Abstract
Angiogenesis was evaluated by immunohistochemistry for platelet endothelial cell adhesion molecule (CD31) in a series of benign and malignant canine mammary neoplasms. Computer image analysis was used to measure the intratumoral microvessel density (number of microvessels per square millimeter) and the area and perimeter of immunolabeled vascular structures. A higher intratumoral microvessel density and lower values for area and perimeter were found in malignant neoplasms compared with benign tumors and among the malignant tumors, in less differentiated phenotypes and in more anaplastic histological types (solid and squamous carcinomas), most of which had metastasized. These findings are consistent with an increase in angiogenesis in multistage neoplasia. Therefore, in more anaplastic malignant neoplasms, numerous but small and often malformed new vessels can be identified. The correlation of angiogenesis data with differentiation grade and histological type of mammary neoplasms is consistent with the findings in human medicine and demonstrates that angiogenesis can also have a prognostic value in veterinary medicine.
Angiogenesis is a complex multistep process characterized by the formation of new capillaries from the preexisting vascular network. In neoplasms, it is essential for tumor growth and metastasis. 1 , 8 , 12
Endothelial cells proliferate 30–40-fold faster in tumor blood vessels than in the vasculature of normal tissues. 17 Furthermore, several studies have demonstrated a significant correlation between marked angiogenesis, evidenced by a high microvessel density (number of microvessels per square millimeter), metastasis, and poor prognosis in several human tumor types, including breast, 10 , 18 , 30 prostate, 31 and non-small cell lung carcinomas, 13 cutaneous melanomas, 27 and testicular germ cell tumors. 22 Therefore, the angiogenic characteristic of a neoplasm is considered a powerful prognostic tool in human medicine. 26 , 31 On the other hand, angiogenesis and its relation to biologic behavior of neoplasms are still poorly understood in veterinary oncology.
Unfortunately, the methods for measuring angiogenesis still represent a problem. The method suggested by Weidner et al., 30 in which the microvessel density was assessed by counting the immunolabeled vessels only in the most vascularized areas of neoplasm (hot spots), has given conflicting results. Recently, new methods of tumor angiogenesis measurement using computerized image analysis, which allow us to evaluate not only the number of microvessels but also other vascular parameters, such as area and perimeter, have been proposed. 2 , 3 , 11 , 28
Therefore, the aim of the present study was to use computerized image analysis to measure angiogenesis in a series of canine mammary tumors to assess microvessel density and, in addition, area and perimeter of each vessel, and to correlate these parameters with differentiation grade and histological type of neoplasia.
Material and Methods
Samples
Forty samples of canine mammary tumors, 10 benign and 30 malignant, were examined. Tumors were classified by World Health Organization criteria 15 as listed in Table 1 and were graded as moderately to poorly differentiated (grades 1 to 3) by two independent observers. The parameters used were 1) extent of tubule formation (for tubular and papillary adenocarcinomas), 2) nuclear hyperchromatism and number of mitoses, and 3) irregularity of size, shape, and staining of nuclei. 19
WHO classification of canine mammary tumors used in this study.
Among the malignant tumors, two of eight tubular adenocarcinomas, three of six solid carcinomas, and two of three squamous cell carcinomas, all grade 3 (58.3%), had already metastasized to the inguinal lymph nodes at the time of diagnosis. Two of three early metastatic solid carcinomas metastasized to the lung after 2 years. Three of eight tubular adenocarcinomas, grade 2 (30%), and two of six solid carcinomas, grade 3 (16.6%), which had not metastasized at the time of diagnosis, spread to lungs, liver, and bones after 2 years.
Immunohistochemistry
All samples were fixed in 10% neutral buffered formalin and embedded in paraffin. Sections 5 µm thick were deparaffinized in xylene, dehydrated in graded alcohols, and washed in 0.01 M phosphate-buffered saline (PBS), pH 7.2–7.4. Endogenous peroxidase was blocked with 0.3% hydrogen peroxide in absolute methanol for 30 minutes. Before the immunohistochemical procedure, a proteolytic treatment with 0.4% pepsin in 0.01 m HCl was applied for 30 minutes at 37 C. The primary antibody was a monoclonal mouse anti-human platelet endothelial cell adhesion molecule (PECAM), also called CD31 (clone JC70; Dako, Denmark), a 100-kd glycoprotein that participates in the adhesion between platelets and endothelial cells. 21 PECAM antigen is regarded in humans as a useful marker for endothelial cells because it is consistently expressed on all types of endothelial cells, and it is expressed only by endothelial cells, platelets, and macrophages. 25 The antibody against human PECAM antigen also recognizes the canine antigen. 5
The antibody was diluted 1:20 in Tris-HCl buffer containing 0.5% bovine serum albumin and 0.015 M sodium azide (Dako). The sections were incubated overnight at 4 C. The immunolabeling procedure included negative control sections incubated in PBS without primary antibody. Biotinylated anti-mouse, anti-rabbit, and anti-goat immunoglobulins (labeled streptavidin biotin [LSAB] Kit; Dako) diluted in PBS were used as secondary antibodies; they were applied for 30 minutes. After washing in PBS, the sections were incubated in streptavidin conjugated to horseradish peroxidase in Tris-HCl buffer containing sodium azide 0.015% (LSAB kit; Dako) for 30 minutes. To demonstrate the immunolabeling, 3,3-diaminobenzidine tetrahydrochloride was used as chromogen, and hematoxylin was sued as counterstain.
Image analysis
Intratumoural microvessel density was assessed randomly by choosing immunolabeled vessels on a 400× field (40× objective and 10× ocular) using an automated (Image Pro-Plus 1) image analysis system (Sistema MONO, Immagini e Computer, Milan, Italy). Twenty fields per tumor were examined.
Images were captured using a microscope (Nikon Eclipse E-400, Tokyo, Japan) coupled to a video camera (JVC TK-C1380E, Japan), stored in the digital memory, and shown on the monitor. Manual outlining of intratumoral microvessels was performed; areas, perimeters (expressed in millimeters), and number of vessels per square millimeter were then calculated based on image analysis. Every immunolabeled endothelial cell separate from adjacent microvessels, tumor cells, and other connective tissue elements was counted as a single microvessel. All the vessels in the stroma outside tumors were considered as normal controls.
Statistical analysis
The vascular parameters expressed by mean and standard deviation were correlated with histological type and differentiation grade of mammary neoplasms by analysis of variance.
Results
The microvessel number per 0.304 mm2 of image analysis was higher in malignant than in benign neoplasms (
Correlation of number, area, and perimeter of vessels with differentiation grade of neoplasms.
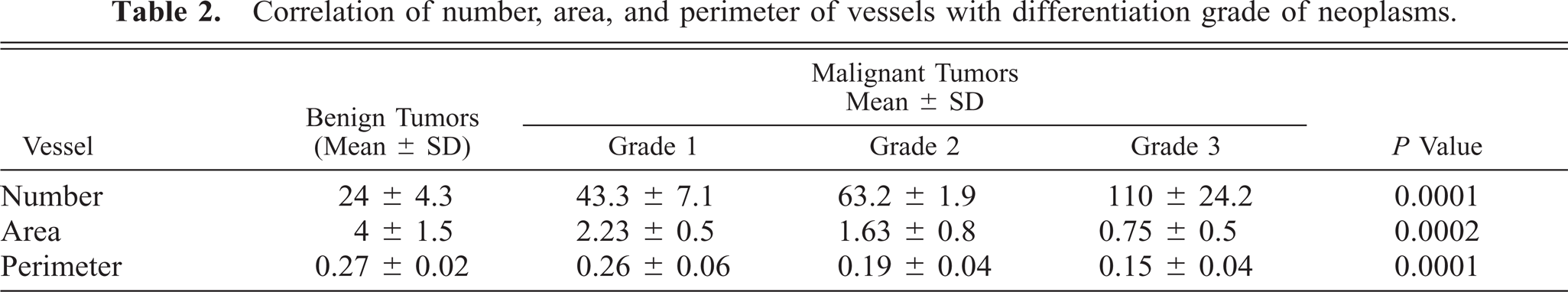
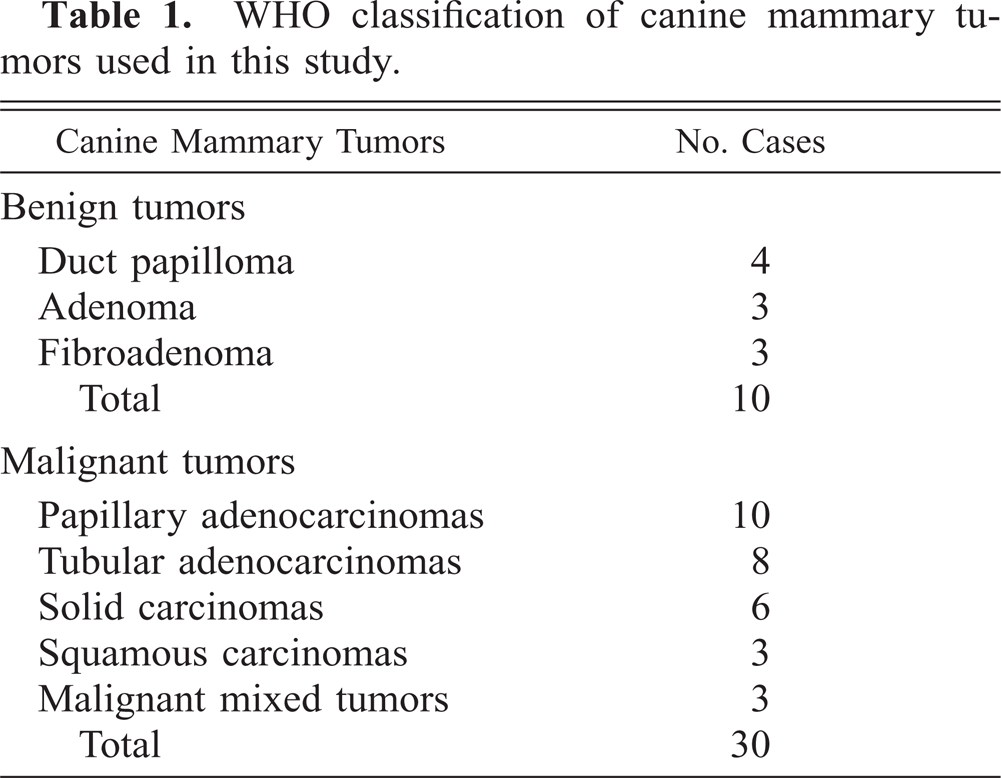
In benign neoplasms and grade 1 papillary and tubular adenocarcinomas, intratumoral microvessels were wide and regular in shape, with erythrocytes evident in the lumens (Fig. 1). In grade 2 and 3 neoplasms and in more anaplastic histological types, the microvessels were small and irregular in shape often without a distinct lumen (Fig. 2). In addition, in two solid carcinomas that metastasized early, mostly isolated endothelial cells were present.
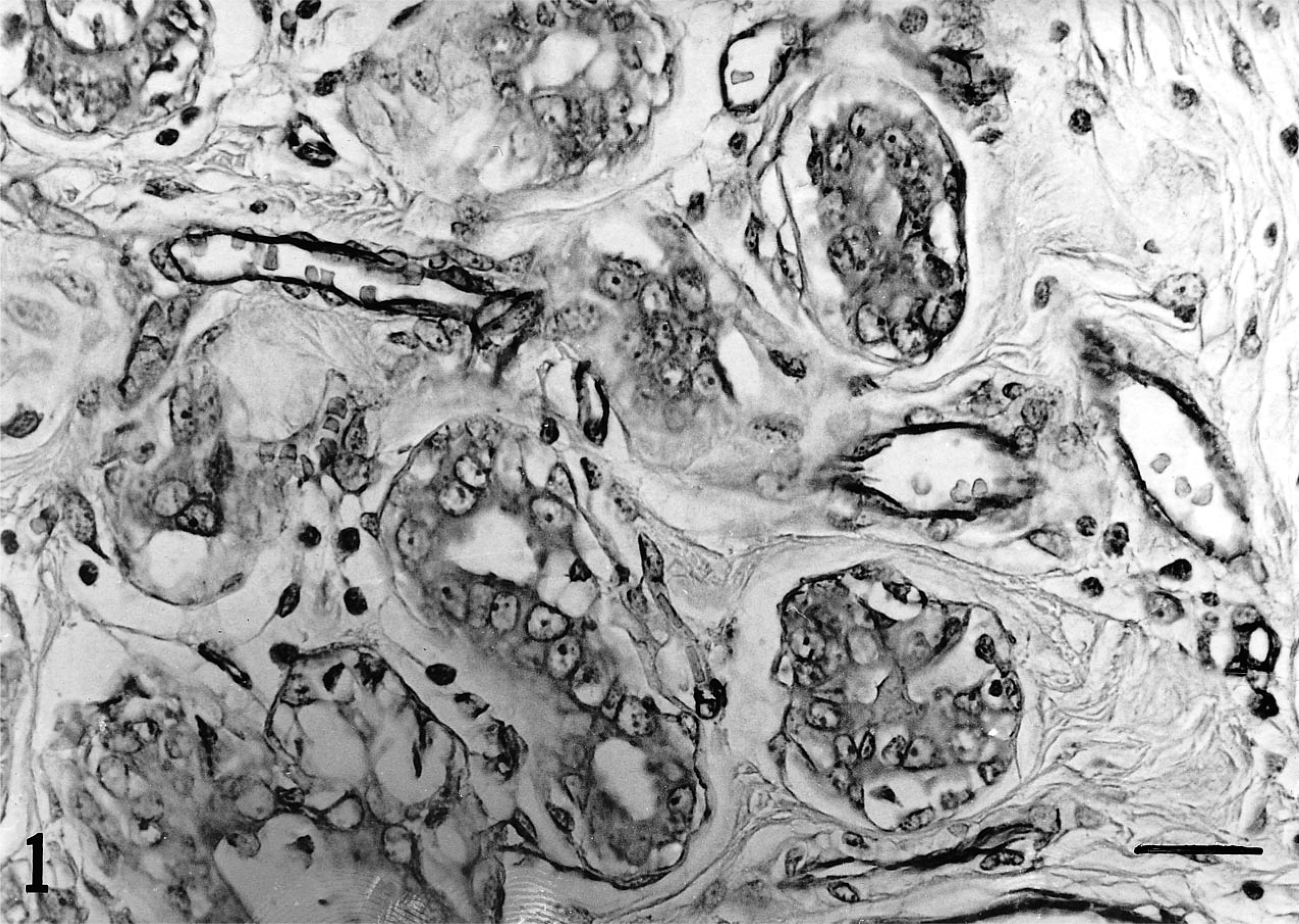
Canine tubular adenocarcinoma, grade 1. Immunostaining for PECAM shows wide vessels, regular in shape, containing erythrocytes. Streptavidin–biotin–peroxidase. Bar = 15 µm.
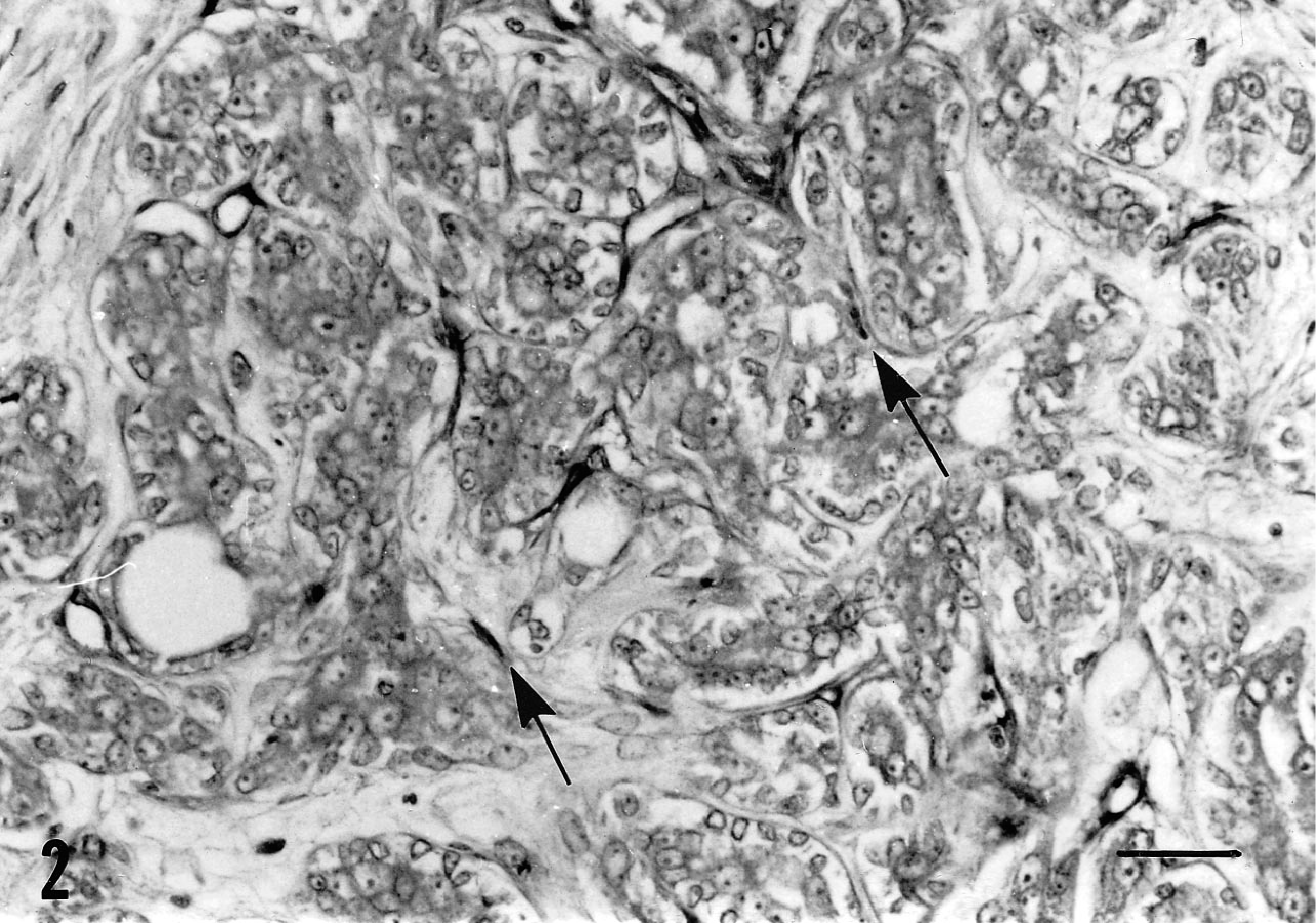
Canine tubular adenocarcinoma, grade 2. Immunostaining for PECAM shows small vessels, irregular in shape and without lumen. Some isolated positive endothelial cells are evident (arrows). Streptavidin–biotin–peroxidase. Bar = 25 µm.
Areas and perimeter were smaller in malignant than in benign neoplasms (area
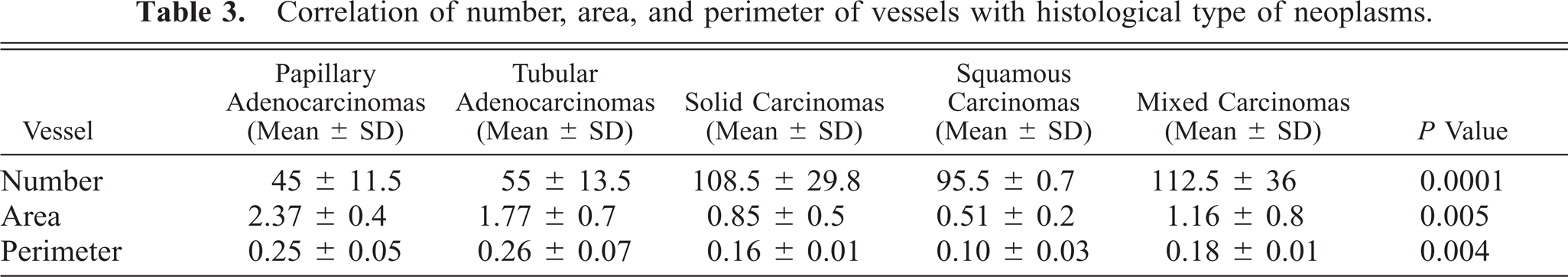
Correlation of number, area, and perimeter of vessels with histological type of neoplasms.
Discussion
In normal tissues, new vessel formation is regulated by many stimulatory and inhibitory factors, which function in a delicate balance. 8 Any change in tissue homeostasis, either physiological or pathological, can perturb the balance and result in acquisition of an angiogenic phenotype. 9 , 16 On the other hand, in experimental and clinical studies, a tumor generally acquires characteristics of malignancy, such as rapid growth and metastatic capability, when angiogenesis increases. 7 , 14 , 30 , 31
In this study, in a series of canine mammary tumors, as in comparable human studies, a higher microvessel density was detected in malignant neoplasms. In addition, in poorly differentiated and metastasizing carcinomas, increased angiogenesis was demonstrated.
One hypothesis for increased angiogenesis is that, as mutations accumulate in tumors, some clones of neoplastic cells switch to an angiogenic phenotype. 16 The molecular basis of the angiogenic switch is not entirely clear but may involve an increase in angiogenesis stimulators, a decrease in angiogenesis inhibitors, or a combination of the two. 6 More precisely, the malignant transformation could be linked to the loss of suppressor genes that encode angiogenesis inhibitors such as angiostatin or thrombospondin-1, 24 to the activation of oncogenes encoding angiogenesis inducers such as vascular endothelial growth factor or fibroblast growth factor, 4 , 23 or both.
The decrease in size of the intratumoral vessels detected in malignant neoplasms and, among these, in less differentiated tumors, can be a consequence of the increased angiogenesis, which can give rise to numerous but small and often abnormal new vessels. For example, in our study, two less differentiated and early metastasizing carcinomas showed many isolated endothelial cells, probably reflecting a very high angiogenic and metastatic potential. Some authors have demonstrated that endothelial cell proliferation index and intratumoral microvessel density were both correlated with poor prognosis and metastasis in many neoplasms, but one was independent from the other. 10 , 29 In this regard, it is worthwhile to remember that endothelial cells can foster metastasis not only by inducing new vessel formation, but also by producing collagenase and other degradative enzymes, thus making it easier for neoplastic cells to escape into the neovasculature. 20 In addition, endothelial cells can also stimulate the growth of tumor cells. 32
Therefore, with an increase in vessels, there are more endothelial cells and a greater risk for tumor growth and dissemination. A key step in this process is the angiogenic switch of the neoplastic cells. This suggests that in tumor therapy, inhibition of angiogenesis by administration of inhibitors could prevent the growth of neoplastic cells and metastasis. 6 , 16 In this regard, canine mammary tumors may be useful models for therapeutic trials.
In conclusion, in veterinary medicine, the microvessel density as an expression of angiogensis may be used as a sensitive parameter correlated with malignancy. In addition, the evaluation of microvessel size may give useful adjunct information about the angiogenic potential of a neoplasm.
Footnotes
Acknowledgements
This work was supported by grants from Ministero della Ricerca Scientifica e Tecnologica 1997. We thank Mr. R. Ilsami for his technical assistance.
