Abstract
Feline primary osteosarcomas involving the skull are extremely rare. When they occur, orbit, mandible, and maxilla are the most common sites. Microscopically, scattered multinucleated giant cells (MGCs) are not an uncommon occurrence in osteosarcoma (OSC), but they are generally in low number. Only in a rare variant, the giant cell-rich OSC, are MGCs the prevalent cell type. Although osteoclast and osteoblast origin have been postulated in human and veterinary literature, the origin of MGCs in osteosarcomas is poorly understood. This report describes a giant cell-rich OSC in the calvarium of a 13-year-old spayed female shorthair cat. The animal exhibited a range of neurologic signs, including left circling, compulsive gait, lack of proprioception, and bilateral absence of menace reaction, with indication of left forebrain involvement. Gross lesions were characterized by a multilobate, spherical mass located in the left calvarium, compressing the left forebrain. Histologically, the tumor was characterized by scattered nests of MGCs separated by small bundles of pleomorphic, fusate to polygonal cells. Between spindle cells, osteoid was very sparse and arranged in thin strands. immunohistochemical stains for vimentin were positive, with no detectable cellular staining for cytokeratin, S-100 protein, or Class II major histocompatibility complex. Ultrastructurally, MGCs contained profiles of rough endoplasmic reticulum; no lysosomes were observed. The origin of MGCs in osteosarcoma remains obscure, and our results confirm their ambiguous identity.
Feline osteosarcomas (OSCs) occur most commonly in the appendicular skeleton and hind limbs appear to be affected more often than forelimbs. 1, 3, 13 Axial OSC can occur in the skull and in the pelvis. Feline tumors of bone involving the skull are extremely rare and within OSCs, only a few cases involving mostly orbit, mandible, and maxilla have been reported. 1, 12 OSC can be variable; lytic, productive, and mixed forms are recognized. Microscopic features can also vary widely; several subtypes are recognized. In all cases, a definitive diagnosis is based on the production of osteoid by neoplastic mesenchymal cells. 6 The most frequent histologic variants are osteoblastic, fibroblastic, chondroblastic, and telangiectatic. Giant cell OSC is extremely rare in veterinary and human literature. 6, 9, 13 These histologic variants do not seem to correlate with any particular prognosis. 14
A 13-year-old spayed female domestic shorthair indoor cat presented with a 2-week history of ataxia and visual deficits. Physical examination revealed diffuse and severe muscular atrophy and poorly groomed coat. Neurologic examination detected left circling, compulsive gait, transitory and short events of head pressing, lack of proprioception in the right forelimb, and bilateral absence of menace reaction with normal pupillary light reflex. The lesion was localized in the left forebrain and was suspected to be an inflammatory or neoplastic problem. Hematology and serum biochemistry demonstrated no relevant changes. Feline immunodeficiency virus antibodies and feline leukemia virus antigen were not detected in serum. Computerized tomography (CT) images showed a single space-occupying lesion (24 × 16 × 20 mm) located in the left temporal region. (Fig. 1). The spherical lesion had distinct margins. It compressed and displaced rightward the intracranial structures. Osteolysis of the temporal bone and involvement of the soft tissue were also seen. CT precontrast density was 62.4 ± 10.8 Hounsfield Units (Hu) and the administration of intravenous contrast medium (iodixanol, 640 mg/kg, Visipaque 320®) produced a strong enhancement (postcontrast density 88.2 ± 9.14 HU) with nonuniform pattern. The anatomic site and the CT features of the lesion were consistent with an extra-axial tumor. The owner refused any surgical procedure and asked for the cat to be euthanatized. Removal of the brain and the mass was allowed.
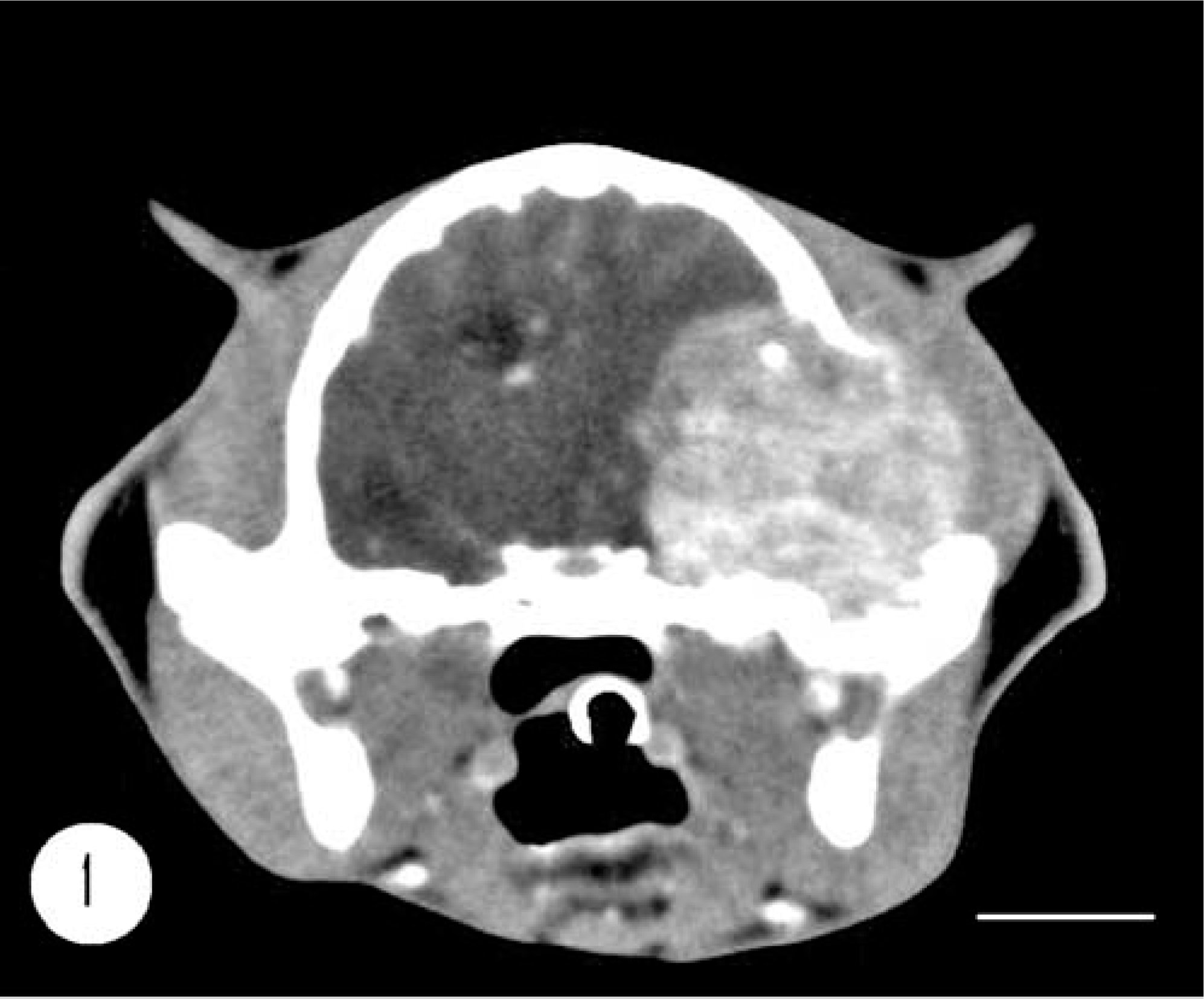
Cranium; feline. CT scan of cranium shows a single space-occupying lesion located in the left temporal region and characterized by spherical shape and distinct margins. The lesion compresses and displaces the intracranial structures. Bar = 1 cm.
Gross examination revealed the left parietal lobe and the rostral part of the left occipital lobe to be compressed by a firm, expansive, multilobate, spherical, white and tan mass measuring 1.5 × 2 cm (Fig. 2). The mass was attached to the calvarium and partially covered by dura mater, which appeared as a fine, whitish, fibrous membrane. The mass was fibrous and firm, with a few gritty areas; the cut surface was yellow, white, and tan, with few whitish areas of mineralization (0.1–0.2 cm). On the surface of the compressed part of the left hemisphere, the meningeal vascular structures appeared prominent because of severe hyperemia. In coronal section, the brain was characterized by a severe asymmetry of the cerebral hemispheres involving most of the left diencephalic and prosencephalic structures. Tissue samples from the brain and the mass were fixed in 10% neutral buffered formalin, embedded in paraffin, and sectioned at 5 or 7 µm according to standard procedure for histopathologic evaluation. All sections were stained with hematoxylin and eosin, Masson trichrome, and tartrase-resistant acid phosphatase (TRAP) stains. Immunohistochemical staining technique was performed with the antisera S-100 (DAKO, polyclonal rabbit anti-S100, code No. Z0311), vimentin (DAKO, monoclonal mouse anti-vimentin, clone V9), cytokeratin (DAKO, monoclonal mouse anti-human cytokeratin and AE1/AE3), and Class II major histocompatibility complex (MHC II, DAKO, monoclonal mouse anti–HLA-DR, clone TAL.1B). The EnVision system (DAKO EnVision™, peroxidase, mouse) was used for amplification. Selected tissue was also processed routinely for transmission electron microscopy.

Cranium; feline. Gross morphology of the lesion visualized in Fig. 1. A multilobate, spherical, white-tan mass, partially covered by dura mater. Bar = 1 cm.
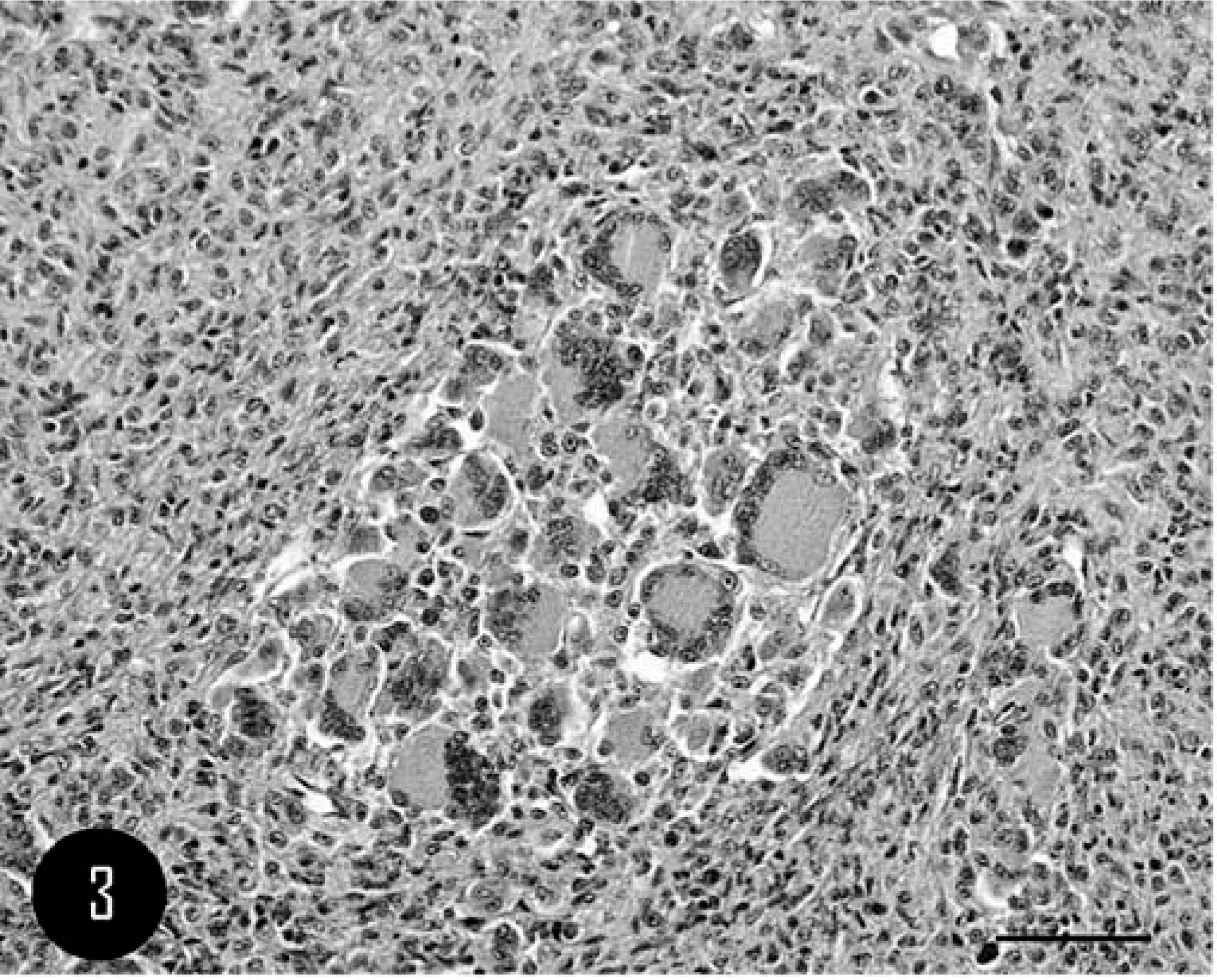
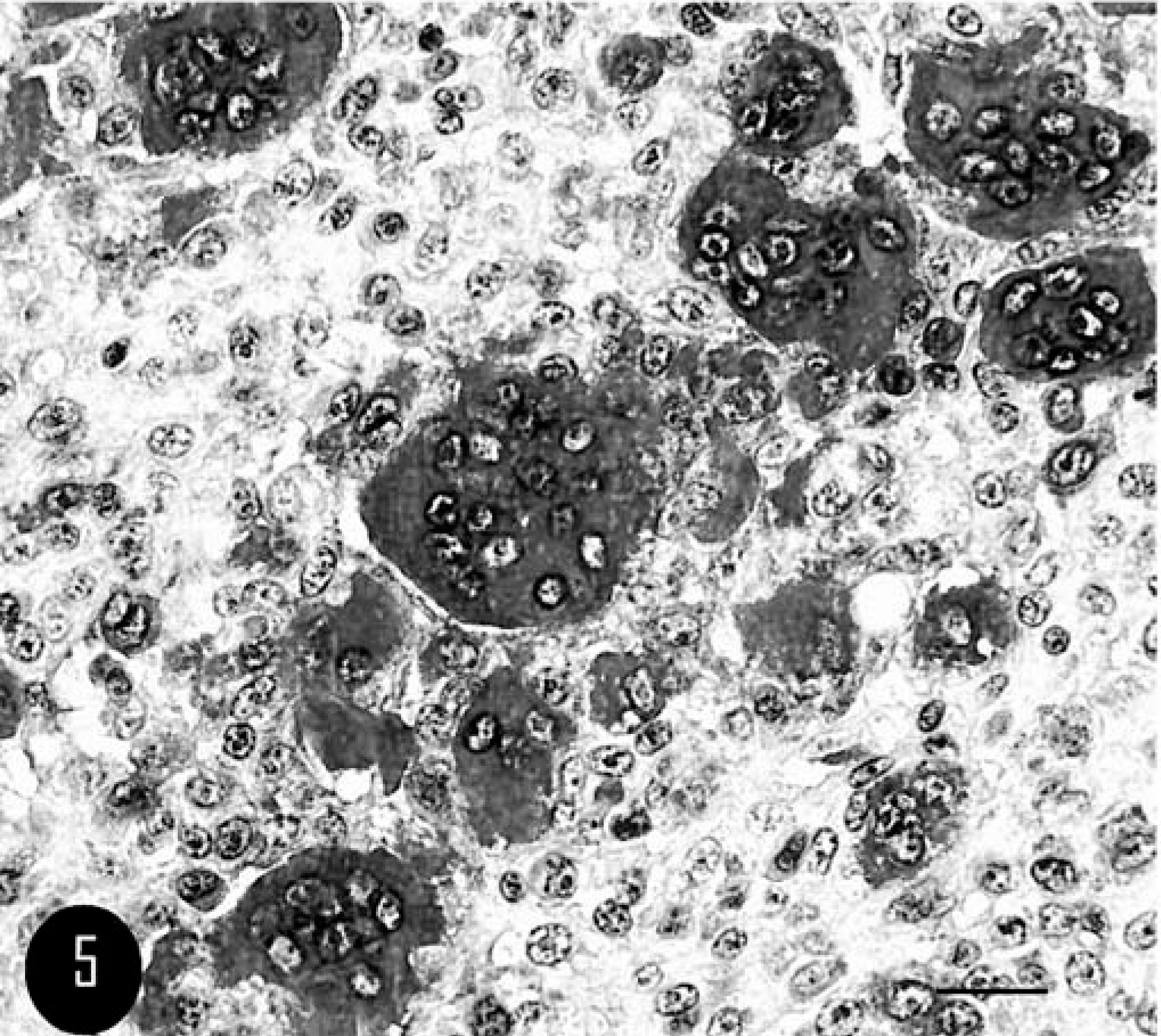
Microscopically, the mass was characterized by moderate cell density and composed of numerous scattered nests of multinucleated giant cells (MGCs); the nests were separated by small bundles of pleomorphic cells, from fusate to polygonal in shape, with marked variation in morphology and size (Fig. 3). Distinct MGC nests were composed of 20 to 10 MGCs characterized by an irregular polygonal shape, distinct cell borders, and abundant homogeneous eosinophilic cytoplasm. The majority of nuclei were oval to round, mostly in an eccentric position, and contained a vesicular pattern of chromatin dispersion; a single, irregular, centrical to paracentrical nucleolus was usually present (Fig. 4).
Calvarium; feline. Nests of multinucleated giant cells (MGC) surrounded by mononucleated polygonal cells. HE. Bar = 100 µm.
Calvarium; feline. MGCs characterized by polygonal shape with distinct cell borders and abundant homogeneous eosinophilic cytoplasm and many oval to round nuclei. HE. Bar = 20 µm.
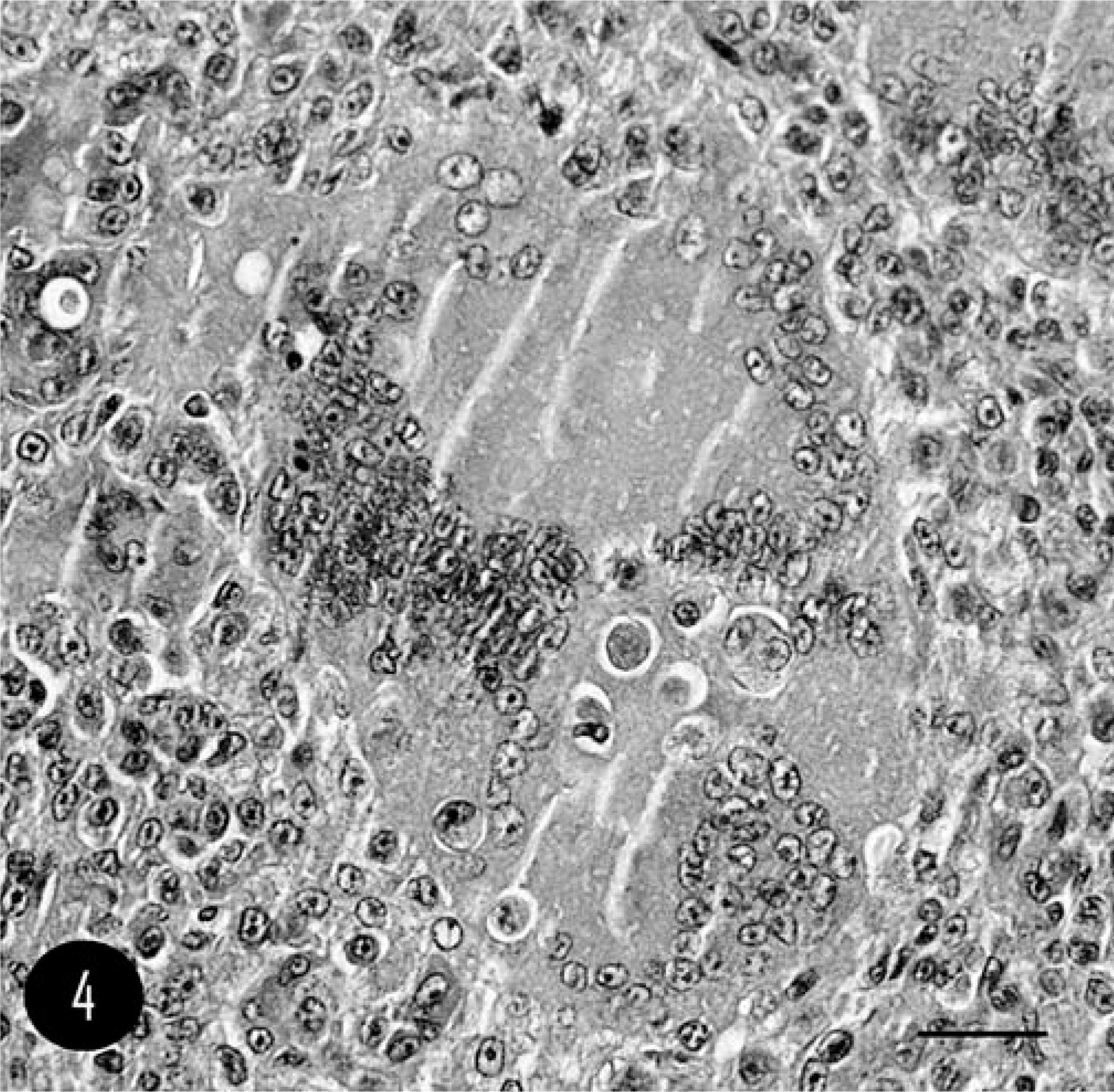
The remaining pleomorphic cell population was composed of both oval to round polygonal cells, with cytoplasm and nuclear features similar to MGCs, and spindle-shaped cells resembling fibroblasts. Osteoid was sparse and arranged in thin strands mixed with spindle cells. Multifocal islands of cartilage differentiation and mineralized foci of osteoid were present. In the periphery of the mass, irregular trabeculae of woven bone were also present. Microscopic changes in the compressed part of the brain included mild, and multifocal lymphoplasmacellular infiltration in the leptomeninges. The cerebral cortex presented mild, diffuse glial proliferation and moderate, diffuse hyperemia. No sign of infiltration was present in leptomeninges or neuroparenchyma. According to World Health Organization classification, 12 the tumor was identified as giant cell–rich OSC. TRAP stain exhibited diffuse and strong stain uptake in MGCs (Fig. 5). No cellular staining was observed in polygonal cells in stroma.
Calvarium; feline. Strong positivity for MGCs, whereas mononucleated cells show no negativity. TRAP stain. Bar = 40 µm.
Immunohistochemical stains for vimentin exhibited diffuse and moderately cytoplasmic labeling in MGC cytoplasm and faint staining in polygonal cells within the stroma. No cellular staining against cytokeratin and S-100 protein was observed. MGCs failed to stain with MHC II, however many polygonal cells mixed in the stroma appeared moderately MHC II positive.
Ultrastructurally, all MGCs contained many irregularly shaped nuclei with dense clumped chromatin, and a single central nucleolus was prominent. The cytoplasm was densely filled with small mitochondria and moderate, short profiles of rough endoplasmic reticulum (Fig. 6). Lysosomes were rarely observed, and free ribosomes were scattered throughout the cytoplasm. No desmosomes were present between the MGCs (see summary in Table 1).
Immunohistochemical and ultrastructural findings.∗

∗ +++ = strongly positive; ++ = moderately positive; + = faintly positive; − = negative; np = not performed; TRAP, tartrase-resistant acid phosphatase; MHC II, Class II major histocompatibility complex; ∗RER, rough endoplasmatic reticulum.
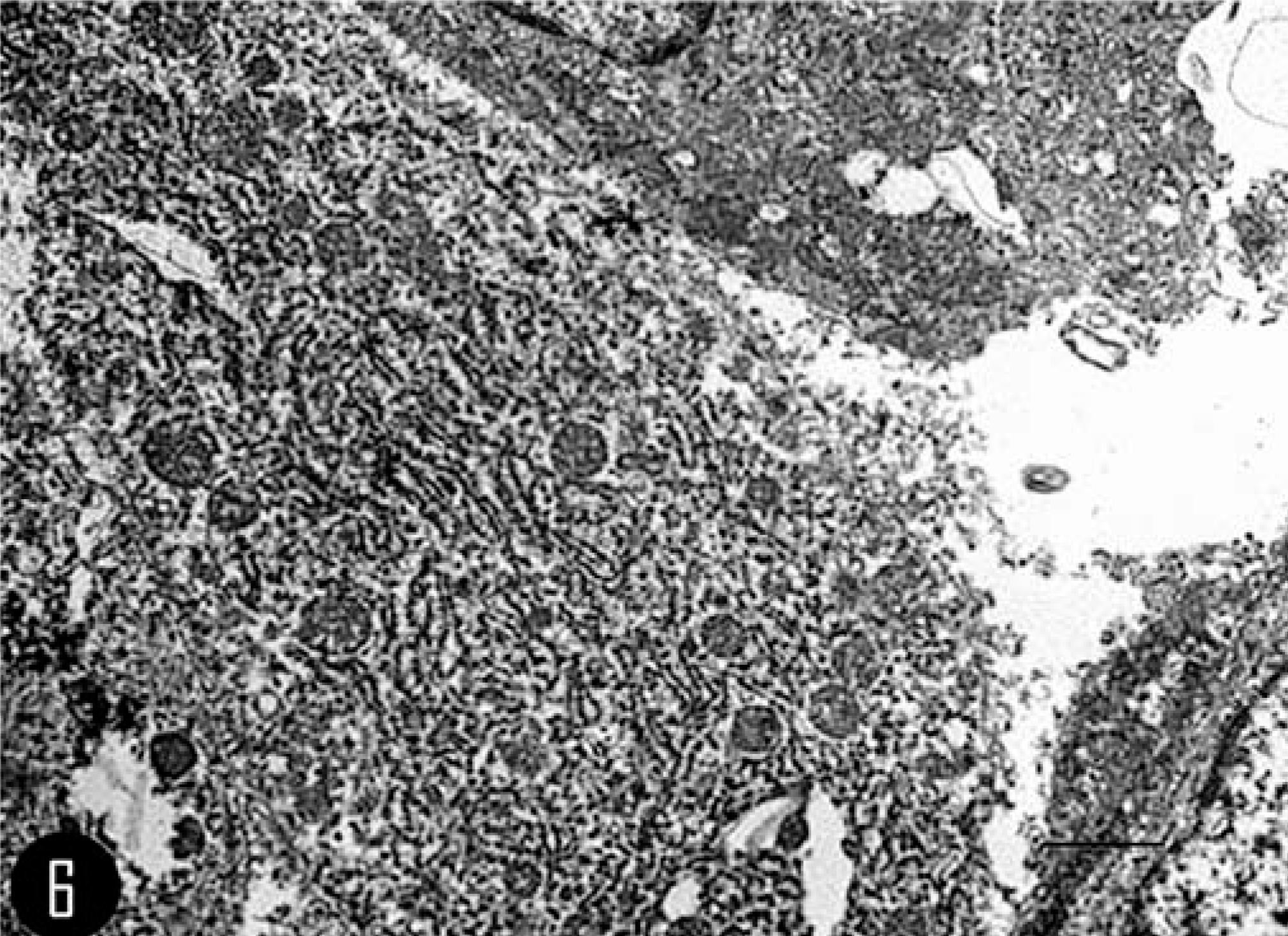
Calvarium; feline. Transmission electron photomicrograph of a MGC, illustrating the prominent rough endoplasmic reticulum. Bar = 2 µm.
Primary bone tumors are uncommon in the cat, with a reported incidence of 3.1 to 4.9 per 100,000 cases; 3, 10 67–90% are histologically malignant. OSCs account for 70–80% of primary feline malignant bone tumors, 7, 14 and OSC of medullary origin is most common. 3 Feline OSC arising from the cranium is extremely rare; those reported involve mostly orbit, mandible, and maxilla. 1, 12 The primary differential diagnosis for tumors arising in the calvarium of the cat is meningioma. However, considering the histologic features of the mass, with the large number of MGCs, giant cell tumor of bone is the primary differential diagnosis. Giant cell tumor of bone is characterized by many MGCs distributed uniformly through the tumor, many cavernous vascular spaces, hemorrhage, and the lack of osteoid production. 6, 13 Giant cell tumor of bone was ruled out on the basis of the absence of these features. The differential diagnosis of meningioma was excluded on the basis of the histologic architecture, cytomorphology, and immunohistochemical results. 5, 13
The origin of MGCs in OSCs is poorly understood in human and veterinary medicine; osteoclast and osteoblast origins have been postulated. Some authors 6, 13 assert MGCs could arise by fusion of osteoclasts, whereas others 14 consider an osteoblastic origin more likely. In this case, ambivalent features of MGCs were observed: MGCs showed some osteoclast-like features, such as TRAP-positive staining and the immunohistochemical pattern (vimentin-positive staining and S-100– and cytokeratin-negative staining), as well as some osteoblast-like features, such as the MHC II–negative reaction and ultrastructural findings. In human and veterinary literature, MHC II is used to differentiate osteoblasts from osteoclasts: osteoblasts usually do not express MHC II, 11 whereas osteoclasts express it at the surface. TRAP reactions have been also used as a cytochemical marker of osteoclasts in several studies. 2, 4, 8 Therefore, in our case as in the literature, MGCs show no evidence of a clear and unequivocal origin; the two most important and ambiguous findings are TRAP-positive staining versus MHC II–negative staining associated with ultrastructural findings. The poor differentiation of MGCs, therefore, could be an explanation for this ambivalence. Being anaplastic cells, they could lose or acquire some features, functions, or both, but in this case, their origin is difficult to recognize.
Footnotes
Acknowledgements
We acknowledge Prof. C. Benazzi and Dr. A. Kipar for their invaluable contribution, and Mr. E. Gallo and Mr. D. Trez for their valuable technical support.
