Abstract
Marginal zone lymphoma (MZL) and mantle cell lymphoma (MCL) belong to a subgroup of indolent B-cell lymphomas most commonly reported in the canine spleen. The goal of this study was to characterize the immunophenotype of splenic MZL and MCL in comparison to their human counterparts. Ten MCLs and 28 MZLs were selected based on morphology. A tissue microarray was generated, and expression of CD3, CD5, CD10, CD45, CD20, CD79a, Pax-5, Bcl-2, Bcl-6, cyclin D1, cyclin D3, MCL-1, MUM-1, and Sox-11 was evaluated. Neoplastic cells in all MCLs and MZLs were positive for CD5, CD20, CD45, CD79a, and BCL2 and negative for CD3, CD10, Bcl-6, cyclin D1, and cyclin D3. Positive labeling for Pax-5 was detected in 8 of 10 MCLs and 26 of 28 MZLs. Positive labeling for MUM-1 was detected in 3 of 10 MCLs, and 27 of 28 MZLs were positive for MUM-1. No MCLs but 8 of 24 MZLs were positive for MCL-1. Canine splenic MZL and MCL have a similar immunophenotype as their human counterparts. However, human splenic MCL overexpresses cyclin D1 due to a translocation. A similar genetic alteration has not been reported in dogs. In addition, in contrast to human MZL, canine splenic MZL generally expresses CD5. Following identification of B vs T cells with CD20 and CD3, a panel composed of BCL-2, Bcl-6, MUM-1, and MCL-1 combined with the histomorphological pattern can be used to accurately diagnose MZL and MCL in dogs. Expression of Bcl-2 and lack of MCL-1 expression in MCL may suggest a therapeutic benefit of BCL-2 inhibitors in canine MCL.
Indolent lymphomas account for roughly 29% of all canine lymphomas. 19 They are composed of T-zone lymphomas and follicular-derived B-cell lymphomas, which include follicular lymphoma, marginal zone lymphoma, and mantle cell lymphoma.
Mantle cell lymphoma (MCL) and marginal zone lymphoma (MZL) are subsets of follicular-derived, indolent lymphomas that are characterized by neoplastic B lymphocytes with low mitotic activity and slow rates of progression. 59 These entities are easily differentiated from one another, as the proliferating cell populations are morphologically distinct. Mantle cells lie within the mantle cuff surrounding lymphoid follicles in the spleen and lymph nodes. 19,57 They are characterized as intermediate-sized cells (nucleus approximately 1.5× the width of a red blood cell) with scant cytoplasm, indented to round nuclei with dense chromatin, and an inconspicuous nucleolus. Neoplastic mantle cells proliferate and cause paracortical atrophy and the fading of hyalinized germinal centers. 6,7 Some MCLs can be difficult to differentiate from follicular hyperplasia, as both consist of increased numbers of mantle cells. However, dropout of the germinal center is not seen in follicular hyperplasia, and this can be useful for differentiating MCL from follicular hyperplasia.
Marginal zone cells are found in lymph nodes, spleen, and bronchus- and mucosa-associated lymphoid tissue as a result of follicular hyperplasia. They are characterized as intermediate-sized cells with abundant lightly stained cytoplasm and nuclei with peripheralized chromatin, as well as a single, large, central nucleolus. Neoplastic marginal zone cells cause perifollicular proliferation with paracortical atrophy and dropout of germinal centers. 11,12,40 Differentiating some MZLs from marginal zone hyperplasia can be challenging as both display marginal zone lymphocyte proliferation and germinal center regression. 56 A key difference is that marginal zone hyperplasia forms discontinuous cuffs of marginal zone lymphocytes mixed with normally occurring mantle cuff cells and centroblasts, whereas MZL forms homogeneous cuffs of marginal zone lymphocytes accompanied by paracortical atrophy.
To assist in distinguishing MZL and MCL from other types of lymphomas, the World Health Organization (WHO) recommends immunophenotyping in combination with histopathology. 53 Both canine MZL and MCL are positive for B-cell markers CD20 and CD79a and negative for the T-cell marker CD3. 57,58 To our knowledge, a detailed immunophenotypic characterization has not been reported for canine MCL or MZL. The morphologies of human splenic MCL and MZL mirror their canine counterparts, but the human variants have been extensively immunophenotyped, whereas the canine variants have not. 43,51 In general, both human splenic MZL and MCL are positive for CD45, CD20, CD79a, Bcl-2, and Pax-5 and negative for CD3, CD23, BCL-6, and CD10.* In addition, MCL is generally positive for CD5, CD38, cyclin D1, and sex-determining region Y box 11 (SOX-11) while MZL is generally positive for Bcl-10 and MUM-1. 17,30,33,34,38,41,60 In both entities, some of the reportedly negative antibodies are expressed in a subset of histologically confirmed cases. For example, MUM-1 is expressed by approximately 35% of MCL cases, and an even smaller percentage of MCL cases expresses BCL-6 or CD10. 23,41 Similarly, 11% to 35% of MZLs have been found to express CD5. 22,33
Human MCL has been found to have zero to low expression of MCL-1, whereas MZL has low to moderate expression of MCL-1. 1 MCL-1 has been used in human lymphomas as a therapeutic indicator in combination with BCL-2. Both proteins originate from the same family and inhibit cellular apoptosis. 25 One study revealed that deletion of both MCL-1 alleles in mice sensitized plasma cells to BCL-2 inhibitors. 61 Therefore, it is important to investigate BCL-2 expression in combination with MCL-1 expression in canine follicular-derived lymphomas.
Human MCL tends to behave more aggressively than canine MCL, but the clinical data are limited. Most human MCLs harbor a t(11:14) chromosomal translocation mutation at t(11;14)(q13;q32) that results in overexpression of cyclin D1. A subset of human MCLs is translocation negative and does not express cyclin D1 but rather expresses cyclin D2 or cyclin D3. 14,20,38 Activation of cyclin D has not been reported in canine MCL. Human MZL behaves similarly to canine MZL and follows an indolent course. Unlike MCL, the exact pathogenesis of MZL is unknown. Cytogenetic studies in humans have revealed that 72% of splenic MZLs demonstrate aberrant karyotypes predominated by gains of 3/3q and 12q, deletions of 7q and 6q, or translocations involving 8q/1q/14q, with the most frequent being deletion of 7q (7q21–36) seen in 40% of cases. 13,49,64 Synonymous chromosomal abnormalities have not been identified in canine MZL.
Of the described markers used for immunophenotyping of human MCL and MZL, CD3, CD10, CD20, CD45, CD79a, Bcl-2, Bcl-6, Pax-5, and MUM-1 have been routinely used in formalin-fixed canine tissues. † CD5, cyclin D1, cyclin D3, and MCL-1 have been used scarcely via different canine immunophenotyping methods. 5,27,36,37,39,44,63 While many of these markers have not been validated via Western blot in canine formalin-fixed, paraffin-embedded tissue, they have demonstrated apparent cross-reactivity based on patterns of immunolabeling in canine tissues. To the authors’ knowledge, SOX-11 immunohistochemistry has not been used at all in canine tissues.
The goal of this study was to characterize the immunophenotype of canine splenic MZL and MCL in relation to their human counterparts to develop a thorough diagnostic immunohistochemical panel.
Methods
Case Selection
A total of 35 splenic MZL and 13 splenic MCL cases were retrospectively selected from cases submitted between 2010 and 2015 to the Veterinary Diagnostic Laboratory at Michigan State University. Of these, paraffin-embedded blocks of tissue were available for 28 MZLs and 10 MCLs. Cases had been diagnosed based on morphologic characteristics and immunophenotyping for CD3 and CD20. We only selected cases that had morphologic characteristics of either entity and avoided any cases that were difficult to distinguish from hyperplasia or other follicular entities as described above.
Histopathology and Immunohistochemistry
For each case, hematoxylin and eosin (HE)–stained sections and sections immunohistochemically labeled for CD3 and CD20 slides were evaluated to confirm the previous diagnosis. Tissue microarrays were generated with five 1-cm diameter punches for each case. Punches were taken from the original paraffin block from multiple regions of each lymphoma. Specifically, we sampled marginal zone cells or mantle cells of each representative lymphoma. These cells were easily identified in HE-stained slides based on their characteristic histologic features (Figs. 1, 2), especially when also combined with immunophenotyping for CD3 and CD20. Completed tissue microarray (TMA) blocks were stained with HE to confirm the proper selection of neoplastic cells. Punches that missed the targeted areas or had poor representation of the neoplastic cells were omitted. Immunohistochemistry (IHC) for CD5, CD10, CD45, CD79a, Bcl-2, Bcl-6, Pax-5, MCL-1, MUM-1, cyclin D1, cyclin D3, and SOX-11 was performed on serial sections of the TMA. The choice of these antibodies was made based on their diagnostic and prognostic value in human MCL and MZL. IHC for the antibodies were carried out in accordance with the guidelines set forth by the American Association of Veterinary Diagnosticians (AAVLD) Subcommittee on Standardization of Immunohistochemistry. 45 IHC was performed on a BOND-MAX Automated Staining System (Vision BioSystems; Leica, Bannockburn, IL) using the Bond Polymer Detection System (Vision BioSystems; Leica) with 3,3′-diaminobenzidine (DAB) as the chromogen for CD3, CD20, CD45, CD79a, and MCL-1; on the Dako link 48 Automated Staining System (Agilent Technologies, Santa Clara, CA) using the peroxidase-conjugated EnVision Polymer Detection System (Agilent Technologies) for CD5, CD10, Bcl-6, MUM-1, cyclin D1, cyclin D3, and SOX-11; or on the Discovery Ultra Automated Staining system (Ventana Medical Systems, Tucson, AZ) using the UltraMap alkaline phosphatase red detection system (Ventana Medical Systems) for Bcl-2 and Pax-5. Details of the antibodies, retrieval methods, and detection are shown in Table 1. Normal canine lymph node and spleen were used as positive controls for all antibodies except SOX-11 based on previous reports demonstrating their apparent and expected cross-reactivity in canine tissues. Normal human tonsil, normal canine spleen, lymph node, and tonsil, as well as nonhuman primate brain and heart, were used as positive controls for SOX-11 based on expected cross-reactivity reported in human tissues. IHC-labeled microarray slides were blindly reviewed by 2 authors (L.S., M.K.) and immunoreactivity was subjectively scored as an average percentage of positive neoplastic cells per punch per case for each lymphoma entity. A case was called positive for CD3, CD10, CD20, CD45, CD79a, Bcl-2, Bcl-6, or Pax-5 if the majority of neoplastic cells showed strong, diffuse, marker-specific labeling. For CD5, cyclin D1, cyclin D3, MCL-1, and MUM-1, a case was called positive if the percentage of immunoreactive neoplastic cells matched the percentage patterns reported in the human literature. A positive cutoff percentage of 50% was used for cyclin D1 20,29,60 and cyclin D3. 20 While the reported expression patterns of CD5, 21,22,33 MCL-1, 1,26 and MUM-1 17,23 in human MCL and MZL as well as in canine tissues vary among studies, 5,27,35,37,56,63 in our study, a case was called positive for these markers if at least 10% of the neoplastic cells showed positive labeling. The data analyzed in this study are available upon request to the corresponding author.
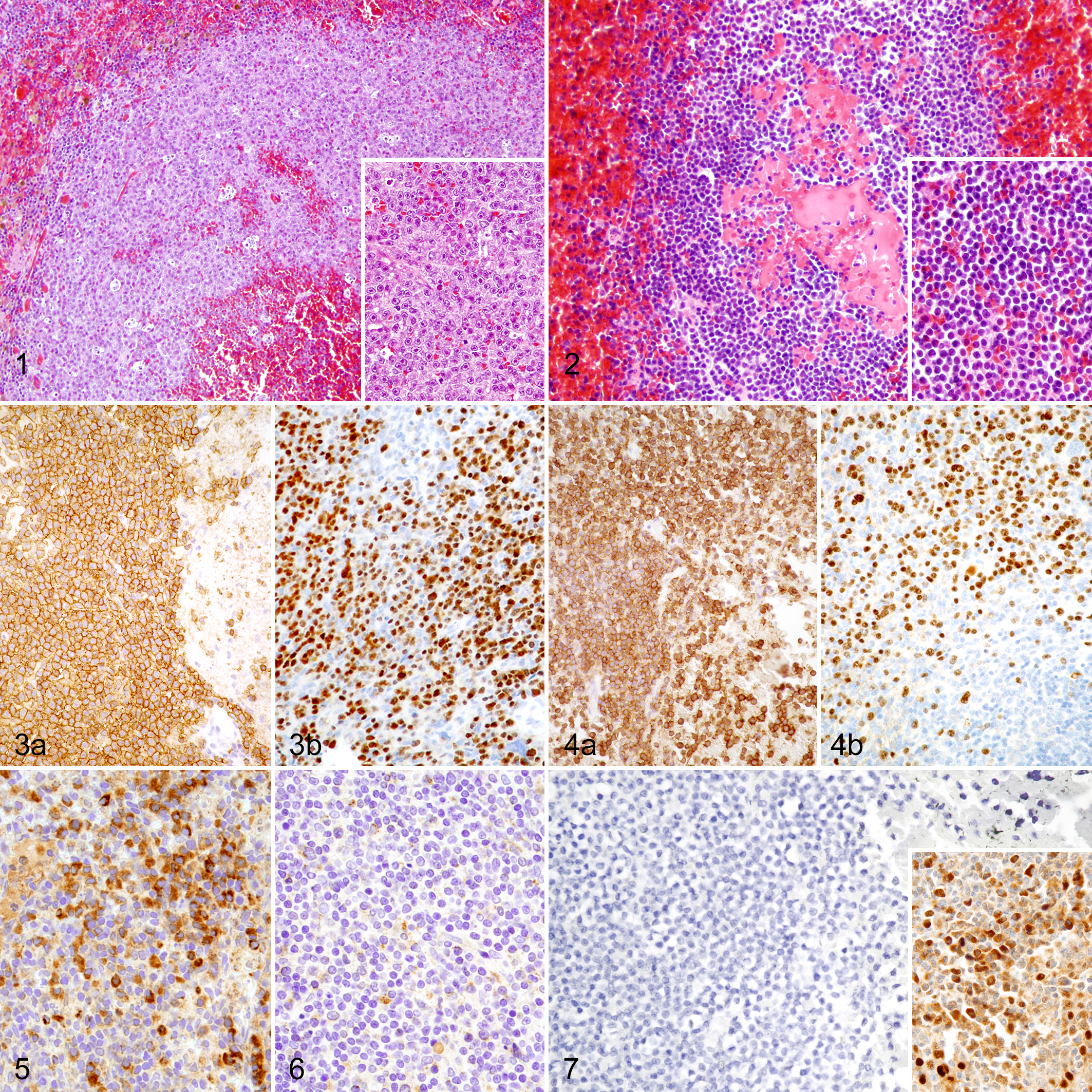
Marginal zone lymphoma (MZL) and mantle cell lymphoma (MCL), spleen, dog.
List of Antibodies With Source, Clone and Manufactures, Concentration Used, Retrieval Information, and Detection Method.
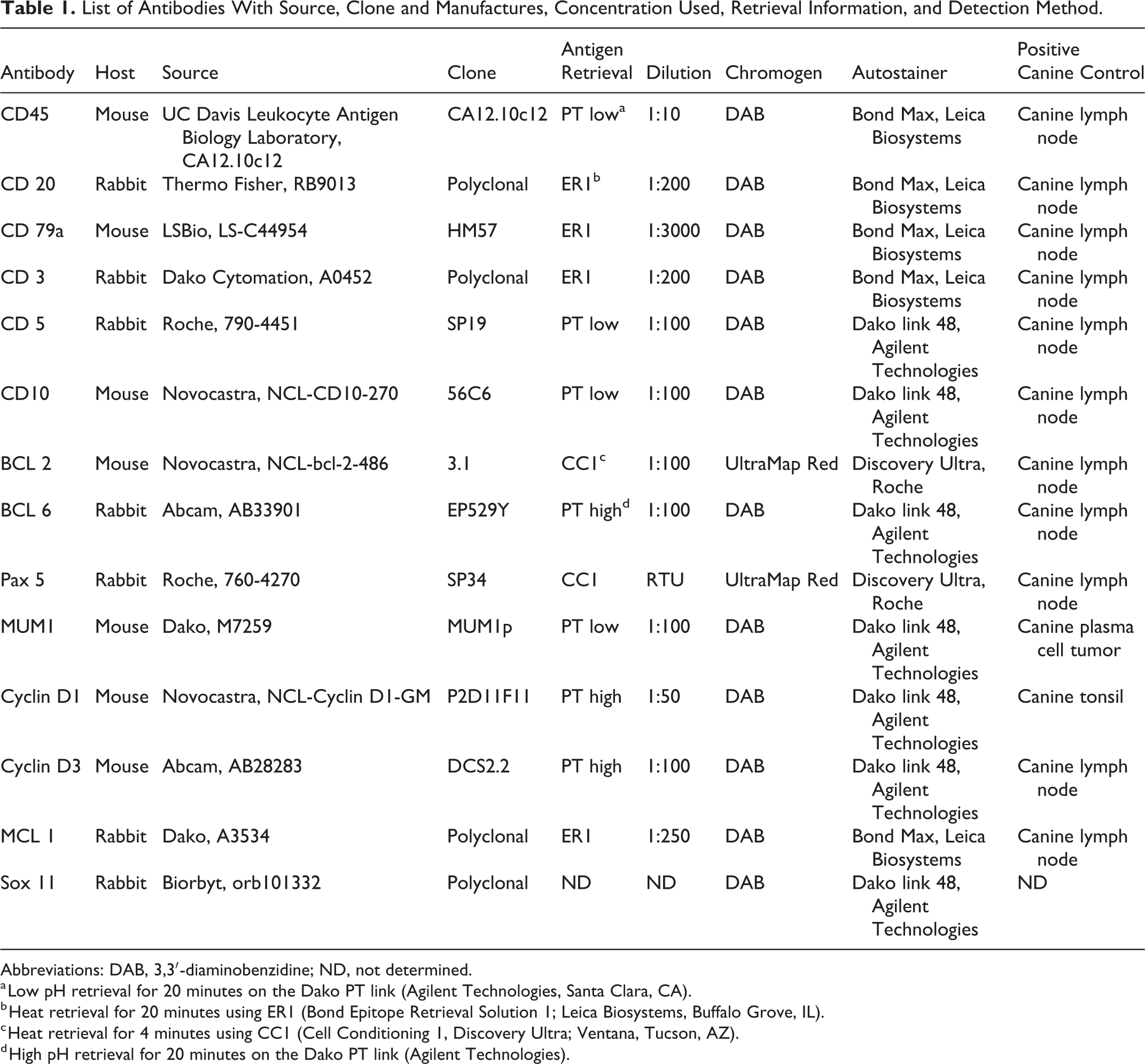
Abbreviations: DAB, 3,3′-diaminobenzidine; ND, not determined.
a Low pH retrieval for 20 minutes on the Dako PT link (Agilent Technologies, Santa Clara, CA).
b Heat retrieval for 20 minutes using ER1 (Bond Epitope Retrieval Solution 1; Leica Biosystems, Buffalo Grove, IL).
c Heat retrieval for 4 minutes using CC1 (Cell Conditioning 1, Discovery Ultra; Ventana, Tucson, AZ).
d High pH retrieval for 20 minutes on the Dako PT link (Agilent Technologies).
Results
Histopathology
MZLs were characterized by proliferating intermediate-sized B cells with abundant, lightly stained cytoplasm and mildly vesiculated nuclei with marginated chromatin and a large single, central nucleolus (Fig. 1). Cells formed homogeneous cuffs around fading germinal centers. MCLs also surrounded fading germinal centers, but the neoplastic cells were intermediate-sized cells with scant cytoplasm and chromatin-dense nuclei with variable degrees of indentation and angulation and inconspicuous nucleoli (Fig. 2).
Immunohistochemistry
All antibodies were positive in canine control tissue, except SOX-11. SOX-11 was expressed in normal human tonsil, but was there was no expression in normal canine lymph node, spleen, tonsil, or nonhuman primate brain or heart. All MCLs and MZLs had strong immunoreactivity for CD20, CD45, CD79a, and Bcl-2 in at least 90% of neoplastic cells (Table 2) (Figs. 3a, 4a). CD3, CD10, Bcl-6, and cyclin D1 did not label any neoplastic cells in either the MCLs or the MZLs (Fig. 7). All MZLs and MCLs had at least a third of all neoplastic cells labeled for CD5. Eight of the 10 MCLs had at least 70% of neoplastic cells labeled for Pax-5, and 26 of the 28 MZLs had nearly all neoplastic cells labeled for Pax-5. While 27 of the 28 MZLs had at least 10% of neoplastic cells labeled for MUM-1, only 3 of 10 MCLs had at least 10% of neoplastic cells labeled for MUM-1 (Figs. 3b, 4b). Eight of 24 MZLs had at least 10% of cells label for MCL-1, whereas no MCLs labeled for MCL-1 (Figs. 5, 6). Less than 10% of neoplastic cells in all MZL and MCL cases labeled for cyclin D3. The number of cells expressing the different markers in a given punch was either very high or very low depending on the antibody and thereby made it simple to determine whether or not an individual punch was considered positive based on the described criteria. There was minimal variation in the percentage of cells expressing a specific protein among punches of the same tumor.
Number of Canine Marginal Zone Lymphomas (MZLs) and Mantle Cell Lymphomas (MCLs) With Positive Immunolabeling for the Different Antigens.
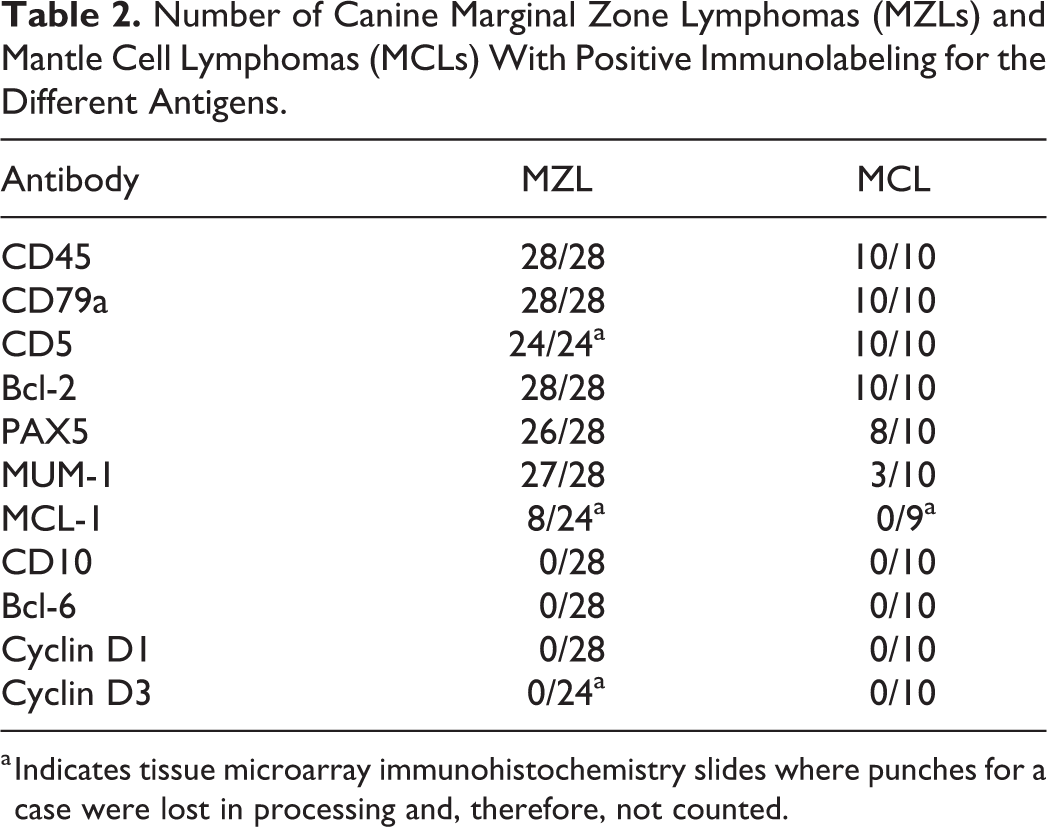
a Indicates tissue microarray immunohistochemistry slides where punches for a case were lost in processing and, therefore, not counted.
Discussion
Canine splenic MCL and MZL have distinct immunophenotypes. Neoplastic mantle cells are positive for CD20, CD79a, CD45, CD5, Bcl-2, and PAX-5; occasionally positive for MUM-1; and negative for CD3, CD10, Bcl-6, cyclin D1, cyclin D3, and MCL-1. Neoplastic marginal zone cells are positive for CD20, CD79a, CD45, CD5, Bcl-2, PAX-5, and MUM-1; one-third are positive for MCL-1; and all are negative for CD3, CD10, Bcl-6, cyclin D1, and cyclin D3. The key differences in between MCL and MZL are their immunoreactivity patterns for MUM-1 and MCL-1. Unfortunately, SOX-11 could not be validated in canine tissue; therefore, it was not evaluated in either lymphoma entity. It is important to recognize that the reported expression patterns are entirely descriptive, and functionality of the detected protein target has not been investigated in this study.
In conjunction with histopathology, an immunohistochemical panel using the reported markers could prove beneficial in accurately diagnosing canine splenic MZL and MCL. The identified immunophenotypes could assist in differentiating these entities from other lymphoma entities that have different reported immunophenotypes. A routine panel composed of CD3 and CD20 for general phenotyping in combination with BCL-2 and Bcl-6 to differentiate follicular lymphomas and MUM-1 and MCL-1 for marginal vs mantle cells is recommended for diagnostic purposes.
The observed immunophenotypes of canine splenic MZL and MCL are similar to their human counterparts, with a few exceptions. Both human and canine MCLs are positive for CD45, CD20, CD79a, CD5, Bcl-2, and PAX-5; variably positive for MUM-1; and largely negative for CD3, Bcl-6, CD10, and MCL-1. Human and canine MZLs are positive for CD45, CD20, CD79a, Bcl-2, PAX-5, and MUM-1; variably positive for MCL-1; and negative for CD3, Bcl-6, CD10, cyclin D1, and cyclin D3. The differences between the human and canine entities revealed in this study are that canine MCLs are negative for both cyclin D1 and D3, and canine MCL and MZL both express CD5. Interestingly, 30% of the MCLs in this study were positive for MUM-1, which is consistent with previously published results of MUM-1 expression in 30% to 35% of human MCLs. 23 Mantle cell development does not involve MUM-1 induction, but expression in both human and canine MCL cases may represent an earlier pregerminal or germinal transition state, since MUM-1 has been shown to be expressed during early B-cell development. 32 The close resemblance of MUM-1 expression patterns supports consistency between the entities in both species.
Bcl-2 inhibitors have been evaluated in humans to target lymphoid malignancies such as follicular lymphoma, mantle cell lymphoma, diffuse large B-cell lymphoma, and some T-cell lymphomas. 2 Lack of MCL-1 expression has been shown to make neoplastic cells more susceptible to Bcl-2 inhibitors. 1 Human MCLs often have minimal to no expression of MCL-1, which is thought to correlate with their promising response to Bcl-2 inhibitors in some studies. 48 Interestingly, all of our canine MCLs also had no expression of MCL-1. We are unaware of the current use of Bcl-2 inhibitors to treat canine indolent lymphomas, but our results may warrant investigation, including additional validation of Bcl-2 expression in these canine tumors.
The primary immunophenotypical difference between human and canine splenic MCL is the expression of cyclin D1 in most human MCLs, while none of the canine MCLs were found to be positive for cyclin D1. Cyclin D1 is overexpressed in most human MCLs due to a translocation, but no such translocation has been identified in dogs. 31 Subsets of human MCLs that are cyclin D1 negative alternatively overexpress cyclin D2 or cyclin D3. 20 These genes also function as regulators of CDK kinases and have been found to be expressed at high levels in some of the cyclin D1–negative MCL cases. 24 Cyclin D1–negative MCL is clinically and morphologically identical to cyclin D1–positive MCL, but it fails to exhibit chromosomal translocations and is thought to be caused by epigenetic mechanisms. 20 Investigation of cyclin D3 in this study revealed weakly positive expression in all MCL and MZL cases with less than 10% of neoplastic cells in all lymphomas being immunopositive. However, positive expression of cyclin D1 and cyclin D3 in human MCL has been defined as strong nuclear labeling in more than 50% of neoplastic cells. 18 For this reason, the lack of expression observed in the lymphomas in our study is not surprising. The lack of cross-species immunoreactivity was excluded as a cause of weak labeling since the positive controls demonstrated strong nuclear labeling in normal canine spleen and lymph nodes. Regardless, the overexpression of cyclin D isoforms most likely does not represent the primary driver in MCL oncogenesis, and murine models have failed to demonstrate that cyclin D1 overexpression alone can induce lymphoma, and mice in these studies required additional genetic alterations to develop MCL. 4,31,47 While the morphology and in large parts the immunophenotype of MCL mirror each other between dogs and humans, there are distinct genetic differences in MCL between both species.
Sox-11 is also used in humans to detect MCL and to identify MCLs of the cyclin D1–negative subtype. 38 Cyclin D1–negative MCLs that express Sox-11 follow a more indolent disease course that carries a better prognosis. 18 We were only able to evaluate 2 clones of antibodies to Sox-11: MRQ-58, a mouse monoclonal antibody, and ORB101332, a rabbit polyclonal antibody. We were unable to label cells in normal canine or nonhuman primate control tissues.
The primary immunophenotypical difference between human and canine MZL is the expression of CD5 in all canine cases, while CD5 is less commonly expressed in human splenic MZL. 51 Eleven percent to 35% of human splenic MZLs have been shown to weakly express CD5. 33 Interestingly, this subset of CD5-positive splenic MZLs is thought to have a more aggressive clinical course. 22 The significance of CD5 immunoreactivity in our canine splenic MZL cases is unclear.
There were several limitations to this study. The overall number of reviewed MCLs was low, which was expected based on their infrequent occurrence in dogs. While generating a tissue array allowed us to screen a larger number of cases with an extensive antibody panel under identical analytic conditions, the nodular architecture of the studied lymphomas was not ideal for array selection. By selecting 5 punches from every donor block, we minimized variations within individual cases. However, the large number of serial sections resulted in loss of a few cases in some sections, as indicated in Table 2. We were also unable to evaluate all the antigens that can be applied to phenotype human MZL and MCL. Numerous antibodies are not available for use in formalin-fixed, paraffin-embedded canine tissues such as Bcl-10, immunoglobulin superfamily receptor translocation-associated 1 (IRTA1), myeloid cell nuclear differentiation antigen (MNDA), or myeloid differentiation primary response gene 88 (MYD88) that are used for the diagnosis of human MZLs or SOX-11 and CD38 that are used for the diagnosis of human MCLs. This limited which antibodies we could use in this study, and attempts to validate SOX-11 antibodies were unsuccessful. Regardless, we were able to use a large panel of antibodies that closely resembled panels commonly used for the diagnosis of human MCL and MZL and demonstrated their accuracy in canine tissue as previously reported. 16,19,35,52,57
In conclusion, the observed immunophenotype for canine MZL and MCL is similar to their human counterparts with the primary difference that human MCLs overexpress cyclin D1 due to a translocation mutation. A similar mutation has not been reported in dogs. Furthermore, canine but not human splenic MZLs generally express CD5. Following identification of B vs T cells with CD20 and CD3, a panel composed of BCL-2, Bcl-6, MUM-1, and MCL-1 combined with the histomorphological pattern can be used to accurately diagnose MZL and MCL in dogs. Consistent expression of Bcl-2 and lack of MCL-1 expression in MCL suggest potential therapeutic use of BCL-2 inhibitors, as used in human MCL, pending additional validation studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
