Abstract
Canine tonsillar polyps are uncommon. We describe 14 tonsillar polyps in dogs and review their classification and pathogenesis. All dogs were adult (3–13 years old). Females (10/14) were more affected than males (4/14). Most of the lesions were asymptomatic (10/14). All lesions were unilateral, pedunculated (9/14), or sessile (5/14), with a smooth (12/14) or papillary/verrucous surface (2/14). Histologically, polyps consisted of benign proliferation of lymphatic vessels, blood vessels, fibrous tissue, and lymphoid tissue in variable proportions, with occasional adipose tissue (4/14). According to the main stromal components, polyps were categorized as lymphangiomatous (5/14), lymphangiolipomatous (2/14), lymphangiofibromatous (2/14), angiofibromatous (1/14), angiofibrolipomatous (1/14), lymphoid (2/14), and myxomatous (1/14). As the pathogenesis of these polyps remains unclear, we propose to replace the term inflammatory tonsillar polyp by a morphological diagnosis based on the stromal characteristics of the lesions. Simple surgical excision was curative in the 9 cases with available follow-up information.
Benign proliferative tonsillar lesions are infrequent in dogs. Only 10 canine tonsillar polyps and an epithelial cyst have been reported, among 3 publications and a conference proceeding. 3,5,11,13 Histologically, most of the lesions are composed of mature fibrovascular stroma with multifocal aggregates of lymphocytes and plasma cells, and have been defined as inflammatory polyps. 11,20 Predominance of dilated lymphatic vessels within a dense fibrovascular stroma have been described only rarely. 3,13
Benign tonsillar polyps in humans are mostly considered hamartomatous growths containing vascular, connective, lymphoid, and fat tissue in variable proportions. 7,8,12 As in dogs, human tonsillar polyps are infrequent but the real incidence is unknown. 7,12 Histologically, polyps are recommended to be classified according to the most prominent stromal component and, in contrast to dogs, different histotypes have been reported including lymphangiomatous, lymphangiectatic, fibrous or fibrolipomatous, lymphoid, fibroepithelial. 2,7,12,17
The objective of this report is to describe the clinicopathological characteristics of 14 tonsillar polyps in dogs, review their classification, and compare the canine lesion to known pathogenesis in humans.
Our records included 402 canine cases with tonsillar histopathology from 1995 to 2020. From those, 14 were selected because the descriptions were consistent with tonsillar polyps. Samples had been fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin. Microscopic characteristics of each case were reexamined by 2 board-certified pathologists (JM, GAR). Vascularization, edema, and degree of inflammation were graded on a 0 to 3 scale (0, absent; 1, mild; 2, moderate; 3, marked). Vascular spaces, lymphoid, fibrous, and fat tissue were graded on a 0 to 4 scale based on percent (0, absent; 1, 1% to 25%; 2, 26% to 50%; 3, 51% to 75%; 4 >75% stroma). Clinical data and follow-up information were obtained through e-mail and telephone interviews with the referring clinicians.
Clinical and macroscopic data are shown in Table 1. Age of the dogs ranged from 3 to 13 years (mean 9.3). Females (10/14) were more affected than males (4/14). All patients presented with a unilateral, variably sized polyp emerging from the surface of palatine tonsil. Polyps were located on the right (6/14), left (4/14), or unreported (4/14). Most dogs were asymptomatic; only 4/12 (33%) cases showed clinical signs consisting of mouth bleeding (case 7), cough (case 10), retching (cases 10, 14), and dyspnea (case 13). Follow-up information was available in 9/14 (64%) cases, none of which showed local recurrence after simple surgical excision.
Clinical and Macroscopic Characteristics of Canine Tonsillar Polyps.
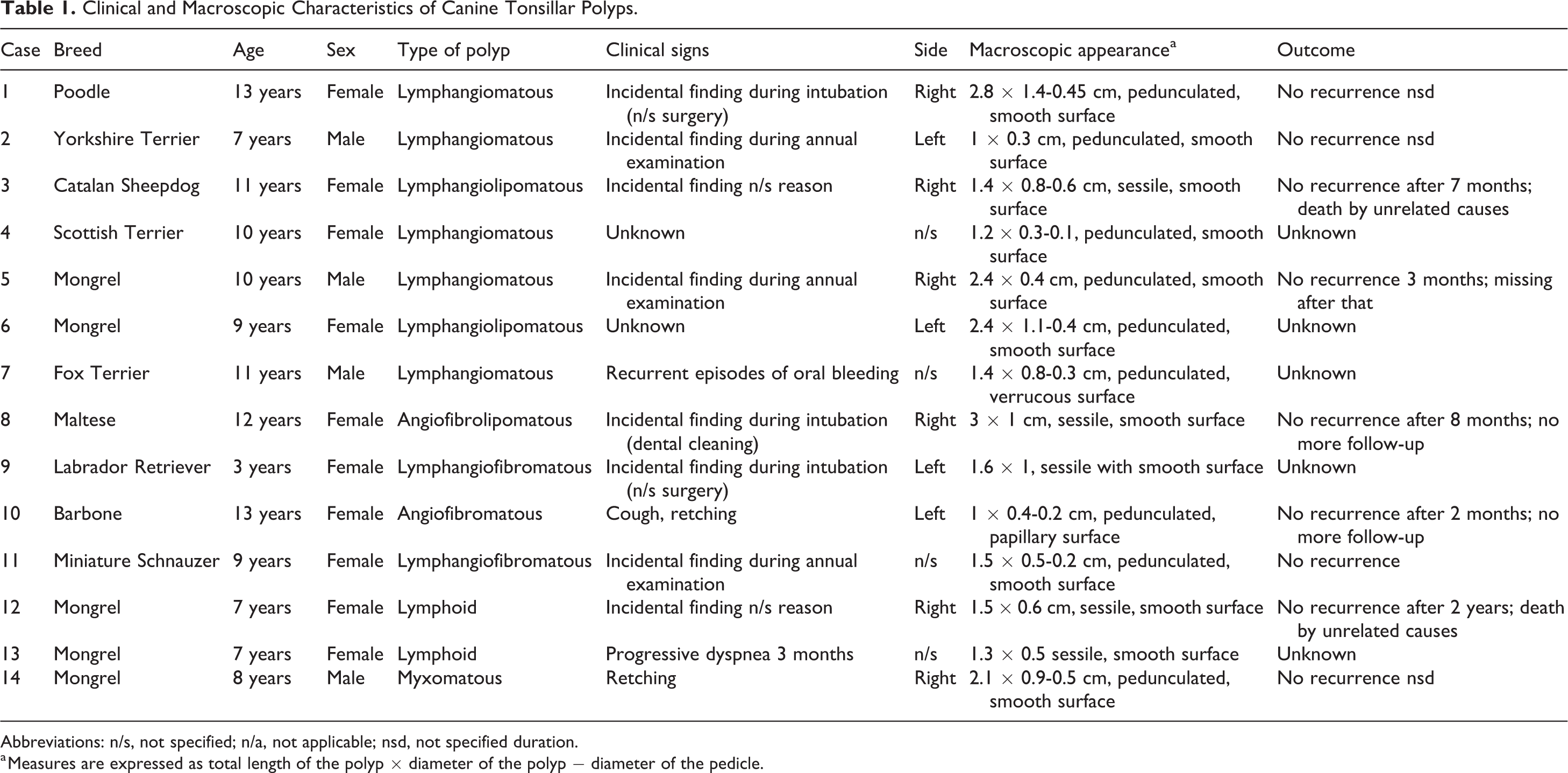
Abbreviations: n/s, not specified; n/a, not applicable; nsd, not specified duration.
a Measures are expressed as total length of the polyp × diameter of the polyp − diameter of the pedicle.
Nine of 14 (67%) polyps were pedunculated and 5/14 (36%) were sessile. The total length of the polyps ranged from 1 to 3.8 cm (mean, 1.93 cm) and the diameter from 0.3 to 1.4 cm (mean, 0.69 cm). Polyps showed a smooth (12/14), papillary (1/14), or verrucous (1/14) surface. Cut section was white to yellow and varied from soft and spongy (9/14) to firm and fibrous (4/14) to gelatinous (1/14).
Polyps were categorized into different histological subtypes according to the main constituents of their stroma. Histologic characteristics of each case are shown in Supplemental Table S1.
Seven of 14 polyps (57%) were diagnosed as lymphangiomatous. Lesions showed a highly vascularized stroma with abundant dilated endothelial-lined cavities and interconnected vascular channels, compatible with lymphatic vessels (Figs. 1, 2). Lymphatics were empty or filled by eosinophilic fluid, and were separated by hypocellular, dense fibrovascular tissue occupying <50% of the stroma (Fig. 2). In 2/14 lesions, 25% to 50% of the stroma was infiltrated by adipose tissue and were subclassified as lymphangiolipomatous (Supplemental Fig. S1). Expanded subepithelial lymphoid follicles were multifocally present (Supplemental Fig. S2). Polyps were covered by stratified squamous epithelium with mild to moderate irregular hyperplasia (Supplemental Figs. S2–S4). Multifocally, the stroma elevated the surface forming small nodular projections and, in one case, causing a verrucous appearance (Supplemental Fig. S4). Acute necrotic changes including fibrinoid necrosis of blood vessel walls and intravascular fibrin thrombi were rarely seen (case 1; Supplemental Fig. S5), without associated clinical signs.
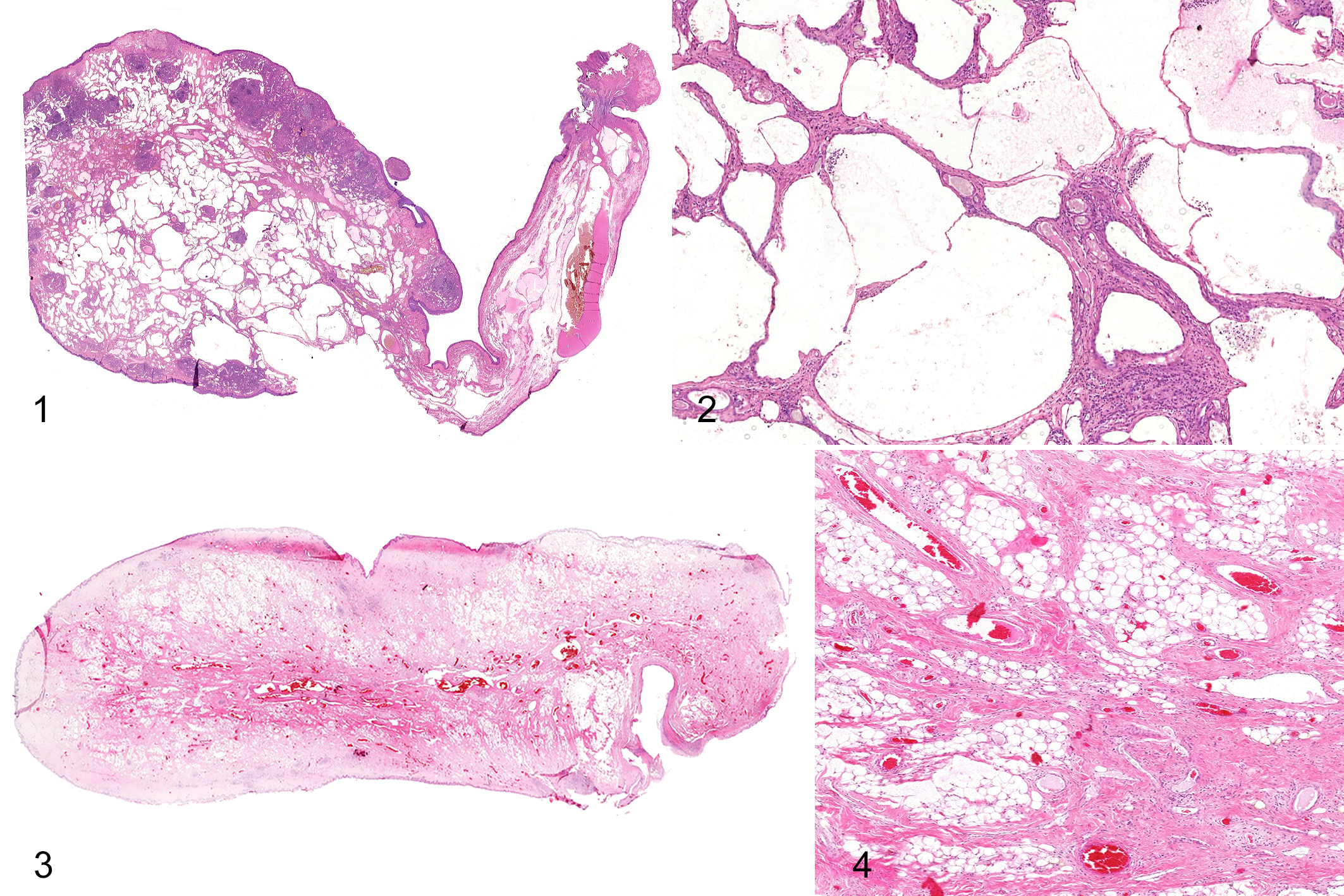
Polyps, tonsil, dog.
Four of 14 cases (29%) showed a predominantly fibrous stroma with numerous, slightly dilated, and non-anastomosing vessels. In 2 of the 4 polyps, lymphatics predominated and were categorized as lymphangiofibromatous (Supplemental Fig. S6). The other 2 of these 4 polyps showed a predominance of blood vessels and were classified as angiofibromatous (1/4) or angiofibrolipomatous (1/4) based on adipose tissue occupying more than 25% of the stroma (Figs. 3, 4).
Two of the 14 cases (14%) were classified as lymphoid polyps. More than 80% of the stroma was occupied by lymphoid tissue organized in coalescing, variable-sized follicular structures with reactive germinal centers surrounding a fibrovascular core (Figs. 5, 6). The presence of reticular epithelium transmigrated by large numbers of lymphocytes was more abundant in lymphoid polyps than in other histological subtypes (Fig. 5, inset).
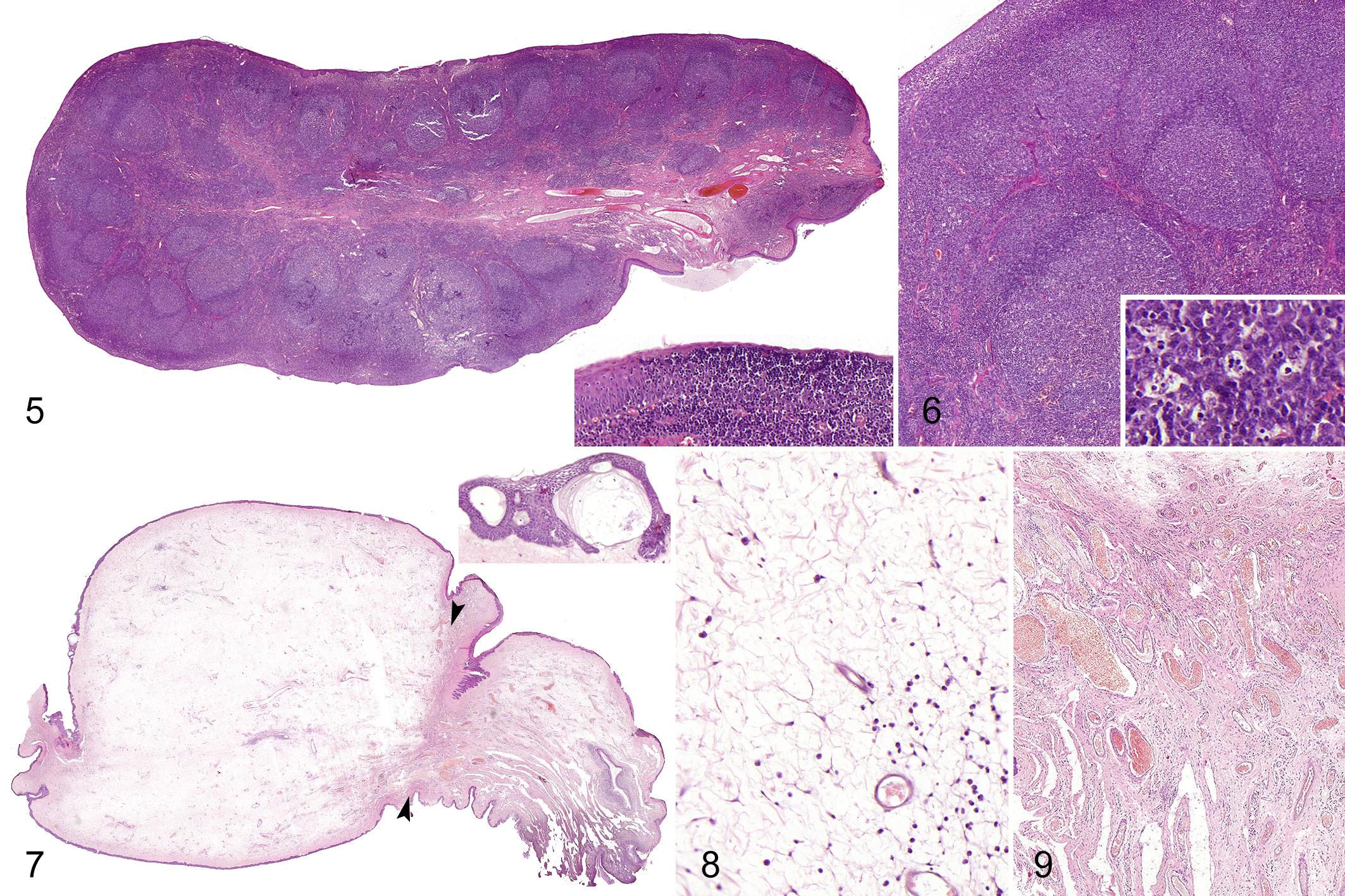
Polyps, tonsil, dog.
Finally, 1 of 14 cases (7%) was categorized as myxomatous (case 14). The stroma was occupied by severe hypocellular myxomatous edema containing few interspersed spindle cells without atypia on loose fibrovascular stroma (Figs. 7, 8). At the base of the polyp, the vascular density was increased (Fig. 9). The surface epithelium showed moderate hyperplasia, intracellular and intercellular edema, and occasional intraepithelial vesicles (Fig. 7, inset).
All polyps showed a mild to moderate inflammation with multifocal infiltrates of lymphocytes and plasma cells, with occasional presence of neutrophils (Fig. 8 and Supplemental Figs. S1, S6).
Ten cases of canine tonsillar polyps were previously reported 3,11,13,14 and are described herein. Although tonsillar polyps mainly affect adult dogs, they can affect dogs as young as 3 years (case 9). 3,11,13 Human lymphangiomatous polyps are more frequent in young adults, while lymphoid polyps commonly affect children. 2,7,8,12 No sex predisposition has been reported for canine or human tonsillar polyps, except for the lymphoid subtype which is more common in males. 2,7,8,11,12 In dogs, females were more affected than males but the overall numbers were too few to determine a sex predilection. All tonsillar polyps reported in dogs were unilateral without side predilection. 3,11,13 In humans, most polyps are unilateral but bilateral involvement has been rarely observed. 6 –8,12 Canine tonsillar polyps are mostly asymptomatic. Only 8/22 (36%) dogs, including the 12 with clinical information in this series, showed clinical signs of lethargy, chronic dyspnea, coughing, gagging, retching, or episodes of oral bleeding. 3,11,13 Human tonsillar polyps might be asymptomatic or associated with dysphagia, dyspnea, foreign body sensation, sore throat, tonsillitis, and cough, depending on the size of the lesion. 7,8,12
Canine and human tonsillar polyps can present as pedunculated or sessile masses with smooth or papillary surface. 3,7,8,11 –13 Canine tonsillar polyps in this series were mostly pedunculated with smooth surface. This contrast with observations from the previously reported case series in which only 3/8 polyps were pedunculated. 11
As in humans, the stroma of canine tonsillar polyps was composed by variable proportions of lymphatic and blood vessels, fibrous tissue, lymphoid tissue, and occasionally fat tissue, leading to different histomorphology that allow their classification into different histological subtypes. 2,7,8,12
Lymphangiomatous polyps were the most common histological subtype in this case series. They show identical features to those previously reported in an adult dog and in humans. 7,13 These type of polyps have also been referred as tonsillar lymphangiomas by human pathologists. 12 However, tonsillar lymphangiomatous polyps have smaller lymphatic spaces and more fibrous and lymphoid stromal elements than lymphangiomas found elsewhere. 12 Canine lymphangiomas usually affect the skin of young dogs, and involvement of internal tissues is rare and mostly associated to a lymphangiomatosis syndrome with systemic involvement. 16
The histomorphology of angiofibromatous polyps in our series is similar to those described by Lucke et al 11 as canine tonsillar inflammatory polyps, and to the stroma of canine and feline nasopharyngeal and middle ear polyps. 9,15 In contrast to tonsillar polyps, nasopharyngeal and middle ear polyps are usually partially covered by pseudostratified ciliated columnar epithelium and the stromal core and peduncle are less vascularized. 9,15 The main differential diagnosis considered for angiofibromatous and angiofibrolipomatous polyps was angiofibroma. Due to the lack of infiltrative growth, the stromal fat infiltration, and the lower cellularity in these polyps when compared to canine nasal angiofibromas, this diagnosis was excluded. 4
One polyp was classified as myxomatous. To the best of our knowledge, such histological appearance has not been described in human or canine tonsillar polyps. The highly edematous appearance of this polyp might be secondary to inflammation, as suggested for similar stromal changes in nasopharyngeal and laryngeal canine and feline polyps. 9,18,19
The pathogenesis of benign tonsillar polyps is unclear. While an inflammatory origin has been suggested for canine lesions, the most accepted theory in human pathology considers these polyps as hamartomas. 2,7,8,11,12,20 This is further supported by the evidence of a disorganized distribution of fibronectin and collagens I and III within the stroma of tonsillar polyps regardless of their histomorphology. 1 Therefore, tonsillar hamartomas could present a variable histologic spectrum including polyps with fibrous/fibrovascular, lymphangiomatous, lymphoid, and/or, more rarely, lipomatous appearances. 2,7,8,12 A second theory involves chronic inflammatory hyperplasia with irreversible lymphatic obstruction and lymphangiectasia, eventually causing mucosal prolapse and formation of a polyp. 8,12 Evidence against this explanation is that chronic tonsillitis occurs more commonly than polyps, and many patients with polyps lack a history of tonsillitis. 7,8,12 A clinical history of previous episodes of tonsillitis was not indicated in any of our cases or those previously reported. 3,11,13 Partial polyp torsion with subsequent lymphangiectasia and acute inflammatory and necrotic changes is possible in lesions with a long peduncle, as observed in our series (case 1).
As in humans, the pathogenesis of canine tonsillar polyps may involve a multistep process with an initial hamartomatous growth followed by secondary inflammatory and degenerative changes. 1,12 Secondary chronic inflammation in human tonsillar polyps causes stromal remodeling and lymphoid hyperplasia, occasionally producing lymphoid polyps. 1,2,12 These are characterized by a stroma composed predominantly of lymphoid tissue (>80%) organized in follicles around a fibrovascular core, similar to that observed in cases 12 and 13. 1,2 To the best of our knowledge, tonsillar lymphoid polyps have not been previously described in veterinary medicine.
Fat tissue was present in 4 of the lesions and, when occupying >25% of the stroma, the term lipomatous was added to the morphological diagnosis. Adipose tissue infiltration within some polyps probably reflects chronic degenerative and metaplastic stromal changes. 7,10,14
In conclusion, canine tonsillar polyps are benign unilateral and usually asymptomatic lesions of adult dogs, with excellent response to surgical resection. They are composed of lymphatics, blood vessels, fibrous, lymphoid, and fat tissue in variable proportions, producing different histomorphology. As in humans, a hamartomatous origin with superimposed inflammatory changes may be considered. We propose replacing the term inflammatory tonsillar polyp with morphological diagnoses based on the stromal characteristics.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Molin_et_al - Canine Tonsillar Polyps: Characteristics, Classification, and Review of the Pathogenesis
Supplemental Material, Combined_supplemental_materials-Molin_et_al for Canine Tonsillar Polyps: Characteristics, Classification, and Review of the Pathogenesis by Jéssica Molín, Miquel Vilafranca, Alejandro Suárez-Bonnet, Jaume Altimira and Gustavo A. Ramírez in Veterinary Pathology
Footnotes
Acknowledgements
Ms Teresa Giró, from the Department of Animal Sciences (UdL), and Dr Maria Santacana, from the Biomedical Research Institute of Lleida (IRBLleida), are gratefully acknowledged for technical assistance. We are grateful to the referring clinicians for their cooperation in this study.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
