Abstract
Chlamydia psittaci has not been reported to cause disease in domestic cats, to our knowledge. In contrast, C. felis infection is common in domestic cats and typically results in conjunctivitis, upper respiratory tract infection, and less frequently pneumonia. Herein, we report the pathologic findings and diagnostic features of a fatal case of psittacosis in a 7-wk-old domestic kitten. The animal was 1 of a litter of 5 that, together with the queen, were yielded to a pet rescue center in Wyoming. Over a period of ~3 wk, the kittens and queen became sick, thin, and icteric prior to death, despite antimicrobial treatments. Postmortem evaluation of a kitten revealed necrosuppurative hepatitis with Gimenez stain–positive intracellular bacteria, nonsuppurative pneumonia, and mild leptomeningitis. The diagnosis of psittacosis was made by 16S rRNA PCR using multiple primer sets and sequencing from liver. Psittacosis should be considered a differential diagnosis in domestic cats with intracellular bacterial hepatitis and interstitial pneumonia.
Keywords
Chlamydia psittaci infection is common in diverse avian species, 3 and has also been reported in dogs, pigs, small ruminants, cattle, and horses.1,6,9,11 The bacterium is an important zoonotic agent that causes asymptomatic infection to severe atypical pneumonia in humans. 5 To our knowledge, feline disease has not been reported. Five 4-wk-old kittens and the queen were submitted to a cat shelter in Wyoming. The queen was thin. Over the following ~3 wk the kittens and queen became icteric, inappetent, and died or were euthanized. The last kitten to die was clinically evaluated at the shelter 1 d before death. Clinically, the kitten was emaciated and had diarrhea and opisthotonus. A complete blood count revealed leukopenia (1.4 × 109/L; reference interval [RI]: 4–19 × 109/L) with marked neutropenia and lymphopenia. Serum chemistry revealed elevated conjugated and unconjugated bilirubin values of 27 and 14 µmol/L (RI: 0–1, and 0–3, respectively). Serologic testing at the clinic for feline immunodeficiency virus and feline leukemia virus was negative (SNAP FIV/FeLV combo test; Idexx).
The kitten died, and an autopsy performed the next day at the Wyoming State Veterinary Diagnostic Laboratory (Laramie, WY) revealed an emaciated animal with marked icterus. Serosanguineous fluid (~5 mL) was present in both pleural spaces. The liver was subjectively of normal size and color. The gall bladder was moderately distended with normal-appearing bile; the bile duct was patent.
Microscopic evaluation of the liver demonstrated small (<500 µm) foci of acute lytic necrosis of hepatocytes with neutrophilic infiltrates (Fig. 1A). There was also prominent sinusoidal leukocytosis. Hepatocytes around the necrotic foci contained dense clusters of gram-negative bacteria (Fig. 1B). Given the presence of intracellular bacteria, fluorescent antibody tests on fresh liver for Yersinia pestis and Francisella tularensis (the causative agents of plague and tularemia, respectively) were performed and were negative. The intracellular bacteria were subsequently found to be Gimenez stain positive (Fig. 1C). Evaluation of the lung demonstrated moderate diffuse interstitial pneumonia with infiltrating macrophages and lymphocytes. There were also patchy areas of alveolar hemorrhage and edema (Fig. 1D). Additional histopathologic findings were mild leptomeningeal infiltrates of lymphocytes and macrophages over neocortices, numerous renal glomerular capillary thrombi, and mild colonic cryptitis. Immunohistochemistry for feline infectious peritonitis virus and feline parvovirus, on brain and colon respectively, was negative.
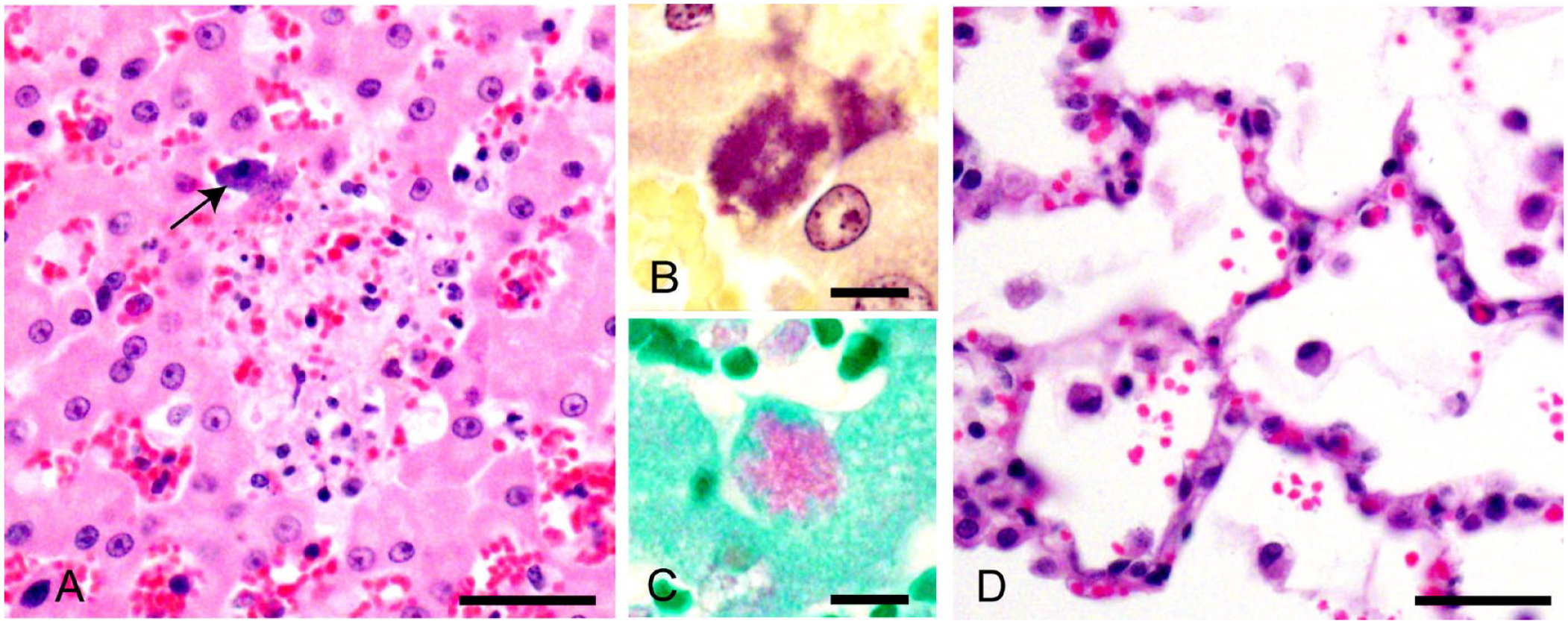
Microscopic findings in a kitten with psittacosis.
Additional testing on liver focused on intracellular pathogens, and included culture and PCR for salmonellosis, PCR for Coxiella burnetii, and immunostaining for Brucella abortus and Toxoplasma gondii. All tests were negative, and we proceeded to use a molecular approach to identify diverse bacterial 16S rRNA sequences. 10 This newly described method 10 utilizes 16 primer pairs designed using a computation approach to collectively amplify all known bacterial 16S rRNA genes, and provides an alternative to the use of degenerate primers. In brief, DNA was isolated from fresh-frozen liver (DNeasy kit; Qiagen), and PCR was completed using the 16 primer pairs, as independent reactions, in a volume of 20 µL as described. 10 Agarose gel electrophoresis revealed a DNA band of ~800 bp in 4 of the 16 primer pairs used; these primer pairs each shared a forward or reverse primer. The primer pair that generated the strongest band was forward CTACGGTGGGCTGCAGT and reverse TCACGGCACGAGCTGAC. PCR was repeated using this primer pair and identical conditions, except with a 60-µL reaction volume. The PCR mix was cleaned up (PureLink quick gel extraction, PCR purification kits; Invitrogen), then submitted for Sanger sequencing. Reverse and forward sequences were aligned then searched against the NCBI nucleotide database using BLASTN (http://blast.ncbi.nlm.nih.gov/Blast.cgi). The amplified sequence was a 100% match with the C. psittaci 16S rRNA gene (GenBank HF545614). No other bacterial 16S rRNA sequences provided an exact match with this sequence. DNA was extracted from deparaffinized tissue sections (DNeasy kit; Qiagen) per the manufacturer’s instructions. Subsequent nested PCR specific for C. psittaci on paraffinized tissue block scrolls was positive on liver, tracheobronchial lymph node, lung, and intestine, but not brain. 8 PCR on scrolls from paraffinized tissues were negative for felid herpesvirus 1 (brain and lung) and feline calicivirus (lung).
Our case demonstrates that C. psittaci should be considered as a differential diagnosis in domestic cats with gram-negative sepsis, acute hepatitis, and/or interstitial pneumonia. Although only 1 of the 6 cats that died was examined postmortem, all had developed icterus and progressive weight loss, suggesting the same disease.
Although C. psittaci has not been reported as a cause of disease in cats, conjunctivitis and rhinitis were described in a 5-y-old domestic cat 1 mo after introduction of a macaw to the household; a chlamydia agent presumed to be C. psittaci was found in both animals, and the cat responded to doxycycline therapy. 7 C. felis and C. psittaci were historically grouped as one species (Chlamydia psittaci group), but were reclassified as separate species in 1999. 2 Given the historic classification of C. felis and C. psittaci as the same species, it is possible that previous cases of C. psittaci in domestic cats have occurred and were reported, but were not distinguished from C. felis because of the molecular testing limitations of the time. 12 Although C. psittaci and C. felis have distinct host preferences and disease profiles, based on whole-genome sequence analysis, these pathogens are closely related. 13 In domestic cats, C. felis infection is a common cause of upper respiratory tract disease and conjunctivitis, and a less common cause of nonsuppurative interstitial pneumonia. C. felis is also an uncommon cause of conjunctivitis in humans. 4 In contrast, C. psittaci is an important zoonotic agent that can cause pneumonia and flu-like disease in humans. 5 The cat in our case had nonsuppurative interstitial pneumonia indistinguishable from C. felis pneumonia. The findings of hepatitis with bacteria, and leptomeningitis, have not been described with C. felis. However, the possibility of milder C. psittaci infection without hepatitis and leptomeningitis needs to be considered. Hepatitis similar to our findings is common in avian psittacosis. We found no evidence of coinfections contributing to our disease findings.
Our findings indicate the importance of C. psittaci–specific testing in domestic cats with consistent clinical or pathologic findings. The presence of the same clinical findings in the queen and kittens is unusual but is consistent with exposure to a virulent pathogen to which there is poor immunity. C. psittaci in dogs and horses has been associated with reproductive loss consistent with increased susceptibility given the physiologic immune suppression of pregnancy.6,11 We speculate that the queen became infected during gestation as a consequence of wild bird hunting and then infected the kittens. However, the source of infection was not determined. Poor nutritional status of the queen may have contributed to the predisposition to infection.
We used a recently described approach to identify the involved bacterial species. 10 Although this approach was developed to amplify diverse bacterial species in the study of microbial communities, here we demonstrate its value as a tool in veterinary laboratory medicine. Potential benefits include the ability to detect diverse bacteria, low cost, speed, and ease of PCR amplicon sequencing without the need for plasmid subcloning. Although requiring 16 initial PCR reactions, this approach avoids problems associated with the use of degenerate primers, including synthetic bias, which can result in failure to amplify target DNA.
Footnotes
Acknowledgements
We thank Julia A. Fox for providing training in the PCR method used.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
