Log 10 TCID50 (50% tissue culture infectious dose) per milliliter swab, with DPI in parentheses.
Organs from the tested passerines showed a range of RNA loads, with pancreas and nasal cavity representing the highest copy numbers in the quelea groups (Table 2). In contrast, both groups of blackcaps showed extraordinarily high genome loads in the central nervous system (CNS) and pancreas (Table 3).
Distribution of Influenza A Antigen and Viral Genomic Load in Tissues of Queleas After Experimental Infection With Highly Pathogenic Avian Influenza Virus H5N1 a
a Queleas inoculated oculo-oronasally with 106 EID50 (50% egg infective dose) per animal (11 birds, QHIGH) and 104 EID50 per animal (11 birds, QLOW).
b Affected organs and their maximum antigen staining: +++, diffuse; ++, multifocal; +, focal; –, negative. Incongruent animal numbers are due to tissues not being considered, because of autolysis or size. CNS, central nervous system.
c Viral RNA detected by real-time reverse transcription polymerase chain reaction in birds after challenge infection with highly pathogenic avian influenza virus strain A/Cygnus cygnus/Germany/R65/06 H5N1. Data are presented as cycle of threshold (CT) values: > 40 scored as negative.
d Cumulative reverse transcription polymerase chain reaction result: adrenal/gonads.
Distribution of Influenza A Antigen and Viral Genomic Load in Tissues of Blackcaps After Experimental Infection With Highly Pathogenic Avian Influenza Virus H5N1 a
a Blackcaps inoculated oculo-oronasally with 106 EID50 (50% egg infective dose) per animal (6 birds, BHIGH) and 104 EID50 per animal (5 birds, BLOW).
b Affected organs and their maximum antigen staining: +++, diffuse; ++, multifocal; +, focal; –, negative. Incongruent animal numbers are due to tissues not being considered, because of autolysis or size. CNS, central nervous system.
c Viral RNA detected by real-time reverse transcription polymerase chain reaction in birds after challenge infection with highly pathogenic avian influenza virus strain A/Cygnus cygnus/Germany/R65/06 H5N1. Data are presented as cycle of threshold (CT) values: > 40 scored as negative.
d EN: marked antigen staining in endothelial cells of 1 bird of
e Cumulative reverse transcription polymerase chain reaction result: adrenal/gonads.
Gross Pathology
At necropsy, the predominant lesions in most birds were mild to moderate edema and congestion of the lungs. Three blackcaps (2 in BHIGH, 1 in BLOW) showed multiple, sharply demarcated, pale white foci (up to 2 millimeters in diameter) in the pancreas, and the surrounding parenchyma was diffusely gray-red.
Histopathology
Tables 2 and 3 give an overview of the observed histopathologic results. Explicit description is restricted to findings in succumbed animals. We focused on predominantly affected organs and extraordinary findings in the CNS (cerebellum, cerebrum, spinal cord), pancreas, lung, heart, spleen, and eye.
Influenza virus antigen was found in 100% of the experimentally infected blackcaps. In queleas, the detection was restricted to birds that succumbed to the disease.
Immunoreactivity was observed within the CNS of all succumbed birds. Immunostaining was detected in neurons and glial and ependymal cells associated with neuronal necrosis, neuronophagia, neuropil vacuolization, and partly, microgliosis (Figs. 2–4). Viral antigen was also present in neurons of the peripheral nervous system. In particular, immunostaining was observed in neurons of intramural ganglia of the intestinal tract, in ganglia adjacent to the adrenal gland, and in the nervus trigeminus. Pancreatic acinar cells were affected in all succumbed queleas and in 2 out of 4 BLOW birds and in 4 out of 4 BHIGH birds. Pancreatic necrosis was widespread and inconstantly accompanied by mixed cellular infiltrates.
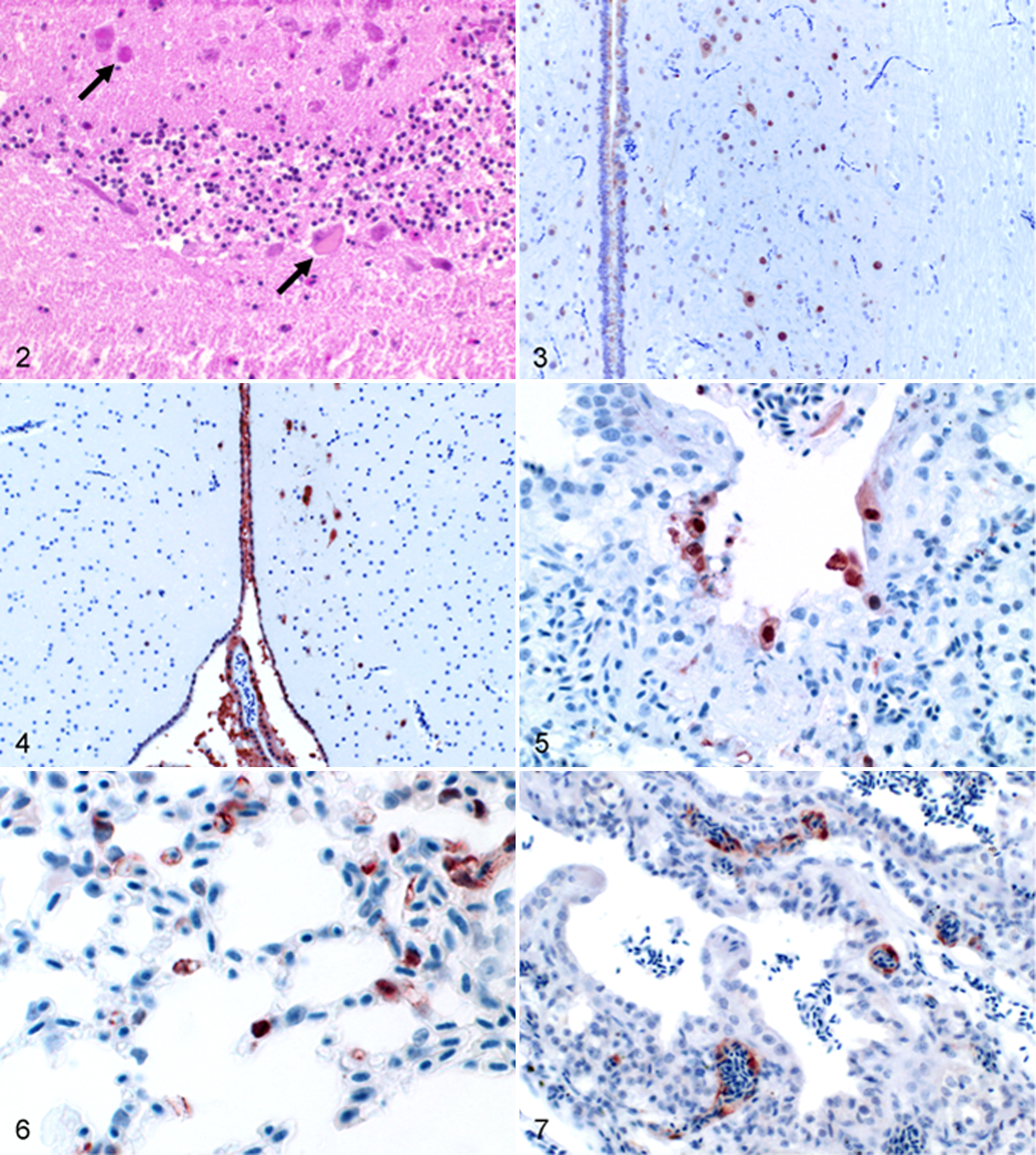
In the lungs, H5N1 viral antigen distribution differed, depending on species and dosage. In blackcaps, 5 out of 6 birds in BHIGH and 2 out of 5 birds of BLOW exhibited influenza antigen in parabronchial epithelium and, to a lesser extent, in pneumocytes of air capillaries (Figs. 5, 6). The queleas were affected far less, with antigen detection only in 1 bird of QLOW and 2 birds of QHIGH. In both species, alveolar edema and congestion were obvious, and bronchial-associated lymphoid tissue within the lungs showed abundant necrosis of lymphocytes, even in birds without antigen signals in the parenchyma. The blackcaps exhibited interstitial pneumonia with predominant mononuclear infiltrates and a focal expanding bronchial necrosis. Hypereosinophilic cardiac myocytes with vacuolar degeneration accompanied by mononuclear myocarditis were detected in most blackcaps. Immunostaining was present in cardial myocytes (2 of BHIGH and 6 of BLOW) and endothelial cells (1 of BHIGH). In queleas, myocarditis and myocardial degeneration were exclusively found in 3 birds, including both groups, and antigen was present in the aortic media of 2 birds. Regarding the lymphatic system, no antigen was found within the spleen. Whereas the blackcaps exhibited an immune response with an increased number of germinal centers and reticuloendothelial hyperplasia, no reaction could be ascertained in queleas. Besides this, 2 blackcaps exhibited lymphocyte necrosis in the spleen. Concerning the eye and adnexa, we detected immunostaining in endothelial cells of the pecten oculi and in corneal epithelium in 1 quelea (QLOW). One additional bird (QHIGH) was affected in the epithelium of a lacrimal gland. Particularly, the Harderian gland was affected in individual birds (3 in BHIGH, 2 in QHIGH, 1 in BLOW, 1 in QLOW,) with necrotic glandular epithelium staining strongly positive and a dense plasma cellular infiltration. Remarkably, 1 blackcap of the BHIGH group, which died at 3 DPI, showed strong and widespread intracytoplasmatic and nuclear immunostaining in endothelial cells of the CNS, esophagus, eye, gizzard, Harderian gland, intestine, kidney, liver, lung, spleen, and trachea (Fig. 7).
All samples of control animals and the following samples of infected birds stained negative and did not reveal histologic lesions: bone, bone marrow, skeletal musculature, skin with adnexes, and proventriculus.
Discussion
Our results strongly suggest that the virulence of a recent HPAIV H5N1 virus strain is determined by the dose and has considerable variability depending on the species—even for species belonging to the same order. In accordance with experiments in Passeriformes performed by Brown et al, the mean time to death increased with reduction of the doses, and the mortality rate decreased. 3 Queleas and blackcaps are both susceptible, but queleas are to a much lesser extent. Whereas all infected blackcaps succumbed to the disease, 2 queleas survived the infection with 106 EID50 and 9 survived infection with 104 EID50.
The virus was shed in respiratory secretions and feces up to 6 DPI, where oropharyngeal swabs contained generally higher viral titers (and genomic load), compared with fecal samples. These findings are in accordance with data from other Passeriformes species. 3 Interestingly, viral excretion could not be demonstrated for every individual bird, and swab samples taken from succumbed birds clearly revealed the highest amount of infectious virus. Furthermore, our necropsy findings confirmed that pancreatic necrosis is the most obvious and consistent lesion in Passeriformes.3,12,17,23,28
Immunohistologic investigation provided detection of influenza virus antigen in 100% of the succumbed birds. Affected organs underwent degeneration, and parenchymal necrosis was prevalent. Remarkably, neurotropism was constantly observed in both species and both doses. In general, viruses can enter the CNS via ascending neuritic infection, via the blood circulation, or via the cerebrospinal fluid. Regarding the staining pattern in the CNS (particularly in blackcaps), one could speculate that the infection of the ependymal cells was followed by a spread throughout the brain (Fig. 4). Ependymal cells of the ventricles and the central canal were affected expansively, and neurons adjacent to the ventricles or the central canal stained positive. We therefore hypothesize that the infection of the brain is a consequence of virus transmission via the cerebrospinal fluid. To test this hypothesis, further experiments are needed with time-dependent euthanasia. In addition to ependymal infection, epitheliotropism was observed in a large variety of organs. In queleas, the pancreas acini cells were constantly affected in the 11 deceased birds. Approximately 50% showed immunoreactivity in the intestine (crypt epithelium, 1 bird; intramural ganglia, 5 birds), nasal cavity, adrenal gland, and ovary. The heart, trachea, and lower respiratory tract were of minor relevance in queleas. In contrast, the predominant organs affected in blackcaps were the heart, lungs, and pancreas with more than 60% positive staining in succumbed birds. Furthermore, there was 1 blackcap with widespread endotheliotropism (Fig. 7), which died 3 DPI, the earliest time point. However, with regard to the small number of animals per group and to recently published data of Kalthoff et al 12 into account, the causal relationship of endotheliotropism to a rapid course of disease should be interpreted with caution. The observations in stonechats imply that the endotheliotropism is not related to the survival time.
Lymphocyte necrosis has been reported in gallinaceous species after HPAIV infection. 22 Tumor necrosis factor α, transforming growth factor β, and other cytokines were discussed to play a role in inducing lymphocytolysis via apoptosis, culminating as lymphocellular necrosis. Virus-infected macrophages releasing cytokines accompanied by a decreased ability to phagocyte degenerating lymphocytes might facilitate the outcome of marked lymphocyte necrosis in lymphoid tissues. 22 Regarding the role of macrophages in cytokine release and their inability to phagocyte degenerated lymphocytes efficiently, we follow Perkins and colleagues' line of argument. 22 However, to the best of our knowledge, tumor necrosis factor α has not been detected in chickens to date. 27
Our data suggest that neurotropism should be considered the main factor for the fatal course of disease in Passeriformes after infection with HPAIV. This assumption is confirmed by published data from experimental infection as well as natural infection.12,17,21,23,28
However, Brown et al 3 hypothesized that death was likely due to failure and/or dysfunction of multiple critical organs, including the pancreas, adrenal gland, heart, or liver. They experimentally infected house sparrows with a recent H5N1 HPAIV isolate in three different dosages. Histopathologic investigation was restricted to the group infected with 106 EID50. They showed viral antigen in several tissues, including the pancreas, brain, heart, air sacs, adrenal gland, and liver.
In the context of surveillance studies, more than 2,400 Passeriformes birds in Europe, Asia, America, and Africa tested negative for AIVs during 1993 and 2006.4,6,18,19,20,25 In contrast, Gronesova et al8,9 detected several LPAIV subtypes in passerines, particularly in blackcaps. The birds, captured at Trnava ponds, nested in the same locality with waterfowl, the reservoir hosts for LPAIV. Additionally, HPAIV H5N1 has been detected in live tree sparrows (Passer montanus) in China, in dead large-billed crows (Corvus macorhynchos) in Japan, in dead Korean magpies (Pica pica sericea).16,17,28
In summary, although the prevalence of AIV in Passeriformes is rather low and although passerine contact with more affected species (eg, ducks) is also low, the relevance of these birds in viral spread remains unclear. Considering the close contact of passerines to poultry and humans and their role as a food source for humans, future studies aimed at clarifying the role of passerines are badly needed. To date, there are only a few studies available regarding viral shedding and morphologic changes in response to infection (in particular after using different dosages). Therefore, further investigations regarding contact transmission and viral shedding would be extremely helpful to assess the epidemiologic role of Passeriformes. The combination of immunohistochemistry and molecular virologic data obtained by rRT-PCR in tissue samples and swab samples resulted here in a much better understanding of the epidemiologic aspects and pathogenesis following infection with HPAIV. The importance of neurotropism in the course of the disease should be reflected in tissue sample evaluation. We suggest that, besides cerebrum and cerebellum, the spinal cord and peripheral nervous system be included for evaluation. The distribution pattern within the CNS might give a critical hint to the route of infection.
Footnotes
Acknowledgements
We thank Gerda Busch and Mareen Grawe for excellent technical assistance and Thorsten Arnold for very skillful animal husbandry.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was financially supported by Forschungs-Sofortprogramm Influenza des Bundes project Nos. FSI 4.1 and FSI 2.42.
