Abstract
Sixty-six cases of indolent canine lymphoid proliferation were reviewed. Age ranged from 1.5 to 16 years (median 9.0 years). Dogs of 26 breeds, plus 13 of mixed breeding or unknown lineage, were represented. B-Cell lymphomas (CD79a+) predominated. Marginal zone lymphoma (MZL), the largest group, involved lymph node (33 cases) and spleen (13 cases), with both tissues involved in five of these cases. Follicular lymphoma (FL) involved lymph nodes (five cases), and mantle cell lymphoma (MCL) occurred as solitary splenic masses (three cases). Nodal CD3+ T-zone lymphomas (TZL) (10 cases), were included since they resembled late-stage MZL at the architectural level. Two cases of marginal zone hyperplasia (MZH) were included to aid in differentiation of early MZL. Clonality status was determined in 54 cases by analysis of immunoglobulin heavy chain (IGH) and T-cell antigen receptor gamma (TCRG) gene rearrangement. Clonal rearrangement of IGH was detected in 28 of 35 MZL cases (80%), four of four FL cases (100%) and three of three MCL cases (100%). Concurrent cross lineage rearrangement of TCRG was detected in six MZL and two FL cases. Clonal rearrangement of TCRG was documented in five of eight TZL cases (63%). Limited survival data obtained for 18 dogs indicated that the B-cell lymphomas (MZL, MCL, and FL) and the T-cell lymphoma (TZL) were associated with indolent behavior and long survival. Although to the authors' knowledge, the true incidence of canine indolent lymphomas is unknown, the tumors are not rare and may have been underrecognized. Recognition of their architectural features, routine application of immunophenotyping, and molecular clonality assessment should alleviate this.
In animals, indolent lymphomas, similar to the low-grade, B-cell lymphomas of humans, are seldom recognized as specific diseases. 8, 23, 26, 37, 42, 43 In animals, a generic diagnosis of lymphoma is most often made, and is based on cell morphology, often without phenotype, and assessment of architectural integrity. 8, 26, 37, 42, 43 The linkage of specific lymphoma type and type-specific therapy, as seen in human clinical trials, is therefore missing in most veterinary studies. Trials of therapy of lymphoma need to be conducted on biologically similar tumors to document differences in therapeutic strategies.
Indolent lymphomas arise on a background of follicular lymphoid hyperplasia, and include follicular, mantle cell, and marginal zone lymphomas of B-cell type, and T-zone lymphoma of T-cell type. 4, 14, 18, 30, 45 The indolent lymphomas of B- and T-cell types share a low mitotic rate and slow rate of clinical progression. Indolent lymphomas in advanced stages retain the same cytomorphologic features, but lose the follicle-related architecture, without which their indolent nature may not be apparent on histologic examination. These lymphomas usually arise after a long period of benign hyperplasia and tend to contain capsular and medullary sclerosis.
The aim of the study reported here was to alert oncologists and pathologists to the less commonly observed indolent types of lymphoma, which require recognition and appropriate management. The treatment of hematopoietic neoplasia in dogs and cats requires a specific, rather than general diagnosis that is based on architecture, cell morphology, and phenotype. 17, 21, 22, 31, 34 Chemotherapeutic trials require determination of subtypes of lymphoma as part of the protocol. 5, 11, 34, 36, 54 The remarkable conservation of genetic information in mammals suggests that the same type of lesion would respond to a similar type of therapy in humans and in dogs. 37, 39, 48, 51, 53
Materials and Methods
Study population
From 1992 to the present, 66 cases of indolent canine lymphoid proliferation were received from state and university diagnostic services. The study group included mantle cell (MCL) and marginal zone (MZL) lymphomas as were defined in humans over this period. 4, 29, 36, 41 Phenotype was determined for 51 cases by immunohistochemical reaction to CD3 and CD79a antibodies for T- and B-cell lineages, respectively (DAKO, Carpinteria CA). Clonality was determined for 52 cases where DNA was available, including nine cases where phenotype was not determined (Table 1). Diagnoses for the 66 cases with all information considered were: five cases of follicular lymphoma (FL) (Table 1, cases 1–5); 46 cases of MZL (Table 1, cases 6–51); three cases of MCL (Table 1, cases 52–54); and 10 cases of T-zone lymphoma (TZL; Table 1, cases 55–64). The 46 cases of MZL included primary splenic MZL in 13 dogs and nodal MZL in 33 dogs. Five primary splenic cases also had lymph node involvement (Table 1, cases 11, 14, 24, 38, 44). The three cases of MCL were located in the spleen. T-Zone lymphomas were included since, in the absence of phenotyping, they closely resembled late-stage MZL at the architectural level. Two cases of marginal zone hyperplasia (MZH) were included to aid in differentiation of early MZL (Table 1, cases 65, 66).
Canine indolent lymphoma: summary of diagnosis, demographics, immunophenotype, molecular clonality, therapy, and survival data.∗
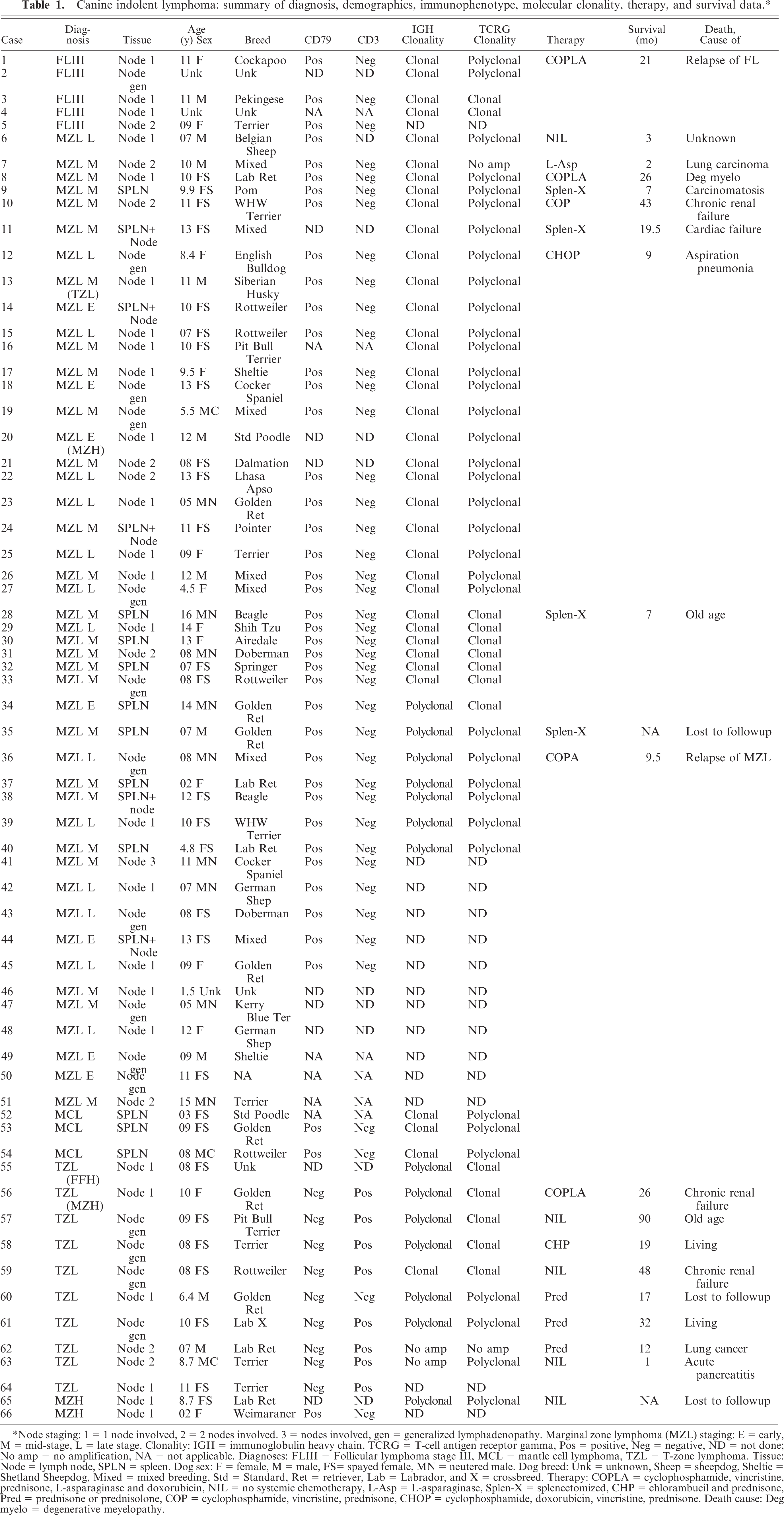
Node staging: 1 = 1 node involved, 2 = 2 nodes involved. 3 = nodes involved, gen = generalized lymphadenopathy. Marginal zone lymphoma (MZL) staging: E = early, M = mid-stage, L = late stage. Clonality: IGH = immunoglobulin heavy chain, TCRG = T-cell antigen receptor gamma, Pos = positive, Neg = negative, ND = not done; No amp = no amplification, NA = not applicable. Diagnoses: FLIII = Follicular lymphoma stage III, MCL = mantle cell lymphoma, TZL = T-zone lymphoma. Tissue: Node = lymph node, SPLN = spleen. Dog sex: F = female, M = male, FS= spayed female, MN = neutered male. Dog breed: Unk = unknown, Sheep = sheepdog, Sheltie = Shetland Sheepdog, Mixed = mixed breeding, Std = Standard, Ret = retriever, Lab = Labrador, and X = crossbreed. Therapy: COPLA = cyclophosphamide, vincristine, prednisone, L-asparaginase and doxorubicin, NIL = no systemic chemotherapy, L-Asp = L-asparaginase, Splen-X = splenectomized, CHP = chlorambucil and prednisone, Pred = prednisone or prednisolone, COP = cyclophosphamide, vincristine, prednisone, CHOP = cyclophosphamide, doxorubicin, vincristine, prednisone. Death cause: Deg myelo = degenerative meyelopathy.
Diagnostic criteria
Follicular-derived lymphomas frequently develop in the presence of germinal centers that may be involuting to “fading” follicles. 22, 28, 38, 44 Involution of germinal centers may result in collapse of the mantle cells into the follicular dendritic cell center, with the formation of clusters of small dense cells in place of the original germinal centers (see late MZL). This is called fading follicular hyperplasia (FFH). Generalized FFH was present in virtually all cases of MZL. The presence of defined mantle cell cuffs is characteristic of lymphoid hyperplasia (Figs. 1–3). In follicular lymphoma (FL), the follicles tend to be large, but uniform in size; lack a mantle cell cuff (“bare”); and are tightly faceted throughout the entire area of the node in an advanced lesion (Fig. 2). 14, 44 Cytologically, the proliferative follicles of FL lack the antigen-related polarity that serves to distinguish FL from lymphoid follicular hyperplasia (Figs. 1, 3a). In FL, the high endothelial venules of the paracortex are compressed between opposing follicles and are not found within them. The FL cases were graded using human criteria for proportions of centrocytes and centroblasts. 44
Lymph node; dog, follicular polarity. A peripheral cortical germinal center with an eccentric cuff of small mantle cells characteristically thicker over the lighter or superficial pole area. The superficial pole (upper) contains a mixture of lymphocytes with many small cells of centrocytic type that have abundant pale-staining cytoplasm. The dark or deep pole area (lower) contains large dividing cells of centroblastic type that have a narrow cuff of highly basophilic cytoplasm. HE. Bar = 10 µm.
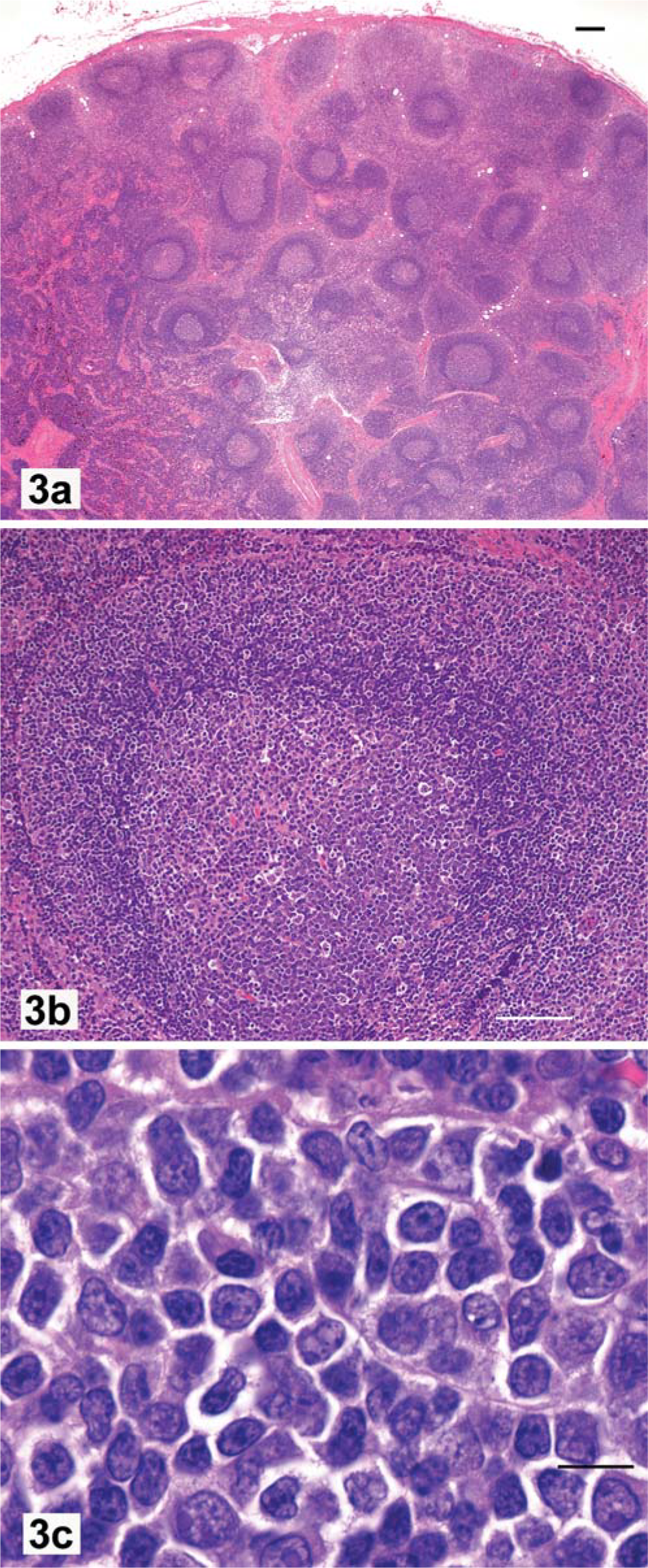
Lymph node; dog, follicular lymphoma. Solid nodules of closely packed lymphocytes that lack an antigen-dependent polarity and with incomplete mantle cell cuffs occupy the entire node with atrophy of the paracortical and medullary areas. HE. Bar = 300 µm.
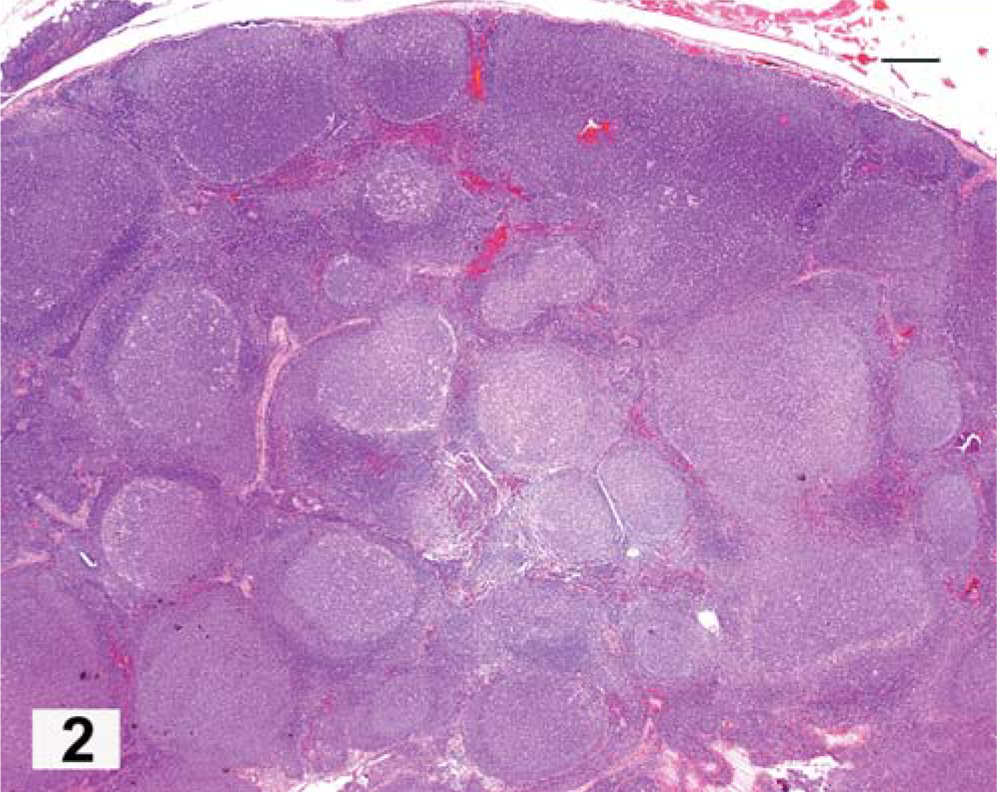
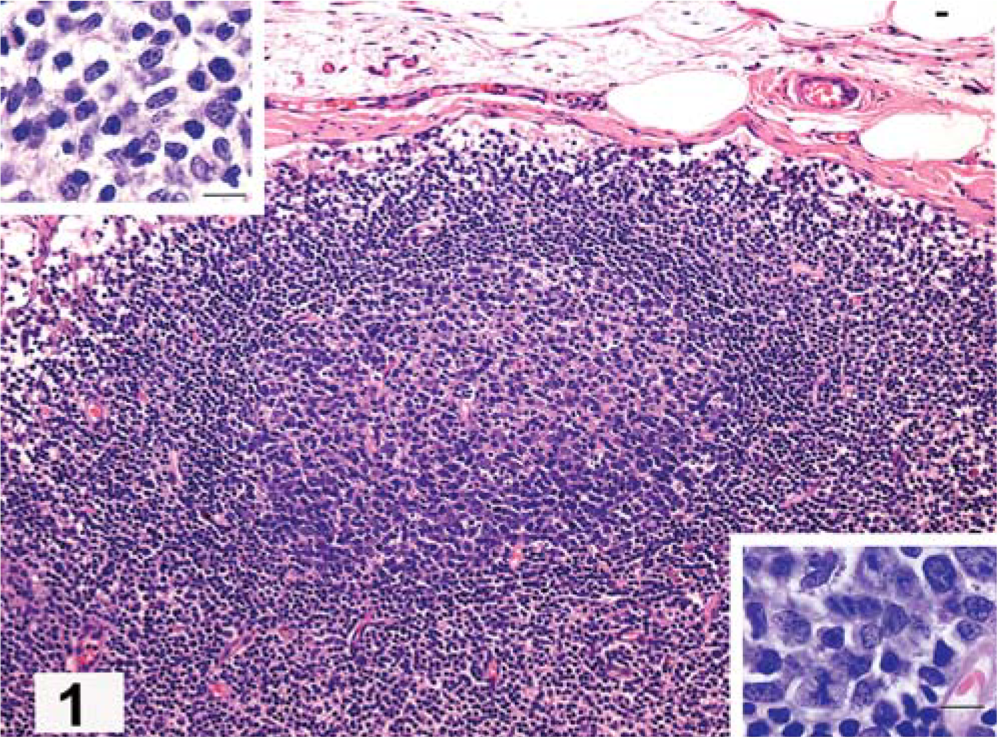
Lymph node; dog, marginal zone hyperplasia.
Marginal zone hyperplasia (Fig. 3) and MZL (Figs. 4, 5) occupy a region within the node or spleen that lies outside of the mantle cell cuff and is clearly related to germinal centers that usually are in regression. 18, 41 Prolonged, vibrant follicular hyperplasia may be accompanied by some degree of MZH that stains more lightly than the mantle cell cuff (Fig. 3a). Marginal zone hyperplasia consists of cuffs, often discontinuous, of larger cells with mild vesiculation of nuclei and single prominent nucleoli (Fig. 3b). The MZH cells are intermixed with smaller mantle cells and large “centroblasts” with multiple small nucleoli that lie near the nuclear membrane (Fig. 3c).
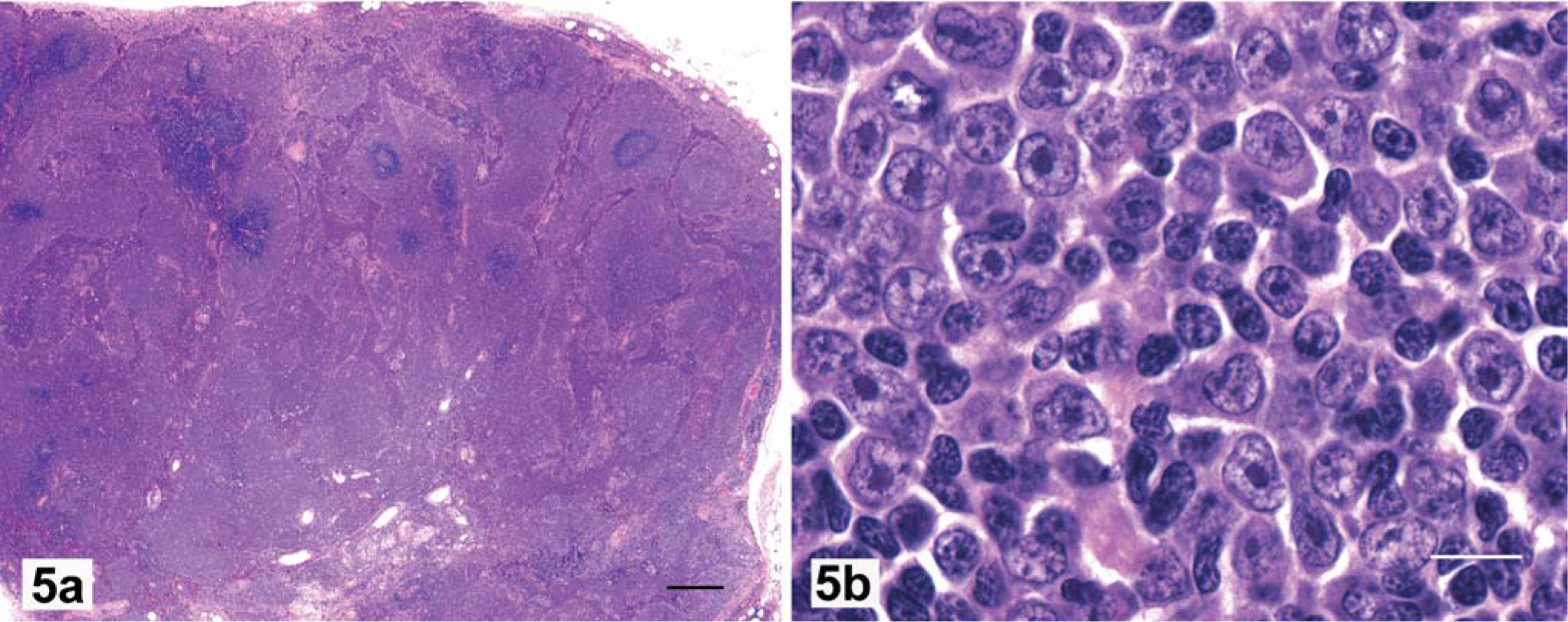
Marginal zone lymphoma is characterized by coalescing areas of perifollicular proliferation usually accompanied by paracortical atrophy (Fig. 4a). MZL can be graded for “early, mid, or late” stage of development, on the basis of area of tissue involved, degree of coalescence of focal areas of neoplastic proliferation, and number of cells in mitosis. 18, 19, 29, 32, 38 In early MZL, the cytologic homogeneity suggests neoplasia before there is coalescence of the marginal zone cuffs. Neoplastic transformation of marginal zone lymphocytes results in an increase in nucleolar size, but not larger nuclei. The chromatin pattern becomes finer, with chromatin more uniformly peripheralized and lining the nuclear membrane (Fig. 4b). The mitotic rate remains low, with none found at 400× magnification in most fields: a key factor in the diagnosis of MZL. Immunophenotypically, marginal zone lymphocytes strongly and uniformly express CD79a with prominent membrane marking. In advanced MZL, the perifollicular cuffs coalesce (Fig. 5a), and cytologically contain a homogeneous cell population (Fig. 5b). Late-stage MZL is characterized by paracortical atrophy with marked and irregular sinus dilatation. These large cavities are filled with red blood cells and neoplastic lymphocytes (Fig. 5a). As a consequence, cases of late MZL have poorly cohesive tissues that may fragment on sectioning.
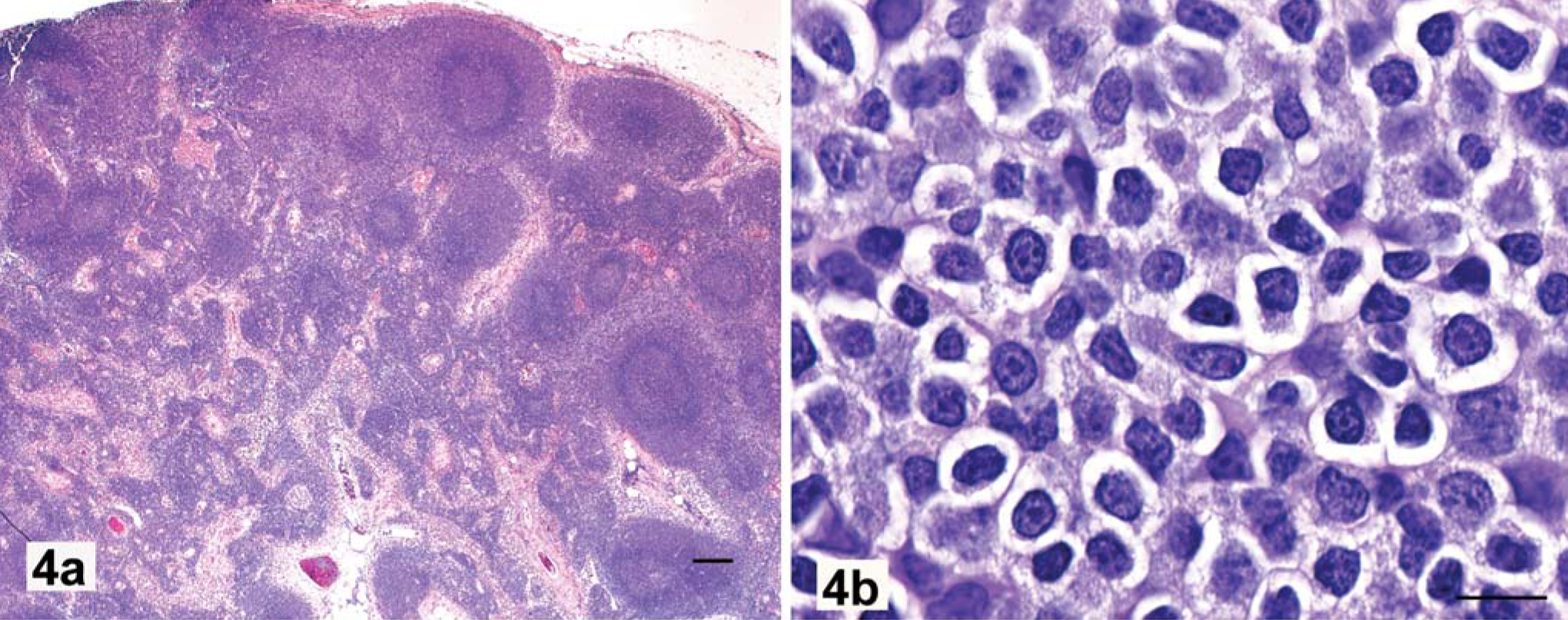
Lymph node; dog, early marginal zone lymphoma, clonal immunoglobulin heavy chain (IGH) gene rearrangement.
Fading follicular hyperplasia is being replaced by marginal zone proliferation that is becoming confluent and invading mantle cell cuffs. Notice multifocal dilation of cortical sinuses that are filled with marginal zone lymphocytes. The node capsule is invaded and irregularly thinned, but the peripheral sinus is largely intact. HE. Bar = 500 µm.
Mantle cell lymphoma (MCL) consists of round masses of deeply stained cells, with regularity of spacing that suggests their anatomic relationship to small arterioles in the spleen and germinal centers in the lymph node outer cortex. 10, 16 Some foci can be seen to surround hyalinized and hypocellular germinal centers, which indicates their relationship to fading follicles (Figs. 6a–c). Cytologically, neoplastic mantle cells have nuclei slightly larger and with more internal nuclear detail than their benign counterparts. They lack nucleoli, and mitoses are rarely found (Fig. 6d). The diagnosis of MCL is more difficult than that of MZL, because the intermediate cells of MCL overrun the fading germinal centers and the benign mantle cells.
Spleen; dog, mantle cell lymphoma.
T-Zone lymphoma (TZL) is characterized by cellular proliferation between fading follicles, rather than surrounding them, as in MZL (Fig. 7a). 22, 36, 45, 49 Cytologically, TZL is composed of homogeneous population of CD3+ small lymphocytes that have sharp, shallow nuclear indentations, inapparent nucleoli, and a moderate volume of pale cytoplasm (Fig. 7b). In contrast, T-zone hyperplasia consists of a heterogeneous population of small lymphocytes, macrophages, and dendritic cells, which imparts a moth-eaten appearance at low magnification (not shown).
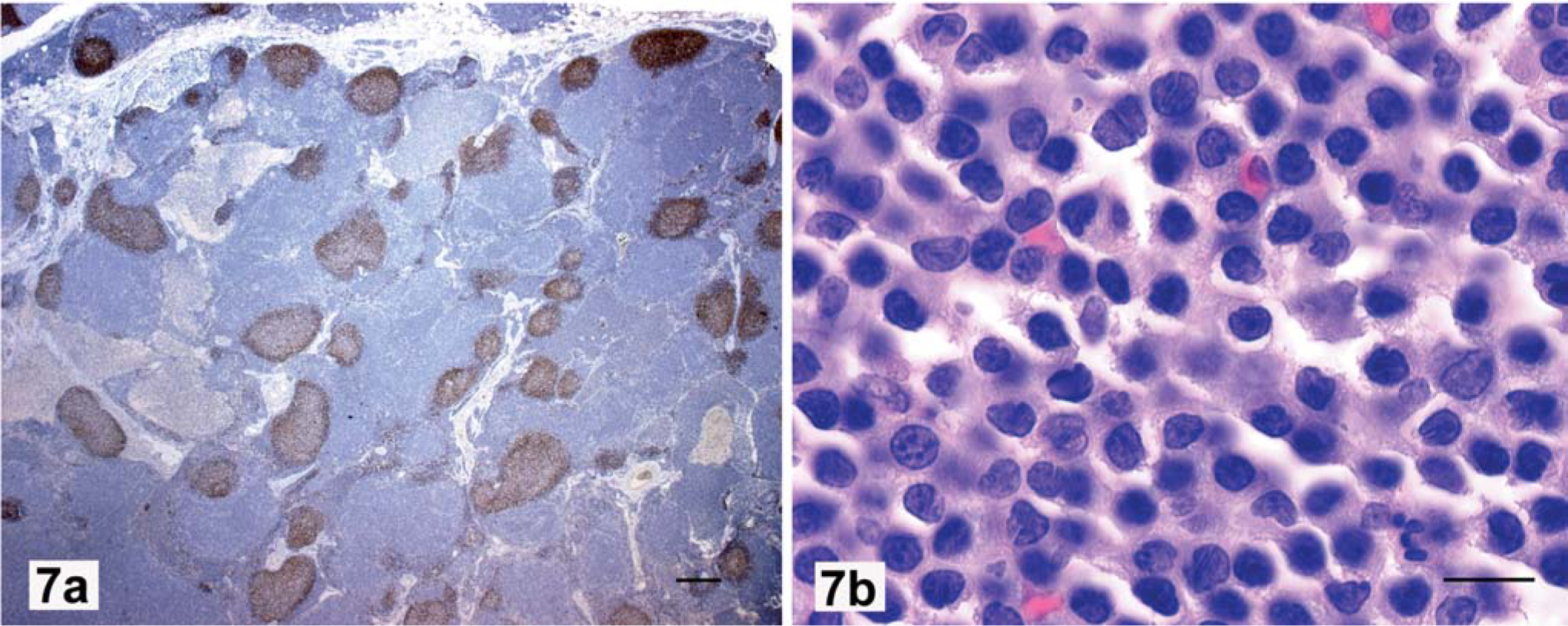
Lymph node; dog. T-zone lymphoma, clonal TCRG gene rearrangement.
Assessment of clonality by polymerase chain reaction (PCR) analysis
The DNA from three to four (25-µm-thick) sections of formalin-fixed paraffin-embedded tissues was extracted and collected into 1.5-ml Eppendorf tubes. Genomic DNA was extracted (DNeasy tissue kit, Qiagen, Valencia, CA) using the manufacturer's recommended protocol, with the exception that tissue lysis with proteinase K was performed overnight or until lysis was complete, whichever was longer. Extracted genomic DNA was quantified spectrophotometrically using an Ultraspec 2100 pro spectrophotometer (Amersham Pharmacia Biotech, Uppsala, Sweden).
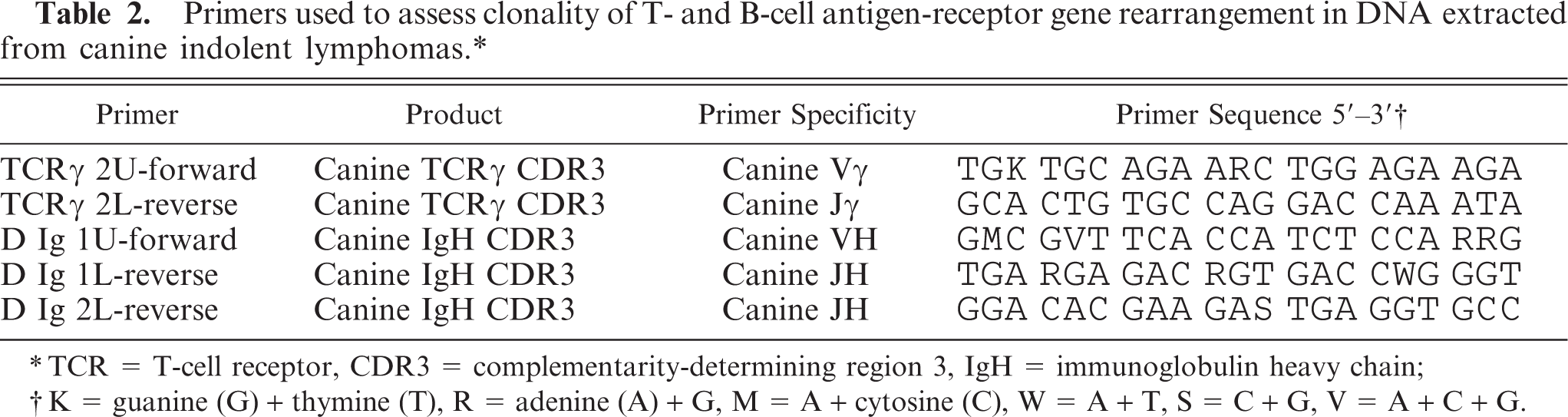
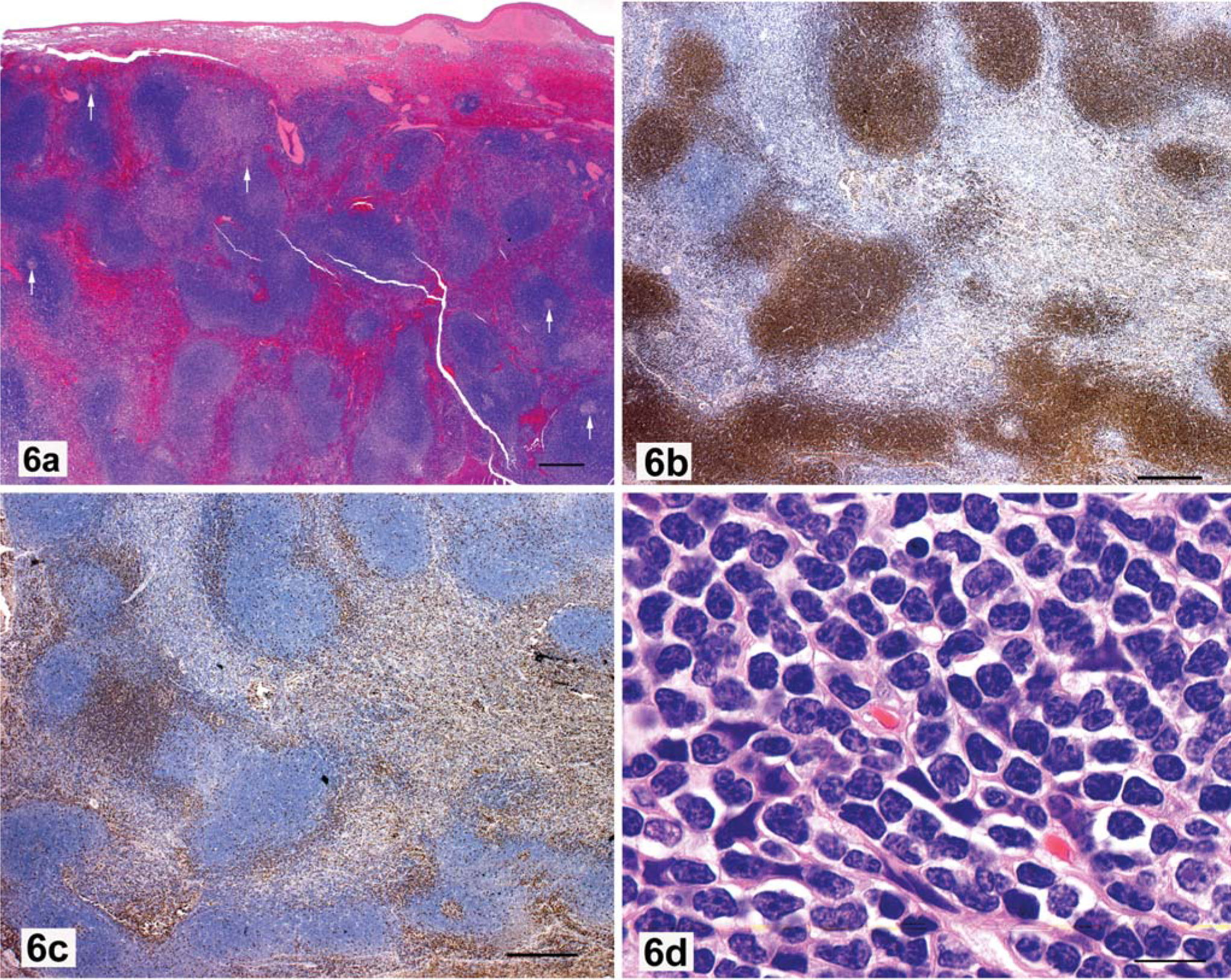
Primers for assessment of T-cell clonality were designed as described. 52 Primer sequences are listed in Table 2. A single set of primers was used for assessment of T-cell clonality (T-cell receptor gamma [TCRG] locus), and a single canine sequence-specific sense primer was used in combination with two canine sequence-specific antisense primers for assessment of B-cell clonality (immunoglobulin heavy chain [IGH] locus); manuscript in preparation). The T-cell clonality primers yield a product(s) approximately 111 basepairs (bp) in size, whereas the B-cell clonality primers amplify a product(s) of approximately 180 bp. The PCR mixes contained approximately 100 ng of genomic DNA template in a total reaction volume of 50 µl. All PCR analyses were performed with an Applied Biosystems GeneAmp PCR system 2700 thermocycler (Foster City, CA). Amplification conditions involved use of a two-step, touch-down protocol to increase specificity of the reactions. 52 Briefly, an initial activation step of 95°C for 15 minutes (HotStar Taq DNA polymerase, Qiagen, Valencia, CA) was followed by five cycles of 94°C for 30 seconds and 72°C for 2 minutes, five cycles of 94°C for 30 seconds and 70°C for 2 minutes, and 35 cycles of 94°C for 20 seconds and 68°C for 2 minutes. A final extension of 68°C for 10 minutes was performed. All PCR-analyzed samples were run in duplicate. All PCR products were screened on 2% agarose 1X Tris acetate–EDTA gels for the presence of a band. Subsequently, polyacrylamide gel electrophoresis was performed using native and denatured 15-µl samples from each PCR. Native samples were loaded directly onto precast, 10% nondenaturing polyacrylamide Tris borate–EDTA (TBE) gels (Criterion Precast gels, Bio-Rad, Hercules, CA). Denatured samples were heated at 95°C for 10 minutes, then were allowed to reanneal at 4°C for 1 hour prior to polyacrylamide gel electrophoresis (heteroduplex analysis). Polyacrylamide gels containing native and denatured PCR products were assayed on ice in 1X TBE at 150 V for 2 hours. Duplicate samples were always run side by side in the PCR analysis (Fig. 8). Gels were then stained for 30 minutes with Gel Star (BioWhitaker Molecular Applications, Rockland, ME) and were visualized with a UV transilluminator (Fisher Scientific, Pittsburgh, PA). The determination of a sample being clonal required the presence of the appropriately and same-sized one or two sharp bands in duplicate samples run in adjacent lanes (Fig. 8). Duplicate PCR assays were used to assist distinction between true clonal samples and pseudoclonal samples. 50 If the result was equivocal on the basis of the duplicate native samples, the denatured heteroduplex result was used to determine the clonality status of the sample. The presence of a broad band, smear, or ladder of bands covering a range of product sizes was interpreted as a polyclonal or reactive lymphoid sample.
Primers used to assess clonality of T- and B-cell antigen-receptor gene rearrangement in DNA extracted from canine indolent lymphomas.∗
TCR = T-cell receptor, CDR3 = complementarity-determining region 3, IgH = immunoglobulin heavy chain
K = guanine (G) + thymine (T), R = adenine (A) + G, M = A + cytosine (C), W = A + T, S = C + G, V = A + C + G.
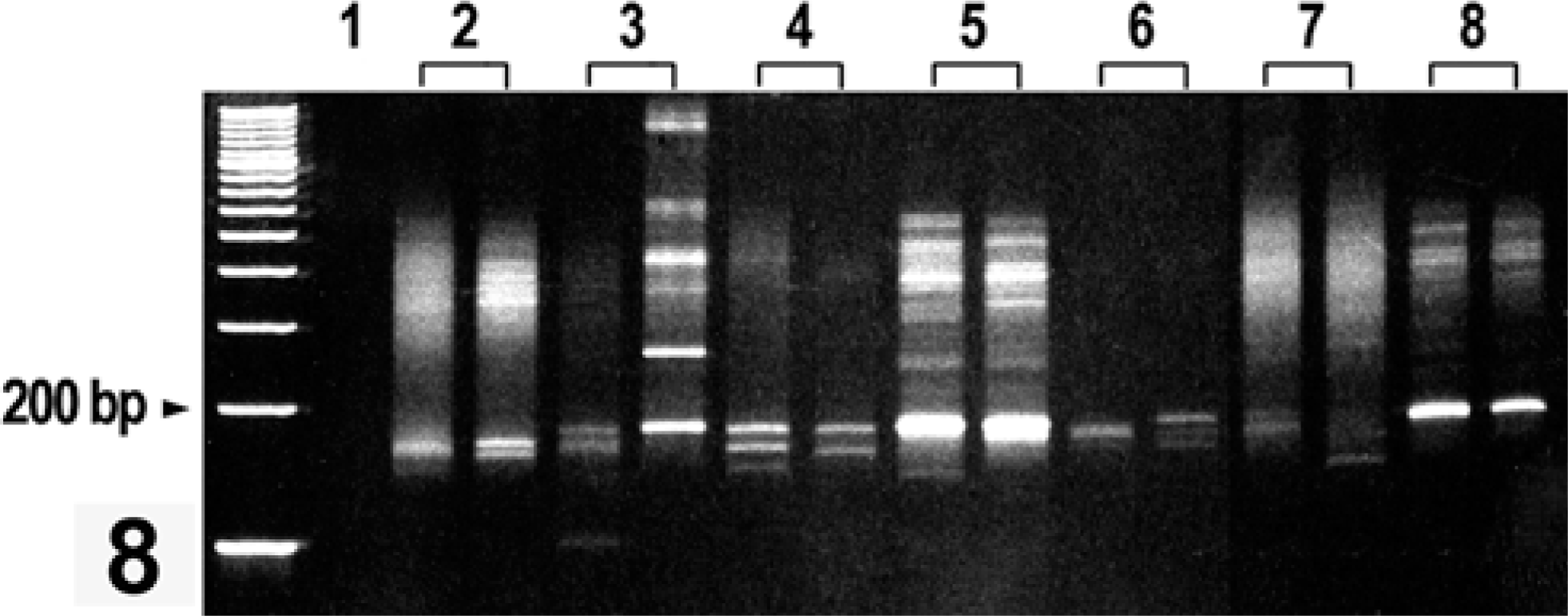
Heteroduplex analysis of B-cell clonality (IGH gene rearrangement) polymerase chain reaction (PCR) products run on a GelStar-stained 10% polyacrylamide gel. Lanes 2–8 are duplicate analyses (separate reaction tubes) of DNA extracted from paraffin blocks of samples from dogs with a diagnosis of marginal zone lymphoma (MZL). The DNA was amplified with canine-specific,B-cell “clonality” primers that produce a product(s) approximately 180 base pairs (bp) in size. Lanes: 1, negative control; 2, polyclonal with pseudoclonal bands; 3, pseudoclonal; 4 and 5, clonal with one or two repeatable sharp bands of appropriate size; 6, pseudoclonal; 7, polyclonal control; and 8, clonal positive control. Heteroduplex products have retarded gel mobility and “run” approximately 300–400 bp in size.
Results
Demographics
The study population consisted of: dogs of mixed breeding (n = 7); Golden Retrievers (n = 7); Labrador Retrievers (n = 6); dogs of unknown breeding (n = 5); Rottweilers (n = 5); terrier types (n = 5); West Highland White Terriers (n = 2); Cocker Spaniels (n = 3); two each of Beagle, pit bull type, Shetland Sheepdog, German Shepherd Dog, and Doberman Pinscher; and one each of 15 other breeds. Large-breed dogs totaled 32, with 21 of small breed, eight dogs of mixed breeding, and five dogs of unknown size or breed. The sex distribution was 23 spayed female, 19 reproductively intact female, 11 neutered male, and 10 reproductively intact male, with 3 of unreported sex. Age of the 66 dogs ranged from 1.5 to 16 years, with median age of 9.0 years (Table 1). The median age distribution by diagnosis was 7.7 years for MZL (n = 46), 8.0 years for MCL (n = 3), 11.0 years for FL (n = 3; 2 age unknown), and 8.9 years for TZL (n = 10).
Presentation
Four dogs with FL had a history of chronic enlargement of one or more lymph nodes, and two of these had chronic inflammatory skin disease. One dog with FL presented with a single enlarged node that regressed on treatment with steroids, and re-presented 2 years later with diarrhea and melena.
The animals with MZL presented with generalized lymph node enlargement (10 cases), one or two nodes enlarged (22 cases), and three nodes enlarged (one case). The remaining 13 cases had focal splenic involvement which, in most cases, was noted on palpation, or was an incidental finding on ultrasonography. The 10 cases of MZL with generalized node involvement included three cases at an early stage (Table 1, cases 18, 49, 50), three cases at mid-stage (Table 1, cases 19, 33, 47) and four cases at late stage (Table 1, cases 12, 27, 36, 43). All 13 cases with splenic involvement had multifocal areas of neoplastic proliferation within a solitary splenic lesion. The remaining 23 animals that presented with one to three enlarged nodes were found to be at an early stage (one case), mid-stage (12 cases), and late stage (10 cases).
All three dogs with MCL presented with a splenic mass that appeared to be the only site of tumor at the time of surgery. In these cases, the foci of cellular proliferation were apparently arising in association with an end arteriole.
The 10 dogs with TZL had a history of chronic node enlargement, present or developing, for 7 months, 9 months, 2 years, and 3 years, and for an unspecified time in the other 6.
Correlation between morphologic diagnosis and clonal analysis
Cases for which the histologic diagnosis was altered on the basis of clonality results have the initial diagnosis following in brackets (Table 1, cases 13, 20, 55, and 56).
For FL, DNA was available from four of five cases. Two cases were clonal for B-cell immunoglobulin gene rearrangement (Table 1, cases 1 and 2), and two were clonal for rearrangement of T- and B-cell genes (Table 1, cases 3 and 4).
With DNA available from 35 of the 46 cases interpreted to be MZL, 22 cases had clonal rearrangement of IGH only (Table 1, cases 6–27). With inclusion of the six cases that had clonal rearrangements for IGH and TCRG genes, (Table 1, cases 28–33) the level of agreement between histologic diagnosis and molecular clonality was 80% (28/35). The single case found to have a clonal TCRG gene rearrangement and included in the MZL category (Table 1, case 34; spleen) had almost complete atrophy of periarteriolar lymphoid sheaths with multiple foci typical of MZL. The neoplastic lymphocytes expressed CD79a. There was no evidence of T-cell lymphoma, and the diagnosis was interpreted to be early MZL. The lack of a clonal IGH gene rearrangement was interpreted as a false-negative result. One case of MZL, initially diagnosed as TZL (Table 1, case 13), had clonal IGH gene rearrangement only. On review, it had architecture typical of MZL, with areas of cellular proliferation around the foci of fading germinal centers. Cytologically, the areas of proliferation were composed of large cells with prominent nucleoli typical of MZL. The immunophenotype was equivocal; the CD3 staining result was favored, the cell type typical of MZL was ignored, and an incorrect initial diagnosis of TZL was made.
The DNA from all three cases interpreted histologically to be MCL was available. All three had a clonal IGH gene rearrangement (Table 1, cases 52–54).
The DNA from 8 of the 10 cases diagnosed as TZL was available (Table 1). Five cases had a clonal TCRG gene rearrangement (Table 1, cases 55–59), and three were polyclonal (Table 1, cases 60, 61, 63). Case 55 was originally diagnosed as FFH. Case 56 was initially interpreted to be MZH. The tissue from case 56 was small and fragmented, and on review, there was small cell proliferation eccentric to the fading follicles, which that was typical of TZL. Immunophenotyping was not available to aid interpretation. In this case, a clonal TCRG gene rearrangement was definitive. The polyclonal results for TZL cases were considered falsely negative.
Only one of two cases of MZH (Table 1, case 65) had molecular clonality assessment, and as expected, the result was polyclonal.
Biological behavior
Clinical information was available for 18 dogs (Table 1). The diagnoses for this group of dogs included MZL (nine cases), TZL (eight cases), and FL (one case).
A dog with clinical stage-IIIa FL (Table 1, case 1) responded with complete remission to initial treatment with COPLA, and rescue therapy with the same protocol at first relapse, with further good responses to CCNU (lomustine), DTIC (dacarbazine), and
Follow-up data were available for three cases of splenic MZL (Table 1, cases 9, 11 and 28). Case 11 also had involvement of the splenic hilar node. All were treated solely by splenectomy, and none died because of MZL in the follow-up period (7 to 19 months).
Follow-up information was available for six cases of nodal MZL (Table 1, cases 6–8, 10, 12, 36). The six cases of MZL were in various clinical stages, as follows: case 6, stage 1a; cases 7 and 10, stage IIIa; cases 8 and 36, stage IVa; and case 12, stage Vb. When treated with standard multi-agent chemotherapy protocols, 8 the animals tended to be responsive to therapy, with longstanding complete clinical remissions (Table 1, cases 7, 8, 10, 12, 36). Relapses were observed in two of these dogs, but lymphoma was the documented cause of death in only one dog (Table 1, case 36). Interestingly, one of the dogs with stage-IIIa disease that was treated sporadically because of poor owner compliance had repeated clinical remissions followed by relapses (Table 1, case 10). This dog had good quality of life until progression of chronic renal failure, 43 months after initial diagnosis of MZL.
Clinical history was available for eight dogs with TZL (Table 1, cases 56–63). All dogs with TZL had generalized lymphadenopathy, without clinical signs of illness (stage- IIIa disease), and none were hypercalcemic. Only two dogs received systemic therapy other than prednisone alone. Therapy did not appear to influence survival or quality of life in this subgroup of dogs, given that the two longest survivors (both clonal for TCRG) were untreated and maintained a normal quality of life (Table 1, cases 57, 59). No dogs with TZL were reported to die of causes related to the lymphoma. The median survival time for dogs with TZL was not yet reached at 22.5 months (range 1–90 months), with two dogs still alive at 19 and 32 months, and one dog lost to follow up at 17 months.
Discussion
An important finding of this study was that indolent lymphomas are not rare in dogs, although the true incidence remains unknown. The current emphasis on B- versus T-cell type in the management of canine lymphomas is based on extensive experience. In terms of survival, it may be as important to distinguish between the aggressive and indolent types of B- and T-cell lymphomas. Reliable information about the biological behavior and response to therapy of the various subtypes of canine lymphoma is needed. The major change in approach of the Revised European-American Lymphoma/World Health Organization (REAL/WHO) system of classification of hematopoietic tumors is that it is based on categorizing neoplasms as single disease entities with definable biological characteristics anchored by topographic, morphologic, immunophenotypic, and molecular genetic evaluation. 1, 28, 34 The first step in this process for animal oncology is for neoplasms to be accurately defined for diagnostic purposes before they can be managed as treatment entities. There is need for collaboration between pathologists and clinicians to determine the boundaries of animal disease categories if they are to be based on diagnostic criteria and response to therapy. Interpretation of nodal architecture is required for the diagnosis of indolent nodular lymphoma, and this requires an adequate biopsy specimen.
In general, the FL, as they occur in animals, appear to be directly comparable to the human counterparts in cytomorphologic and biological progression. Twenty percent of human lymphomas are of the follicular type, but FL are less common in dogs and cats, and are infrequently reported. 3, 20, 21, 25, 30, 31 Recognition of neoplasms related to the germinal center has been facilitated by routine immunophenotyping of animal lymphomas. Immunophenotyping may document the “footprint” of a follicle-related heritage in diffuse lymphomas by outlining fading follicles with a mixture of smaller B and T lymphocytes. In the absence of immunophenotyping, follicular lesions that have progressed to a diffuse architecture are likely to be categorized generically as “lymphoma.” Follicular lymphoma needs to be distinguished from follicular hyperplasia, and this requires careful morphologic assessment aided by molecular clonality determination.
Human splenic MZL is a rare form of lymphoma that make up <1% of lymphoid tumors. The prevalence of canine MZL is unknown. Human MZL is the most common primary splenic lymphoma, and this may be the case in dogs. Splenic MZL in humans always involves the whole spleen in a micronodular pattern attributable to uniform involvement of all follicular areas. 12, 29, 41 In sharp contrast, canine splenic MZL is a focal or locally extensive lesion that projects abruptly from the normal contour of the splenic capsule. These tumors are palpable and evident on ultrasound examination. 46 Cytologically, neoplastic marginal zone lymphocytes in dogs have a more immature, immunoblastic appearance than their counterparts in MZH. A major feature of canine MZL is the initial low mitotic rate, despite an immunoblastic appearance. With disease progression, the cells are morphologically similar, but there are many apoptotic cells and up to 5–10 mitoses/400× field.
In terms of presentation, nodal and splenic MZL in humans are described as separate diseases. 18, 19 Human primary splenic MZL usually also involves splenic nodes and liver, bone marrow, and blood. 12, 29 By contrast, in canine splenic MZL, the spleen is usually the only site of involvement, with occasional spread to the splenic hilar nodes. The focal areas of neoplastic lymphocytes form irregular clusters that are larger than germinal centers and that, similar to human MCL and MZL, surround end arterioles. These areas are separated by small CD3+ lymphocytes and occasionally by solid sheets of plasma cells that appear cytologically similar to cutaneous plasmacytoma. These diseases are reported to occur together in humans. 10
Human nodal MZL accounts for <2% of all lymphoid neoplasms. A third of cases involve extranodal soft tissue and, occasionally, bone marrow and blood. 27 A second form of human nodal MZL has cells that are more irregular and slightly larger and resemble a nodal form of mucosa-associated lymphoid tissue-type (MALT) lymphoma. 38 MALT lymphoma is not well defined in the dog, but a single case in this series (Table 1, case 3 FL III,) had submandibular node involvement with complete destruction of an adjacent salivary gland and multiple areas of “lymphoepithelial lesions” characteristic of human MALT lymphoma. The nodal lesion was typical of FL and not of MZL.
Human MCL constitutes 3–10% of all human non-Hodgkin lymphomas. In animals, MCL appears to be rare or largely undetected. 20, 21, 26, 40 Unlike MZL, which appears to follow a long period of marginal zone hyperplasia, MCL appears not to be detected at a stage of hyperplasia. Cytologically, canine MCL is similar to that of humans, but a major difference appears to be that canine MCL develops more frequently in the spleen than in the nodes. The “blastoid” variant of human MCL has larger, more aggressive-appearing nuclei that have nucleoli, and tends to become leukemic with progression. 2, 6, 16 This variant was not observed in the dogs of this study. Cyclin D1 is overexpressed in most cases of human MCL; detection of cyclin D1 is useful diagnostically, but this reaction is difficult to carry out on human tissue. 7, 13, 35 Detection of cyclin D1 was attempted without success in our cases of canine MCL. Human MCL tends to present as a disseminated disease with lymph node, spleen, bone marrow, and gastrointestinal tract frequently involved. 16
The diagnosis of canine splenic fibrohistiocytic nodule or nodular lymphoid hyperplasia is common, and cases of MZL and MCL may have been included in this designation. 46, 47 Similarly, on cytologic examination of specimens, the population of uniform small lymphocytes of canine splenic MCL would likely be considered benign. 15 This underscores the need for architectural assessment in establishing a diagnosis of these lymphomas. Hence, the current trend toward fine needle aspiration biopsy for the diagnosis of canine lymphoma will lead to continued underrecognition of MZL and MCL.
T-Cell lymphomas of large cell type in lymph nodes, skin, and gut of dogs have been well described. 17, 22, 24, 32, 33, 42 Indolent forms of nodal T-cell lymphoma in humans have been described. 1, 28, 36 The “T-zone lymphoma,” as recognized in the Kiel classification, has also been referred to by others as “small clear cell” lymphoma. 21 The T-zone designation (TZL) is preferred here because the architectural identification of the paracortex as a T-cell domain gives more indication of the expected immunophenotype of the lesion. Even when well advanced, TZL may be difficult to recognize as a malignant neoplasm because of the small mature-appearing cell type and paucity of mitotic activity. The inclusion of 10 cases of TZL in this study was intended to highlight the difficulty that may occur in distinguishing TZL and late-stage MZL. The report that human peripheral T-cell lymphoma (TZL) may mimic MZL is highly applicable to the dog. 49 A subtle difference in architecture exists between MZL where the proliferation is concentric and encircles the fading germinal centers, whereas in TZL, the proliferation is eccentric to the residual B-cell foci.
The detection of clonality in lymphoid proliferations of the dog is a useful adjunct to histologic examination. 9, 52 Clonal rearrangement of IGH was confirmed in 28 of 35 cases (80%) diagnosed as MZL, supporting the diagnosis of B-lymphoid neoplasia, despite the nodular appearance of the tumor and indolent biological behavior. Additionally, the initial histologic diagnosis was altered on the basis of clonality results (and reassessment of other results) in four instances (Table 1, cases 13, 20, 55, 56). Failure to detect clonality in 7 of 35 cases (20%) is consistent with the sensitivity of PCR-based clonality assays. 50 This can be attributable to inadequate or inappropriate selection of tissue for PCR analysis as well as lack of primer coverage of all possible IGH rearrangements, partial rearrangements of IGH, and chromosomal defects (deletion or translocation). 50 In view of convincing histologic findings, the seven negative IGH results in canine MZL were interpreted as falsely negative. Careful histologic examination of tissues and immunophenotyping are necessary for the accurate interpretation of clonality results. This is underscored by the finding that 6 of 28 cases (21%) of MZL with a clonal IGH gene rearrangement had a concurrent TCRG gene rearrangement. Clonal rearrangement of B- and T-cell receptor genes from the same DNA sample may occur under a number of circumstances. These include true cross-lineage rearrangement, in which some B-cell lymphomas will aberrantly rearrange TCRG. 50 Additionally, the presence of a benign population of T lymphocytes of limited diversity, reacting against the B-cell lymphoma, may result in an oligoclonal or clonal TCRG gene rearrangement. 50 In the absence of duplicate PCR clonality analysis, amplification of TCRG from scant residual reactive T cells could produce a false-positive result (“pseudoclonality”). This possibility was excluded because the PCR assays were conducted in duplicate and were analyzed side by side in the same gel. With these caveats recognized, molecular clonality assessment integrated with clinical, morphologic, and immunophenotypic data provides an important advance in the assessment of canine lymphoma. This is especially so when doubt still exists after routine morphologic and immunophenotypic assessment.
Limited follow-up information for dogs of this study indicated that indolent nodal MZL tends to respond well to systemic chemotherapy, and prolonged or repeated clinical remissions may be achieved. It also suggests that, as in humans, the benefit for postsplenectomy adjuvant cytotoxic chemotherapy for purely splenic MZL may be limited. 5 It appears that some canine indolent B-cell lymphomas, when in late histologic stage, will behave clinically like high-grade lymphomas and, in fact, may have made that transformation. Results of follow-up evaluation of eight cases of TZL suggest that canine indolent nodal T-cell lymphomas (TZL) have long survival time, despite marked lymphadenopathy and limited clinical response to therapy. This is in marked contrast to the typical observation associated with high-grade lymphomas, where a B-cell immunophenotype generally predicts more favorable response to therapy, remission duration, and overall survival time. 48 Traditional combination systemic chemotherapy is still recommended for indolent lymphomas in late histologic stage, when the clinical behavior mimics that of high-grade lymphomas.
In conclusion, the level of heterogeneity in canine lymphomas is deserving of greater attention by pathologists and oncologists. Greater precision in diagnosis will permit more accurate prognostication and therapy better tailored to high-grade and indolent lymphomas. Current methodologies, which include immunophenotyping and molecular clonality assessment, provide the means to better recognize the gray zone between hyperplasia and neoplasia, and for better recognition of specific subtypes of lymphoma. Recognition of homogeneous subtypes of lymphoma is essential for future studies designed to discover the specific underlying genetic defects that define these disease entities and their biologic behavior.
