Abstract
Accidental intra-airway exposure of dogs with pure oleic acid produced bronchiolitis obliterans and bronchopneumonia. Pulmonary changes included multifocal to coalescing necrosis of bronchioles and adjacent alveoli, hemorrhage, inflammation, and exudation of fibrin. Hyperplasia of bronchiolar and alveolar epithelial cells and proliferation of loose fibrovascular connective tissue formed polyps or plugs of variable size and shape. Polyps in the airways primarily consisted of fibroblasts with loose or myxoid stroma and were variably covered with attenuated epithelial cells. Some polyps had prominent vasculature, mixed inflammatory cell infiltration, and/or necrosis. Polyps or plugs variably effaced bronchioles and adjacent alveoli. The changes closely resembled human bronchiolitis obliterans-organizing pneumonia (BOOP). Controlled intra-airway delivery of oleic acid in dogs may be a potential animal model of obstructive pulmonary diseases such as BOOP or bronchiolitis obliterans.
Oleic acid is a monounsaturated dietary fatty acid, and its dietary supplementation is essential in some animal species (e.g., rats). 3 Ironically, oleic acid induces acute pulmonary edema in animals, including dogs, after intravenous administration. 1, 2, 6, 8, 12 Oleic acid–induced pulmonary injury is a well-described animal model of human acute adult respiratory distress syndrome (ARDS). 1, 2, 12 Oleic acid injures pulmonary vascular endothelium and damages the alveolocapillary barrier, with consequent dose-dependent increases in pulmonary vascular permeability, protein leakage, and edema. This report describes bronchiolitis obliterans and bronchopneumonia in dogs after oral dosing of pure oleic acid, likely introduced by oral gavage accident or aspiration. The pulmonary injury was morphologically similar to human bronchiolitis obliterans–organizing pneumonia (BOOP). 4, 5
Due to their poor aqueous solubility, many hydrophobic drug candidates often have low oral bioavailabilities. Oleic acid was evaluated as a potential vehicle to solubilize these water-insoluble drug candidates in beagle dogs, three males and three females per group, at doses of 1 ml/kg, 2 ml/kg, and 5 ml/kg body weight by oral gavage once daily for 28 consecutive days. Oleic acid was well tolerated at 1 ml/kg and 2 ml/kg.
Bronchiolitis obliterans and bronchopneumonia were observed in four of six dogs dosed with 5 ml/kg pure oleic acid. One affected dog was euthanatized on Day 24 due to inappetence, dehydration, and lethargy. The remaining three affected dogs survived the study period and were euthanatized on Day 29. Discoloration (e.g., slight to moderate, focal or multifocal, unilateral or bilateral), consolidation, and atelectasis of middle lung lobes were observed in four affected dogs at necropsy (Fig. 1).
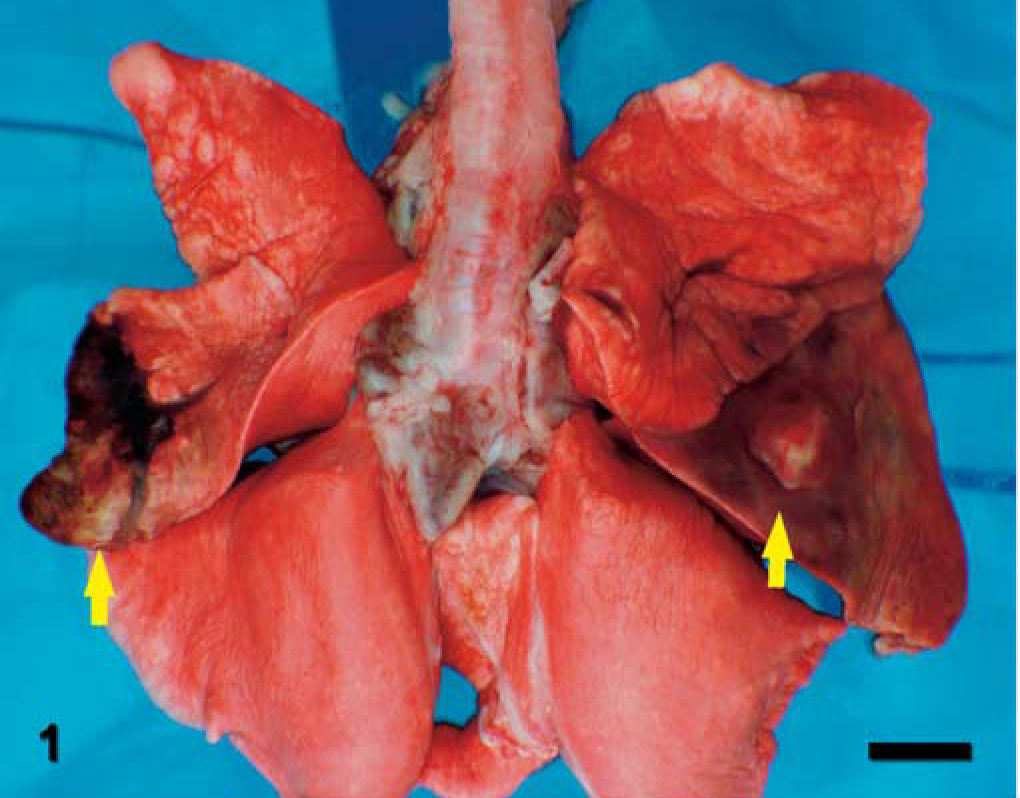
Lung; dog. Bilateral discoloration, consolidation, and atelectasis of the middle lung lobes (arrows).
Microscopic changes in affected lung lobes were primarily observed in distal bronchioles and adjacent alveoli. Affected lung lobes were characterized by multifocal to coalescing necrosis of bronchioles and adjacent alveoli with erythrocytes, mixed inflammatory cells, and fibrin exudates in bronchiolar and alveolar walls and lumens. Epithelial necrosis, erosion, or ulceration in the affected bronchioles and alveoli was accompanied by epithelial cell regeneration, fibrovascular tissue proliferation, and mixed inflammatory cell infiltration (Fig. 2). Numerous clear and sharply demarcated round or oval vacuoles of variable sizes were observed in the lumens of airways and alveoli and in the cytoplasm of epithelial cells, macrophages and, to a lesser degree, neutrophils. Ultrastructurally, intraluminal and intracytoplasmic vacuoles were nonmembranous and variably osmiophilic material (lipid) that was compatible with oleic acid (Fig. 3). Bacteria and feed debris were observed in the lung of one affected dog.
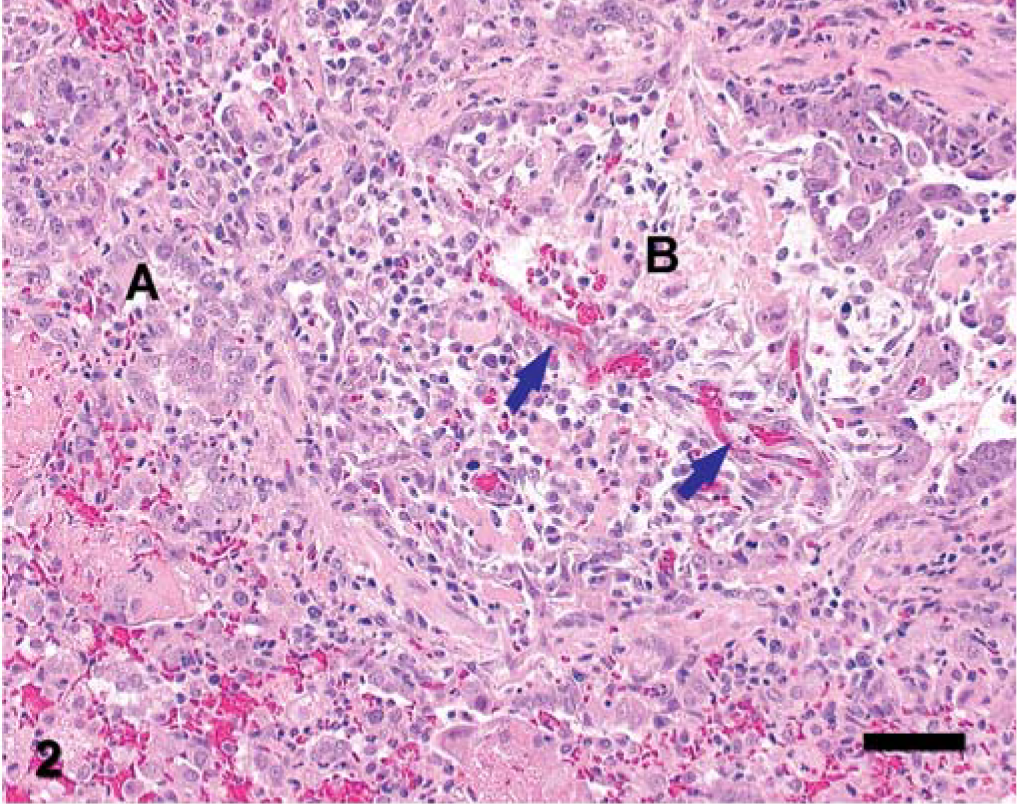
Lung; dog. Inflammation, necrosis, and epithelial cell regeneration in a bronchiole (B) and adjacent alveoli (A) were accompanied by proliferation of loose fibrovascular tissue with prominent vasculature (arrows). Note the mixture of necrotic debris and fibrin in bronchiolar and alveolar lumens. HE stain. Bar = 52 µm.
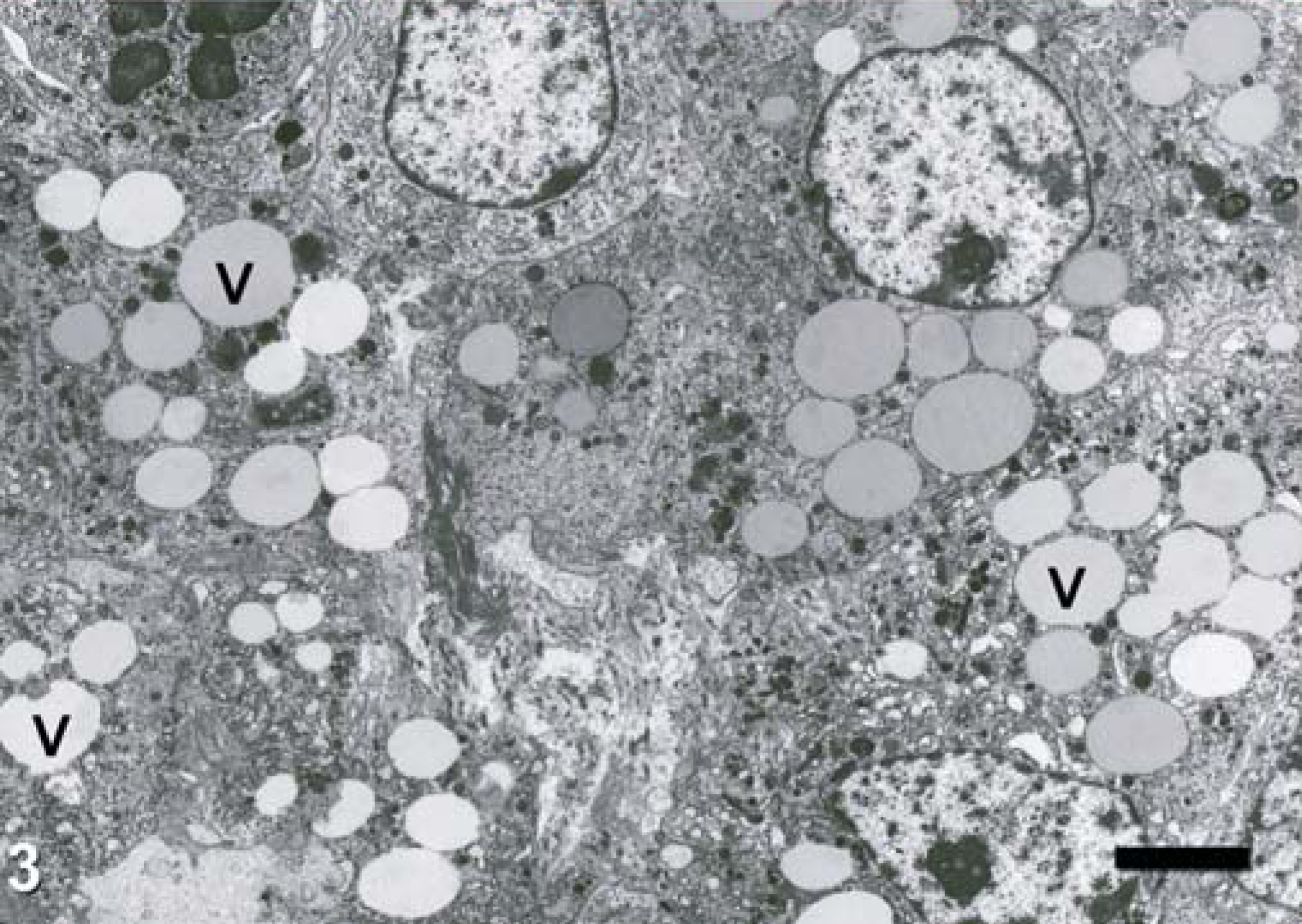
Lung; dog. Nonmembranous osmiophilic vacuoles (V) in the cytoplasm of macrophages, neutrophil, and epithelial cells are compatible with oleic acid (lipid). Lead-uranium stain. Bar = 3.2 µm.
Many distal bronchioles were filled with fibrovascular tissue polyps of variable size and shape (Fig. 4). The exudates in the adjacent alveoli formed irregular plugs. The affected bronchioles and alveoli were variably effaced or obliterated by polyps or plugs with epithelial erosion, ulceration, and regeneration. The polyps were irregularly attached to the bronchiolar wall and variably extended into adjacent alveolar lumens (Fig. 4). The polyps were primarily composed of fibroblasts with loose or myxoid stroma, and many were partially covered with attenuated epithelial cells. Prominent vasculature, necrosis, and mixed inflammatory-cell infiltration were observed in many polyps. In more mature lesions, fibrous polyps consisted of mature fibrovascular tissue cores covered with cuboidal or low columnar epithelial cells. Mixed inflammatory cell infiltration and epithelial attenuation were variably observed in some bronchi and submucosal glands.
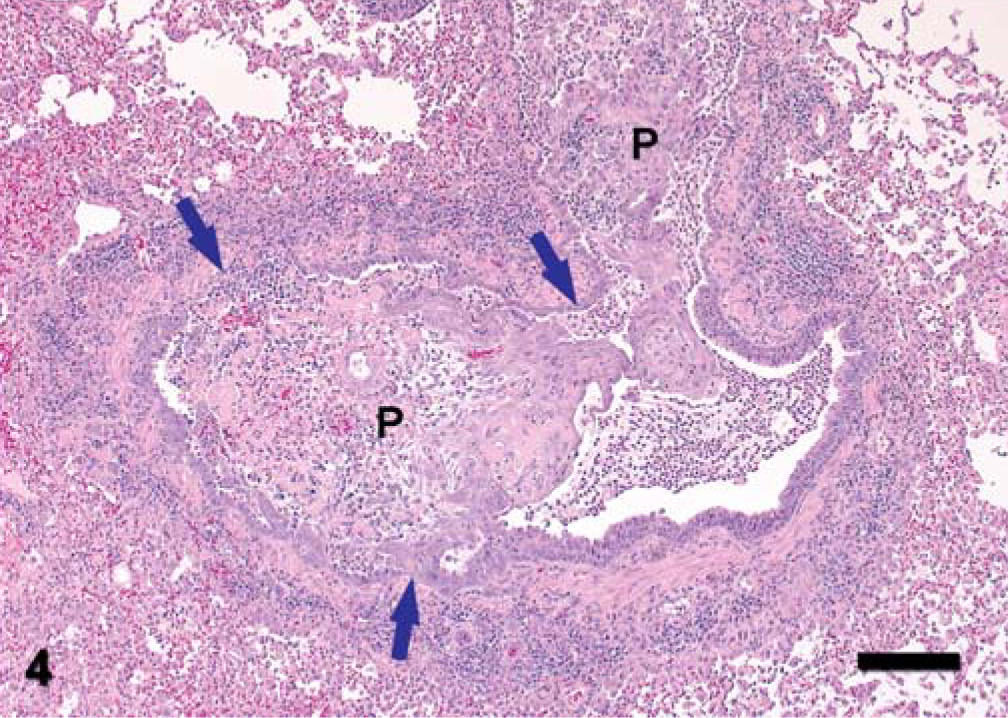
Lung; dog. Multiple fibrovascular polyps (P) of irregular size and shape in a bronchiole are accompanied by necrosis and mixed inflammatory cell infiltration. Note the bronchiolar epithelial erosion, ulceration, and epithelial hyperplasia (arrows). HE stain. Bar = 142 µm.
Oleic acid–associated bronchoalveolar lesions in dogs, as described in the present report, were morphologically similar to BOOP. 4, 5 Gross and microscopic distribution patterns of the bronchoalveolar lesions primarily in the middle lung lobe suggest a local pulmonary response to oleic acid, likely introduced by oral gavage accident or aspiration. When mixtures containing oleic acid are administrated by oral gavage to animals, care must be taken to minimize accidental pulmonary exposure. In these studies, lung lesions should be interpreted with caution because airway exposure to oleic acid can produce necrotizing and proliferative lung lesions. However, controlled intra-airway delivery of oleic acid could be explored as a potential animal model of obstructive pulmonary diseases such as BOOP or bronchiolitis obliterans.
Oleic acid can cause injury following different routes of administration. Oleic acid given intravascularly concentrates preferentially in the lung, and the pulmonary tissue is vulnerable to oleic acid–induced injury. 1, 10, 12 In animal models, single intravascular doses of pure oleic acid cause diffuse pulmonary edema that is morphologically and physiologically similar to human ARDS. 1, 2, 12 In humans, accidental oral ingestion of oleic acid products can cause respiratory distress. 10 Delayed and progressively severe onset of the respiratory syndrome in the patient suggested that the pulmonary injury was due to the systemic circulation, not direct aspiration, of oleic acid. Airway injury (e.g., hemorrhage, fibrosis) has been observed in humans and animals with oleic acid as the excipient or absorption enhancer in metered-dose inhalation. 9, 11 The bronchoalveolar lesions observed in this report were likely associated with a direct local effect of oleic acid.
ARDS and BOOP represent two distinct pulmonary responses to diverse local or systemic conditions in humans with distinct clinical, radiographic, and prognostic features. 4, 5, 7 ARDS involves diffuse injury to pulmonary capillary endothelium and/or alveolar epithelium, resulting in increased capillary permeability, protein leakage, fibrin exudation, and edema. 4 Alveolar capillary injury and repair are the key features, with formation of characteristic hyaline membranes in alveoli.
Although exact causes are unclear, BOOP is primarily an inflammatory response that affects distal airways and adjacent alveoli. Bronchiolar injury and repair are the key features, with loose fibrous or fibrovascular polyps filling both bronchioles and alveoli. 4, 5, 7 Fibrous or fibrovascular polyps in BOOP can be resolved, and apoptosis may play an important role in the resolution of polyps. 4, 5, 7 Abundant capillary vasculature in polyps may facilitate the apoptosis.
Oleic acid induces ARDS-like injury via intravenous administration and BOOP-like injury via local exposure. 1, 2, 12 Bronchioalveolar epithelial necrosis and inflammatory exudation are prerequisites for the fibrogenesis. 7 Oleic acid–associated BOOP-like injury in dogs could result from initial injury to epithelial cells and/or endothelial cells, followed by exudation and interaction among various inflammatory cells and mediators. 1, 6, 8 Hopefully, further exploration of oleic acid–induced bronchoalveolar injury in dogs could shed some light on the pathogenesis of BOOP.
