Abstract
A 14-y-old Miniature Pinscher bitch was admitted to a veterinary clinic because of inappetence and a distended abdomen; ultrasound examination revealed a fluid-filled uterus with a single 1-cm hyperechoic nodule in its lumen. Ovariohysterectomy was performed. Grossly, the uterine horns were distended irregularly and asymmetrically, and the uterine lumen contained 200–300 mL of brown watery fluid. A single white polypoid 0.9-cm diameter nodule was present at the site of the distended uterine horn and arose from the endometrium with a narrow stalk. Histologically, the polyp consisted of dense, smooth muscle fascicles admixed with glandular components; its surface was covered by simple cuboidal epithelium with areas of squamous metaplasia. The myomatous cells expressed the myogenic markers smooth muscle actin and desmin. We interpreted the mass as an adenomyomatous uterine polyp, which is a rare variant of an endometrial polyp.
An adenomyomatous polyp (AP) has not been reported in veterinary medicine, to our knowledge, and is a rare lesion in women. The gross appearance of an AP can be similar to a conventional endometrial polyp (EP), but the histologic features of the 2 types of polyps are different. EPs are focal sessile or pedunculated hyperplastic nodules composed of endometrial stroma and glands,1,9 and in veterinary medicine, they have been reported in various animals, including bitches, hedgehogs, an African wild dog, goat, cats, and a horse.3–7,12,22 EPs are common in women; however, APs have been detected in <2% of previously examined polyps. 19 An AP is composed chiefly of smooth muscle cells; an EP is composed of inactive endometrial stromal cells. 18 We describe here the histologic features of an AP using immunohistochemistry as well as the associated clinical presentation in a bitch.
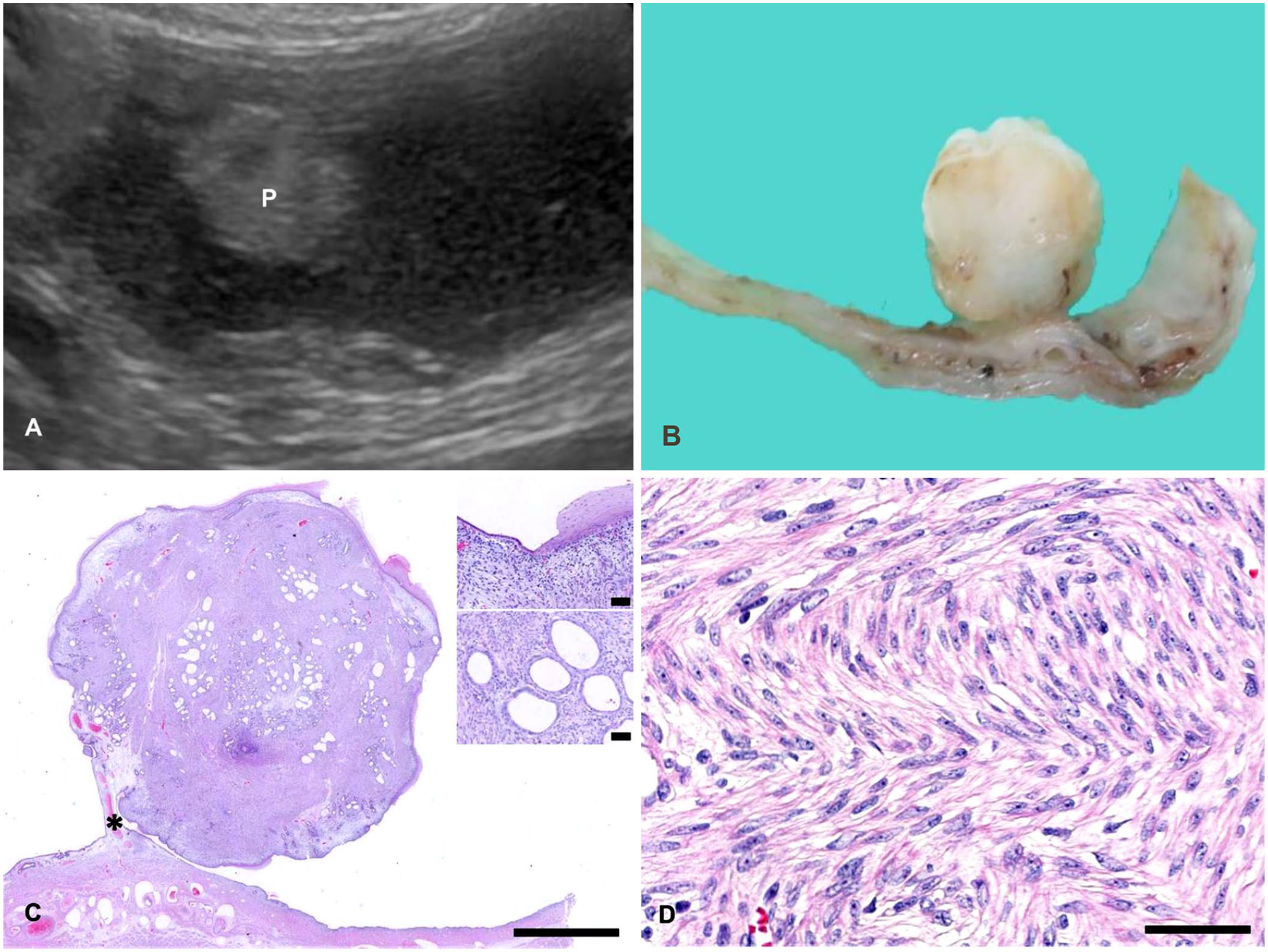
A 14-y-old nulliparous Miniature Pinscher bitch was presented to a local animal hospital with a 2–3 d history of soft stool, vomiting, and loss of appetite that progressed to anorexia. A complete blood count revealed mild leukocytosis (29×109/L; RI: 6–17×109/L); white blood cell differentials were not performed. Blood chemistry showed elevated C-reactive protein (CRP; >21 mg/L, RI: 0–0.7 mg/L), hyperproteinemia (78 g/L; RI: 50–72 g/L) related to hyperglobulinemia (46 g/L; RI 24–32 g/L), elevated hepatic enzyme activities (alanine aminotransferase 4.43 µkat/L, RI: 0.28–1.3 µkat/L; aspartate aminotransferase 0.97 µkat/L, RI: 0.28–0.73 µkat/L; alkaline phosphatase 47.5 µkat/L, RI: 0.78–4.24 µkat/L). Urea (20.2 mmol/L; RI: 3.3–10.4 mmol/L), creatinine (244 µmol/L; RI: 36–124 µmol/L), and phosphorus (3.13 mmol/L; RI: 0.61–1.61 mmol/L) concentrations were elevated; therefore, renal disease was suspected. Ultrasonographic examination revealed an enlarged, fluid-filled uterus; a 1-cm hyperechoic nodule was identified in the lumen (Fig. 1A). Although a vaginal smear was not performed, the patient was considered to be in estrus given that the owner noted a mild hemorrhagic discharge. The patient also had an oronasal fistula at the left upper canine tooth and several mammary masses. An ovariohysterectomy was performed, and uterine content was submitted for bacterial culture with no growth. Formalin-fixed ovaries and uterus were submitted for histologic examination.
Adenomyomatous uterine polyp in a Miniature Pinscher.
Grossly, the uterine horns were 20 cm in length, with diameter up to 7 cm. The uterine horns had several distended segments and contained 200–300 mL of dark-brown watery fluid (hydrometra). A 4–5-cm diameter dilated area contained a single, round, white, 0.9-cm diameter polypoid mass, attached to the endometrium with a short narrow stalk. This polypoid mass was located 4–5 cm distant from the uterine body. The polyp was firm, pedunculated, and had a slightly irregular but smooth and shiny surface; the cut surface was white (Fig. 1B).
Formalin-fixed paraffin-embedded sections of the uterine mass and ovaries were stained with H&E and Masson trichrome stains. Immunohistochemistry (IHC) was performed using the following antibodies: pancytokeratin AE1/AE3 (AE1/AE3; Dako); vimentin (Dako); desmin (clone D33; Nichirei Bioscience), and smooth muscle actin (SMA; clone 1A4, Dako). Heat-induced antigen retrieval was performed by incubation with citrate buffer, pH 6. All slides were immersed with 0.3% H2O2 in methanol solution for 30 min for endogenous peroxidase blocking and in Blocking One solution (Nacalai Tesque) for 30 min to block nonspecific binding. The slides were incubated with a primary antibody for 1 h at 37°C in a humidified chamber. The secondary antibody was the Histofine simple stain MAX-PO (Nichirei Bioscience). The 3,3′-diaminobenzidine substrate (MilliporeSigma) and hematoxylin were used to visualize IHC stains.
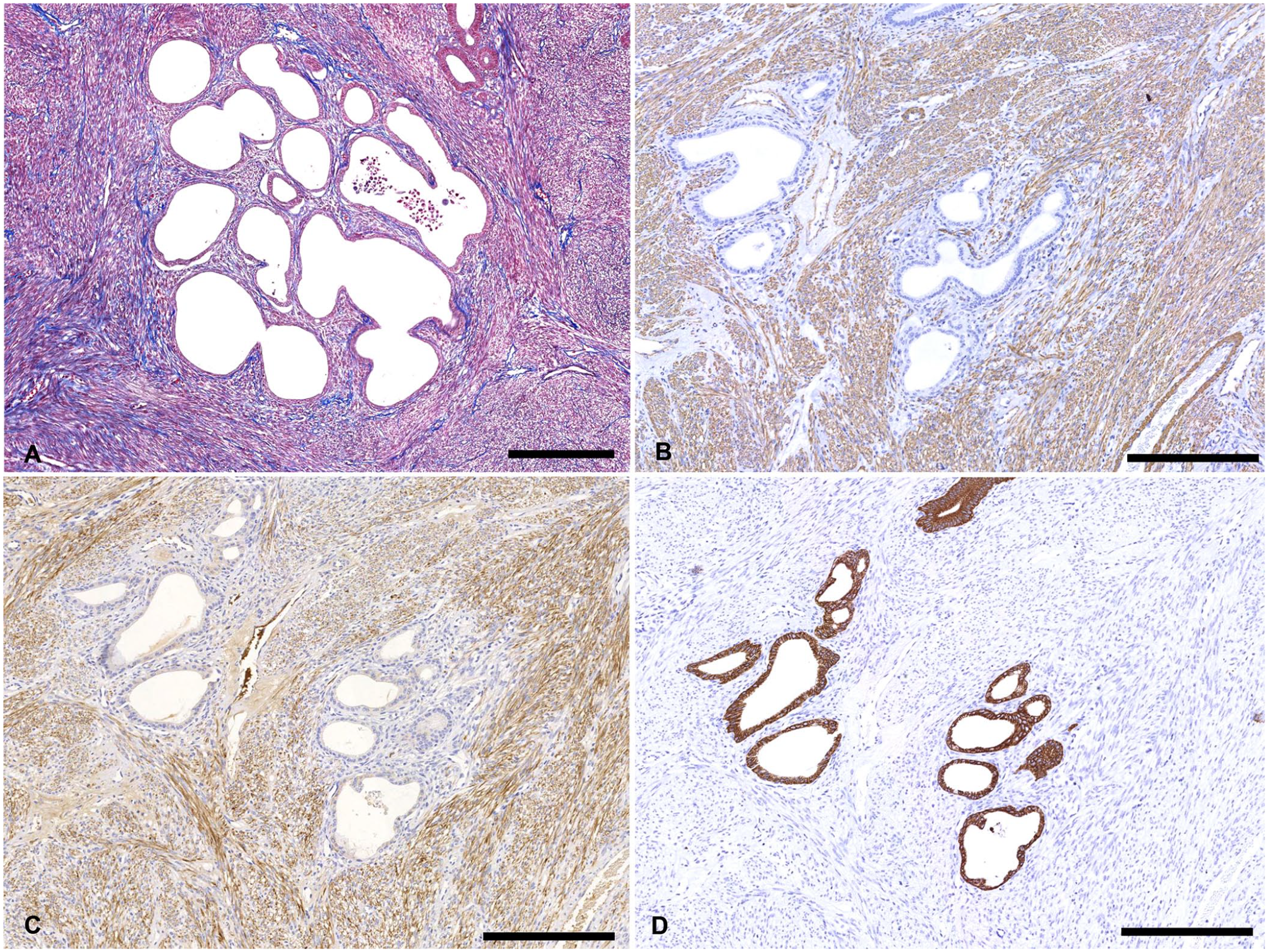
Histologic examination revealed that the polyp arose from the endometrium (Fig. 1C) and was covered by simple cuboidal epithelium with wide areas of squamous metaplasia and hyperkeratosis (Fig. 1C, inset, upper). The epithelium that covered the polyp was continuous with the normal endometrial epithelium. The polyp was composed predominantly of dense bundles of long spindle-shaped cells admixed with glandular components (Fig. 1C, inset, lower). The spindle cells were in interwoven fascicles with a frequent herringbone proliferative pattern. The spindle cells have moderate amounts of eosinophilic cytoplasm, elongated “cigar-shaped” nuclei, clumped chromatin, and prominent nucleoli, with indistinct cell borders (Fig. 1D). With Masson trichrome staining, spindle cells exhibited prominent red staining with red to dark-red nuclei; the collagenous material stained blue (Fig. 2A). Spindle cells were not present in the stalk. The glandular components were variable in size and shape but relatively small. Rare glands were irregularly dilated and filled with proteinaceous eosinophilic material. Most of the glands were lined by simple cuboidal-to-columnar epithelium. At the periphery of the polyp and around some glands were loosely arranged spindle-to-stellate cells. The spindle cells and glandular components were monotonous and showed no cytologic atypia; no mitotic figures were evident. Small blood vessels were distributed throughout the polyp; thick-walled blood vessels existed prominently at the base of the stalk (Fig. 1C, asterisk). Similarly, large, well-developed vessels were present in the myometrium under the polyp. The endometrium of the entire uterus was severely attenuated as a result of hydrometra; cystic endometrial glands were not recognized. Both ovaries had small granulosa cell tumors, of 3-mm and 9-mm diameter.
Adenomyomatous uterine polyp in a Miniature Pinscher.
In IHC, SMA was diffusely labeled in the bundles of elongated spindle cells and vascular walls (Fig. 2B). The cytoplasm of these spindle cells showed strong dot-like positivity. Desmin was also labeled in these smooth muscle–like spindle cells (Fig. 2C). The endometrial epithelial cells and glandular epithelial cells were strongly positive for pancytokeratin AE1/AE3 (Fig. 2D). Vimentin was lightly labeled in the spindle cells but not in the glandular epithelium. In accord with reports of human APs, our findings were compatible with an AP of the uterus.
EPs are polypoid overgrowths of endometrial glands and endometrial stomal cells, and they are typically found in postmenopausal women. 18 EPs are also recognized in older animals; their prevalence is ~3% in dogs and cats, 13 although many EPs in animals may be undetected. An AP of the endometrium is a rare variant of EPs in women, and its cellular component is largely composed of smooth muscle compared to the fibrous stroma in conventional polyps.2,16,18,19 Reported histologic features of EPs in animals are of polyps with endometrial glands surrounded with variable amounts of endometrial stromal cells or fibrocollagenous stroma.3–6,12,22 There may be some SMA-positive fibers within the fibrous stroma; these smooth muscle fibers had been interpreted as a metaplastic change. 13 Our case is different from those of previously reported EPs in animals, given that the polyp was composed predominantly of smooth muscle cells that were clearly visualized by IHC for SMA and desmin. The features were analogous to human APs.
Clinical signs of APs in humans range from asymptomatic to symptomatic; vaginal bleeding is the most common sign. 17 In our case, the bitch had nonspecific clinical signs, such as lethargy and inappetence, which may have been related to its renal disease, oronasal fistula, and/or abdominal enlargement that was the result of hydrometra. The owner recognized a mild bloody discharge from the vulva, which was considered a sign of estrus. It is difficult to determine if this minor bleeding was the result of estrus or related to the polyp, but no obvious sign of abnormal discharge was recognized at the clinic.
A study of 21 canine EPs demonstrated that >50% of bitches had pyometra, indicating that pyometra was the major critical complication. 13 EPs have also been associated with the cystic endometrial hyperplasia (CEH)-pyometra complex. 8 In polyp cases, CEH occurs in 50% of dogs and 70% of cats. 7 The pathogenesis of polyp formation in CEH-pyometra complex in animals is unknown; small aggregates of cystic endometrial glands may stimulate interstitial fibrous connective tissue deposition, which may progress to form a polyp. 20 In our case, neither CEH nor endometritis were recognized. Instead, hydrometra was observed, and fluid accumulation in the uterus could stimulate endometrial stromal cells. Although hydrometra has been reported in a young cat with EPs, this condition was not recognized as a concurrent condition in the retrospective study of 21 canine EP cases.11,13
The etiopathogenesis of EPs is not completely understood in women. One hypothesis of EP formation is hormone excess. In women, a history of hormone replacement therapy for menopause and the use of tamoxifen are recognized conditions associated with EP formation.16,21 Tamoxifen, a drug used to treat breast cancer, acts as an estrogen antagonist in breast tissue and estrogen agonist in the lower genital tract, and thus has an estrogenic effect on the myometrium. 16 Concentrations of estrogen and progesterone receptors in the glandular epithelium were significantly higher in EPs than in normal human endometrial tissue, 10 which could explain the development of EPs in a hormonally imbalanced state. In zoo animals, the use of melengestrol acetate, a progesterone-like compound used to control breeding, has been related to the development of EPs. 15 In our case, the patient also had bilateral ovarian granulosa cell tumors; ovarian granulosa cell tumors can produce excessive estrogen or progesterone. 14 Therefore, the ovarian neoplasia could induce hormonal imbalance that may have led to polyp formation and hydrometra in this bitch.
Veterinary pathologists should be aware of the existence of APs in animals. We emphasize the importance of using myogenic markers to correctly recognize APs among EPs.
Footnotes
Acknowledgements
We thank Drs. Shou Takamine and Ryousuke Namiki for providing clinical information and data.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
