Abstract
A 12-year-old male domestic shorthair cat with a history of depression, aggression, and fever was presented for necropsy, and formalin-fixed tissues from a second male domestic shorthair cat of unknown age were submitted for histopathologic examination to the Athens Veterinary Diagnostic Laboratory. Both cats had granulomatous pneumonia associated with large numbers of parasite eggs and larvae, consistent with Aelurostrongylus abstrusus. Both cats also had larvae within glands in the colon that were identified morphologically as A. abstrusus first-stage larvae.
Aelurostrongylus abstrusus is one of the most common lungworms of cats. Adults live in the bronchioles and terminal ducts, and the eggs form nodular deposits in the alveoli. 12 This parasite has an indirect life cycle, and cats become infected by eating an intermediate or paratenic host that is infected with third-stage larvae. Various species of snails and slugs serve as intermediate hosts, and numerous species of birds, rodents, frogs, and lizards can serve as paratenic hosts. Third-stage larvae are ingested and migrate from the gastrointestinal tract to the lung where they mature to adults and establish an infection. Within the lung, eggs develop and hatch to first-stage larvae that are coughed up, swallowed, and eventually passed in feces. Although it has been reported that infective third-stage larvae penetrate the mucosa of the esophagus, stomach, and small intestine and travel via the bloodstream and lymphatics to the lungs within 24 hr, 12 convincing data documenting this are lacking in the veterinary literature. The prepatent period in experimental infections is 5–10 weeks. 6–8,10,11
Diagnosis of A. abstrusus infection can be challenging, and parasite detection in fecal samples via the Baermann method is generally considered the technique of choice. However, this has limitations because no larvae are passed during the prepatent period, larvae may be intermittently shed, and adult worms can persist in the lungs after the end of the patent period, which is typically 2-3 months, although much longer patent periods are occasionally reported. 8,10 One study successfully used polymerase chain reaction to detect A. abstrusus and differentiate it from other parasites in fecal samples and pulmonary swabs. 15 At least partly as a result of the challenges of diagnosis, the true prevalence of A. abstrusus infection is unknown. One study in California reported a prevalence of 1.9%, and a second study in Alabama reported a prevalence of 18.5% among stray cats euthanized at a humane shelter. 9,16 The Alabama study used Baermann fecal examination and gross and histopathologic examination of lung tissue. The method of diagnosis was not specified in the California study, although cats were necropsied. A study in Croatia reported prevalences, based on histopathologic diagnosis, ranging from 3.9% to 22%, depending on geographic region. 4 However, most reports of A. abstrusus involve single or small numbers of animals. 2,3,5 In none of these reports has A. abstrusus ever been reported in the intestinal mucosa of a cat. The present study reports the occurrence of 2 cases of A. abstrusus infection with larvae in colonic tissue of the affected cats.
In the first case, a 12-year-old domestic shorthair cat was presented to the Athens Veterinary Diagnostic Laboratory (University of Georgia, Athens, Georgia) for necropsy. The cat had a history of alternating episodes of depression and aggression over the previous 3 days with a variably elevated temperature ranging from 39.2°C to 41.1°C. Gross and histologic findings were indicative of nodular thyroid hyperplasia and hypertrophic cardiomyopathy; the clinical signs in this cat were attributed to hyperthyroidism. As an additional finding, pulmonary alveoli contained clusters of parasite eggs and larvae that were morphologically consistent with A. abstrusus (Fig. 1). Adult nematodes were not observed. Eggs had thin walls and were embryonated or contained morulae. Eggs with morulae contained 3-8 visible blastomeres that were 15-20 μm in diameter. Larvae had a maximum diameter of 14.5 μm, and lateral alae were visible on rare cross-sections. Mild granulomatous inflammation with rare eosinophils was associated with these foci. Similar larvae were occasionally observed in glands in the colon (Fig. 2) and along the mucosal surface. No inflammatory reaction or degenerative changes were associated with these larvae. Other incidental findings included pancreatic islet amyloidosis and mild interstitial nephritis.
In the second case, a cat was found dead outside and was necropsied by the referring veterinarian. Gross changes reported by the referring veterinarian included congested lungs with purulent material in the bronchi, an enlarged liver with nutmeg appearance, a flabby heart wall, and 1 ml of yellow-tinged fluid in the abdomen. Samples of kidney, lungs, large and small intestine, liver, stomach, spleen, heart, and urinary bladder were submitted in 10% buffered formalin to the Athens Veterinary Diagnostic Laboratory for histologic evaluation. Within sections of lung, there was marked smooth muscle hypertrophy of the pulmonary artery. Large groups of alveoli were filled with parasite eggs and larvae that were morphologically similar to those described in the first case and were consistent with A. abstrusus. Adult nematodes were not observed. Parasites were surrounded by large numbers of epithelioid macrophages and fewer multinucleated giant cells. Scattered bronchioles were filled with neutrophils and few larvae. Similar larvae were present in the colonic lumen and in glands in the colon (Figs. 3, 4). These glands were often mildly dilated and filled with mucus and scant necrotic cellular debris. The lamina propria contained small numbers of lymphocytes, plasma cells, and globule leukocytes. The liver was congested with few areas of centrilobular necrosis. Significant microscopic abnormalities were not observed in other examined tissues, and the cause of this cat's death was not determined.
A larva that was teased out of fresh colon from the first case had a kinked tail with a dorsal spine and was 306 μM in length (Fig. 5). Based on morphometric characteristics, the larva was considered a first-stage larva (L1) of A. abstrusus. 1,10,14
Archives of the Athens Veterinary Diagnostic Laboratory were searched, and an additional 11 cases of feline pulmonic A. abstrusus were identified that also had intestinal tissue available. Of these 11 cases, 8 had large intestinal sections available and 3 had only small intestinal sections available for evaluation. Hematoxylin and eosin-stained sections of intestine were examined, and A. abstrusus larvae were not identified in intestinal sections of any of the 11 cats.
Larvae of lungworms have not been reported in the colonic mucosa of any species. However, most studies focus on pulmonary pathology, and intestines are rarely examined. Evaluation of intestinal sections was reported in only 2 studies. In one of these, 11 cats were experimentally infected with A. abstrusus and were sequentially necropsied over a 41-day period. Sections of stomach, duodenum, jejunum, ileum, and large intestine were collected for histologic examination, and no abnormalities were noted. 13
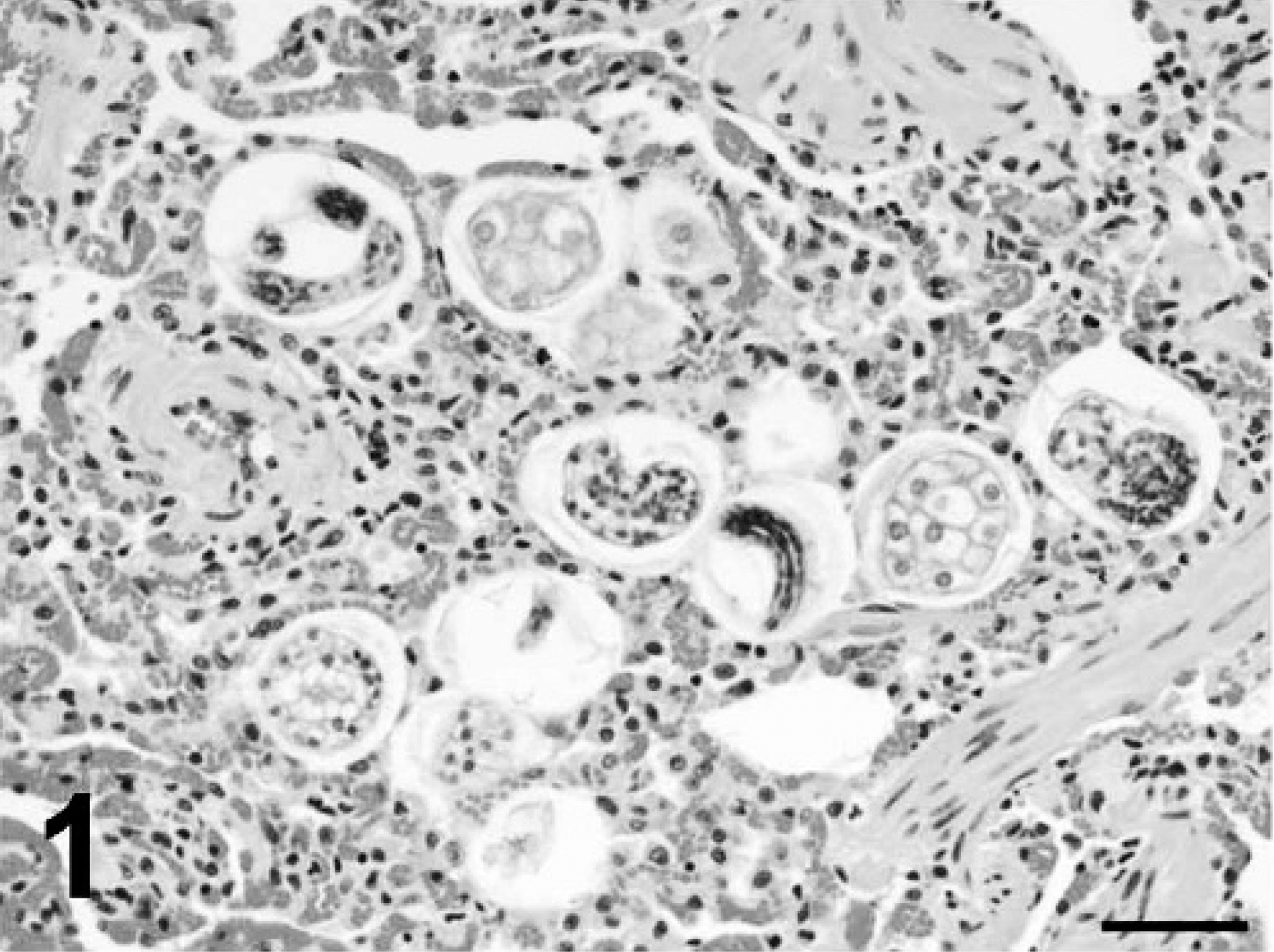
Lung; case 1. Aelurostrongylus larvae and eggs with morulae are present within alveoli. Hematoxylin and eosin. Bar = 50 μm.
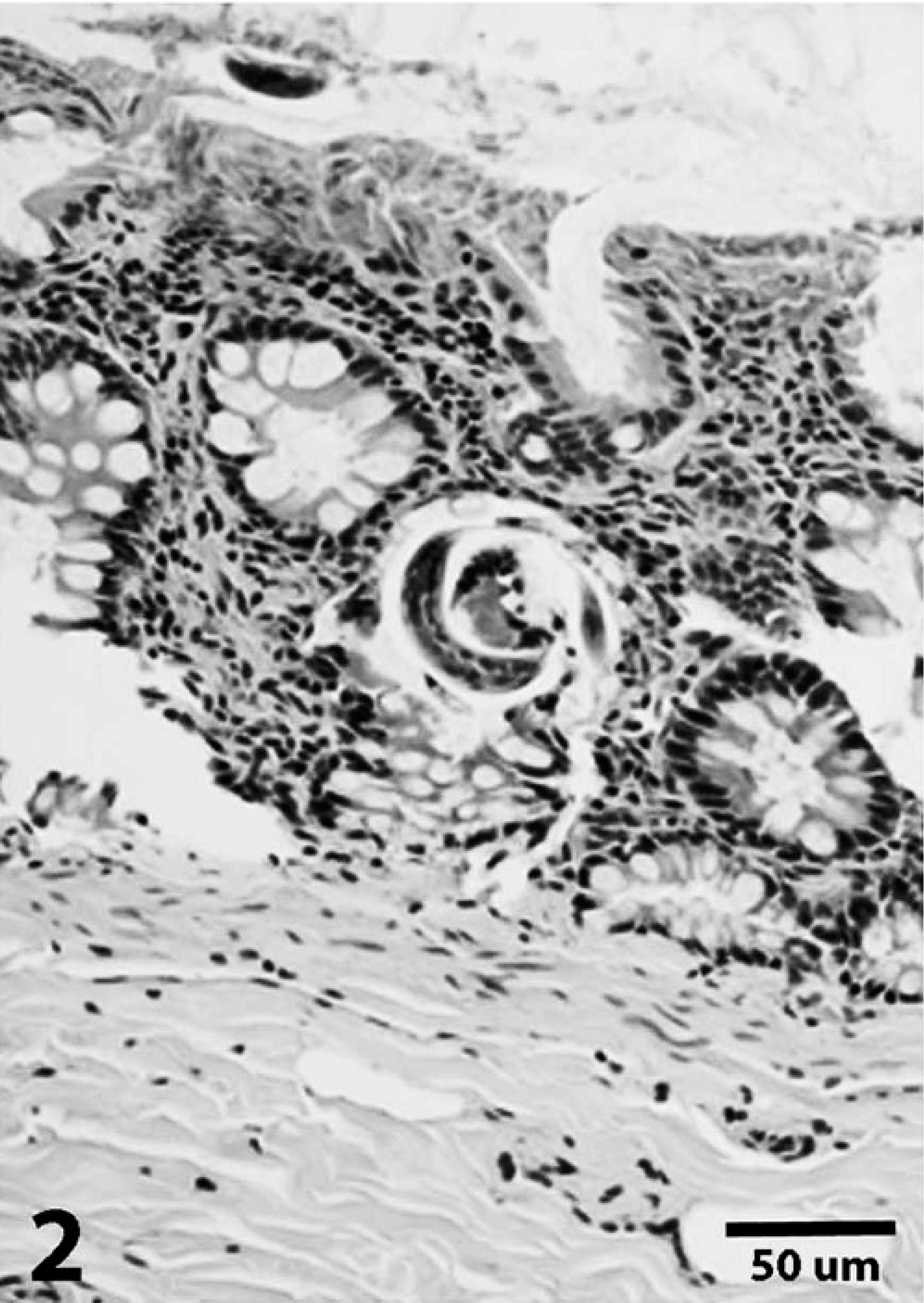
Colon; case 1. Aelurostrongylus larvae are noted in glands and along the mucosal surface. Hematoxylin and eosin. Bar = 50 μm.
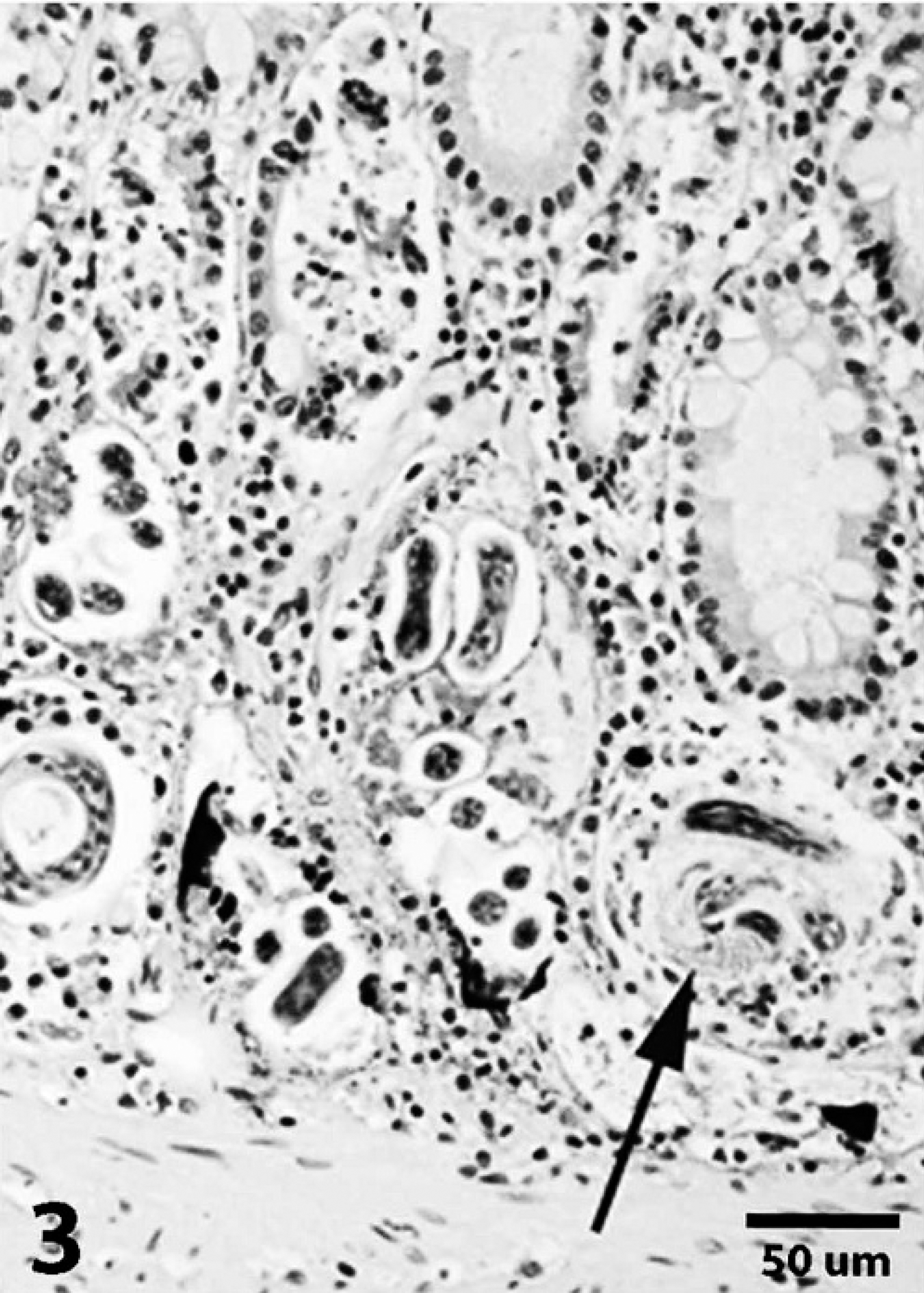
Colon; case 2. Numerous Aelurostrongylus larvae are present in glands. Some glands are dilated and contain mucus and cellular debris (arrow). Hematoxylin and eosin. Bar = 50 μm.
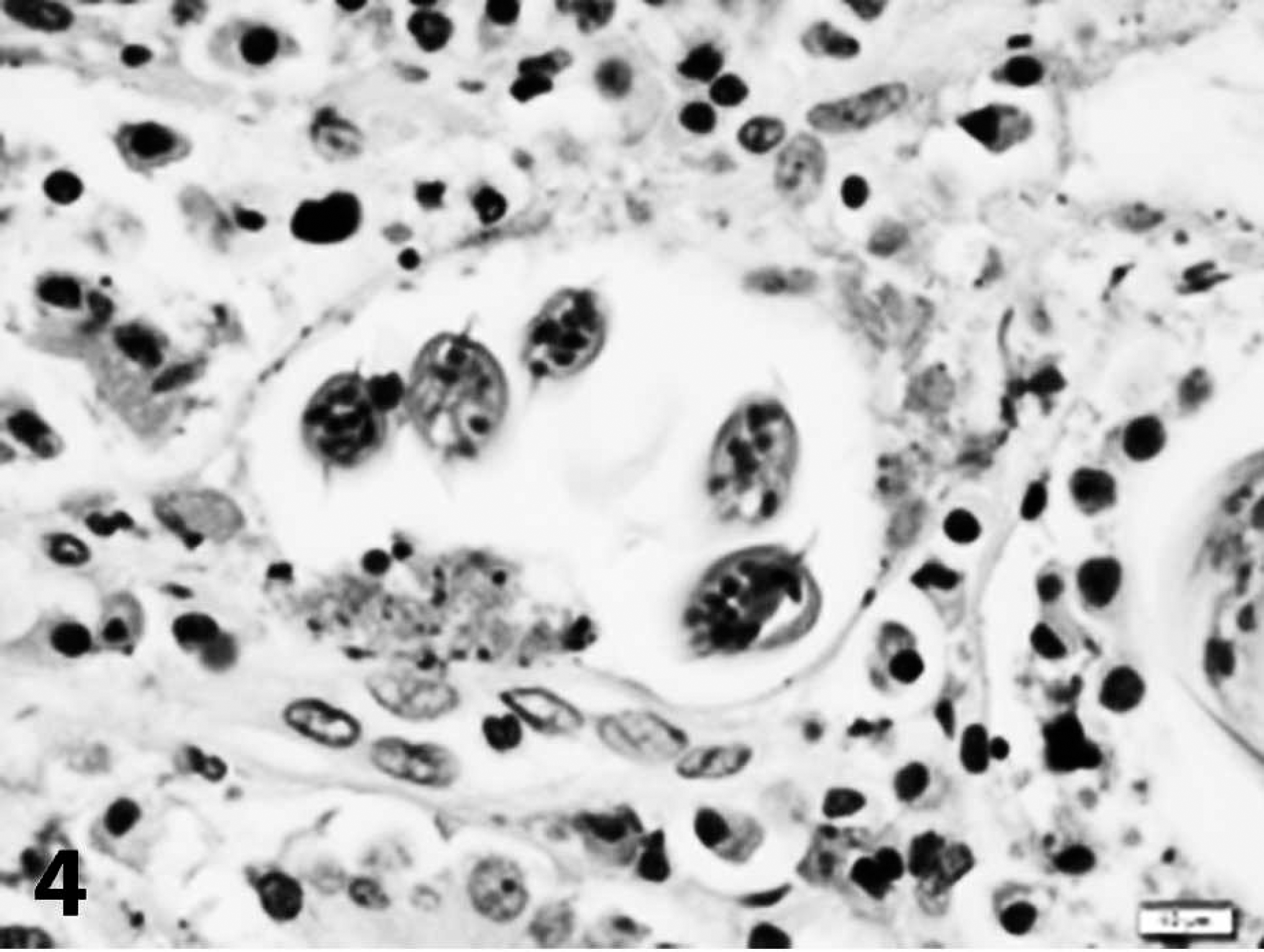
Colon; case 2. Five cross-sections of a coiled larva are present within a dilated gland containing scant cellular debris. The larva has prominent lateral alae. Hematoxylin and eosin. Bar = 10 μm.
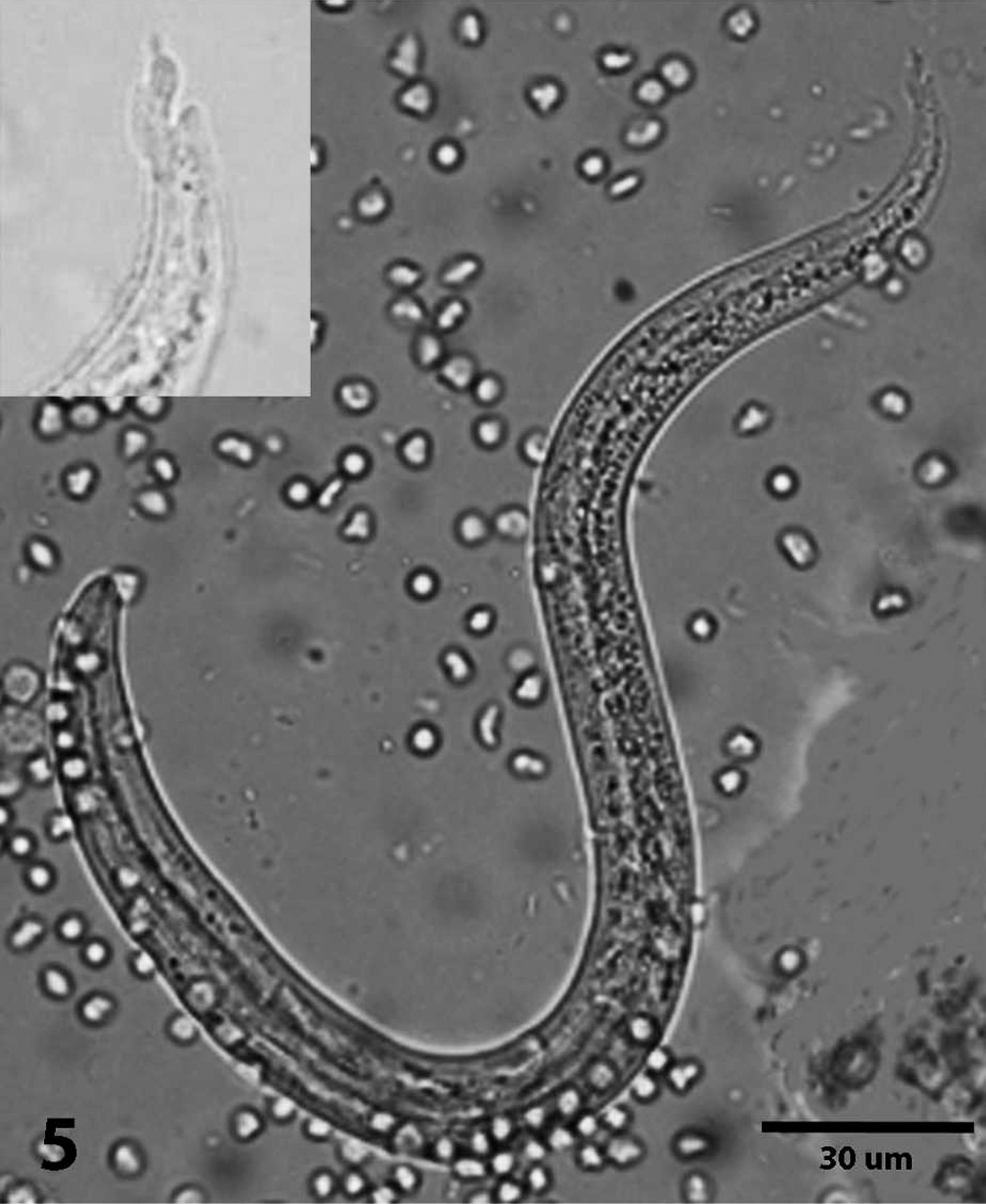
Photomicrograph of L1 teased from colon; case 1. Inset: Photomicrograph shows details of the characteristic s-shaped kinked tail. Bar = 30 μm.
Another study experimentally infected 9 cats with A. abstrusus and examined all organs histopathologically. The 9 cats were necropsied 12 weeks or more following infection, and abnormalities were only described in lungs. 7
Identification of the parasites in the 2 cases in the current study was based solely on histologic examination and morphometric characteristics. However, the notched tail on the larva is characteristic of Aelurostrongylus spp., of which A. abstrusus is the only species that has been reported in cats. Other pulmonary parasites reported in cats include Paragonimus kellicotti and Eucoleus aerophilus. In addition to other differences, P. kellicotti and E. aerophilus have operculated eggs or eggs with bipolar plugs that are distinctly different from the eggs of Aelurostrongylus spp.
The clinical significance of the intestinal larvae in the 2 cats in the present report is uncertain. The larvae did not appear to cause significant colonic disease. Only rare larvae were identified in the first cat, with no associated inflammatory or degenerative changes, and although the second cat had many more larvae, only mild dilation of glands and accumulation of mucus and scant cellular debris were noted. As first-stage larvae, presumably they were in the process of being excreted in feces. However, their presence in glands is unexpected. This may be a normal but rarely observed occurrence. Other possible causes include decreased gastrointestinal motility or postmortem migration from the lumen into the glands. Neither cat was reported to have decreased gastrointestinal motility, but historical information was limited. Postmortem migration is considered unlikely at least in the second cat because of the presence of other changes in the colon, albeit mild.
