Abstract
Chelonian intranuclear coccidiosis has been reported once, in two radiated tortoises (Geochelone radiata), and is apparently rare. We describe intranuclear coccidiosis diagnosed histologically in two radiated tortoises, three Travancore tortoises (Indotestudo forstenii), two leopard tortoises (Geochelone pardalis), one bowsprit tortoise (Chersina angulata), and one impressed tortoise (Manouria impressa). Infection was systemic and involved alimentary, urogenital, respiratory, lymphoid, endocrine, and integumentary systems. Trophozoites, meronts, merozoites, macrogametocytes, microgametocytes, and nonsporulated oocysts were seen histologically or by electron microscopy. intracytoplasmic and extracellular stages of parasite development also were identified histologically. Sequencing of a coccidial 18S rRNA consensus polymerase chain reaction (PCR) product revealed a novel sequence that provided phylogenetic information and may be useful for further diagnostic test design. intranuclear coccidiosis was associated with variable degrees of inflammation in all cases, was considered the cause of death in six tortoises, and was a substantial contributing factor to the cause of death in two tortoises.
More than 30 species of coccidian parasites have been described in Chelonidae (tortoises and turtles). 3– 6, 11 Eimeria spp. are apparently the most common coccidia in chelonians, but Caryospora cheloniae, Isospora spp., and Mantonella sp. also have been described. 11 Caryospora cheloniae is an important pathogen of mariculture-reared green turtles (Chelonia mydas), 10 but to the authors' knowledge, there are no reports of pathologic changes associated with these other coccidia.
The parasitic coccidia of vertebrates most commonly have endozoic development (merogeny and gamogeny) within the cytoplasm of enterocytes. At least 11 species of Eimeria, Isospora, and Cyclospora are caryotropic, having intranuclear developmental stages. Intranuclear coccidia have been described in all classes of vertebrates, including four species of Isospora that infect lizards. 2 Lesions associated with intranuclear coccidiosis are apparently rare, with only a single report of fatal infection in two radiated tortoises (Geochelone radiata). 9 We describe gross and light and electron microscopic findings of fatal intranuclear coccidiosis in eight additional tortoises representing four species from six sources.
Materials and Methods
Case histories
Case No. 1 was a wild-caught, adult male, impressed tortoise (Manouria impressa) native to Thailand that had been confiscated by the US Fish and Wildlife Service (USFWS) during illegal shipment into the USA. The tortoise was relocated to the Fort Worth Zoo, Fort Worth, Texas. On arrival, the tortoise appeared to be in good physical condition. Over several months, the tortoise became anorectic and emaciated. Anaerobic culture of ascites fluid revealed growth of Clostridium sp. The tortoise did not respond to a variety of antibiotics or anthelmintics (fenbendazole or metronidazole), and died.
Case No. 2 was a 7-month-old leopard tortoise (Geochelone pardalis) bred at a private facility in Baton Rouge, Louisiana. The tortoise died after a 4-week history of lethargy, poor appetite, and apparent icterus.
Case Nos. 3 and 4 were adult, captive-bred, radiated tortoises from a facility on St Catherine's Island, Georgia. Case No. 3 presented with lethargy, weakness, and bilateral ocular and oral mucoid discharges. The tortoise was treated with antibiotics, but died 11 days after presentation. Case No. 4 was an adult male radiated tortoise that presented with bilateral ocular and nasal mucoid discharges. The tortoise was treated with antibiotics, but died 3 days after initial onset of clinical signs of disease.
Case Nos. 5–7 were Travancore tortoises (Indotestudo forstenii) native to Celebes that were confiscated by the USFWS on entry into the USA. The tortoises were relocated to the Wildlife Conservation Society, Bronx, NY. Case No. 5, an adult female, died after a 3-week history of weakness and dehydration. During that time, it had been treated with antibiotics and fluids administered subcutaneously. Case No. 6 was a male that had been clinically normal until 2 days prior to death, when it separated from the group and was found dead. Case No. 7, a male, had a 3-day history of lethargy, weakness, and red conjunctiva. Despite being treated with antibiotics, it died.
Case No. 8 was an adult male Bowsprit tortoise, Chersine angulata, from the zoo in central Florida. It had a history of anorexia and lethargy. Biopsy specimens of liver, kidneys, and small intestine were obtained during exploratory coelotomy. Biopsy results indicated intranuclear coccidia in the renal tubular epithelial cells. Fecal parasite examination revealed coccidian oocysts. The tortoise died 1 day after surgery.
Case No. 9 was an adult female leopard tortoise from a private collection in New York. It presented with a 1-month history of anorexia followed by death. Ancillary diagnostic procedures were not performed on this tortoise.
All tortoises were necropsied within 24 hours of death. All tissues to be examined microscopically (Table 1) were preserved in neutral-buffered 10% formalin, processed in routine manner, sectioned at 5- to 6-µm thickness, and stained with HE. Select tissues from some cases also were stained by periodic acid–Schiff (PAS) and Fite's acid-fast techniques. Case Nos. 1, 2, and 9 were necropsied on site, and sections of selected viscera were submitted to Northwest ZooPath for histologic examination. Sections of kidney from Case No. 1 and tympanic scale, and middle and inner ear from Case No. 2 were further fixed in 4% glutaraldehyde and examined by electron microscopy at the Armed Forces Institute of Pathology, Washington DC (case No. 1), and at the Electron Microscopy Core Laboratory, University of Florida (UF) (case No. 2). Case Nos. 3 and 4 were necropsied on site, and tissues were submitted to the Wildlife Conservation Society (WCS), Bronx, NY for histologic examination. Case Nos. 5–7 were necropsied on site at the WCS. Necropsy and histologic examination of Case No. 8 was performed at the UF. All slides of tissues from all tortoises were subsequently reread by one pathologist (M. Gardner).
Distribution of intranuclear coccidia and severity of related lesions in affected tortoises.
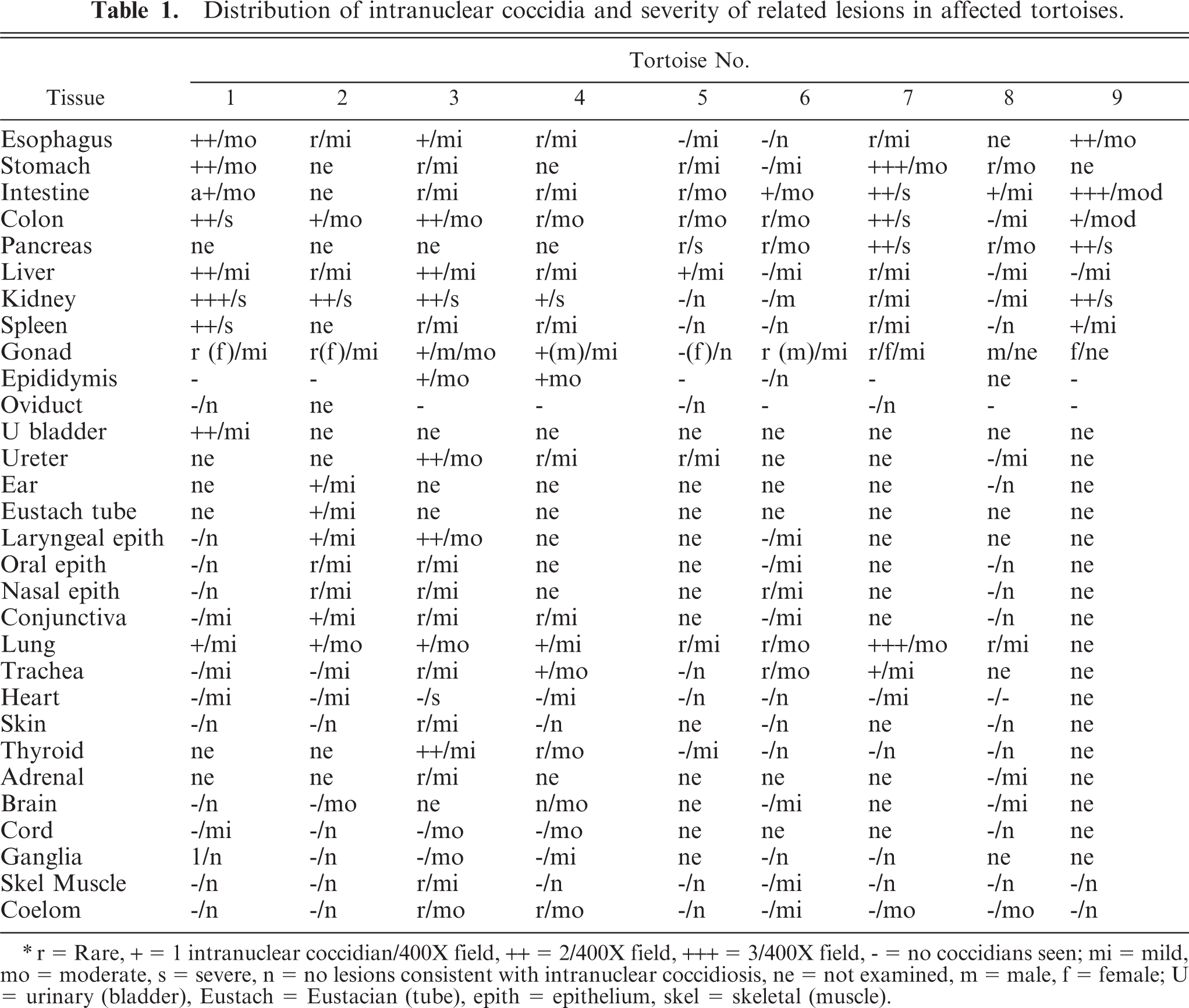
r = Rare, + = 1 intranuclear coccidian/400X field, ++ = 2/400X field, +++ = 3/400X field, − = no coccidians seen; mi = mild, mo = moderate, s = severe, n = no lesions consistent with intranuclear coccidiosis, ne = not examined, m = male, f = female; U = urinary (bladder), Eustach = Eustacian (tube), epith = epithelium, skel = skeletal (muscle).
Polymerase chain reaction (PCR) analysis and sequencing
For each tortoise, DNA was extracted from microtome slices of paraffinized, infected tissues using the DNEasy kit (Qiagen, Valencia, CA). Primers were designed from conserved regions of various coccidian species in various genera targeting an approximately 350-basepair segment corresponding to bases 1,018–1,377 of the small subunit ribosomal RNA gene of Toxoplasma gondii (Genbank accession No. L37415). Fifty-microliter reactions were run, with each containing 4 μl of extracted DNA, 1 µM forward primer 1135F 5′-ACYATAAACTATGCCRACTAGA-3′ (Y = C or T, R = A or G), 1 µM reverse primer 1503R 5′-CYTCCYTRCRTTARACACGCAA - 3′, 200 µM each dATP, dCTP, dGTP, and dTTP, 2.5 U of Pwo DNA polymerase (Thermo Hybaid, Franklin, MA), and 4 µl of PCR buffer (Thermo Hybaid). The mixture was amplified in a thermal cycler (PCR Sprint, Thermo Hybaid) with an initial denaturation at 94°C for 5 minutes, followed by 40 cycles of denaturation at 94°C for 30 seconds; annealing at 48°C for 60 seconds; DNA extension at 72°C for 60 seconds; and a final extension step at 72°C for 7 minutes.
The PCR products were resolved in 1% agarose gels. Bands were excised and purified using the QIAquick gel extraction kit (Qiagen). Products were sequenced directly in both directions using the Big-Dye Terminator Kit (Perkin-Elmer, Branchburg, NJ) and were analyzed on an ABI 377 automated DNA sequencers at the UF DNA Sequencing Core Laboratory.
Analysis of data
The sequences were compared with known sequences in GenBank (National Center for Biotechnology Information, Bethesda, MD), EMBL (Cambridge, UK), and Data Bank of Japan (Mishima, Shiuoka, Japan) databases using BLASTN. 1
Predicted homologous 344–357 nucleotide sequences of representative coccidial 18S rRNA available from GenBank were aligned manually. Phylogenetic analyses of the predicted alignment were performed with the PHYLIP (Phylogeny Inference Package, version 3.61) program package. 8 A maximum likelihood tree was generated using Dnaml with global rearrangements and 10 jumbles. Gaps of all lengths were counted as single events. Babesia caballi (GenBank accession No. AY534883), a noncoccidian apicomplexan, was used as the outgroup. The validity of the tree topology obtained was tested by using bootstrap analysis 7 starting with Seqboot with 100 resamplings from the aligned sequences, followed by maximum likelihood calculations, and calculation of the most probable (consensus) tree using Consense.
Results
Histopathologic changes
The distribution of intranuclear coccidia and associated lesions in these tortoises, on the basis of tissues available for microscopic examination, are summarized in Table 1. Generally, infection in all tissues was accompanied by lymphocytic or lymphoplasmacytic inflammation and variable degrees of necrosis. Lesions suggestive of coccidiosis were also frequently detected in heart and central nervous system (CNS) tissue, but coccidia were not seen in these tissues. Case Nos. 5 and 7 had concurrent amebiasis, and Case Nos. 5, 7, and 8 had bacterial septicemia; the lesions associated with these processes occasionally obscured morphologic features of intranuclear coccidiosis in these animals.
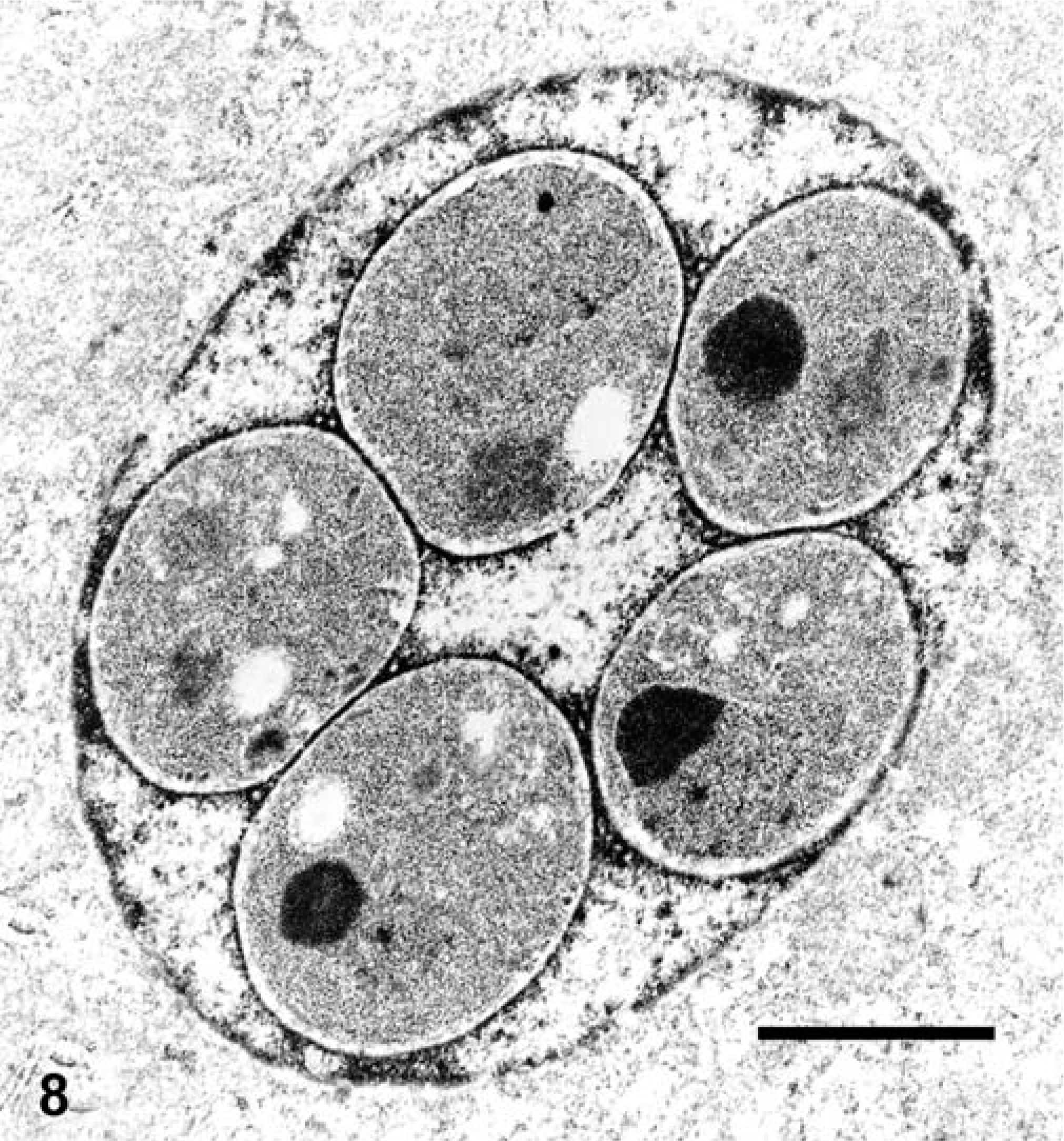
The coccidia were best observed in the HE-stained sections, and selective staining of the organisms was not observed in histologic sections stained by PAS or Fite's acid-fast techniques. Organisms were most abundant in the kidney and pancreas, but could be readily found in other tissues such as liver, intestine, and spleen. The organisms were most often detected in the nucleus of infected cells. Gamete stages were spherical, up to 6 µm in diameter, and had a slightly granular basophilic cytoplasm and a small, slightly eccentric nucleus. The cytoplasm of the gametes sometimes had a pink hue in HE-stained specimens. Macrogametes and microgametes could not be discerned at the light microscopic level. Meronts were spherical, and had a central residual body surrounded by up to several (sometimes >16) merozoites. Many infected cells had more than one gamete, and some had as many as four gametes. Rarely, gametes and meronts were detected in the same nucleus. Infected cells with single small gametes in the nucleus were difficult to distinguish from uninfected cells with hypertrophied nucleoli, especially in partially autolysed tissue. All stages of the coccidia also were detected rarely in the cytoplasm of various cell types.
Alimentary tract
Lesions involving the alimentary tract were similar at all levels of the tract, but were most severe in the small intestine and colon. Intranuclear coccidia in various stages of development were detected in the mucosal epithelium, and rarely in unidentified mononuclear cells resembling macrophages in the lamina propria or submucosa. The number of cells containing coccidia varied considerably (Table 1), and for some animals, a diligent search at 400X magnification was needed to find the organisms. Lesions associated with coccidian infection included necrosis of infected epithelial cells and variable degrees of mucosal epithelial hyperplasia (Fig. 1). The presence of coccidia was accompanied by infiltrates of small to large numbers of lymphocytes and plasma cells, with a few granulocytes and macrophages distributed throughout the lamina propria and submucosa and occasional migration through the overlying mucosal epithelium. Bacterial overgrowth was seen in ulcerated portions of the colon in case No. 1, and case Nos. 5 and 7 had concurrent amebic and bacterial infection in ulcerated portions of the colon. Edema of the submucosa was prominent in all affected tortoises.
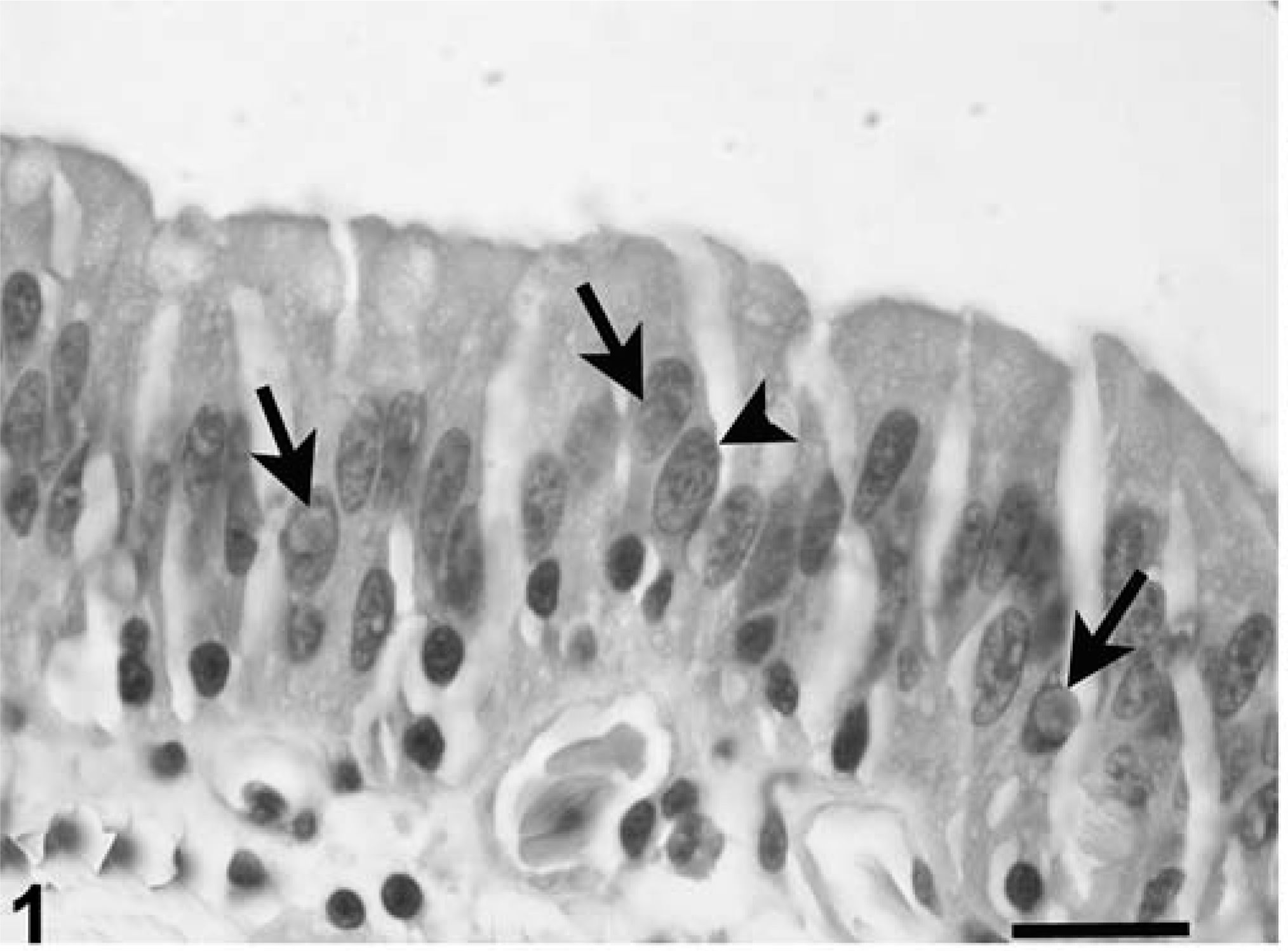
Small intestine, impressed tortoise, case No. 1. Notice intranuclear gametes (arrows), and intranuclear schizont (arrowhead). Only a few lymphocytes and plasma cells are evident in lamina propria and within the epithelial layers. HE. Bar = 30 µm.
Coccidia were detected in the nuclei of acinar and ductular epithelium of the pancreas, and a few intracytoplasmic meronts and merozoites were seen in unidentified cells resembling macrophages and degenerative ductular epithelial cells. Rarely, ducts contained intraluminal nonsporulated oocysts. Parasitism was accompanied by moderate to severe degeneration and necrosis of infected cells; acinar atrophy and loss; interstitial fibrosis, ductular hyperplasia, and dilatation; and acinar cell necrosis. Inflammation was interstitial, multifocal, and predominantly, lymphocytic (Figs. 2, 3).
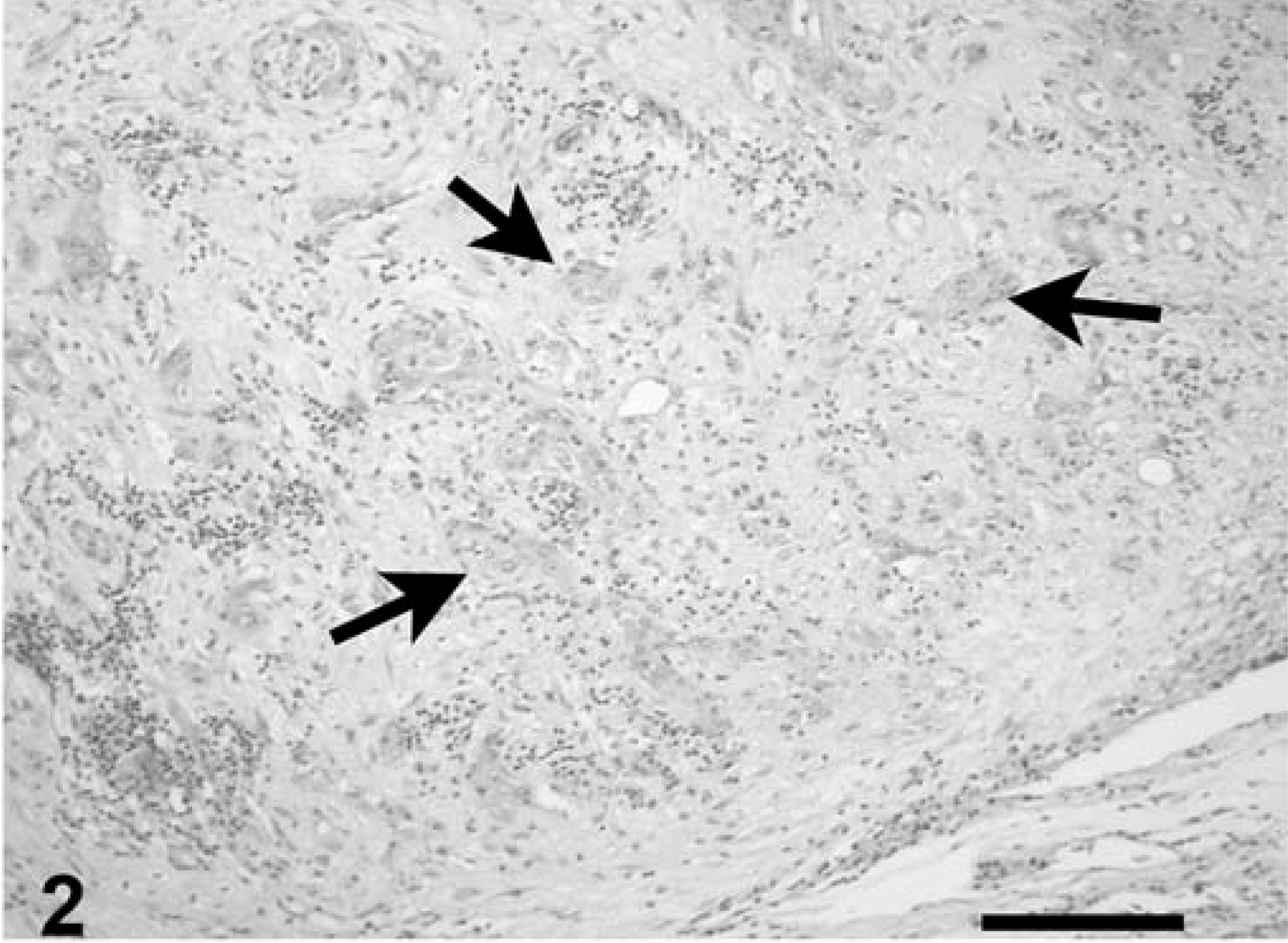
Pancreas, leopard tortoise, case No. 9. Notice complete loss of acinar tissue. Few ducts remain (arrows), widely separated by zones of fibrosis and inflammation. HE. Bar = 230 µm.
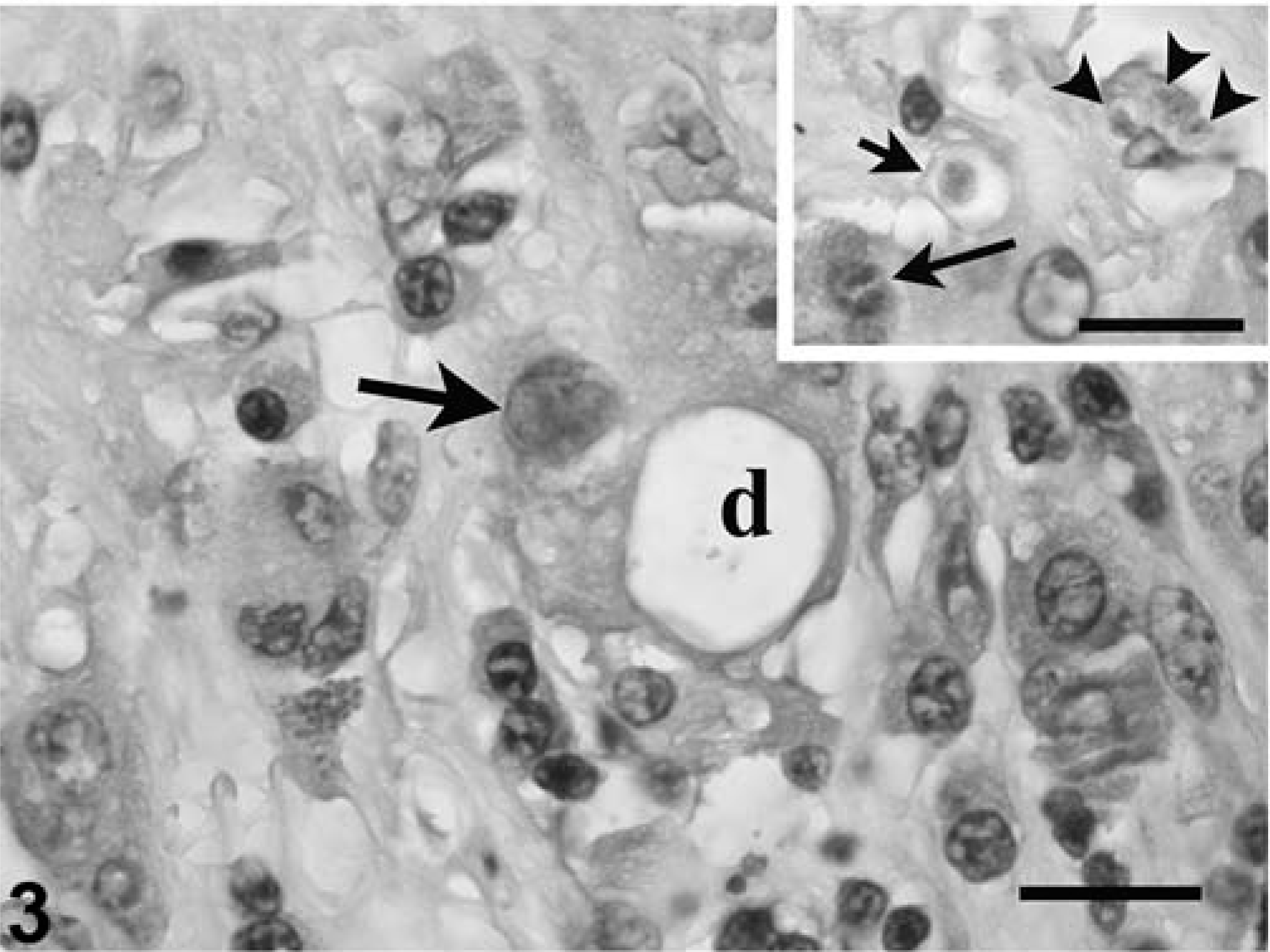
Pancreas, leopard tortoise, case No. 9. Notice two intranuclear gametes (arrow) in an epithelial cell lining a degenerative duct (d). Also notice infiltrate of lymphocytes, plasma cells, and heterophils in the adjacent fibrous stroma. HE. Bar = 28 µm.
Intranuclear coccidia were detected in the bile duct epithelium, hepatocytes, and melanomacrophages of the liver, and intracytoplasmic meronts or merozoites were seen occasionally in the melanomacrophages. Parasitism was accompanied by small random foci of hepatocellular necrosis and periportal to random infiltrates of lymphocytes, plasma cells, and heterophils. Rarely, affected ducts had epithelial cell crowding and hypertrophy (Fig. 4).
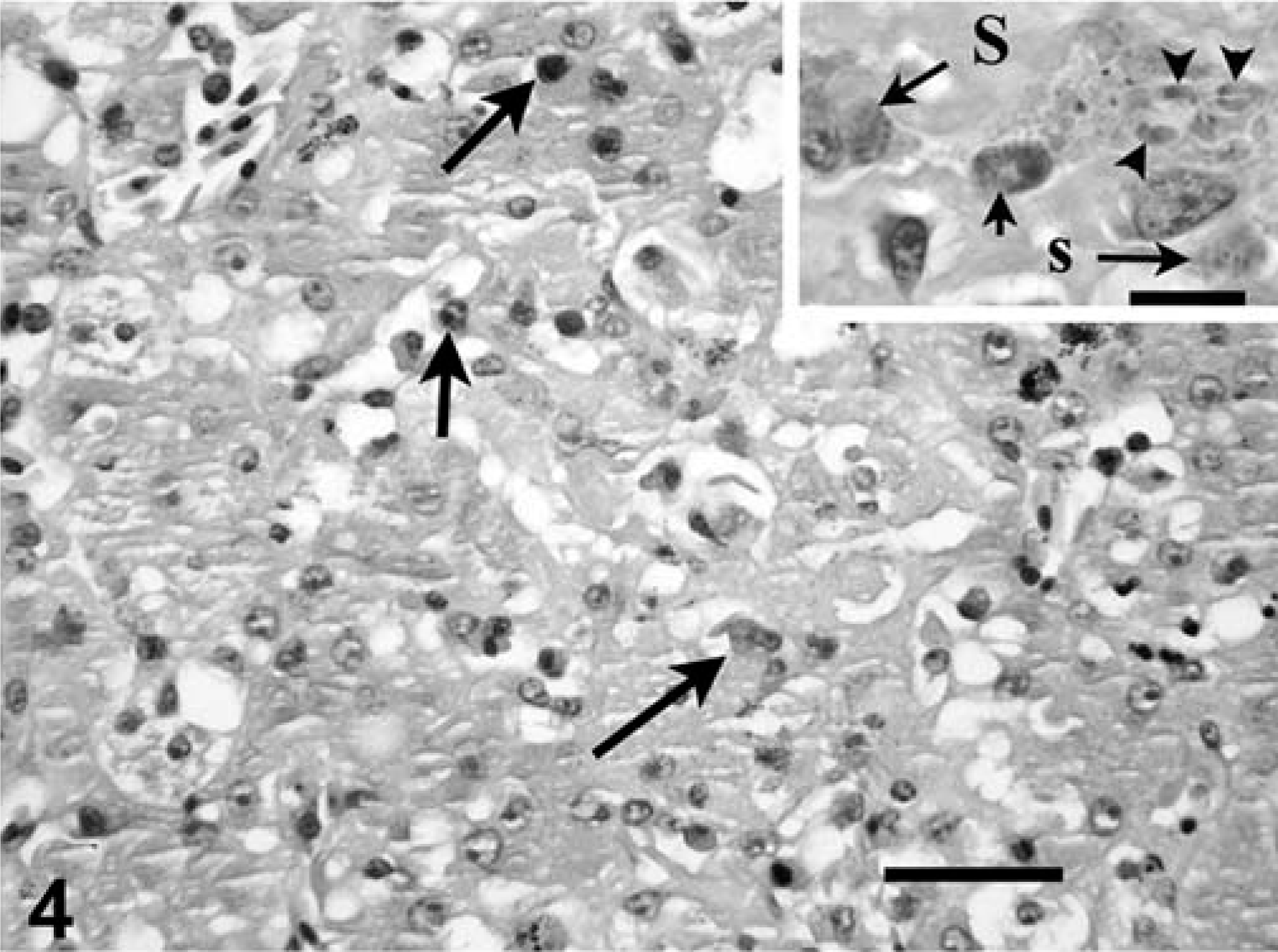
Liver, impressed tortoise, case No. 1. Notice central focus of hepatocellular necrosis admixed with scant infiltrate of inflammatory cells. Intranuclear and intracytoplasmic coccidia are in the field (arrows), but are difficult to discern at this magnification. HE. Bar = 60 µm.
Respiratory tract
Generally, mucosal lesions in the proximal portion of the respiratory tract were similar to those seen in the gut mucosa. Lungs had rare to frequent coccidia in the nuclei of pneumocytes, accompanied by pneumocyte hypertrophy and intestinal infiltrates of lymphocytes and fewer heterophils.
Urinary tract
Kidney tissue was available from all tortoises. Coccidia were detected in the nuclei of cortical and medullary tubules, and intracytoplasmic meronts and merozoites were detected in tubular epithelium. Rare extracellular nonsporulated oocysts were noted in the tubular lumina. Parasitism was associated with moderate-to-severe renal tubular necrosis and mild-to-moderate interstitial lymphocytic or lymphoplasmacytic inflammation (Figs. 5, 6). Glomeruli sometimes had mild mesangioproliferative change. Ureters had rare to moderate numbers of infected mucosal epithelial cells, with mild mucosal hyperplasia and submucosal lymphocyte infiltrates. Case No. 8 had mild mucosal hyperplasia, but coccidia were not seen. Urinary bladder was available from case No. 1, and it had moderate numbers of coccidia in the mucosal epithelium, with associated mild epithelial hyperplasia and moderate submucosal lymphocytic inflammation and exocytosis.
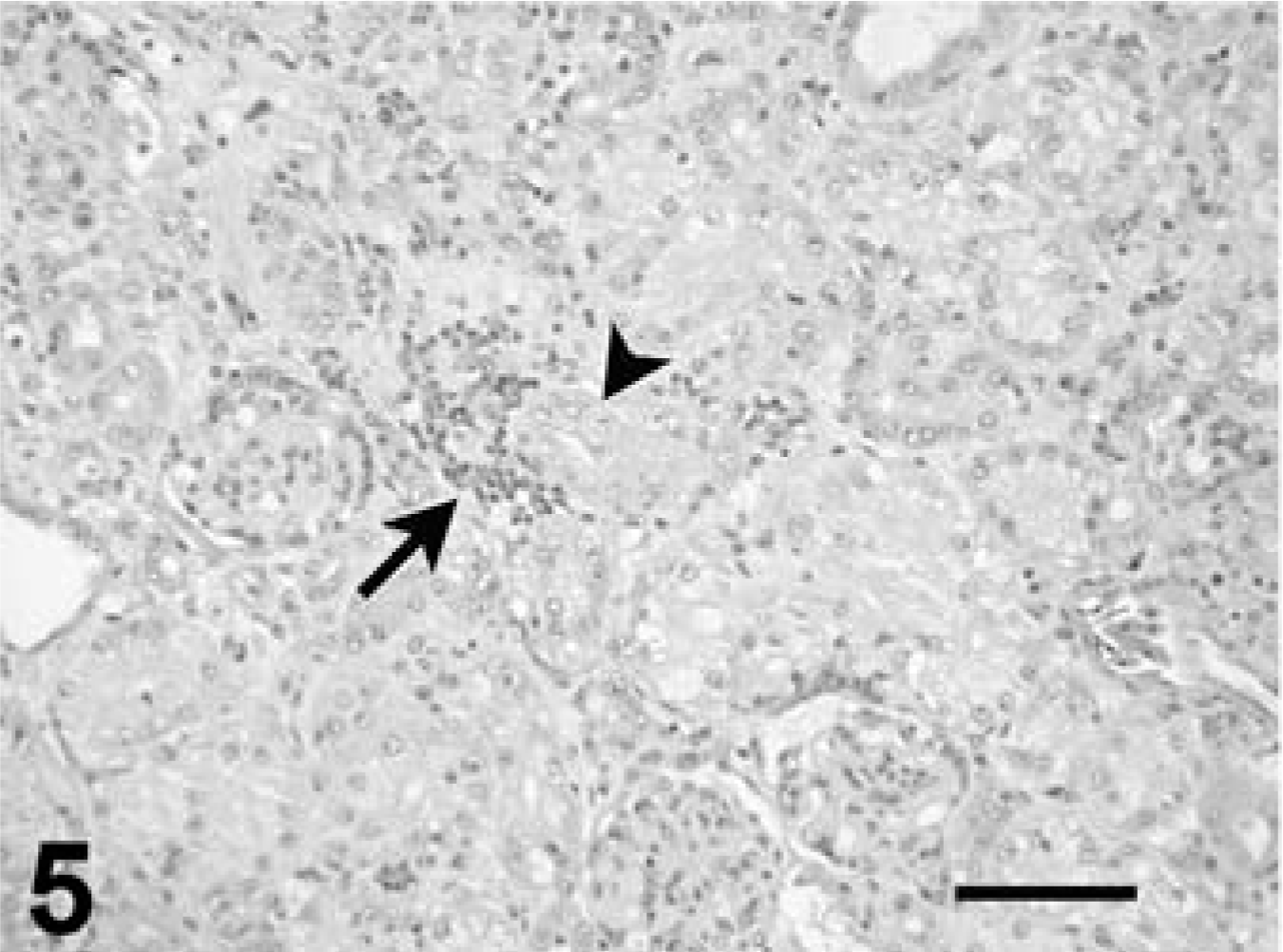
Kidney, leopard tortoise, case No. 9. Notice necrotic tubule (arrowhead) partially delineated by an infiltrate of mononuclear inflammatory cells (arrow). HE. Bar = 100 µm.
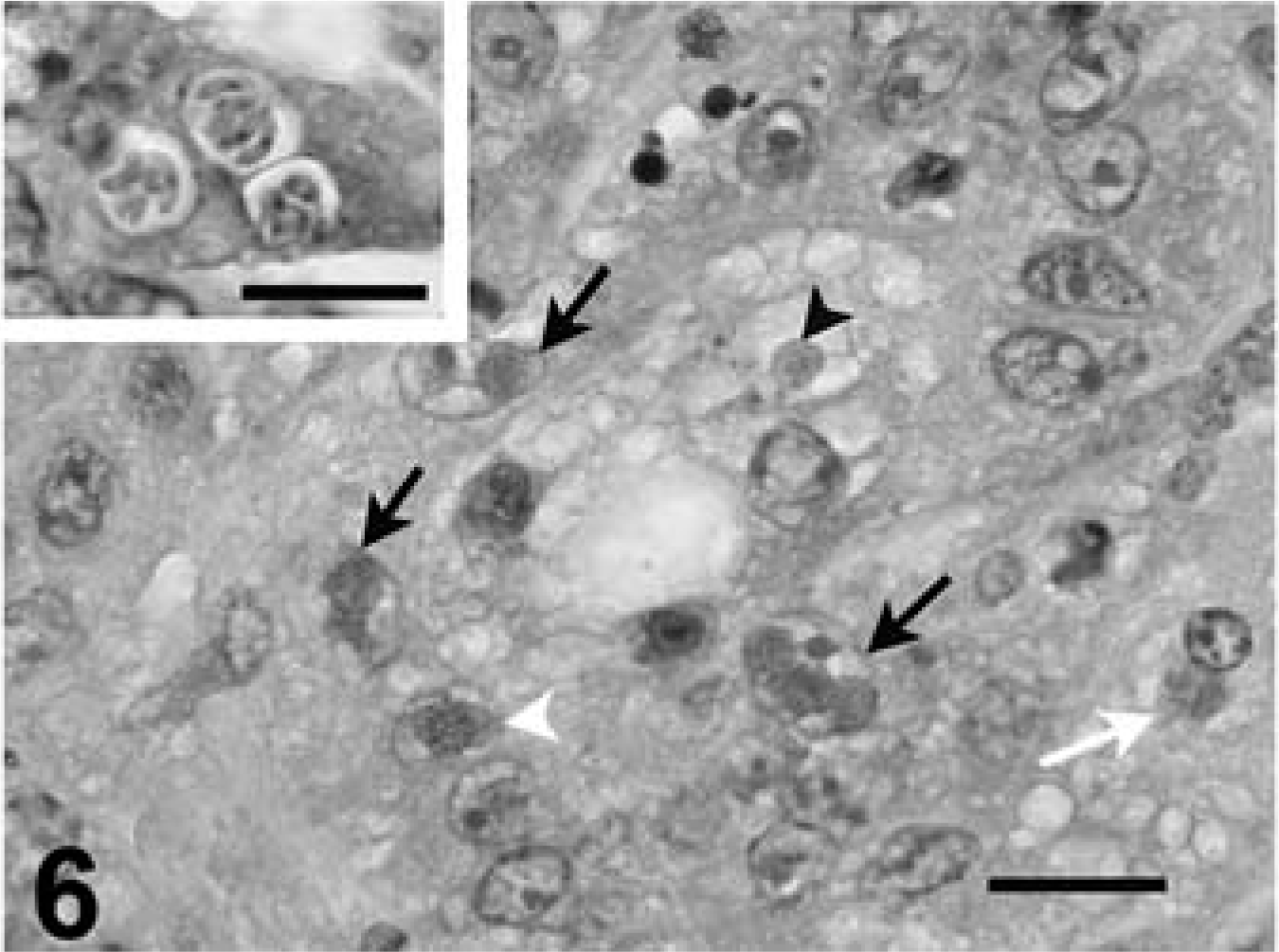
Kidney, leopard tortoise, case No. 9. Degenerative tubules lined by epithelial cells that contain one or more intranuclear gametes (black arrows), intracytoplasmic protozoa (white arrow), or intranuclear schizont (white arrowhead). HE. Bar = 20 µm. The lumen of one tubule contains an oocyst (arrowhead).
Reproductive tract
Ovary tissue had rare coccidia in follicular epithelium and unidentified interstitial cells accompanied by mild interstitial infiltrates of lymphocytes. The ovary from case No. 5 did not contain coccidia or lesions. Coccidia or lesions were not detected in oviduct. Testes had rare to a few infected interstitial cells with mild interstitial infiltrates of lymphocytes. Epididymis had a few infected epithelial cells and moderate submucosal infiltrates of lymphocytes.
Spleen
The spleen had rare to moderate numbers of coccidia in the nuclei of mononuclear cells resembling macrophages or lymphocytes in the red pulp. Foci of necrosis and fibrin deposition were detected adjacent to cells with intranuclear coccidians.
Endocrine system, CNS, and special senses
The thyroid had rare moderate numbers of infected follicular epithelial cells with associated mild-to-moderate interstitial lymphocytic inflammation. Tortoise No. 5 had mild lymphocytic inflammation in the interstitium between follicles, but no associated coccidia. Tortoise Case No. 8 had follicular goiter. Both tortoises, for which adrenal gland tissue was available, had mild focal interrenal (cortical) cell necrosis, and case No. 3 had rare infected interrenal epithelium. The brain had mild multifocal perivascular lymphocyte infiltrates in the meninges and neuropile at various levels, with a few microglial nodules, but coccidia or other organisms were not seen. The spinal cord had mild-to-moderate perivascular inflammation, and microglial nodules were detected in case Nos. 1, 3, and 4, but coccidia were not seen. Eustachian tube and inner/middle ear were available from case No. 2. Frequent coccidia were detected in the nuclei and cytoplasm of mucosal epithelium of the middle ear and Eustachian tube, accompanied by mild epithelial hyperplasia, exfoliation, and submucosal infiltrates of lymphocytes and plasma cells. Numerous nonsporulated oocysts were seen in the lumen of the middle ear (Fig. 7). Middle and inner ear tissue also was available from case No. 8, but lesions or coccidia were not detected.
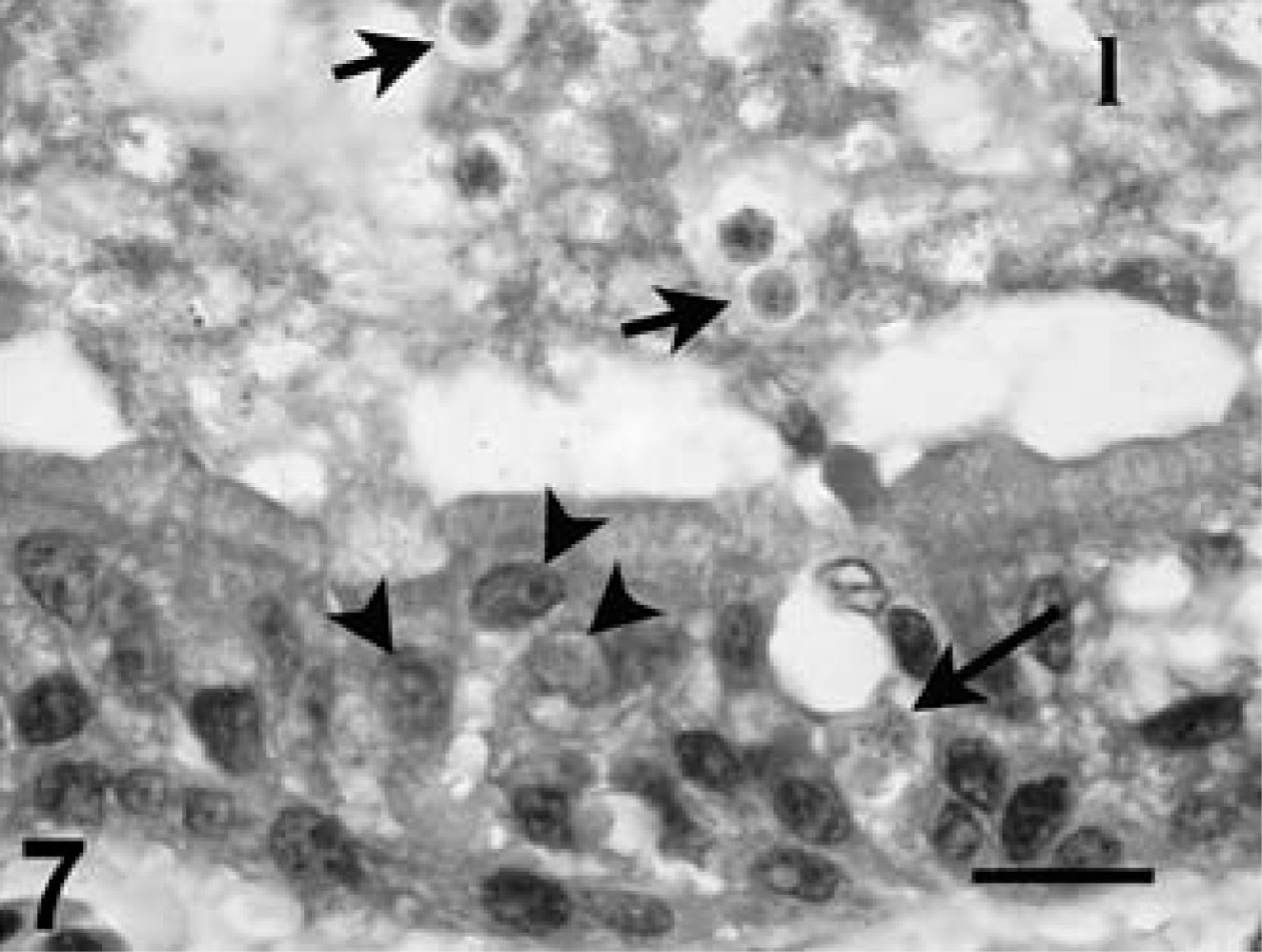
Middle ear, leopard tortoise, case No. 2. Notice intranuclear (arrowheads) and intracytoplasmic (large arrow) protozoa in the epithelium of the mucosa. Also notice the unsporulated oocysts in the lumen (small arrows), admixed with debris and bacteria. HE. Bar = 20 µm.
Skin
Rare epithelial cells with intranuclear coccidia were detected in the epidermis of the skin from the head of tortoise 3. Tortoises 3, 6, and 8 had small foci of ulceration and epidermal necrosis with mild, dermal, perivascular lymphocytic inflammation, and occasional exocytosis.
Musculoskeletal system
Small foci of lymphocytic inflammation were detected occasionally in skeletal muscle of case Nos. 3 and 6, and rare intranuclear coccidia were detected in mononuclear cells in areas of inflammation in tortoise No. 3.
Ultrastructure
Electron microscopy performed on case Nos. 1 and 2 revealed the microanatomy of a intranuclear coccidian (Figs. 8–11). Infected nuclei were often enlarged and contained one or more organisms. Trophozoites measured 2–5 µm in diameter (Fig. 8). Meronts measured up to 7 µm in diameter and contained numerous (>16) merozoites (Fig. 9). Merozoites were banana shaped, measured 4.0 × 1.5 µm, and budded from a residuum. Macrogametes (Fig. 10) and microgametes (Fig. 11) were numerous, and measured up to 6 µm in diameter. Oocysts were unsporulated, and measured 8 µm in greatest diameter.
Kidney, impressed tortoise, case No. 1. Electron micrograph of intranuclear trophozoites in a tubular epithelial cell. Uranyl acetate and lead citrate. Bar = 3 µm.
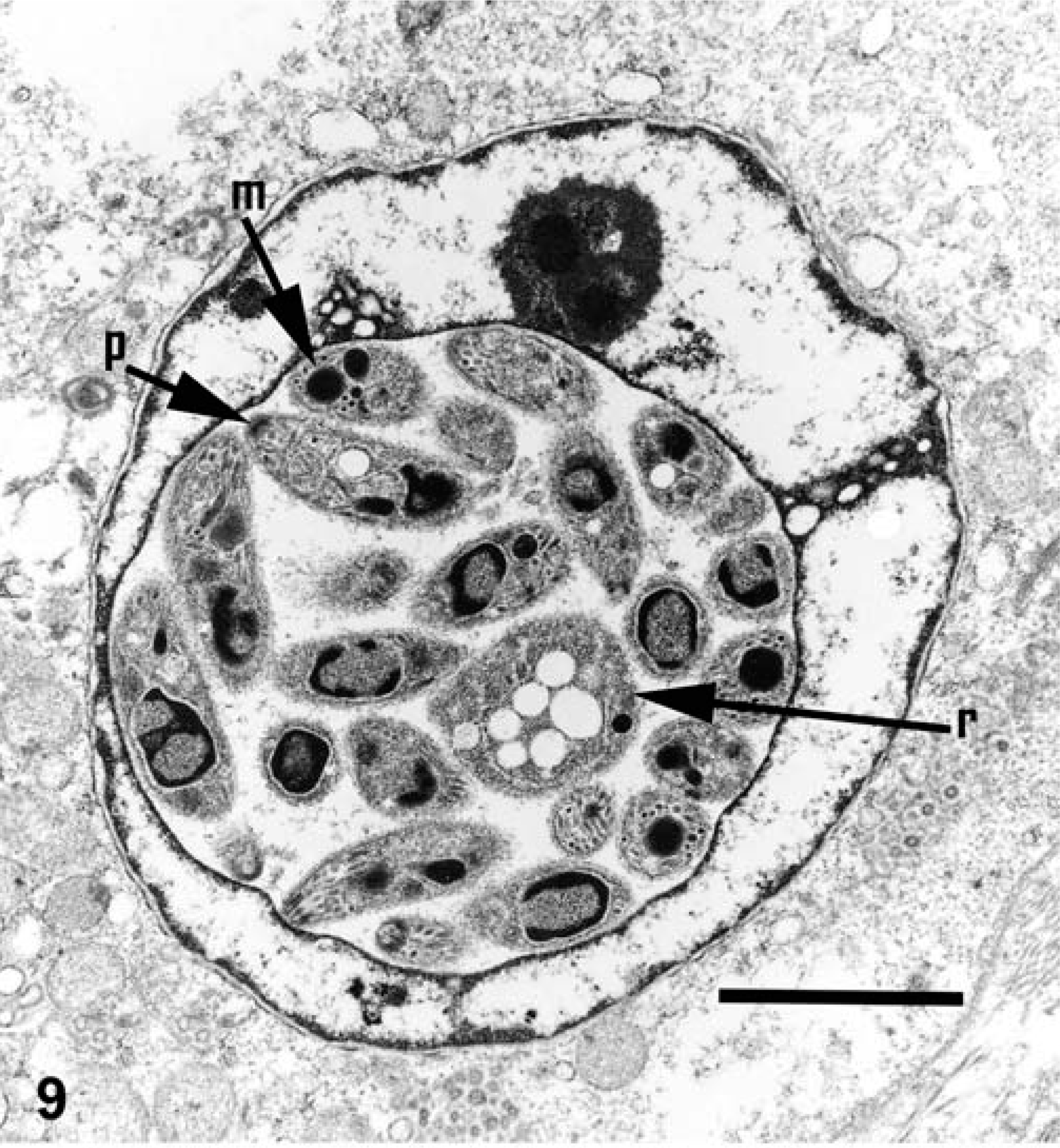
Kidney, impressed tortoise, case No. 1. Electron micrograph of a meront in nucleus of renal tubular epithelial cell. Notice that merozoites have a polar ring (p), micronemes (m), and a large nucleus (n). Also notice the residual body (r) from which the merozoites budded. Uranyl acetate and lead citrate. Bar = 4 µm.
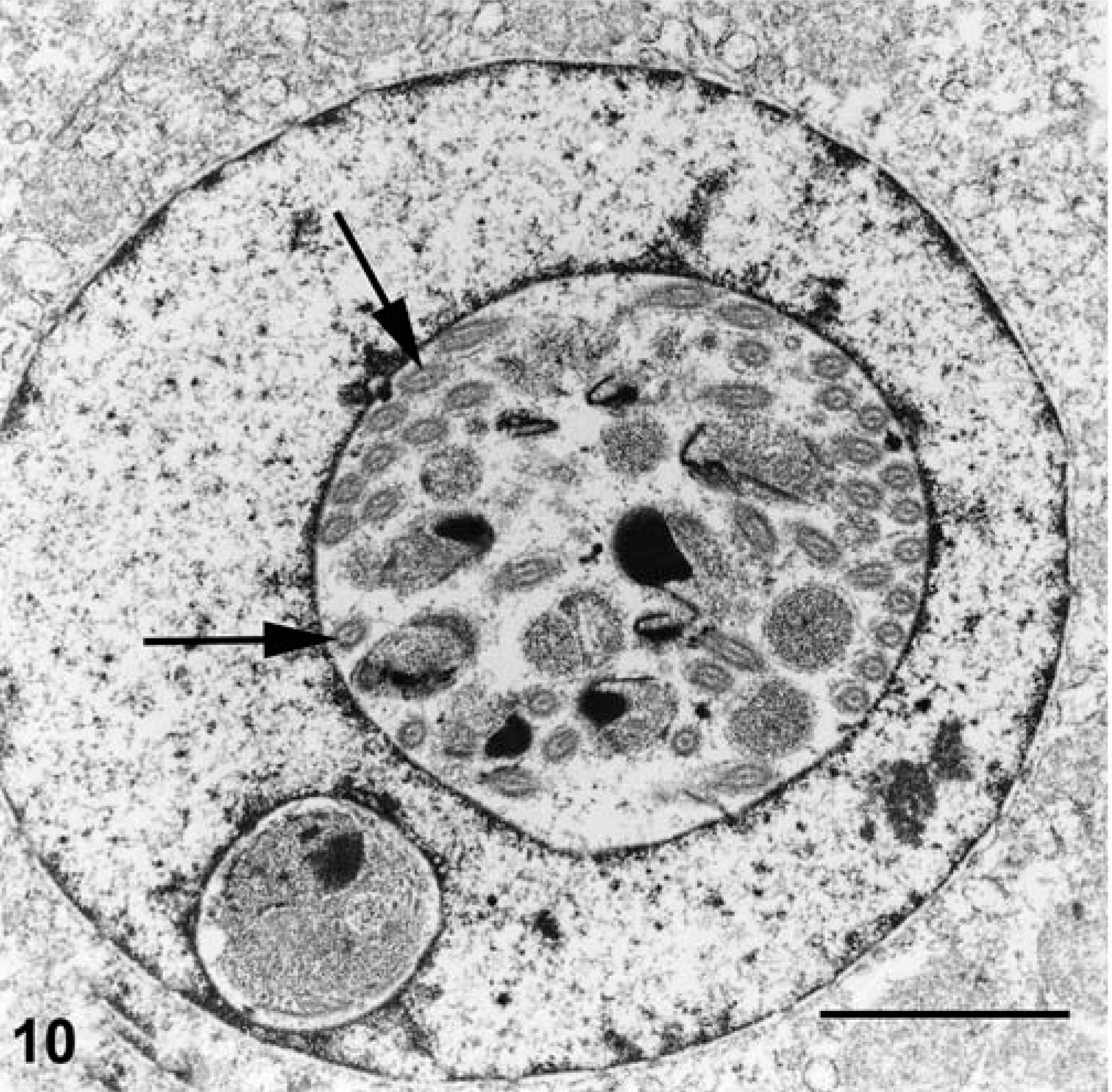
Kidney, impressed tortoise, case No. 1. Electron micrograph of the nucleus of a tubular epithelial cell containing a microgametocyte. Notice the flagella (arrows) of the numerous microgametes. Uranyl acetate and lead citrate, bar = 2.4 µm.
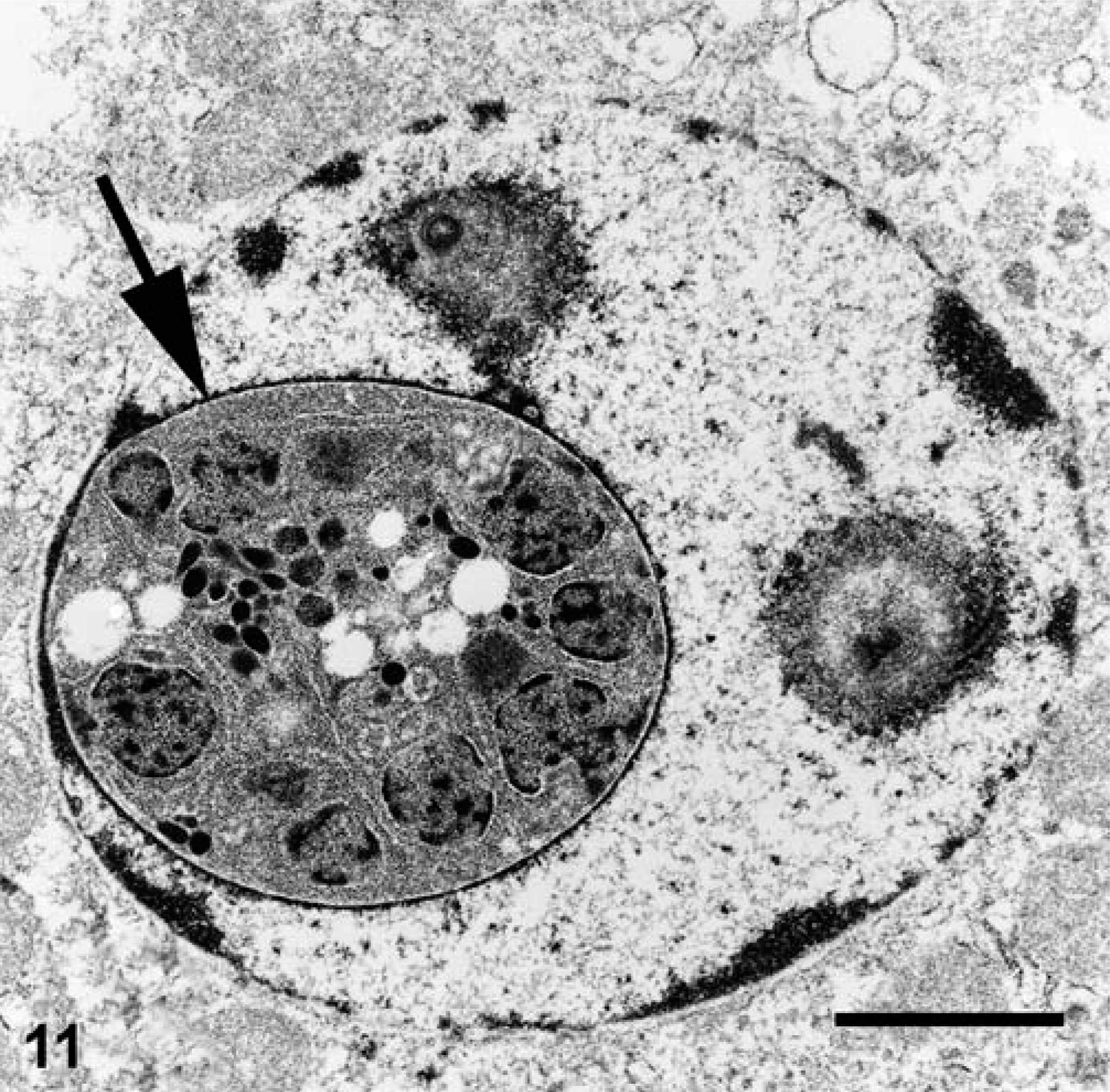
Kidney, impressed tortoise, case No. 1. Electron micrograph of the nucleus of a tubular epithelial cell containing a macrogametocyte (arrow). Uranyl acetate and lead citrate. Bar = 2.1 µm.
PCR and sequence analysis
The PCR products from case Nos. 7 and 9 were amplified and sequenced. The sequence from both tortoises was identical. This sequence was identical to that from several other cases of intranuclear coccidiosis in tortoises (Wellehan and Johnson, unpublished data). The TBLASTN results for the sequence indicated the highest score with the Hyaloklossia lieberkuehni 18S small subunit ribosomal RNA gene (GenBank accession No. AF298623). 12 The maximum likelihood phylogenetic tree with bootstrap values is shown (Fig. 12). Sequence data was submitted to GenBank; the accession number is AY728896.
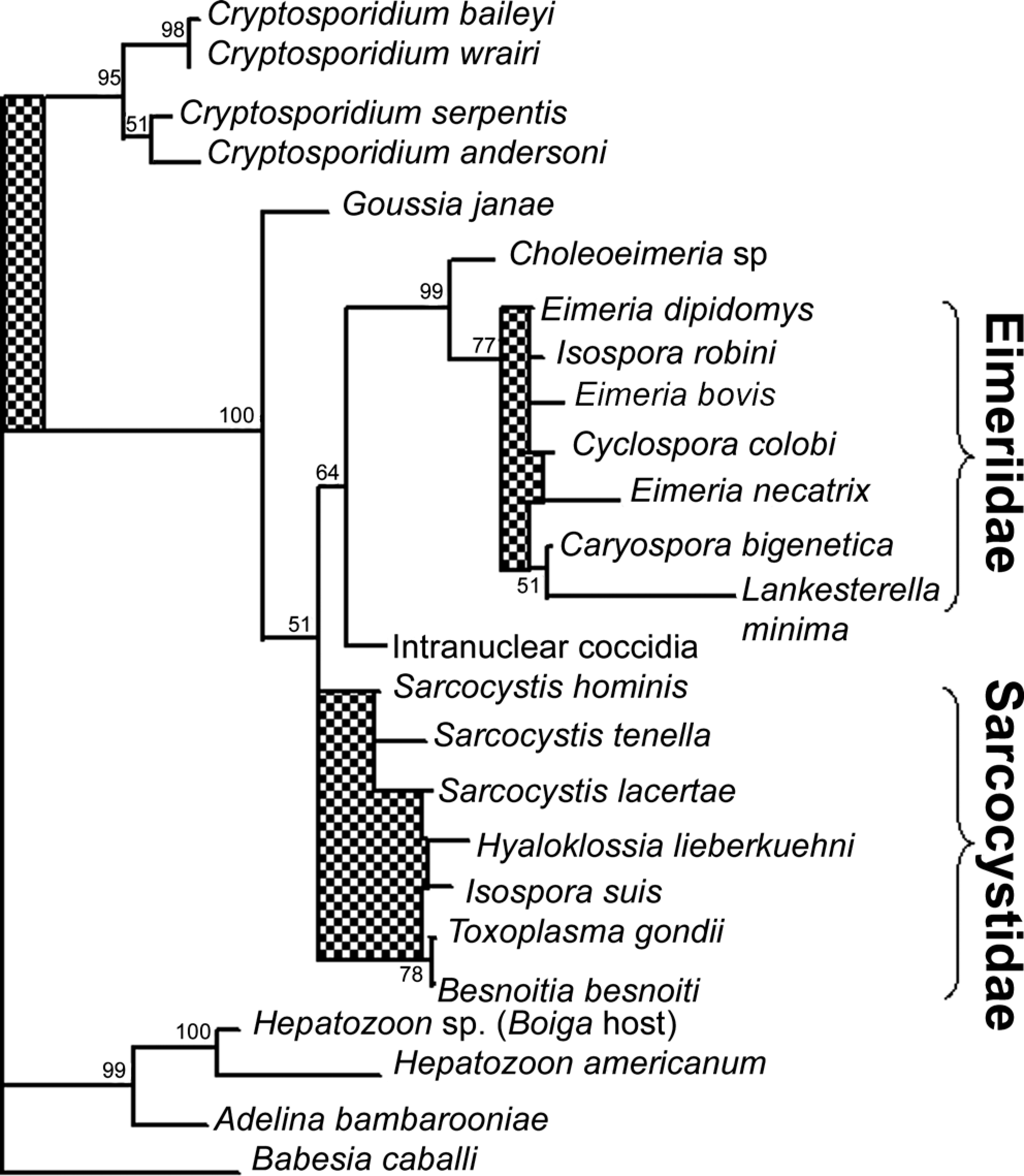
Maximum likelihood phylogenetic tree of partial coccidial 18S rRNA nucleotide sequences. Babesia caballi was used as the outgroup (GenBank accession No. AY534883). The validity of the tree topology obtained was tested by using bootstrap analysis with 100 resamplings. Branchings with bootstrap values <50 are not shown, and areas where these branchings occurred are checkered. Intranuclear coccidia are in bold. Other sequences were retrieved from GenBank: Adelina bambarooniae (AF494059), Besnoitia besnoiti (AF109678), Caryospora bigenetica (AF060975), Choleoeimeria sp. (AY043207), Cryptosporidium andersoni (AB089285), Cryptosporidium baileyi (AJ276096), Cryptosporidium serpentis (AF151376), Cryptosporidium wrairi (AF115378), Cyclospora colobi (AF111186), Eimeria bovis (U77084), Eimeria dipidomys (AF339490), Eimeria necatrix (U67119), Goussia janae (AY043206), Hepatozoon americanum (AF176836), Hepatozoon sp. (Boiga host), Hyaloklossia lieberkuehni (AF298623), Isospora robini (AF080612), Isospora suis (U97523), Lankesterella minima (AF080611), Sarcocystis hominis (AF176945), Sarcocystis lacertae (AY015113), Sarcocystis tenella (L24383), and Toxoplasma gondii (L37415).
Discussion
Lesions associated with intranuclear coccidiosis were considered the principal cause of death in case Nos. 1–4, 6, and 9. The death of case Nos. 5 and 7 was attributable to intranuclear coccidiosis and concurrent bacterial infection and amoebiasis originating in the intestinal tract of these tortoises. Intranuclear coccidiosis may have been an incidental finding in case No. 8.
Intranuclear coccidiosis in these tortoises was a systemic event, involving alimentary, respiratory, urogenital, integumentary, lymphoid, and endocrine systems. Organisms were present in pancreas, lung, and small intestine of all tortoises for which these tissues were available. Lesion severity in various affected tissues generally was proportional to density of infected cells in the tissue. Lesions were most severe in pancreas, colon, kidney, and lung. Lesions were judged to be subacute to chronic in most sites, especially the alimentary tract. Lesions in the pancreas were consistently severe, and loss of functional pancreatic tissue may have contributed substantially to the wasting detected in case Nos. 5, 8, and 9. Chronic inflammation and mucosal alterations associated with intestinal coccidia may have contributed to malabsorption and dysbiosis, and in case Nos. 5 and 7, to bacterial and amebic enteritis and septicemia. Lesions in the kidney were usually severe, and likely were associated with appreciable renal dysfunction, as reported in radiated tortoises. 9 On the basis of morphologic alterations in the renal parenchyma, the renal lesions were less chronic than those in the gastrointestinal tract and pancreas. The tubular necrosis, absence of tubular regeneration, and mild-to-moderate inflammatory response in this organ indicated a shorter course of infection, and suggest that infected tortoises may not survive long after the kidney becomes involved. The inflammatory and proliferative changes in the respiratory tract and lung also seemed more acute than those in the gastrointestinal tract and pancreas. Lung lesions were considered sufficiently severe to contribute substantially to the death of affected tortoises.
Nucleic acid sequences were obtained from only two of nine cases. Possible reasons for failure to obtain sequences from the other seven cases include degradation of DNA over time, cross-linking during formalin fixation of tissue, handling loss in the laboratory, PCR failure, or failure of the primers to bind. As the primers are designed toward conserved areas and have successfully amplified those of other coccidian species, we believe that the last possibility is unlikely, and that the first two possibilities are the most probable explanation.
Sequence data obtained from these isolates provides information for the design of nucleic-acid based diagnostics. Previous phylogenetic analysis of coccidial 18S rRNA found two main clusters, which correspond roughly to the families Sarcocystidae and Eimeriinae, 12, 13 and our results are in agreement. Our results also indicated that the monophyly of the genus Isospora is not supported, and that Goussia is not found within the families Sarcocystidae and Eimeriinae. This is in agreement with other nucleic acid-based coccidial phylogenies, 12, 13 providing support for the usefulness of this subregion of 18S rRNA for limited phylogenetic analysis. Phylogenetic analysis of the intranuclear coccidial sequence does not provide support for inclusion in either the Sarcocystidae or the Eimeriinae, and this organism may be paraphyletic to these families. Although TBLASTN results indicated the highest score with Hyaloklossia lieberkuehni, more rigorous examination in the context of other sequences confuses this relationship. Although there is strong support for clustering of this organism with other coccidia, additional sequences of 18S rRNA and other genes from this organism and others are needed to more firmly resolve the phylogeny of this organism.
To our knowledge, intranuclear coccidia have been reported from tortoises only once previously. 9 The organisms reported here are morphologically distinct from those reported in radiated tortoises. In our tortoises, the meronts were 7 µm in greatest diameter, whereas those in the previously described cases were 15 µm in diameter. Oocysts in our tortoises measured 8 µm in greatest diameter, whereas those in the previously reported tortoises were 12 µm in diameter. Additionally, our meronts contained over 16 merozoites, whereas those of the previous report contained up to 16 merozoites. The route of oocyst shedding is unknown, but is presumed to be the gastrointestinal tract on the basis of presence of gametes and oocysts in the mucosal epithelium; however, the presence of coccidia in epithelium of the urinary tract, respiratory tract, conjunctiva, male reproductive tract, and skin suggest that transmission may also occur after contact with secretions or excretions from these sites. Oocyst isolation and sporulation studies would provide additional useful information on the taxonomic status of the organism. Experimental infectivity studies would be needed to elucidate the complete pathogenesis of the infection.
