Abstract
The vascular system of Cuvier’s beaked whales (CBW) (Ziphius cavirostris; family Ziphiidae), an extremely deep, prolonged-diving cetacean, is increasingly receiving anatomic and physiologic study due to possible anthropogenic interactions; however, vascular pathology rarely has been reported in this species. Thirteen CBW stranded in the Canary Islands from June 2008 to June 2014 were autopsied. A careful dissection of the thoracic and abdominal vasculature was performed on these animals. All had moderate to severe and extensive chronic fibrosing arteritis with aneurysms, hemorrhages, and thrombosis primarily involving the mesenteric and gastroepiploic arteries and the thoracic and abdominal aorta. Microscopically, the lesions varied from subacute subintimal hemorrhages and severe neutrophilic, eosinophilic, and histiocytic dissecting arteritis with intralesional nematode larvae to marked, chronic, fibrosing arteritis with thickening and distortion of the vascular wall with calcification and occasional cartilage metaplasia. In addition, adult nematodes in renal arteries and veins, renal parenchyma and/or ureter were identified morphologically as Crassicauda sp. Nucleic acid sequenced from renal nematodes from 2 animals yielded closest nucleotide identity to C. magna. The pathogenesis is proposed to involve a host response to larval migration from the intestine to the kidney through the mesenteric arteries, abdominal aorta, and renal arteries. Severe consequences for such lesions are possible and could vary from reduced vascular compliance to chronic renal disease and predisposition to the development of disseminated intravascular coagulation and multiorgan failure. Severe chronic arteritis in CBW is associated with renal parasitism by Crassicauda spp.
Beaked whales (BWs) belong to the odontocete Family Ziphiidae, which includes 22 recognized species grouped in 6 genera. 46 Because of their extreme, deep, and long-lasting diving profile, their pelagic habits, and general lack of concentrated populations, these species include some of the least-studied cetaceans. 46
Studies on BWs traditionally have focused on anatomy, 33,41 diet, 39,42 and diving/foraging behavior. 3,22,27,43,47 Recently, their pathophysiology gained more scientific attention due to their involvement in mass strandings linked to military use of midfrequency and high-intensity sonar. 4,13 –15,17,24,26,38,44 In these studies, stranded BWs demonstrated systemic hemorrhages associated with gas and fat emboli, a pathologic picture resembling decompression-like sickness (DCS) previously not reported in cetaceans. 14,25
Among cardiovascular diseases, vasculitis is of major concern in humans and animals. 35 In humans, primary forms of vasculitis are classified according to vessel diameter, the role of immune complexes, the presence of specific autoantibodies, granuloma formation, organ specificity, and even population demographics. 35 The 2 more frequent pathogenic mechanisms of vasculitis are immune-mediated inflammation and direct invasion of vascular walls by infectious pathogens including bacteria or fungi, but also metazoan parasites. 35 Verminous arteritis has been largely recognized in humans with Angyostrongylus cantonensis and A. costaricensis 10,36 and in horses 49 and dogs 1 often with a grave prognosis when heavy burdens of adults or larvae migrate through vital organs (eg, Strongylus vulgaris) or result in a thrombotic crisis (eg, Dirofilaria immitis).
In cetaceans, verminous arteritides have involved baleen whales (Mysticeti) associated with nematodes of the genus Crassicauda, 30,31 and common porpoises (Phocoena phocoena) and Burmeister’s porpoises (P. spinipinnis) associated with Pseudalius inflexus. 7,20,25 In whales, vasculitis was linked to a highly pathogenic species of Crassicauda, C. boopis, which promoted extensive inflammation and sclerosis of the arteries during larval migration in blue whales (Balaenoptera musculus), humpback whales (Megaptera novaeangliae), and fin whales (Balaenoptera physalus). In common porpoises and Burmeister’s porpoises, heavy burdens of Pseudalius inflexus are responsible for necrotizing vasculitis with thrombosis, and proliferative arteritis of pulmonary vessels with partial or complete vascular occlusion and aneurysms. 25 Multifocal mesenteric arterial sclerosis has been rarely reported in CBW. The cause and significance of its occurrence has not been explained to date. 16 This study describes the macroscopic and microscopic characteristic lesions of a vascular pathologic condition involving Crassicauda spp. migration and affecting CBW.
Material and Methods
Stranding Epidemiologic Data and Autopsy Examination
Autopsies were conducted on 13 CBW stranded dead along the coasts of the Canary Islands from June 2008 to July 2014. Life history data (ie, age class, sex, nutritional [NS], and decomposition [DS] status, morphometrics, and stranding conditions of the individuals) were recorded systematically following established criteria (Supplemental Table 1). Three age categories based on total body length (TBL) and gonadal macroscopic and histologic appearance were considered: fetus/neonate/calf, juvenile/subadult, and adult. 19 The NS of each animal was established morphologically based on anatomical parameters such as the osseous prominence of the spinous and transverse vertebral processes and ribs, the mass of the epaxial musculature (longissimus dorsi, multifidus, spinalis), and the amount of subcutaneous and cavitary fat deposits, taking account of the species, and the age of the animal. These parameters allowed us to classify their NS as good, moderate, poor, and emaciated. 2 The DS was classified as very fresh (individuals known to live-strand and necropsied immediately), fresh, moderate autolysis, advanced autolysis, or very advanced autolysis. 29
The autopsy protocol used a more in-depth study of the cardiovascular system implemented since 2008, and included thorough examination, description, and imaging. Following the opening of the peritoneal and pleural cavity, a visual evaluation of the medium and larger caliber arteries and veins was performed. Arterial evaluation included the heart, subclavian, brachiocephalic trunk, carotid arteries, thoracic aorta, intercostal arteries, abdominal aorta, celiac trunk, cranial and caudal mesenteric arteries, renal, colonic, gonadal, and iliac arteries. Similarly, homologous medium and large caliber veins were evaluated including the jugular, azygos, caudal vena cava, portal, mesenteric, and renal veins, and genital plexus. In addition, special attention was paid to the urinary system to establish the presence/absence of Crassicauda sp. adult or larval forms. Grading of Crassicauda parasitization in renal arteries or veins, renal calices, or intra- and extrarenal ureteral segments, was based on adult and larval nematode counts wherein less than 10 individuals represented grade +, 10–20 grade ++, and more than 20 grade +++.
Histopathology and Histochemistry
Selected samples were fixed in 10% buffered formalin, 4 μm sections were prepared and stained with hematoxylin and eosin (HE) (Leica Autostainer XL 2V3 RevC, Leica Biosystems Nussloch GmbH, Nussloch, Alemania). Additional histochemical techniques Congo red, Gram, Masson’s trichrome, modified Movat-Russel pentachrome, osmium tetroxide (postfixation technique), periodic acid-Schiff (PAS), Perl’s, Van-Gieson, and Von Kossa stains were prepared as needed. 34
Morphologic identification of renal Crassicauda nematodes was done as described. 5 DNA of 2 nematodes collected into 70% ethanol from 2 CBs was extracted with modified cetyltrithylammonium bromide method CTAB protocol. 50 The polymerase chain reaction (PCR) was performed using primers 930F and 1200 R for the 18 S ribosomal gene. 48 PCRs were carried out with 500 nM of each primer, 1 U Taq polymerase (Invitrogen by Life Technologies, Carlsbad, California) and 3 µL of template DNA, in a final volume of 25 µL. The amplification was performed under the following conditions: 95°C for 5 min, 9 cycles of 55°C for 45 s, followed by 30 cycles at 50°C and extension for 1 min. The PCR products were analyzed by electrophoresis, purified and submitted to bidirectional sequencing using the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, California) and an ABI 3500 sequencer (Applied Biosystems). A negative PCR control (water), a negative extraction control without DNA, and a positive control (Nematode DNA), were included in each assay.
A BLASTN (GenBank database) search was conducted on the sequences with higher identity. The sequences were aligned using the ClustalW (DNA) software and the identity values of the nucleotide sequences were calculated using the program Bioedit Sequence Alignment Editor version 7.0.5.3 software. 21
Results
Of 13 autopsied animals, 8 were females (62%) and 4 were males (31%). The sex could not be determined in 1 case. Age classes were adult (n = 9), and juvenile/subadult (n = 4) (Supplemental Table 1).
On autopsy examination, all CBW had very hard, noncollapsible, tortuous, and dilated mesenteric arteries, up to 3 times the normal caliber of the distal and proximal branches (Fig. 1). Likewise, the caudal thoracic and abdominal aorta, and renal arteries were hard, distended and noncollapsible, in a diffuse distribution or affecting segments up to 10 cm long. Occasionally, the arterial caliber was greatly widened to form discrete dilatations (aneurysms), particularly prominent at branching points of the abdominal aorta and mesenteric arteries. On section, the arterial wall was thickened up to 2.5 cm in the abdominal aorta and celiac trunk. In addition, the cranial thoracic aorta, gastroepiploic, gonadal, iliac, subclavian, and intercostal arteries were affected (in decreasing order of frequency) (Supplemental Table 1).
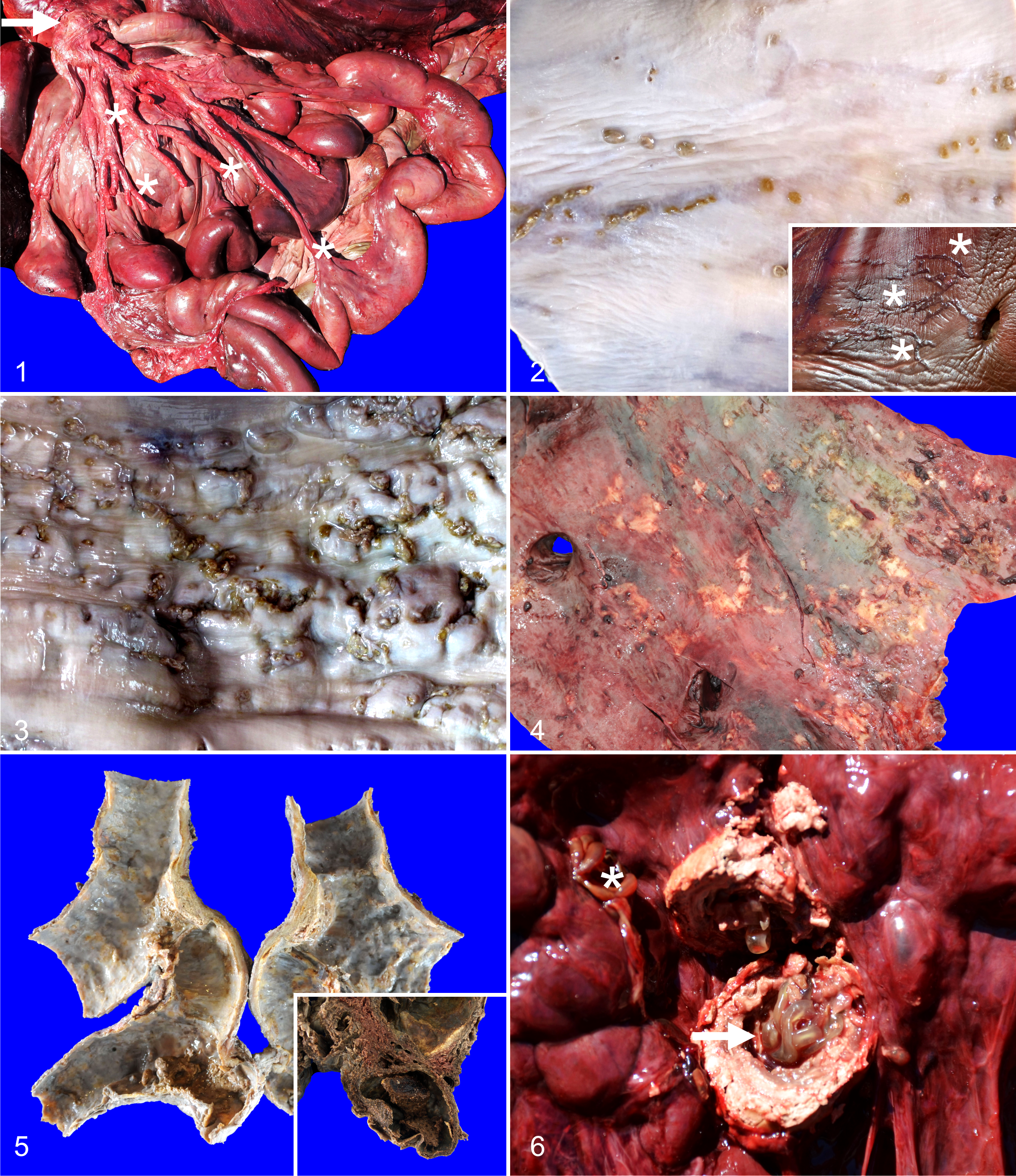
Arteritis, Cuvier’s beaked whale (CBW).
Upon sectioning of affected arteries, the variety of gross lesions was broadly classified into 3 major patterns: (1) discrete, single to compound linear tortuous tracks; (2) well-demarcated, linear to coalescing nodules; and (3) isolated to multifocally coalescing plaques. Linear tracks were characterized by 0.3–5 × 0.1–0.3 cm, red to gray, smooth, and firm lines of raised tunica intima (Fig. 2). On section, they rarely extended deeper than 2 mm from the tunica intima. In contrast, nodular lesions had a wider range of forms, from well-demarcated, 0.3–1.5 cm in diameter, pale tan to gray, smooth, firm overgrowths projecting onto the lumen, to multifocally coalescing, well-demarcated, linear to irregularly shaped, 1.5–3.5 cm, firm, raised projections of the vascular wall into the lumen, frequently pitted or with up to 1 cm in diameter superficial defects, and adherent, pale yellow friable fibrin clots (Fig. 3). On section, nodules were mottled dark red to gray and extended deep into the tunica media and occasionally reached or projected as a red, friable, granular mesothelial surface. Luminal, 0.3–1.5 cm in diameter, raised, yellow to gray, hard, flat intimal plaques frequently had well-organized, adhered fibrin thrombi (Fig. 4). On cut surface, the plaques were mottled white to pale gray, gritty and throughout the thickness of the wall of affected arteries (ie, affecting intima, media, adventitia). Plaques reached the largest size primarily in the branching points of the mesenteric arteries, caudal abdominal aorta, and renal arteries, and along with previous lesions, resulted in luminal surfaces with prominent and tortuous ridges and furrows. Within these areas, the arterial caliber typically was widened forming aneurysms (Fig. 5) in which fibrin thrombi frequently were lodged. Linear tracks were inconsistently observed in the aortic bulb (inset, Fig. 2), thoracic aorta, and cranial abdominal aorta just cranial to the mesenteric and celiac branching system; while, nodules and plaques were more often encountered in all affected vessels.
Eleven of the animals (84%) had heavy parasitic infestation of the kidneys and associated vessels by male and female adult Crassicauda. The urinary tissues were unavailable for dissection in the other 2 animals’ carcasses because of predation or trauma of the caudal abdominal region. Grossly, nematodes penetrated the wall of the renicular calyces and ureters. The nematodes extended into the interrenicular stroma where they were walled off by dense, fibrous connective tissue capsules and formed granulomas up to 4 cm diameter (Fig. 6). Nematodes often obliterated the medium and small caliber renal arteries and veins. Affected kidneys had variably sized reniculi suggesting both atrophy and hypertrophy had occurred. Large variably sized granulomas effacing the renicular parenchyma were only detected in animals with grades ++ and +++ of parasitization.
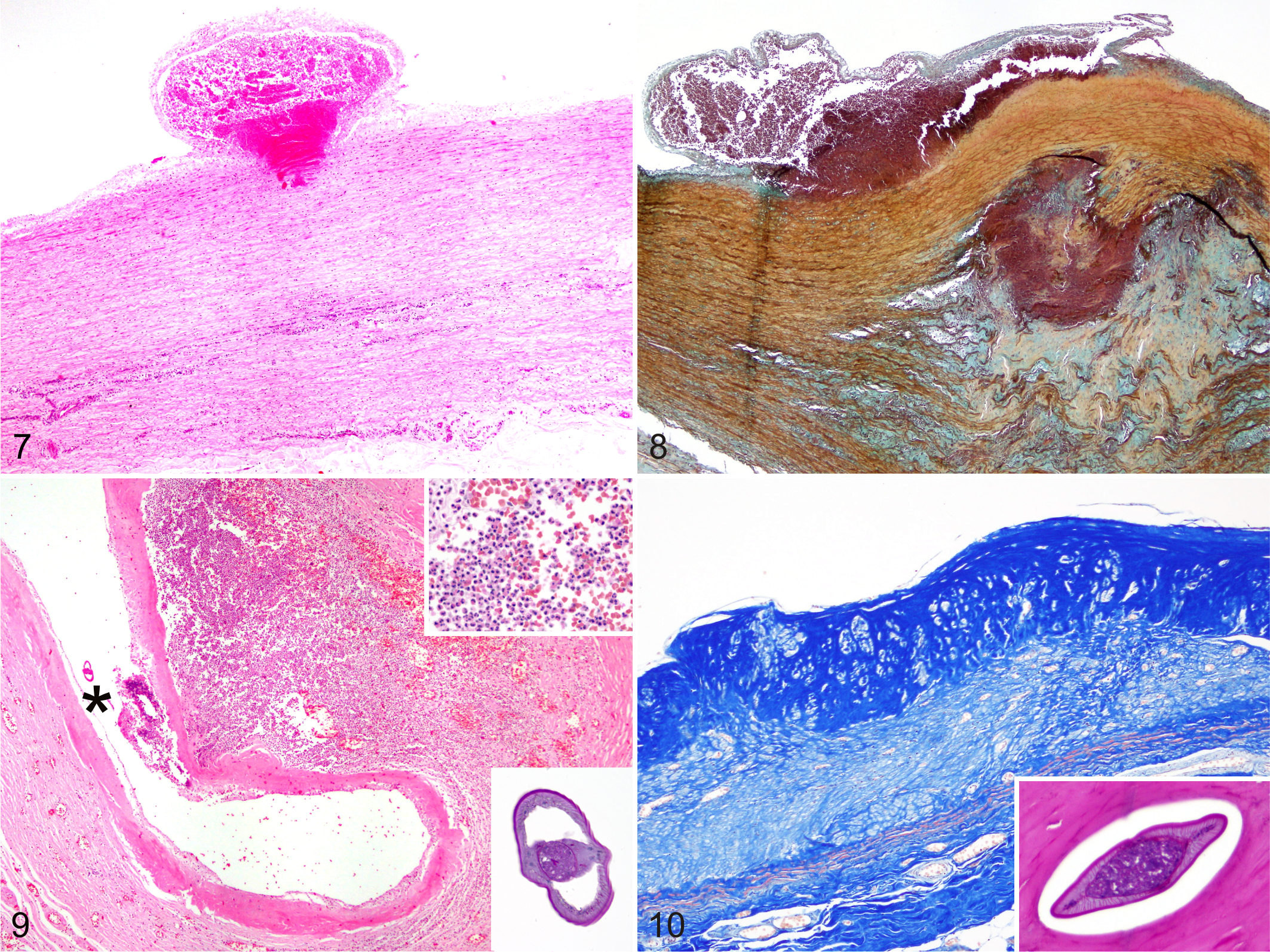
Histologically, there were 3 major macroscopic patterns according to chronicity: discrete single to compound linear tortuous tracks; well-demarcated, linear to coalescing nodules; and isolated to multifocally coalescing plaques. Linear tracks consisted of acute to subacute, subintimal hemorrhages (Fig. 7). The tunica intima was elevated and the subintimal collagen, elastin and reticulin fibers, as well as the internal elastic lamina, were distorted, separated, or indistinguishable as extravasated erythrocytes filled these linear defects. The endothelium was consistently disrupted over these defects. Occasionally, the inner tunica media had distorted collagen fibers and myocytes. The tunica intima occasionally was missing and the exposed subintima had prominent fibrin deposition. Nodular lesions typically reflected a more advanced lesion. These had large numbers of intact and degenerate neutrophils, eosinophils, and reactive macrophages with brown intracytoplasmic granules (hemosiderin), and karyorhectic cellular debris, elevating the subintima, and extending deeper into the tunica media, severely distorting, fragmenting or replacing collagen, elastin, and reticulin fibers. These areas also were expanded by edema, moderate amounts of basophilic granular to finely fibrillar myxoid material (Fig. 8), and confluent aggregates of basophilic, Von-Kossa positive, refractile mineralized material. Nematode larvae occasionally were observed in the vascular lumen (Fig. 9) or in subintimal spaces in affected areas. Nematode larvae had a 2–4 µm thick cuticle, a 1–3 µm hypodermis with prominent, multinucleated lateral chords, and 20–40 µm platymyarian-coelomyerian musculature. Within the pseudocoelom a prominent intestine with uninucleated epithelium and an apical brush border were noted. No reproductive structures were in any of the nematode larvae sectioned, histomorphologic features of Crassicauda sp larvae.
Artery, Cuvier’s beaked whale (CBW).
Frequently, the tunica intima and media, and rarely the entire thickness of these nodules, had been effaced by a dense band of fibrous connective tissue with hypertrophic myofibroblasts and dissecting pleocellular inflammatory cell infiltrates. Inflammation obscured the myocytes, vasa vasorum, nervi vasorum, and serosa. Plaques represented the more advanced lesions and consisted of a diffuse, full-thickness replacement of the normal histoarchitecture of the arterial walls by a mature, dense, paucicellular, fibrous connective tissue (Fig. 10). Re-endothelialization was rare. The smooth muscle cells of the tunica media were replaced by extensive plaques in which isolated remnants of calcified necrotic debris (presumed nematodes) surrounded by a discrete lymphohistiocytic infiltrate. In addition, frequent variably sized areas of myofibroblast hypertrophy and hyperplasia, fibrosis, loss of the vasa vasorum, and rare areas of fibrohyaline-cartilage metaplasia were observed in the severe lesions.
Histologically, parasitized renal lobules had nematodes within the calices and dilated pelvices associated with pyogranulomatous pyelonephritis characterized by a moderate to marked amount of degenerate neutrophils and eosinophils with abundant karyorhectic cellular debris, reactive macrophages, hemorrhage, and mineralization. This inflammation frequently was accompanied by thrombosis, hemorrhage, tubular necrosis, areas of compression-induced coagulative necrosis, or areas of interstitial fibrosis with atrophy and extensive loss of nephrons. Parasitized renal arteries, and to a lesser extent renal veins, showed the previously described range of lesions with clusters of nematodes sometimes enmeshed in organized thrombi. Chronic suppurative and sclerosing ureteritis with intralesional nematodes was seen in the severe cases. Rare intra-arterial nematode larvae compatible with Crassicauda spp. occasionally were observed simultaneously with heavy renal nematodal infestation. Major pathologic findings in the vascular and renal systems are summarized in Supplemental Table 2.
DNA extracted from the 2 nematode samples presented the expected ∼638 bp 18 S fragment, and their sequences (603 bp) were identical to Crassicauda magna (KM233410), the only sequence available for this genus. A broad list of etiologic diagnoses related to cause of death were identified for each individual (Supplemental Table 1).
Discussion
This study provides evidence of a direct association between arteritis and heavy Crassicauda parasitism in CBW. From June 1995 to July 2014, 77 BWs stranded along the coasts of the Canarian archipelago, including 5 different species: Sowerby’s BW (Mesoplodon bidens, n = 1), Blainville’s BW (M. densirostris, n = 7), Gervais’s BW (M. europaeus, n = 12), True’s BW (M. mirus, n = 1), and CBW (n = 56). From these, complete autopsies were performed on 57 animals. Twenty other BWs, including 4 different species (Northern bottlenose whale [Hyperodon ampullatus, n = 1], Sowerby’s BW [n = 8], Gervais’s BW [n = 3], and CBW [n = 8]) from the Iberian Peninsula, Azores archipelago, United Kingdom, and Greece were also autopsied by our research team. From these 6 species of BWs necropsied, the CBW was the only species in which arterial and renal crassicaudiasis were found.
All stranded and autopsied CBW had consistent but variable pathologic arterial changes. They primarily included moderate to severe, segmental to diffuse, extensive fibrosing arteritis with arteriosclerosis, aneurisms, and thrombosis, especially in the mesenteric, gastroepiploic, renal, thoracic, and abdominal aorta. The macroscopic and histologic arterial lesions are similar to those reported in Strongylus vulgaris infections in horses, 11,12,18 as well as Crassicauda sp in blue whales, fin whales, and humpback whales. 30 The high numbers of individuals affected suggest that this parasitic genus is endemic in this host species in the Canarian Archipelago, as suggested in a number of other cetacean populations. 30,40
Histopathologic changes in the arteries of affected horses include fibrosis of the intima and/or media, with or without mononuclear inflammatory cells, neutrophils, eosinophils, and necrotic foci. 37 The severity of the inflammatory changes has been shown to be directly related to the presence of larvae entrapped in intimal thrombi, the intima itself, and less commonly, in the media or adventitia. 37 Similar inflammation of the mesenteric arterial wall has been observed in whales in association with nematode larvae. 30,31 Nematode larvae rarely were observed within the arterial lumen or subintimally in the CBW.
Rare cholesterol clefts were noted in only 1 individual examined suggesting that lipids may not play a role in the pathogenesis of these lesions. Atherosclerosis, as recognized in humans and other animal species, is not a comparable condition.
All CBW with arterial lesions had infestation by male and female adult Crassicauda in the kidney. Two CBW had arteritis, but the urinary tracts were not available to be examined due to predation. The associated lesions were similar to those reported in C. boopis infestation or trauma 30,31 and in other BWs, namely, Baird’s BW (Berardius bairdii), Arnoux’s BW (B. arnuxii), 28 and Stejneger’s BW (Mesoplodon stejnegeri) 45 by C. giliakiana. However, our cases had more severe renal arterial and ureteral involvement, while venous involvement was less pronounced. Venous lesions have been previously described as leading to severe occlusive vasculitis that might extend into the vena cava and form a nidus for venous thrombosis. 30,31 The author of those studies suggested fragmentation of these intravenous renal and vena caval lesions might have led to localized thromboemboli. In our study, such lesions were not identified in the vena cava.
The observation of nematode larvae in mesenteric arteries with adult nematodes in the kidney, suggested migration from the intestine to the kidney through the mesenteric and aortic vasculature as the most plausible pathogenesis to explain the lesion distribution. Based on the chronicity, extension, and severity of the arterial lesions, a life cycle similar to that proposed for C. boopis larvae is likely; 31 that is, migration of ingested larvae from the intestine into tissues would cause arterial lesions, prior to their maturation to adults in the kidney. Histologic detection of nematode larvae in the cranial mesenteric arterial wall, the distribution of aortic scarring, and the pronounced mesenteric, caudal abdominal aortic and renal arteritis were observations further supporting this theory. Final emergence into the renal spaces would complete the cycle. 30,31 In the present study, no aberrant migrating larvae were identified, but their presumed tracks along the intima of other major arteries (eg, celiac trunk, cranial thoracic aorta, aortic bulb, intercostal arteries) were readily evident on gross examination and histologically confirmed. Crassicauda larvae or adults were in arterial walls or kidneys. Their transmission would rely on shedding of larvated eggs and hatched larvae through the urine. An (extrasomatic) indirect life cycle with an intermediate host acting as a mode of infection also has been speculated, with a number of intermediate host species (euphausiids, capelin, herring, sand lance, or other natural food sources of the fin whale). 30,31 It is not yet known whether final larval maturation occurs in the arterial walls or in renal tissues. Nevertheless, larvae are thought to promote mechanical endothelial injury or erosions that would cause vascular disease.
This study did not examine any potential intermediate or paratenic host. In the Canary Islands, the diet of CBW has been shown to consist mainly of oceanic cephalopods, the most frequent being Taonius pavo, Histioteuthis sp., Mastigoteuthis schmidti, and Octopoteuthis sicula. 42 To the authors’ knowledge, Crassicauda sp intermediate stages have not been studied in these species of cephalopods. Further studies on the diet of this species with special emphasis on parasitic carriage in these latitudes are likely to provide more insight into the life cycle and transmission.
Acute, subacute, or chronic lesions of variable location, severity, and extent were only found in adults and juvenile/subadults. This result differs from those of Lambertsen, 31 who found a high prevalence of arteritis and renal crassicaudiasis in calves. This bias of our study could be related to the difference in populations examined, since all the animals in this study were adults and juvenile/subadults.
Lambertsen 31 favored direct transmission through incidental ingestion of C. boopis larvae shed in the dam’s urine during periods of nursing, rather than acquisition of infection from the milk, as has been argued for other host species. 8,9 Because Crassicauda was absent from mammary gland tissues examined in the 8 females included in this study, our data supports Lambertsen’s proposal. Nonetheless, transmission through milk cannot entirely be disregarded. Because cassicaudiasis has been proposed as a cause of mortality of whale calves, a larger number of autopsies in females, neonates, calves and juveniles/subadults would offer a more accurate epidemiological perspective.
Disease attributed to S. vulgaris larval migration through the cranial mesenteric artery, emergence of young adults, or adult nematodes feeding on mucosal material and incidental damage to blood vessel, are varied and include thromboembolism with segmental cecal or colonic ischemic necrosis, progressive exercise intolerance, anemia, hind-leg lameness, intestinal stasis, colic, and rarely intestinal rupture. 6,32 Lambertsen 31 speculated mesenteric arterial lesions would compromise digestive function, impairing vigor and health in whales, as largely described in horses. 49 However, he postulated that the mechanism of death in severe cases would be due to complete occlusion of the renal veins, massive swelling and abscessation of the kidneys, and congestive kidney failure. He also observed pyelonephritis involving both kidneys in conjunction with bilateral occlusions of the renal veins. Our findings are consistent with these proposed consequences of crassicaudiasis in CBW, although the documentation of clinical disease in this free-ranging species is difficult. The lesions here described seem to be of slow and chronic progression, likely dependent on parasitic reinfection and total burden, and affected animals may not experience any clinical signs if lesions are not extensive or if the species have adapted to the parasite. However, dramatic vascular disease with altered vascular resilience and compliance of the greater vessels could impact CBW, especially during their long and deep dives.
Morphology of the renal nematodes was consistent with the genus Crassicauda 5 of which 14 species currently are recognized: C. anthonyi, C. bennetti, C. boopis, C. crassicauda, C. fuelleborni, C. giliakiana, C. grampicola, C. magna, C. pacifica, C. tortilis, C. delamureana, C. pacifica (synonym of C. boopis), C. carbonelli, and C. duguyi (probably a synonym of C. magna). 23 Speciation based on morphologic features was hindered by moderate to advanced decomposition of a large percentage of the specimens. The genome of Crassicauda species has not been sequenced yet; thus, we conducted a comparative genomic analysis of this genus. The first molecular identity of 1 Crassicauda species was only recently defined using the 18 S rDNA, 23 which is a conserved DNA gene and thus may not show variations between species. We must include the possibility that we are dealing with a different species, especially considering that the only sequence available for comparison is from another host species and geographic area.
Determining causes of mortality in these species is highly complex in most instances. Extensive pathologic analyses are required but logistics and decomposition of carcasses by the time of autopsy may preclude pathologic examinations. Although trauma was considered the cause of death in 4 of the CBW of this study, crassicaudiasis with arteritis and no other significant disease was noted in the other 9 CBW. Indeed, the weakened parasitized animals may have been predisposed to trauma. Crassicaudiasis appears to be a leading natural cause of CBW mortality in the Canary archipelago.
In summary, our study suggests a direct association between arteritis and renal disease due to crassicaudiasis in CBW. Severe arterial lesions could result in fatalities from disseminated intravascular coagulation, multisystemic failure and severe renal damage, or major aggravation of coexisting disease. Further pathologic studies coupled with biochemical analysis and parasitologic identification involving wider geographic areas will help to elucidate the real risk this newly recognized pathologic condition poses for populations of CBW.
Footnotes
Acknowledgements
The authors thank Dr S. A. Taniwaki from LABMAS, Departamento de Medicina Veterinária Preventiva e Saúde Animal (FMVZ, USP) for technical assistance and sequencing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by Regional National Research Project CGL2012–39681 and a Technical Assistant Contract by Canary Islands Government (TEC0002955). This study involved a PhD student supported by the Ministry of Education of Spain through an FPU grant (AP2010–0002).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
