Abstract
Multiple endocrine neoplasia (MEN) embodies a group of diseases in human patients and domestic animals that are characterized by hyperplasia or neoplasia, or both, of two or more endocrine tissues. The MEN-1 syndrome is associated with menin gene mutations that induce various combinations of parathyroid, pituitary, and pancreatic endocrine tumors in humans. Two male, Domestic Shorthair cats developed symmetric alopecia, insulin-resistant diabetes mellitus, and pituitary-dependent hyperadrenocorticism at 12 and 13 years of age. Examination of skin biopsy specimens revealed atrophic dermatosis associated with hyperadrenocorticism. In one cat, cutaneous lesions consistent with paraneoplastic alopecia associated with pancreatic adenocarcinoma also were evident. Multiple invasive pancreatic beta cell carcinomas, pituitary corticotroph adenomas, and thyroid C-cell and parathyroid chief cell hyperplasia were diagnosed on the basis of results of gross, histologic, and immunohistochemical findings in both cats. Pancreatic exocrine adenocarcinoma was diagnosed in both cats. one cat also had hepatocellular carcinoma. Exons 1-8 of the feline menin gene were sequenced and were found to bear 93% homology with the human gene sequence, and the corresponding amino acid sequences shared 98% homology. Purification of total RNA and amplification of cDNA from lesional tissues to document mutations in the feline menin gene sequence were unsuccessful. The combination of lesions observed was consistent with the diagnosis of MEN-1-like syndrome in both cats.
Multiple endocrine neoplasia (MEN) syndromes are a group of diseases that are characterized by simultaneous development of hyperplasia or neoplasia, or both, of two or more endocrine organs or tissues in human patients. 22, 28 In humans, MEN is grouped into three major syndromes: MEN-1, MEN-2A, and MEN-2B (MEN-3). The MEN-1 syndrome (Wermer's syndrome) is characterized by development of various combinations of parathyroid, pituitary, and pancreatic endocrine tumors. 26, 28 Furthermore, MEN-1 phenotypic subtypes have been associated with the additional development of carcinoids, thyroid neoplasms, C-cell hyperplasia, adrenocortical hyperplasia/adenoma, and mesenchymal tumors. 15, 26 MEN-2A is characterized by development of thyroid medullary carcinoma in association with pheochromocytoma and parathyroid hyperplasia, whereas MEN-2B defines the familial thyroid adenocarcinoma syndrome. 22 Also, mixed and overlapping MEN-1 and MEN-2 syndromes have been described. 22 Although MEN-2A and MEN-2B derive from different mutations of the rearranged during transfection oncogene, 22 MEN-1 is an autosomal dominant hereditary syndrome resulting from the mutation of the menin gene, which codes for a protein involved in transcription inhibition. 28 The various mutations identified in the menin gene sequence of patients with MEN-1 have indicated correlation with development of different tumor phenotypes. 15
MEN-like syndromes in dogs, 3, 14, 30, 33 horses, 4 bulls, 27 and ferrets 6 have been reported. However, a condition closely resembling MEN-1 has been documented only in dogs 3, 30 and mice. 2 We describe the gross, microscopic, and immunohistochemical findings of MEN–1-like syndrome in two cats and compare the menin gene and protein sequences in cats and humans.
Material and Methods
Case histories
Cat No. 1
A 12-year-old, neutered male, Domestic Shorthair cat was initially referred for evaluation of polyuria, polydipsia, and polyphagia of 1 month's duration. Complete blood cell count (CBC), clinical biochemical analysis, and urinalysis were performed. The only abnormalities were high blood glucose concentration (319 mg/dl; reference interval, 60–120 mg/dl) and severe glycosuria (3+). Other abnormalities were evidenced at that time. Glycemia was controlled by insulin administration. After 3 years, clinical signs of disease recurred and the cat developed episodic convulsions. At that time, the cat had multifocal areas of patchy symmetric alopecia on the flank, abdomen, and extremities (Fig. 1). The skin was atrophic, and multifocal scaling of the four foot pads also was evident. Skin biopsy specimens were characterized by orthokeratotic hyperkeratosis and epidermal atrophy. Most of the hair follicles were in telogen arrest, with excess accumulation of tricholemmal keratin (Fig. 2). Size reduction of follicular units (“follicular miniaturization”) was evident. At that time, the cat had severe hyperglycemia (690 mg/dl), and high creatinine (3.2 mg/dl; reference interval, 0.9–2.2 mg/dl) and urea (352 mg/dl; reference interval, 19–34 mg/dl) concentrations. Also, increased serum cholesterol (271 mg/dl; reference interval, 71–156 mg/dl) and triglyceride (516 mg/dl; reference interval, 27–94 mg/dl) values were detected. A moderate increase in alanine transaminase (103 U/liter; reference range, 25–97 U/liter) also was found. Urinalysis results were characterized by prominent glycosuria (3+). The combination of insulin-resistant diabetes mellitus and increased cholesterol and triglyceride values with the cutaneous histologic findings were indicative of hyperadrenocorticism. Basal blood cortisol concentration (140 nmol/liter) was within the reference interval (20–270 nmol/liter). Cortisol concentration decreased to 61 nmol/liter by 3 hours after administration of a high-dose dexamethazone suppression test; however, the 61 nmol/liter value persisted after 8 hours from initial stimulation (reference interval, 0.4–40 nmol/liter). Abdominal ultrasonography revealed bilaterally symmetric adrenal gland enlargement and multiple hepatic hypoechoic parenchymal lesions, with gall bladder and bile duct distention. The combination of clinicopathologic findings was highly suggestive of pituitary-dependent hyperadrenocorticism with secondary insulin-resistant diabetes mellitus. After 1 month, the cat was euthanized at the owner's request following poor response to therapy and worsening of the clinical signs of disease. A full necropsy was performed.

Cat No. 1. Diffuse truncal and limb alopecia.
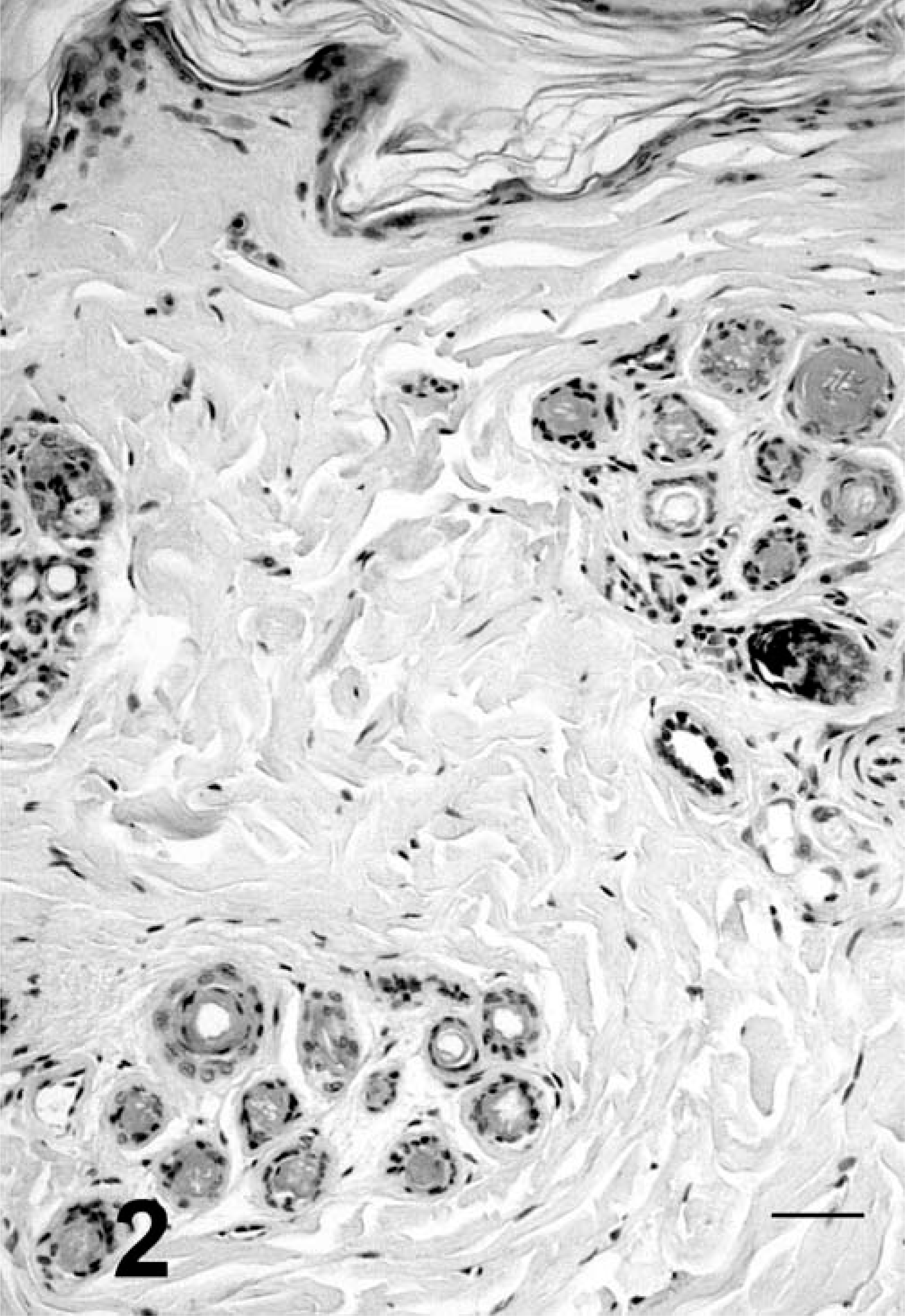
Skin, cat No. 1. Orthokeratotic hyperkeratosis and epidermal atrophy. Hair follicles have miniaturization and accumulation of tricholemmal keratin. HE. Bar = 50 µm.
Cat No. 2
A 13-year-old, neutered male, DSH cat presented with polyuria, polydipsia, and polyphagia of 3 months' duration. Clinical biochemical analysis revealed hyperglycemia (447 mg/dl). Urinalysis results were characterized by severe glycosuria (3+) and mild proteinuria (+). The cat had diffuse truncal hypotrichosis with moderate cutaneous atrophy. Examination of skin biopsy specimens revealed orthokeratotic hyperkeratosis, and epidermal and dermal atrophy. All hair follicles were in telogen phase (bulbs or anagen hairs could not be found) and follicular infundibular distention, with keratosis and a few comedos were present. Follicular miniaturization was not seen in this cat. Despite a progressive increase in insulin dosage, hyperglycemia persisted. Insulin resistance was confirmed by results of glucose tolerance tests, and blood glucose values reached 908 mg/dl after 20 days from initial presentation. Basal blood cortisol values (128 nmol/liter) were increased. Results of the adrenocorticotropic hormone (ACTH) stimulation test were characterized by an excessive response, with increasing cortisolemia, from a basal concentration of 128 nmol/liter to 791 nmol/liter (normal response <600 nmol/liter). Abdominal ultrasonography revealed bilaterally symmetric adrenal gland enlargement and a large (2.5 × 3 cm) pancreatic mass. The combination of clinicopathologic findings was highly suggestive of pituitary-dependent hyperadrenocorticism and pancreatic neoplasia. The cat was euthanized at the owner's request, and a full necropsy was performed.
Light microscopy
Multiple tissues were fixed in buffered 10% formalin, processed in routine manner, and embedded in paraffin wax; then, 5-µm-thick sections were stained with HE. Sections of the hypophysis were stained by use of the periodic acid–Schiff (PAS) technique.
Immunohistochemical analysis
A panel of panendocrine and specific endocrine markers, including antibody dilutions and retrieval conditions, was used (Table 1). Chromogranin A and neuron-specific enolase (NSE) were applied to all endocrine tissues. Expression of insulin, glucagon, and gastrin was evaluated only on pancreatic tissue. Anticalcitonin and antithyroglobulin antibodies were applied to thyroid and parathyroid tissue sections. Anti-ACTH was applied to hypophyseal tissue sections. Additionally, exocrine pancreatic and hepatic lesions were stained with anticytokeratin 8/18. All primary antibodies applied were directed against human antigens, but have been applied and have demonstrated the same tissue reactivity in several mammalian species.
4,
9,
14,
16,
20,
23,
29
Five-micron-thick tissue sections were placed onto poly–
Antibody panel used to characterize feline multiple endocrine neoplasia (MEN-l)-like syndrome.∗

NSE = Neuron specific enolase, ACTH = adrenocorticotropic hormone.
Heating retrieval = continuous microwave heating for 6 minutes at maximal power (750 W) with citrate buffer pH 6.0. Enzymatic retrieval = 1% pepsin for 10 minutes at 37°.
Gene sequencing
To sequence the normal menin DNA sequence, total RNA from fresh feline tissues collected during routine surgery was extracted by use of commercial kits (RNAeasy Mini Kit QIAGEN, Milano, Italy). Complementary DNA (cDNA) was obtained by retrotranscription with commercially available kits (Ready To Go Your-Prime First-Strand Beads, Amersham, Nerviano, Milano, Italy) primers: random hexamers, oligo dT, and gene-specific primer.
The primers used for cDNA sequencing of the feline Men1 gene (Table 2) were designed on the basis of the most conserved portions of the coding regions of known Men1 sequences available in the Genbank (accession Nos.: HUMAN Men1 U93237 and MUS MUSCULUS Men1 AF109390). The single-stranded cDNA was used as a template for polymerase chain reaction (PCR) analysis, which was carried out by use of an Eppendorf Mastercycler (Hamburg, Germany). The PCR products were applied to 2% agarose gel for electrophoresis, and the segments of predicted molecular weight obtained were gel purified using the QIAquick gel extraction kit (Qiagen, Milano, Italy), then were quantified. The fragments were sequenced directly using the Applied Biosystem cycle sequencing technology and a standardized protocol 1 on an automated DNA sequencer (ABI PRISM 310 Genetic Analyzer, Foster City, CA). The predicted amino acid sequence was obtained using the ExPASy proteomic server. 10 Total RNA also was extracted from formalin-fixed, paraffin wax-embedded material in the pancreatic and hepatic tissues from the two cats, was amplified, and was used to examine for the presence of menin gene mutations.
Feline menin gene primer sequences.
F = Forward, R = reverse.
Results
Gross findings
Both cats had multiple, white, pancreatic nodules that varied from 2 mm to 1.5 cm in diameter. Additionally, cat No. 2 had a large (2.5 × 3 cm) pancreatic mass with multifocal areas of hemorrhage and necrosis on cut section. Both cats had severe hepatic lipidosis. cat No. 1 also had a 5-cm hepatic mass with a necrotic center and multiple, white, firm, 0.3-mm to 1-cm, intrahepatic nodules. Bilateral severe adrenal gland enlargement (approx. 2 to 3 times normal size) was evident in both cats. The parathyroid glands of both cats were mildly increased in size. The pituitary gland of both cats was diffusely enlarged and had an irregular contour. All other organs, including the thyroid, were grossly unremarkable.
Microscopic and immunohistochemical findings
The pancreas had similar lesions in the two cats. Multiple neoplastic nodules expanded the pancreatic parenchyma, invaded the capsule, and extended to the peripancreatic mesentery. The nodules were composed of large (15–20 µm in diameter), round to polygonal cells with distinct cell borders, and variably abundant lightly eosinophilic, granular to homogeneous cytoplasm. Nuclei were round to irregularly shaped and hyperchromatic with indistinct nucleoli. Mitotic figures were rare. Neither intravascular neoplastic emboli nor distant metastases were observed.
In both cats, the neoplastic cells expressed chromogranin and synaptophysin with variable intensity, NSE and insulin (Fig. 3), and were anti-CK8/18, antigastrin, and antiglucagon negative. For both cats, a diagnosis of locally invasive beta cell carcinoma was made. Additionally, in cat No. 1, some of the neoplastic nodules were characterized by severe islet cell lipidosis. In the two cats, the pancreas was also characterized by tubular hyperplasia/dysplasia and tubular adenocarcinoma. Hyperplastic and neoplastic tubular structures expressed cytokeratin 8/18 with variable intensity and were negative for all endocrine markers. A diagnosis of exocrine pancreatic tubular adenocarcinoma was made.
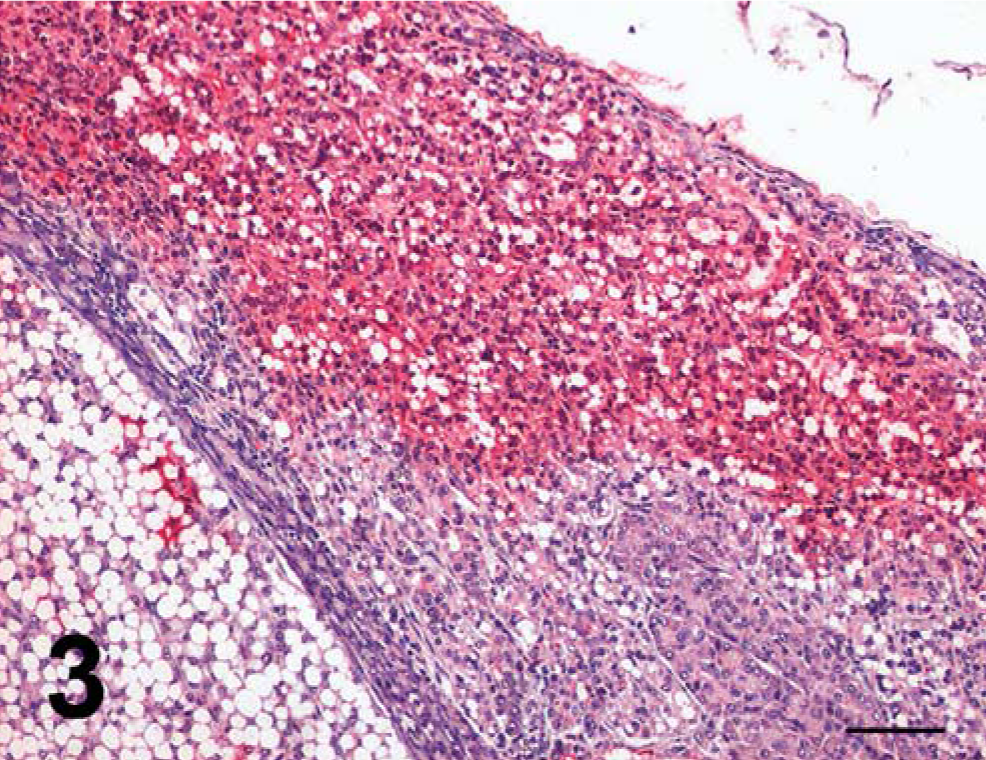
Pancreas, cat No. 1; beta cell carcinoma. Notice invasion of exocrine tissue by insulin-positive neoplastic cells. Antiinsulin immunoperoxidase stain, hematoxylin counterstain. Bar = 50 µm.
In the two cats, the adenohypophysis was compressed by an expansile, well-demarcated, unencapsulated neoplastic nodule composed of dense sheets of large, polygonal cells with distinct cell borders and abundant granular cytoplasm. Nuclei were round to oval with granular chromatin and one single, central to paracentral round nucleolus. Mitotic figures ranged from 0 to 1 per 400×-magnification field. These cells were PAS positive and variably expressed chromogranin A, synaptophysin, and ACTH (Fig. 4). A diagnosis of pituitary corticotroph adenoma was made.
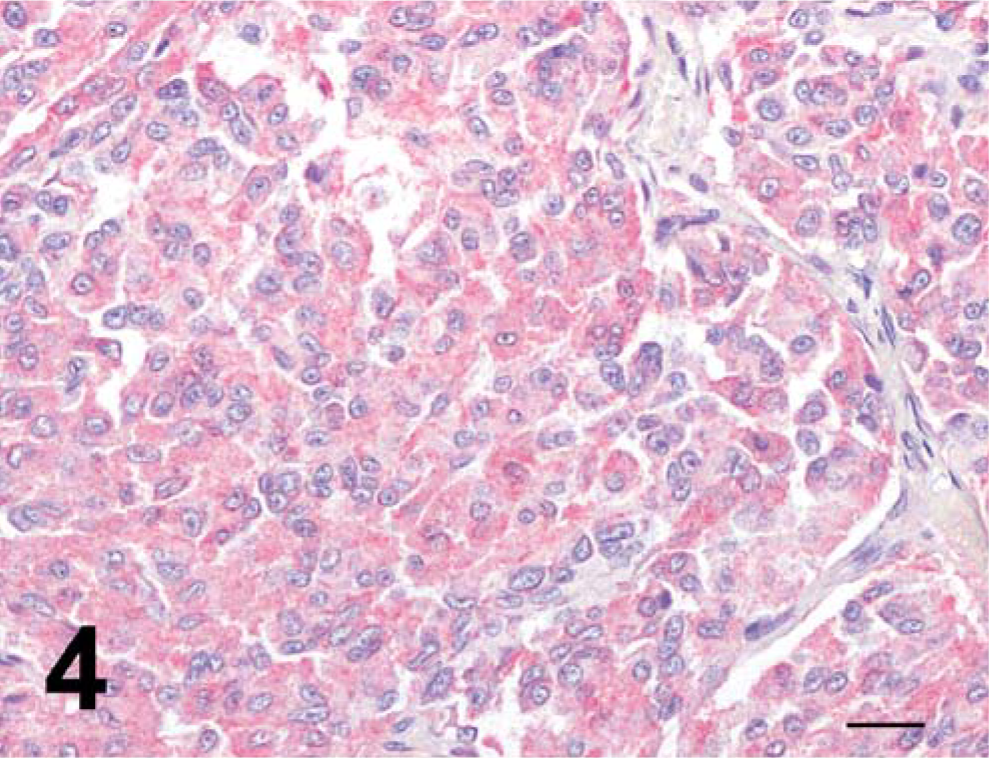
Hypophysis, cat No. 1; corticotroph adenoma. Notice diffuse granular cytoplasmic adrenocorticotropic hormone (ACTH) positivity of neoplastic cells. Anti-ACTH immunoperoxidase stain, hematoxylin counterstain. Bar = 20 µm.
The adrenal glands of both cats were characterized by symmetric adrenocortical hyperplasia of the zonae reticularis and fasciculata. In both cats, the interstitium of the thyroid was multifocally expanded by large aggregates of round to polygonal cells with indistinct cell borders and lightly eosinophilic to clear, finely granular cytoplasm. These aggregates consisted of chromogranin A-positive cells. These findings were consistent with C-cell hyperplasia. The thyroid of cat 2 was characterized by occasional follicle dilatation with flat epithelium and abundant intraluminal colloid. Parathyroid glands of both cats were composed of a monomorphic, uniform population of chief cells. These cells were characterized by distinct cell borders and abundant, lightly eosinophilic cytoplasm, and ranged from 12 to 20 µm in diameter (Fig. 5). Chromatin was granular and often was marginated; one to two round, blue, small nucleoli were evident. Mitotic figures were not found. Inactive chief cells, oxyphils cells, or intermediate cells were not found. Moreover, atrophic subcapsular residual parathyroid parenchyma was not apparent, excluding the diagnosis of diffuse adenoma. These findings were consistent with diffuse chief cell hyperplasia in both cats. Additional findings were hepatic lipidosis in the two cats and hepatic carcinoma in cat No. 1.
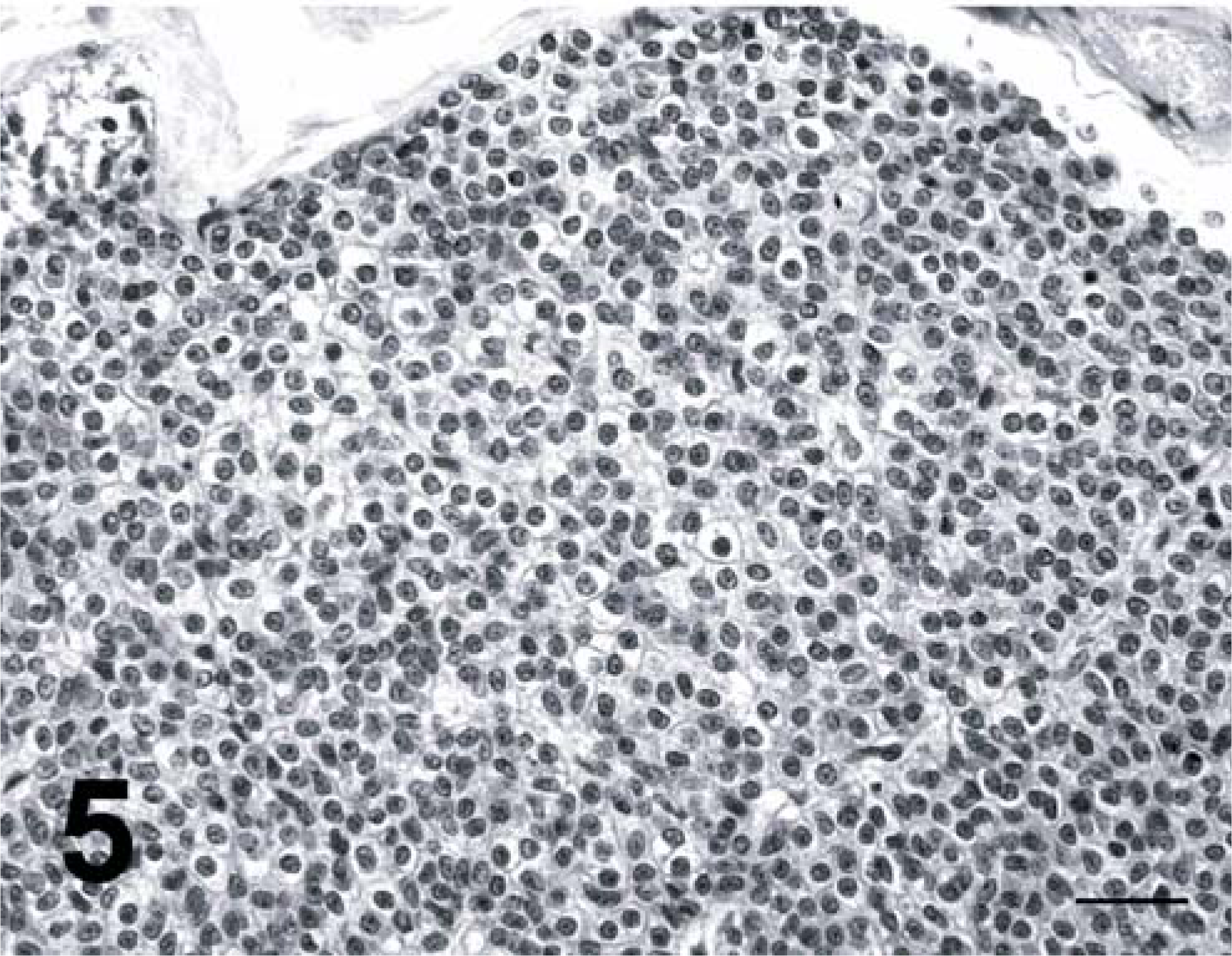
Parathyroid gland, cat No. 2. Diffuse parathyroid chief cell hyperplasia. Hematoxylin and eosin stain. Bar = 30 µm.
Menin gene sequence
The cDNA sequence of feline Men1 has been deposited in the European Bioinformatics Institute's nucleotide sequence database (http://www.ebi.ac.uk/embl/) under accession No. AJ844607. DNA sequencing of feline Men1 cDNA resulted in an incomplete sequence of 1,266 basepairs (bp). The sequence obtained accounted for exons 1–8. Translation of the feline Men1 gave rise to a sequence of 421 residues that shared 98% homology with human menin protein. The feline binding domains for NFkB and Jun D and the first Smad3 binding domain were identical to the human counterparts. Attempts to repeat the sequencing protocol from formalin-fixed and paraffin wax-embedded lesional tissue specimens from the two cats were unsuccessful.
Discussion
The development of endocrine neoplasms is generally considered a rare event in cats; 17 therefore, the simultaneous observation of multiple pancreatic beta cell carcinomas, pituitary corticotroph adenoma, and thyroid C-cell hyperplasia was considered unusual and highly suggestive of a MEN-1 like syndrome in these two cats. In human patients, a diagnosis of MEN-1 requires identification of at least two simultaneous endocrine lesions involving the parathyroid glands, pancreas, and pituitary gland, which typically are associated with the syndrome. 26, 32 Thus, the combination of lesions in the two cats fulfilled the requirements for diagnosis of a MEN-1 syndrome. A previous report describing two cats with insulin-resistant diabetes mellitus, hyperadrenocorticism, and pancreatic exocrine neoplasia, 13 closely resembled the condition in our cats. A pituitary adenoma was found in one of the cats of that previous report, but the hypophysis of the second subject was not examined. Those two reported cats might have developed a MEN-1 or overlapping MEN-like syndrome that was not fully characterized. Furthermore, the combination of the endocrine tumors observed in our two cats was consistent with an MEN–1-like syndrome subtype in humans that has seldom been described. 11, 18
In our two cats, the concurrent finding of symmetric adrenocortical hyperplasia and corticotroph adenomas confirmed pituitary-dependent hyperadrenocorticism. To the contrary, in human patients with MEN-1, nonfunctional adenomas are the most common pituitary neoplasm, whereas corticotroph adenomas are rare, representing only 2% of hypophyseal tumors. 26 Interestingly, in patients with MEN-1 attributable to pituitary adenomas, the most frequent concurrent endocrine lesions are multiple pancreatic insulinomas or glucagonomas. In humans, this is considered an extremely rare combination of lesions, compared with those of classical MEN-1, 15 where gastrinomas or nonfunctional neoplasms are more frequent 26 and insulinomas develop in only 10% of patients. 26 Both cats developed beta cell carcinomas that were characterized by invasion of peripancreatic mesentery, but neoplastic intravascular embolization and distant metastases were not identified. Conversely, in patients with MEN-1, the leading cause of disease-specific mortality is related to pancreatic endocrine metastases. 26
In human beings, the major medical problem associated with MEN-1 is development of hypercalcemia secondary to functional parathyroid lesions. 26, 32 Functional parathyroid hyperplasia is also common in dogs with MEN-1 like syndrome. 3, 30 Despite the finding of diffuse parathyroid chief cell activation, clinical signs associated with hyperparathyroidism were not observed in either cat. The presentation in the two cats differed from that in most human MEN-1 cases where asymmetric parathyroid hyperplasia or adenomas associated with clinical hyperparathyroidism are characteristic. 22, 28 This was the major difference observed when comparing our cats with MEN-1 human patients and dogs. The distinctive feline clinical and lesional phenotype should be taken into account when screening for feline MEN–1-like cases. Actually, the major clinical signs observed in our two cats were the cutaneous changes and the progressive development of insulin-resistant diabetes mellitus. Distinctive skin lesions such as truncal alopecia, cutaneous atrophy, and fragility have been reported in 33–72% of cats with hyperadrenocorticism. 25 Thus, in both cats, skin changes were considered highly indicative of associated hyperadrenocorticism. Additionally, cutaneous changes in one cat paralleled those reported in cases of feline paraneoplastic alopecia linked to exocrine pancreatic tumors or biliary adenocarcinomas. 21 At present, the pathogenetic correlation between skin lesions and pancreatic or hepatic malignancies has not been elucidated, although hypoproteinemia or deficiencies in biotin, zinc, or fatty acids have been proposed. 7 Also, increased or abnormal hormonal production has not been reported in cats with paraneoplastic alopecia. 21
The second and more severe clinical problem was the progressive development of poorly controlled diabetes mellitus. Insulin resistance is considered a feature of feline diabetes mellitus, and diabetic cats generally develop a relative decrease in insulin sensitivity, compared with that in human patients with type-2 diabetes mellitus. 5, 24 Contrary to our cases, a significant number of diabetic cats undergo remission after control of hyperglycemia is instituted. 24 However, cats often develop cortisol-induced insulin resistance 19, 31 ; therefore, hyperadrenocorticism may have aggravated the resistance to insulin treatment instituted in these cats. As a consequence, the poor prognosis in the two cats with MEN–1-like syndrome seemed related to inadequate control of diabetes mellitus, not to metastatic disease such as develops in humans.
In people, MEN-1 syndromes are related to the somatic germline mutation of the menin gene. 26 The menin protein resides in the nucleus and interacts with the N-terminus of the AP1 transcription factor acting as a transcriptional activator of JunD. 12 The high homology observed between the first eight feline exons and the corresponding human exons suggests that human and feline menin genes may share a common function. Specifically, the binding domains for NFkB, Jun D, and the first Smad3 were identical in cats and humans. 8 In humans, various lesional phenotypes have been associated with specific mutation loci of the menin gene. As an example, the combination of pituitary and pancreatic endocrine tumors has been associated with a specific mutation of the menin gene. 15 Patients with frameshift mutations develop pancreatic endocrine tumors, 15 and patients with frameshift mutations of exon 2 also develop pituitary neoplasia. These types of mutations are absent from patients without tumors of the hypophysis. 15 It will be interesting to compare the mutations in the feline menin gene with those in the human gene to confirm a specific lesional phenotype-genotype correlation in cats. Unfortunately, attempts to repeat the same sequencing protocol from lesional tissue specimens were unsuccessful. This was attributed either to RNA degradation or, more likely, to the large size of the sequences to be amplified by the selected primers since the maximal cDNA length that can be efficiently sequenced from formalin-fixed, paraffin wax-embedded material is approximately 200 bp.
In conclusion, the neoplasia combinations described here seems to be the first detailed description of MEN–1-like syndrome in cats. The lesional phenotype observed in the cats closely resembled a specific inherited subtype of human MEN-1. Moreover, the similarities observed at the clinical and pathologic level and the high homology of the protein sequence suggest a similar role of the menin gene in cats and humans. Thus, we are now screening cats clinically for MEN-like syndromes to collect fresh tissue specimens and to evaluate the presence of specific menin gene mutations.
