Abstract
Macaques provide an important animal model for the study of hormonal agents and their effects on risk biomarkers for breast cancer. A common criticism of this model is that spontaneous breast cancer has rarely been described in these animals. In this report, we characterize 35 mammary gland lesions ranging from ductal hyperplasia to carcinoma in situ and invasive ductal carcinoma in cynomolgus and rhesus macaques. Based on a retrospective analysis, we estimated the lifetime incidence of mammary gland neoplasia in aged female macaques to be about 6%. Hyperplastic lesions (n = 19) occurred segmentally along ducts and included such features as columnar alteration, micropapillary atypia, and fibroadenomatous change. in situ carcinomas (n = 8) included solid, comedo, cribriform, and micropapillary elements, encompassing 4 of the major architectural patterns seen in human lesions. invasive ductal carcinomas (n = 8) were generally solid, with prominent central necrosis and mineralization, often on a background of micropapillary ductal hyperplasia and in situ carcinoma. Cytologic changes of invasive lesions included increased mitoses, nuclear pleomorphism, extensive microinvasion, and stromal desmoplasia. Axillary lymph-node metastases were confirmed in 5 of the 8 invasive carcinomas. on immunohistochemistry, intraductal and invasive carcinomas had increased Ki67/MIB1 and HER2 expression and selective loss of estrogen and progesterone receptors. These findings suggest that breast cancer is an underreported lesion in macaques and highlight unique morphologic and molecular similarities in breast cancer between human and macaque species.
Introduction
The mammary glands of macaques are similar to those of women in many aspects of physiology and pathology. 15, 34, 40, 44 Human and macaque mammary glands share similar patterns of development and regression, cytokeratin phenotype, sex steroid receptor expression, mechanisms of glandular secretion, and steroidogenic enzyme expression. 28, 31, 44, 49 Female macaques also exhibit reproductive characteristics comparable with women, including a 28-day menstrual cycle, 15 natural ovarian senescence, 24 and reproductive tissue responses to exogenous estrogens. 12, 13 Despite these similarities, mammary gland tumors have been described only sporadically in macaques, 1, 4, 14, 19, 23, 27, 30, 45, 46, 54 while representing one of the primary causes of cancer death in women. 29 This apparent species difference in breast cancer incidence is a common criticism of the macaque model for studying biomarkers of breast cancer risk. In this report, we describe a collection of spontaneous hyperplastic and neoplastic mammary gland lesions in cynomolgus and rhesus macaques. The goal of this study was to characterize these lesions based on morphology, proliferation, sex steroid receptors, and oncogene expression, and to evaluate similarities with human breast cancer.
Materials and Methods
Animal histories
The animals in this case series were adult rhesus (
Characteristics of macaques with hyperplastic mammary gland lesions.∗
OVX = ovariectomized; Mm = Rhesus macaque,
Biopsy taken 6 months before case No. 21 in Table 2 (same animal).
Received soy isoflavones at doses comparable with those in standard monkey chow.
Characteristics of macaques with neoplastic mammary gland lesions.∗
OVX = ovariectomized; Mm = Rhesus macaque,
Biopsy taken 6 months after J706 in Table 1 (same animal).
Treated with danazol for endometriosis.
Sequential biopsies taken 22 m apart from the same animal-1768 is the recurrent tumor (contralateral to 97B3) collected at necropsy.
Received oral dehydroepiandrosterone 10 years before death.
Treated with E2 benzoate 5 years before death.
Male monkey.
Received dietary soy isoflavones at doses comparable with those in standard monkey chow.
Estimated incidence of mammary gland lesions in macaques.
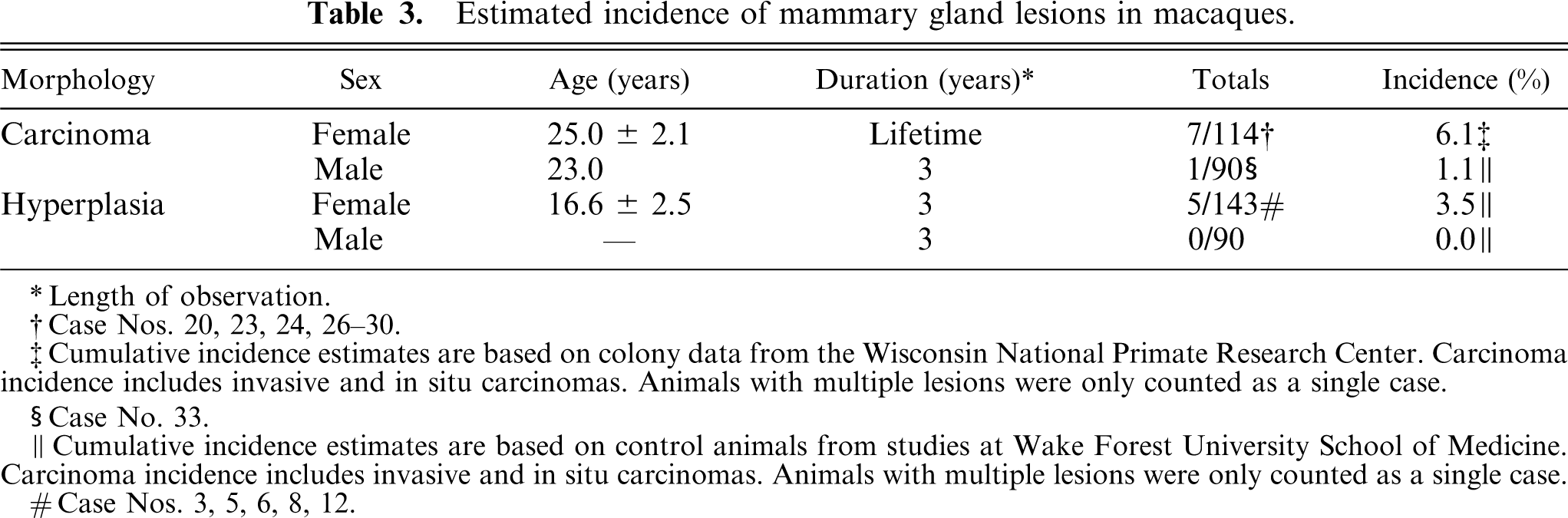
Length of observation.
Case Nos. 20, 23, 24, 26-30.
Cumulative incidence estimates are based on colony data from the Wisconsin National Primate Research Center. Carcinoma incidence includes invasive and in situ carcinomas. Animals with multiple lesions were only counted as a single case.
Case No. 33.
Cumulative incidence estimates are based on control animals from studies at Wake Forest University School of Medicine. Carcinoma incidence includes invasive and in situ carcinomas. Animals with multiple lesions were only counted as a single case.
Case Nos. 3, 5, 6, 8, 12.
Case animals varied in age (range: 5 to 35 years, mean: 18.6 years) and hormonal status, including premenopausal, ovariectomized, and naturally postmenopausal female macaques and 1 male macaque. None of the case animals were pregnant. The animals had been fed various commercial monkey chow formulations supplemented with fruit (CNPRC, WNPRC, UTMDA, NIH) or custom formulated diets (WFUSM), all of which met the nutrition requirements of the National Research Council for nonhuman primates. A subset of monkeys received various oral hormonal agents (Tables 1 and 2), which included conjugated equine estrogens with (
Histopathology
Lesions were identified either grossly or during routine histologic examination of mammary gland tissues. All mammary tissues were fixed in 10% neutral buffered formalin upon removal, although we cannot verify that all fixation procedures (e.g., time in fixative) were uniform across the various contributing institutions. For routine evaluation, the glands were sectioned sagittally through the nipple and trimmed to 2–3 cm in length. Fixed tissues were embedded in paraffin and sectioned to 5 µm in thickness for hematoxylin and eosin (HE) and immunohistochemical staining.
HE stained slides were evaluated qualitatively for histologic changes by multiple investigators (CEW, JMC, and the submitting pathologist for each case) and also were reviewed with an MD pathologist (KRG) for consistency with human terminology. Ductal hyperplasia was defined as increased cellularity of the ductal epithelium beyond that seen under physiologic conditions and in which the epithelial layer is at least 3 cell layers in thickness (including myoepithelium). Ductal hyperplasias were further classified as conventional (DH) or atypical (ADH); a diagnosis of ADH required either cytologic or architectural atypia. Lobular hyperplasia was defined by distention and distortion of <50% of the acini in a lobule by atypical lobular epithelial cells. Focally enlarged lobular units with regular acini were considered physiologic and were not included as lesions. Ductal carcinoma in situ (DCIS) was defined by proliferation of ductal cells that have malignant architectural and cytologic features but have not breached the basement membrane. 32 Atypical architectural features included comedo, cribriform, papillary, solid, and micropapillary patterns; cytologic features of atypia included loss of polarity, uneven distribution of nuclei, increased mitoses, anisokaryosis or anisocytosis, and necrosis. Lobular carcinoma in situ (LCIS) was defined by distention and distortion of >50% of the acini in a lobule by atypical lobular epithelial cells. 35 Human lobular carcinomas frequently undergo a specific chromosomal alteration resulting in loss of the adhesion molecule E-cadherin. 53 Equivocal in situ lesions were thus immunostained for E-cadherin to assess lobular (−) or ductal (+) origin of neoplastic cells. Invasive ductal carcinomas (IDC), the most common type of malignant breast cancer in women, 41 were defined by extensive stromal invasion and/or evidence of lymph-node metastasis and vascular invasion. The modified Scarff-Bloom-Richardson method was used for histologic grading of invasive carcinomas. Under this system, grade is assigned based upon the degree of tubule formation, nuclear pleomorphism, and mitotic counts. 17 For cases with multiple diagnoses (e.g., DCIS with IDC), the highest-grade lesion was used for classification and evaluation. Two case pairs are included (case Nos. 2 and 21, 24 and 30) in which sequential samples yielded different diagnoses.
Immunohistochemistry
Immunostaining procedures were performed on fixed, paraffin-embedded tissues by using commercially available primary monoclonal antibodies for Ki67/MIB1 (Ki67-MIB1; Dako, Carpinteria, CA), progesterone receptor (PR) (NCL-PGR; Novocastra, Newcastle upon Tyne, UK), estrogen receptor alpha (ERα) (NCL-ER-6F11, Novocastra, UK), estrogen receptor beta (ERβ) (MCA 1974, clone PPG5/10; Serotec, Raleigh, NC), HER2/neu (Herceptest, Dako, Carpinteria, CA), and E-cadherin (4A2C7; Zymed, San Francisco, CA). Staining methods included antigen-retrieval with citrate buffer (pH 6.0), biotinylated rabbit anti-mouse Fc antibody as a linking reagent, alkaline phosphatase–conjugated streptavidin as the label, hematoxylin as the counterstain, and Vector Red as the chromogen (Vector Laboratories, Burlingame, CA). Ki67/MIB1, PR, ERα, and ERβ staining was quantified by a computer-assisted counting technique, by using a grid filter to select cells for counting 33 and a modified procedure of cell selection, described previously. 12 Numbers of positively stained cells were expressed as a percentage of the total number examined (200 cells). At least 3 separate microscopic fields were surveyed for each slide. By convention, HER2 immunoreactivity was assessed by using the standard Food and Drug Administration approved Herceptest scoring system, with the following categories: negative (0/1+), if <10% of neoplastic cells had membrane staining; weakly positive (2+), if >10% of the tumor cells had mild to moderate membrane staining; or strongly positive (3+), if >10% of tumor cells showed strong complete membrane staining.
All immunostaining batches included negative staining control slides treated with species-matched nonimmune serum in place of the primary antibody. The majority of slides with lesions (18/19 hyperplasias and 14/16 carcinomas) had at least a small amount of morphologically normal mammary gland present on the margins of the tissue, providing an additional internal staining control for these cases. Slides from a set of normal postmenopausal macaque mammary glands were also stained and counted as reference controls. We cannot exclude that potential differences in fixation may have introduced variation in immunostaining across lesions. Immunohistochemical counting was performed on a subset of hyperplasia cases because of loss of some lesions with sectioning. One carcinoma case (case No. 26) was excluded from immunohistochemical evaluation because of a complete lack of immunostaining in tumor or normal adjacent tissue, presumably because of improper fixation.
Statistics
Data were analyzed by using the SAS statistical package (version 8; SAS Institute, Cary, NC). Group means and intergroup comparisons were determined by using a general linear model. Lesion types were sorted into the following categories: normal, ductal hyperplasia, LCIS, DCIS, and IDC. Correlations between immunohistochemical counts and morphologic criteria were evaluated by using Spearman's rank correlation test. Log10 transformations were performed where appropriate to improve normality and homogeneity of variance. A 2-tailed significance level of 0.05 was chosen for all comparisons.
Results
Incidence and morphology
Macaque mammary gland lesions were classified as ductal hyperplasia (
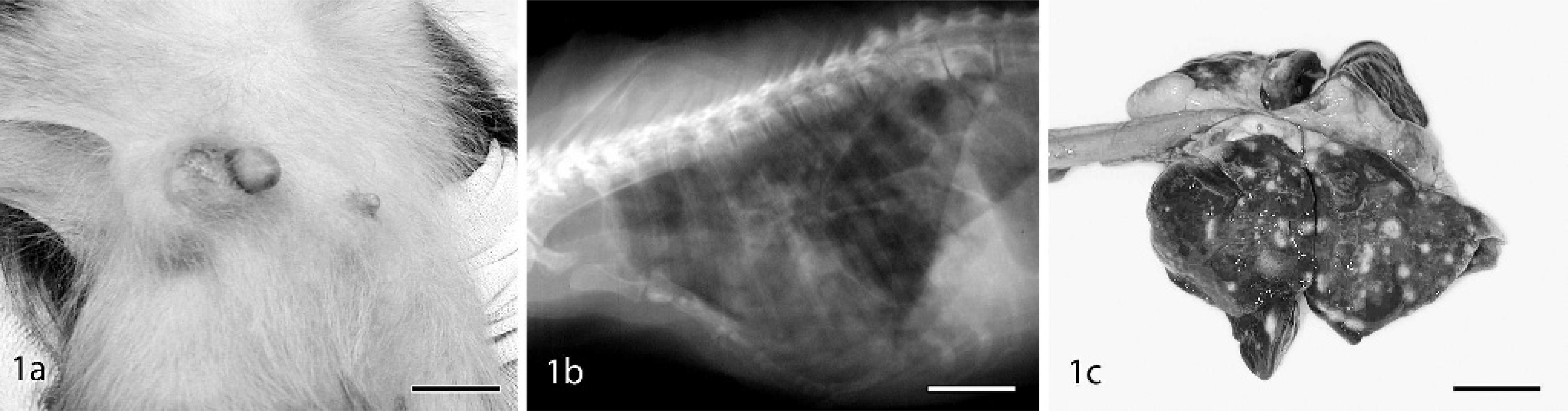
Cynomolgus macaque, case No. 34.
The lifetime incidence of mammary gland carcinoma (in situ and invasive) in female macaques was estimated at 6.1%, based on data from a population of aged animals at the WNPRC (Table 3). The single section incidence of mammary hyperplasia in younger female control animals in studies at WFUSM was 3.5%. While originating from distinct populations, animals with carcinomas were significantly older than those with hyperplastic lesions (
Proliferative lesions showed a high degree of morphologic heterogeneity (Figs. 2–3). Hyperplastic mammary gland lesions ranged from mild to florid and included such general features as piling up of epithelial cells into mounds, tufts, and micropapillary stalks; cystic dilation of ducts; luminal bridging; and variable secretory changes. Architectural atypia was found in 9 of the 19 (47%) hyperplasia cases, with segmental micropapillary change representing the most common atypical growth pattern (Fig. 2b). Several cases included prominent expansion of the stroma in a loose myxomatous pattern resembling fibroadenomatous change (Fig. 2c). A common component of most hyperplastic ductal lesions was columnar alteration (also referred to as columnar cell metaplasia), similar to that described in the human literature. 22, 42 In this change, ductal epithelial cells become densely packed and stratified, with elongate hyperchromatic nuclei, sometimes with apical cytoplasmic “snouts” or secretory blebs (Fig. 3a). Ductal hyperplasia with columnar alteration was a prominent change easily distinguished from the more diffuse and regular nuclear rounding and enlargement produced by hormone treatment. In several cases, hyperplastic ducts were present within or adjacent to regions of ductal and lobular apocrine metaplasia; in the latter case, luminal epithelial cells often contained bright red intracytoplasmic secretory granules (more conspicuous than those seen in the human breast).
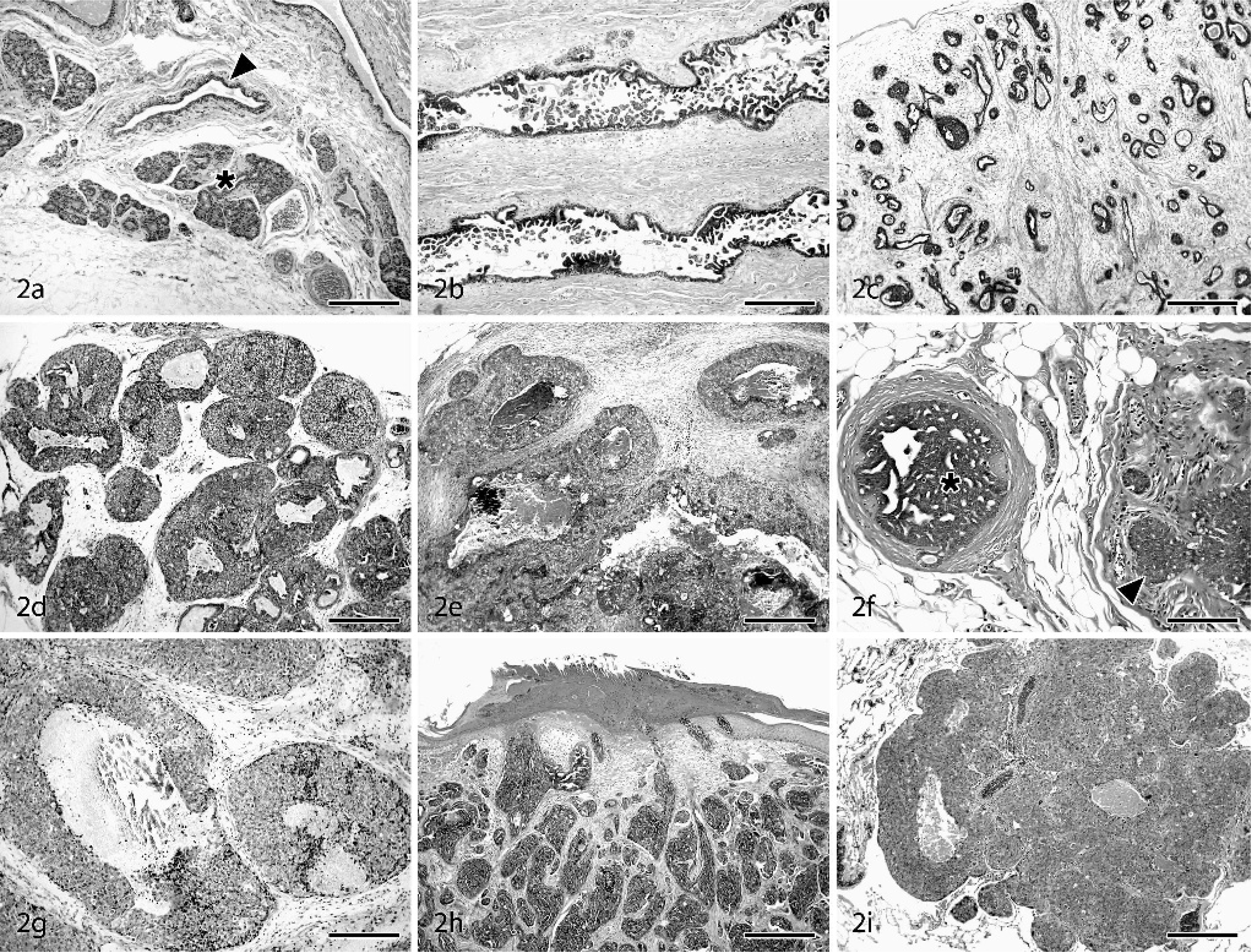
Mammary glands: cynomolgus and rhesus macaques. HE. Bars = 500 µm, unless otherwise stated.
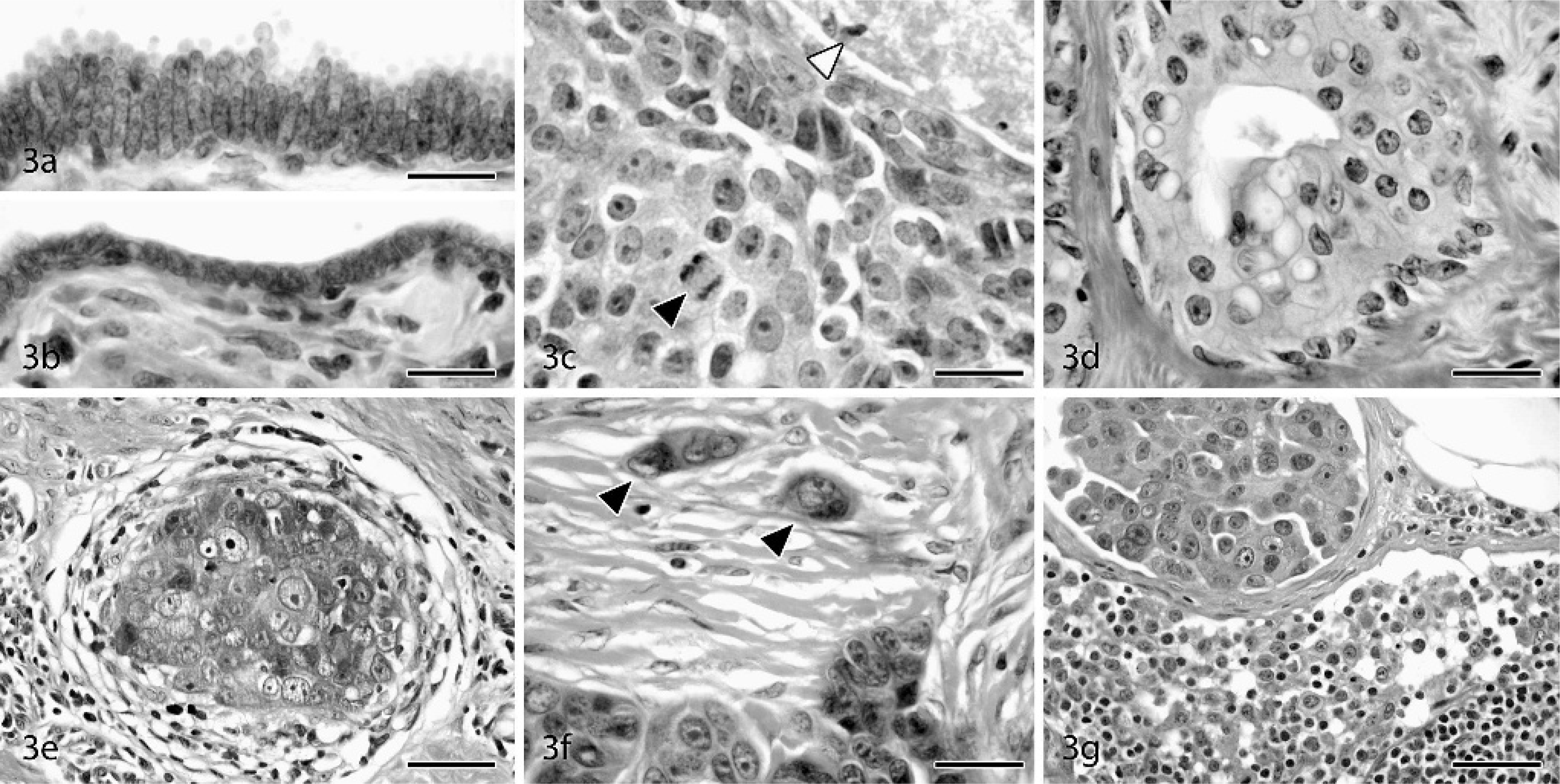
Mammary glands: cynomolgus and rhesus macaques. HE.
In situ ductal carcinomas incorporated 4 of the 5 major morphologies described for human lesions: comedo, solid, cribriform, and micropapillary. Several different growth patterns were often present within a single lesion, with solid and comedo types representing the most common combination. Lower magnification features of DCIS included marked expansion and filling of ducts by sheets of epithelial cells (Fig. 2d) and prominent comedo-type necrosis in 4 of 5 DCIS (case Nos. 21, 22, 24, 33) (Fig. 2e) and 1 of 3 LCIS lesions (Case No. 20). In situ carcinomas were commonly surrounded by a thin rim of capillaries and variable amounts of fibrosis. Cells within DCIS lesions often had increased mitotic rates, greater nuclear pleomorphism, prominent nucleoli, and a general lack of polarity and cohesion (Fig. 3c). Regions of several in situ carcinomas showed a dimorphic pattern with simple cuboidal surface epithelium overlying expansile populations of disorganized neoplastic cells, suggesting intraductal spread beneath the luminal epithelial layer. Five of the DCIS lesions were found within regions of conventional and ADH, and sequential biopsies in 1 case revealed regional progression from ADH to DCIS (case Nos. 2, 21). Extensive apocrine change was present throughout 1 LCIS (case No. 20) and 1 DCIS lesion (case No. 24) (Fig. 3d). Squamous differentiation was also present multifocally in 1 DCIS case along the margins of comedo necrosis (case No. 33). LCIS lesions were characterized by solid sheets of glandular epithelial cells, often interspersed with irregular acinar structures. LCIS cases generally displayed greater cellular uniformity but less cohesion than in situ ductal carcinomas. Neoplastic cells in 2 of the LCIS cases contained abundant flocculent pale eosinophilic cytoplasm, medium-sized round to oval nuclei, and distinct nucleoli (resembling type B human LCIS), 25, 42 while cells in the remaining LCIS lesion (case No. 31) had smaller amounts of amphophilic cytoplasm, small round dark nuclei, and no evident nucleoli (similar to type A LCIS). 25, 42
Invasive carcinomas ranged from low- to high grade morphology. Neoplastic cells were commonly arranged in sheets, with large round to oval euchromatic nuclei, prominent nucleoli, and amphophilic to pale basophilic cytoplasm. IDC lesions had significantly increased mitoses, nuclear pleomorphism (Fig. 3e), and loss of tubular morphology compared with normal control tissues and in situ lesions (Table 4). All IDC cases had regions of prominent central necrosis and mineralization. Thus, 81% (13/16) of macaque carcinomas had some degree of comedo-type change, about twice the 30–50% incidence reported in human breast carcinomas. 3, 36 While comedo type necrosis is an indicator of higher grade lesions in women, no clear associations were detected between necrosis extent and histologic grade in macaque carcinomas. Higher grade carcinomas also showed extensive stromal invasion (Fig. 3f) and marked loss of tubular architecture. In 1 case (case No. 26), neoplastic cells were diffusely expanded by abundant clear cytoplasm resembling clear cell change. Evidence of vascular invasion was found in 5 of the 8 carcinomas (case Nos. 25–27, 34, 35) (Fig. 3g) and nerve sheath invasion in 1 case (case No. 27). Two of the carcinomas involved the primary lactiferous ducts (case Nos. 34, 35). IDC lesions were consistently located on a background of ADH (Fig. 2f) and DCIS. General changes in areas surrounding carcinomas included stromal desmoplasia (Fig. 3e), neovascularization, cystic dilation of ducts, and lymphoplasmacytic infiltration.
Histologic characteristics of mammary gland carcinomas in macaques.∗
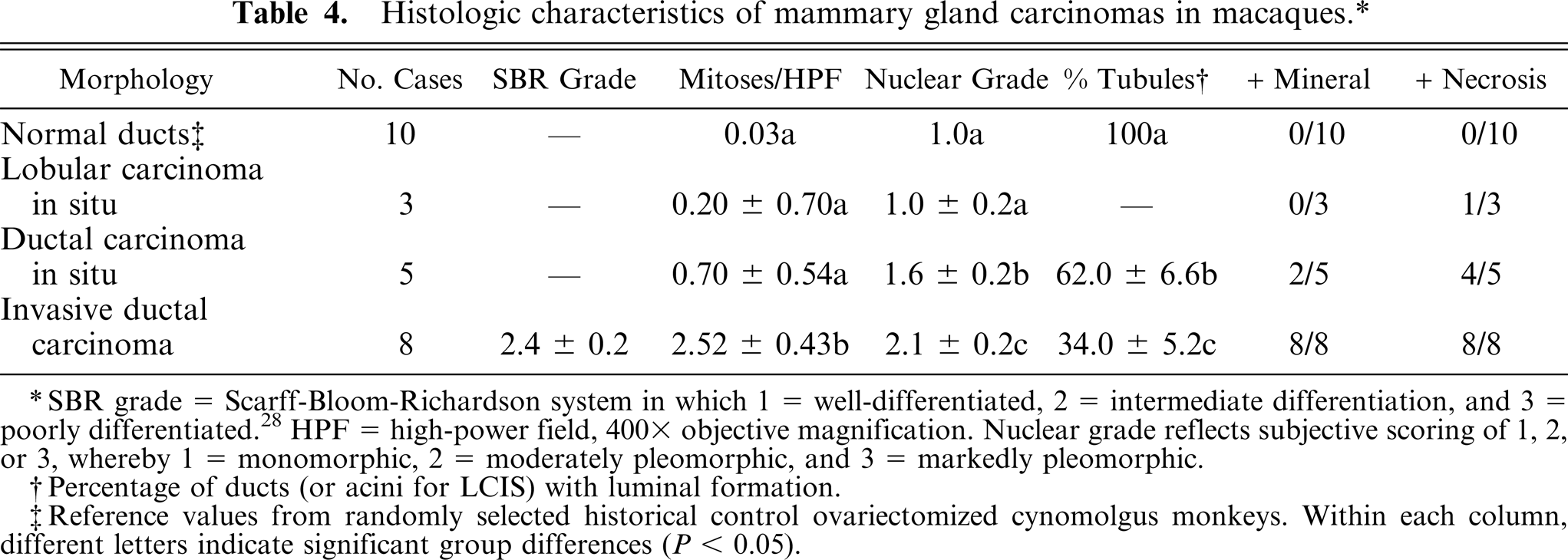
SBR grade = Scarff-Bloom-Richardson system in which 1 = well-differentiated, 2 = intermediate differentiation, and 3 = poorly differentiated.28 HPF = high-power field, 400 x objective magnification. Nuclear grade reflects subjective scoring of 1,2, or 3, whereby 1 = monomorphic, 2 = moderately pleomorphic, and 3 = markedly pleomorphic.
Percentage of ducts (or acini for LCIS) with luminal formation.
Reference values from randomly selected historical control ovariectomized cynomolgus monkeys. Within each column, different letters indicate significant group differences (P < 0.05).
Immunohistochemistry
Tissues were immunostained for several markers of tumor differentiation and aggressiveness, including proliferation (Ki67/MIB1), sex steroid receptors (ERα, ERβ, PR), and epidermal growth factor receptor-2 (HER2) (Figs. 4–9). Normal control breast tissues from a group of untreated ovariectomized cynomolgus monkeys were also evaluated as a reference group. Ki67/MIB1-positive cells were generally present but in low numbers in normal control tissue epithelium (25/27, or 93%, of normal cases had ≤10% positive cells). Among proliferative lesions, Ki67/MIB1 expression increased with lesion severity and was significantly higher within ductal in situ and invasive carcinomas compared with normal breast epithelium (Table 5). Proliferation was also higher in IDC (
Immunohistochemical characteristics of mammary gland lesions in macaques.∗
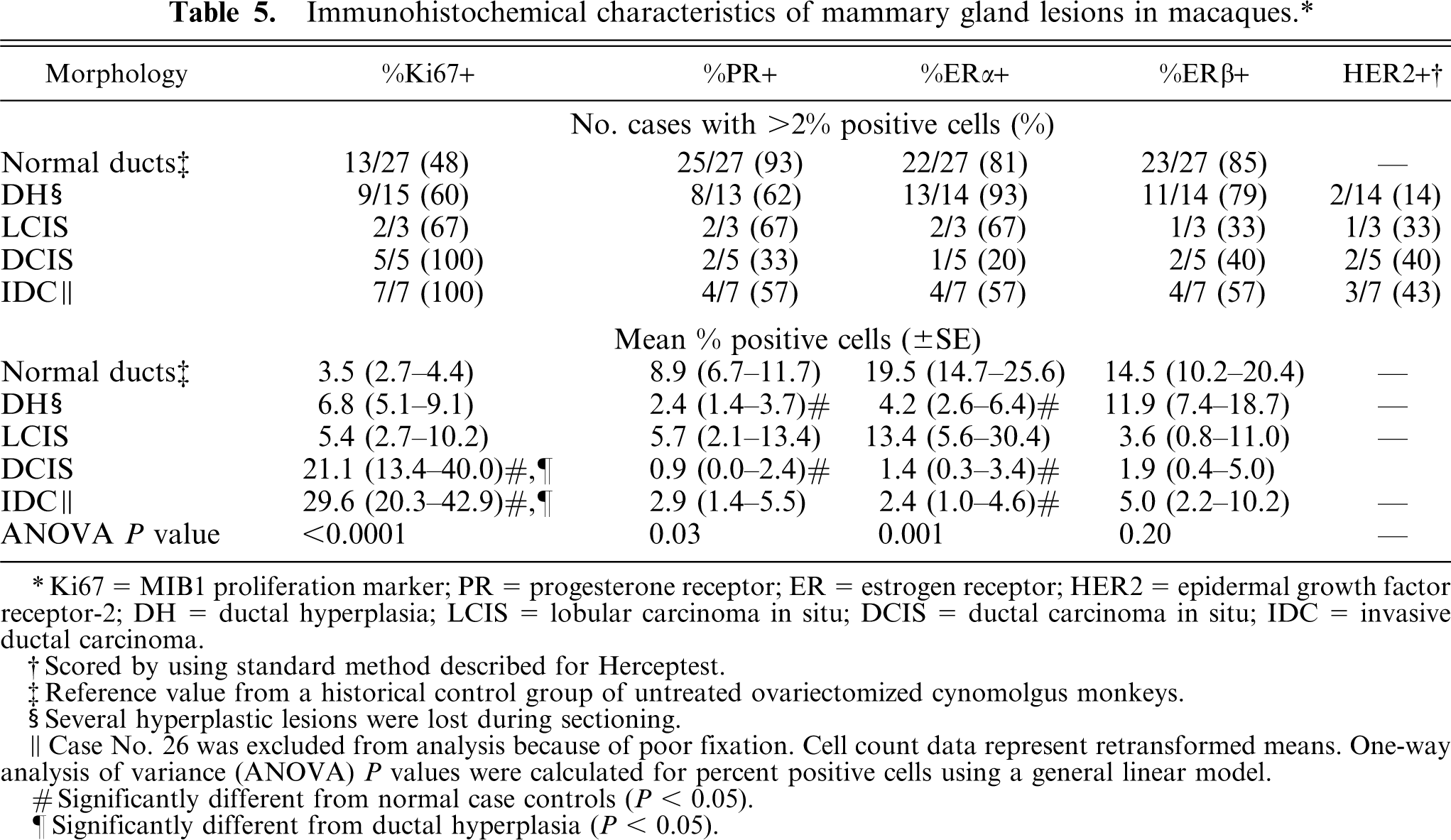
Ki67 = MIB1 proliferation marker; PR = progesterone receptor; ER = estrogen receptor; HER2 = epidermal growth factor receptor-2; DH = ductal hyperplasia; LCIS = lobular carcinoma in situ; DCIS = ductal carcinoma in situ; IDC = invasive ductal carcinoma.
Scored by using standard method described for Herceptest.
Reference value from a historical control group of untreated ovariectomized cynomolgus monkeys.
Several hyperplastic lesions were lost during sectioning.
Case No. 26 was excluded from analysis because of poor fixation. Cell count data represent retransformed means. One-way analysis of variance (ANOVA)
Significantly different from normal case controls (
Significantly different from ductal hyperplasia (
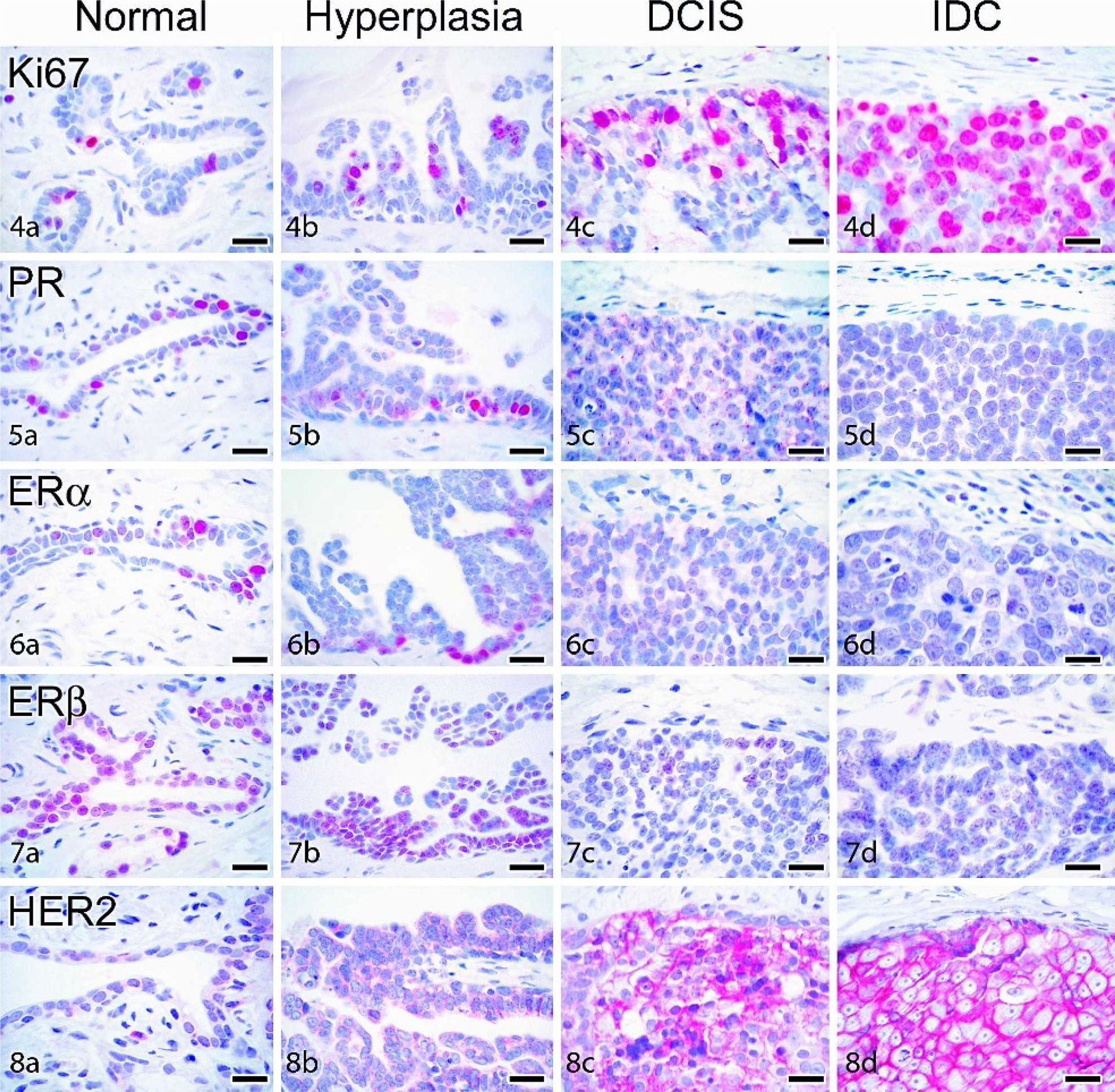
Immunohistochemical staining of cynomolgus and rhesus macaque mammary-gland lesions. Labeled streptavidin biotin, hematoxylin counterstain. All bars = 20 µm. DH = ductal hyperplasia; DCIS = ductal carcinoma in situ; IDC = invasive ductal carcinoma.
Sex steroid receptor expression was present in all normal cases and generally ranged from 5–20% for PR, ERα, and ERβ. Among lesions, sex steroid expression was lower overall compared with normal control tissues (Table 5) but highly variable among individual cases. Expression of at least 1 sex steroid receptor was detected in 14 of 15 (93%) hyperplasia lesions evaluated, while 1/3 (33%) LCIS, 2/5 (40%) DCIS, and 3/7 (43%) IDC lesions were completely sex steroid receptor negative. When present, nuclear reactivity for PR, ERα, and ERβ in carcinomas was present either diffusely (as shown in Fig. 9) or within scattered patches of cells. ERα and PR staining intensity in carcinomas was highly variable but generally lower than that seen in normal and hyperplastic epithelium. ERβ staining, which is typically more diffuse but lower intensity than ERα in normal tissue (Fig. 7a), was slightly higher intensity when present in carcinomas (Fig. 9c). Overall ERα reactivity (total % positive cells) tended to be associated with lower histologic grade within carcinomas, although this relation was not significant (
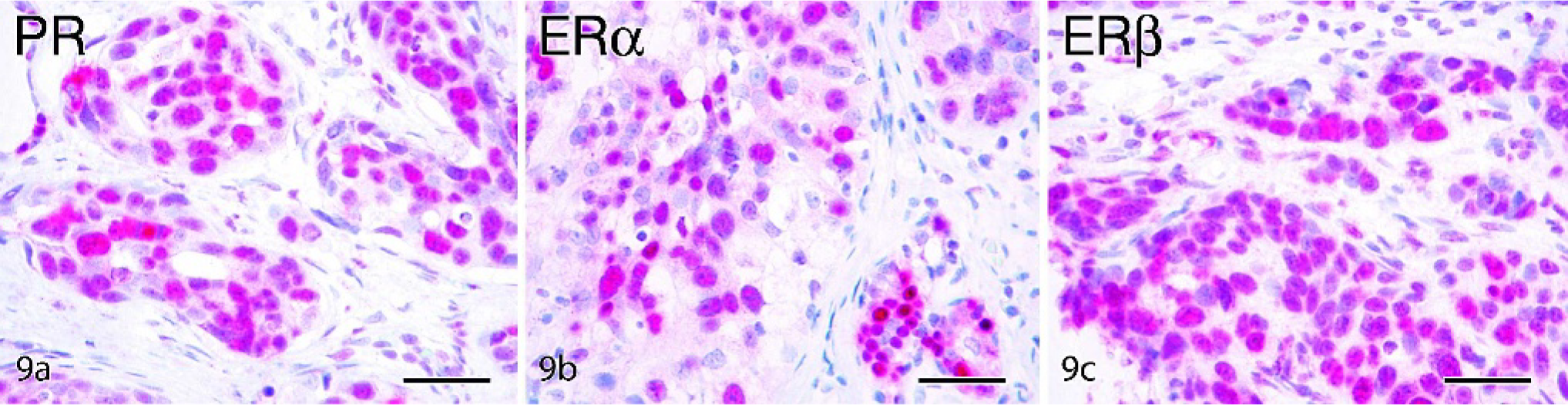
Mammary gland ductal carcinoma, cynomolgus macaque, case No. 35. Labeled streptavidin biotin, hematoxylin counterstain. Bars = 50 µm. Strong positive immunohistochemical staining for progesterone receptor (PR)
The proportion of cases with strong cell membrane immunostaining for HER2 generally increased with lesion grade (Table 5; Fig. 8b–d), while minimal to no staining was found in normal control tissue (Fig. 8a) and normal internal control tissue adjacent to lesions. Positive membrane and cytoplasmic immunostaining for E-cadherin was present in normal ducts and lobuloalveolar units, with stronger reactivity in ducts (Fig. 10a). Among carcinomas, strong E-cadherin staining was present in 4 of 5 DCIS but none of the LCIS lesions (Fig. 10b–c). No immunostaining was observed for any markers in the negative staining control slides.
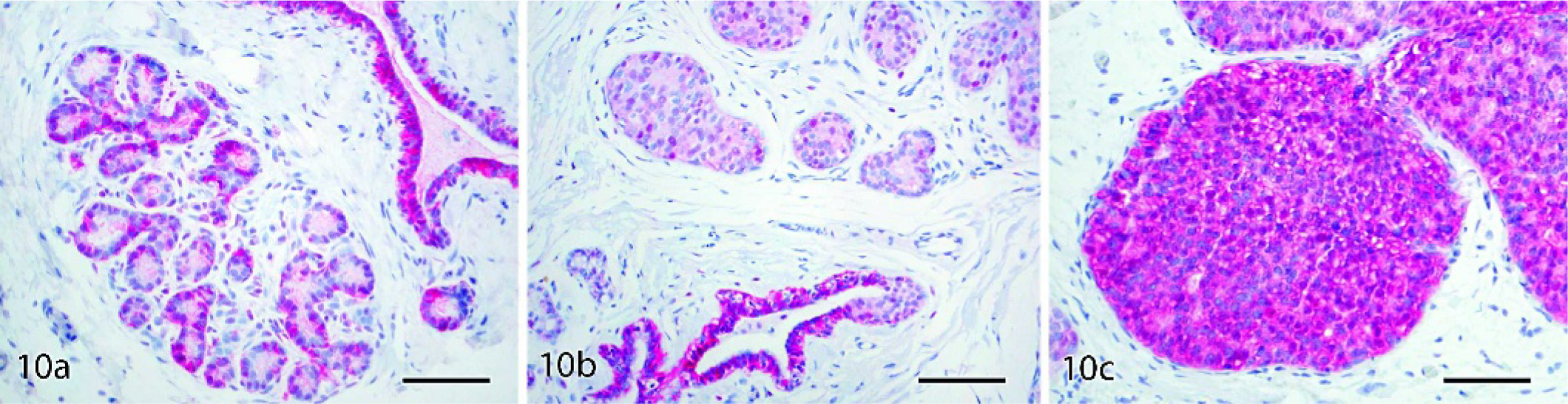
Mammary glands: cynomolgus and rhesus macaques. Labeled streptavidin biotin, hematoxylin counterstain. Bars = 80 µm.
Discussion
Macaques provide an important animal model for evaluation of hormonal factors related to breast cancer risk. In this paper, we demonstrate a diverse range of histologic and immunohistochemical features of proliferative lesions in macaques, many of which resemble those seen in the human breast. Morphologic similarities included ductal hyperplasia with micropapillary and columnar changes; solid, comedo, micropapillary, and cribriform patterns of in situ carcinoma; coincident ADH, carcinoma in situ, and invasive carcinoma; and nodal and pulmonary metastases. Immunohistochemical similarities included increased epithelial expression of the proliferation marker Ki67/MIB1, loss of sex steroid receptors in a subset of carcinomas, increased HER2 oncogene expression in select higher grade lesions, and loss of E-cadherin expression in LCIS lesions.
Findings from this study suggest that mammary gland neoplasms are an underobserved or reported lesion in macaques. Based on available evidence from one of the few aging studies of macaques, rates appear to fall within the 4–8% lifetime incidence range seen in lower-risk populations of women. 20, 37 Sporadic reports dating back to 1940 have identified spontaneous and sex steroid induced mammary lesions in macaques, 1, 4, 7, 14, 19, 23, 27, 30, 45, 46, 51, 54 including one multistudy analysis that found a 42% incidence of intraductal hyperplasia in control animals. 51 Still, few cases of invasive breast cancer in macaques have been characterized, and breast cancer has generally been considered a rare disease in nonhuman primates. 7 This paucity of reports is likely multifactorial. First, the majority of captive macaques are not kept into their natural postmenopausal years (when the majority of breast cancer cases occur in women), contributing to an underestimation of the true incidence. Second, many of the macaques surveyed for this study were multiparous (based on breeding records or uterine histology) and ovariectomized, both of which lower breast cancer risk in women. 10 Female macaques are also generally not as obese as higher-risk human populations and thus have lower endogenous postmenopausal estrogens, a trait also associated with reduced breast cancer incidence in women. 10, 18 Last, the standard monkey chow diet is high in soy protein, which may have protective effects against breast and other reproductive cancers. 2 The preponderance of higher grade lesions in rhesus macaques (Table 2) may relate either to species differences in susceptibility or to the more advanced age of rhesus macaques surveyed.
While specific morphologic trends were identified (e.g., the propensity toward comedo-type necrosis), a distinctive feature of the macaque lesions was their heterogeneity. This characteristic differs from the more widely used animal models of human-breast cancer, which often produce more specific tumor types. For example, chemically induced mammary adenocarcinomas in rats typically express sex steroid receptors, require hormonal stimulation, and rarely metastasize or exhibit necrosis and mineralization. 16, 47 More recent genetically engineered mouse (GEM) models, in contrast, produce oncogene-specific mammary tumors that are frequently sex steroid receptor negative, hormone independent, metastatic (to lungs rather than nodes), and relatively uniform in morphology within a particular model. 5 Descriptive terminology from rodent models may also be problematic, because it is not always similar to that of human breast lesions. 6 The collection of macaque lesions in this study was readily categorized by human terminology and still highly variable in morphologic characteristics (e.g., mineralization in 10/16 or 63% of carcinomas), metastatic behavior (nodal involvement in 5/16 or 31% of carcinomas), age of onset (age range of 5 to 35 years), sex steroid receptor expression (present in 9/15 or 60% of carcinomas), and HER2 overexpression (present in 6/15 or 40% of carcinomas). While lacking the advantages of reproducibility and specificity found in rodent models, these diverse patterns correspond well with those seen in human breast cancer. 26, 36
In recent years, the experimental approach for macaque studies of cancer risk has shifted from longer-term descriptive investigations to more mechanistic biomarker studies. One of the more important biomarkers is the Ki67/MIB1 proliferation antigen. In human breast cancer, higher Ki67/MIB1 expression is significantly associated with increased carcinoma grade, poor clinical response to endocrine therapy, and decreased overall survival, 8, 52, 55 while in macaques this marker has been used extensively to predict risk associated with various contraceptive and postmenopausal hormonal agents. 9, 13 In the current study, Ki67/MIB1 expression in invasive carcinomas (29.6% of cells) was similar to that of human breast carcinomas 48 and was positively correlated with tumor grade. Higher grade carcinomas also showed selective loss of sex steroid receptors and increased expression of the oncogene HER2, both of which are important negative prognostic indicators for human breast cancers. 39, 43 These findings are further supported by recent genetic evidence showing ≥95% identity between human and macaque nucleic acid sequences for key oncogenes, such as the early onset breast cancer 1 gene (BRCA1). 38
Because of their close phylogenetic relation, macaques have long been recognized as an important animal model in the study of human disease. In this paper, we demonstrated unique similarities between macaque and human mammary gland neoplasms. Macaque lesions, while morphologically diverse, followed well-described growth patterns for human breast cancer. Our findings point to key molecular parallels between the macaque and human breast and lend support to the use of risk biomarker analyses in this model. This information highlights the value of aged macaque colonies and how they may potentially provide insight into the natural history of human disease.
Footnotes
Acknowledgements
We thank Hermina Borgerink, Beth Phifer, and Jean Gardin for their technical contributions. This work was supported by the National Institutes of Health Grant AT00639 (JMC), the National Center for Research Resources Grant T32 RR07009 (CEW), the Wisconsin National Primate Research Center Grant P51RR000167 (ALU), and the California National Primate Research Center Grant P51RR000169 (RPT).
