Abstract
Immunohistochemical analysis using paraffin-embedded specimens is the method of choice to evaluate protein expression at a cellular level while preserving tissue architecture in normal and neoplastic tissues. Current knowledge of the expression of terminal differentiation markers in the mouse mammary gland relies on the evaluation of frozen tissues by use of immunofluorescence. We assessed changes in patterns of expression of terminal differentiation markers throughout the development of the mouse mammary gland in paraffin-embedded tissues. The expression of a-smooth muscle actin (SMA) and keratins (K) 5, 8/18, and 14 was influenced by the development stage of the mammary gland. Expression of K5 and SMA was restricted to basal cells. Keratin 14 was consistently expressed by mammary basal cells, and was detected in scattered luminal cells from 13.5 days after conception through puberty. Labeling for K8/18 of luminal cells was heterogeneous at all times. Heterogeneous expression patterns in luminal cells suggest this layer has cells with a variety of biological functions. The absence of K6 expression at any stage of the development of the mammary gland was confirmed by use of reverse transcriptase-polymerase chain reaction analysis, which indicates that this intermediate filament is not a marker of the mammary gland stem cell. Finally, consistent with results of earlier studies, keratins 1, 10, 13, and 15, and filaggrin, involucrin, and loricrin were not detected at any stage of mammary gland development.
Keywords
Breast cancer is the leading type of neoplasia in women in the United States, accounting for about 30% of cancer diagnosed in women from 1995 to 1999, at a cost of about 6 billion dollars annually for treatment. 1 Researchers have turned to the mouse as a model to investigate various approaches to understand mechanisms and improve treatment of breast cancer in humans. The fixed genetic background, controlled environment, and pathogen-free status of inbred strains of mice ensure reproducibility of the model phenotype. The availability of multiple genetically identical individual mice carrying transplantable tumors ensures a continuous supply of reference and tumor tissue in which consistent changes in gene and protein expression can be discerned. Furthermore, the ever-increasing number of genetically engineered mice that develop various types of mammary cancer or exhibit resistance to this disease provides additional research resources. However, comparison between mouse mammary tumor models and human breast tumors is problematic because of differences in the development and morphology of the mammary gland. 9 Similarities exist in molecular and cellular characteristics, 34 but comparative immunohistochemical studies in this field are limited or rely on frozen sections with suboptimal tissue preservation. Use of mouse-specific antibodies and antibodies that cross-react between species in paraffin-embedded tissues makes it possible to better categorize mouse tissues and tumors on the basis of protein expression patterns than can be based on morphologic characteristics alone.
Mammary gland development is controlled by sequential expression of growth regulators by mammary epithelial and mesenchymal cells. 4, 7, 14, 21 Development begins with thickening of the embryonic ectoderm followed by invasion of the underlying mesenchyme. On completion of mammary bud formation from the ectoderm, mammary glands in female mice go through three distinct periods: 1) development of a mammary bud by postconception day (PCD) 10.5 and, subsequently, of the rudimentary ductal tree from the epidermis, which occurs from PCD 13.5 through birth; 2) invasion of the mammary pad by terminal end buds and early alveolar development starting at 3 weeks of age and extending through puberty; and 3) cycles of lobulo-alveolar development resulting in milk production and secretion during late pregnancy and lactation. 4 Cycles of lobulo-alveolar development in the adult are separated by periods of involution of the mammary gland. 11, 48 These marked changes in the architecture of the mammary gland are associated with changes in keratin expression in humans 2, 5, 35, 62 and in mice. 3, 57 These changes in keratin expression during mammary gland development are important because the formation of cancer in humans and in mice is influenced by developmental changes in the mammary gland at the time of oncogenesis. 13, 51
The postpubertal mammary gland in rats, mice, and humans is composed of a branching ductal system ending in specialized lobules. The entire mammary tree is lined by an inner layer of luminal cells and an outer layer of basal cells, which often are referred to as myoepithelial cells. Results of immunofluorescent, ultrastructural, transplantation, and in vitro studies indicate that these two layers originate from a common ectodermal stem cell located in the ductal luminal compartment. 10, 16, 17, 25, 42, 58, 59, 67 These luminal stem cells give rise to luminal and myoepithelial progenitor/transient amplifying cells, which gradually differentiate into luminal and myoepithelial cells, respectively. Cap cells of the terminal end bud, which have been proposed as stem cells of the mammary epithelium, 71 may actually represent more committed cells than do stem cells located in mammary ducts (i.e., transient amplifying cells). 24, 52 The concept that ductal stem cells are the progenitor cells of the entire mammary gland is supported by results of autoradiographic studies indicating that these cells cycle at a lower pace than does any other population of mammary cells, 56, 69, 73 and can regenerate a complete mammary tree when transplanted into mammary fat pads. 69
In the study reported here, mammary gland development was characterized in 129P3/J, C57BL/6J, and FVB/NJ mice by immunohistochemical analysis using a panel of terminal differentiation markers. The results established that terminal differentiation proteins are expressed sequentially in the mammary gland and can be used to distinguish luminal and basal cells in paraffin-embedded tissues. More importantly, they confirm that luminal mammary cells are composed of heterogeneous populations.
Materials and Methods
Mice
Representative mammary tissue specimens were collected from female mice of the 129P3/J, C57BL/6J, and FVB/NJ strains (The Jackson Laboratory, Bar Harbor, ME) at key developmental stages 48 : PCD 13.5, pups at birth, 10-day-old pups, 21-day-old pups, 42-day-old pups, virgin adults (63–65 days old), and adults at 7 days' gestation, 14 days' gestation, time of parturition, 14 days of lactation, 4 days after weaning, and 3 weeks after weaning. These strains were chosen because C57BL6/J is one of the most commonly used strains in research today, 129P3/J and C57BL6/J are the primary strains used for targeted mutagenesis, and C57BL/6J and FVB/NJ are the primary strains used for transgenic studies.
Mice were maintained in humidity-, temperature-, and 12 : 12–hour light : dark cycle-controlled vivaria under specific-pathogen-free conditions. Mice were caged in double-pen polycarbonate cages (330 cm2 floor area), with sterilized white pine shavings as litter. Cages were washed once a week. Mice were allowed ad libitum access to autoclaved food pellets (NIH31 modified with 6% fat, diet code 5K52, PMI Nutrition International, Brentwood, MO) and acidified sterile water (pH 2.8–3.2). Mice were euthanized by CO2 asphyxiation and were necropsied.
Indirect immunohistochemical analysis
A section of each mammary gland was preserved in five fixatives: Fekete's acid-alcohol-formalin, neutral-buffered 10% formalin, 4% paraformaldehyde, IHC Zinc fixative™ (BD Biosciences, San Jose, CA), and Bouin's fixative. Samples were fixed overnight, transferred to 70% ethanol, and embedded in paraffin, then 5- to 6-µm-thick serial sections were prepared. Skin covering the mammary tissue was included in all sections, making the epidermis and dermis positive controls for immunohistochemical analysis. Slides were deparaffinized and rehydrated through a series of xylenes and graded ethanol. Endogenous peroxidase activity was then quenched by incubation with 3% hydrogen peroxide in methanol for 30 minutes at room temperature. Antigen retrieval was performed for samples tested with antibodies against keratins (K) 15 (K15) and K8/18. Briefly, slides were placed in citrate buffer (pH 6.0) and submitted to five cycles of microwave boiling followed by periods of 2 to 3 minutes' slow cooling at room temperature. Slides were washed and incubated for 30 minutes with blocking serum (10% normal fetal bovine serum [FBS] diluted in phosphate-buffered saline [PBS]). Excess blocking serum was blotted, and the slides were incubated overnight at 4°C with primary antibodies (Table 1) diluted in PBS with 1% FBS. The secondary biotinylated antibody was applied for 30 minutes at room temperature followed by a 45-minute incubation with the peroxidase-labeled avidin-biotin complex (Dako, Carpinteria, CA). Reaction of the avidin-biotin complex with diaminobenzidine (Sigma, St. Louis, MO) and H2O2 produced a brown precipitate. The slides were counterstained with Mayer's hematoxylin. Control sections for nonspecific reactions were treated using the same incubation protocol but omitting the primary antibody in each series.
List of antibodies used.
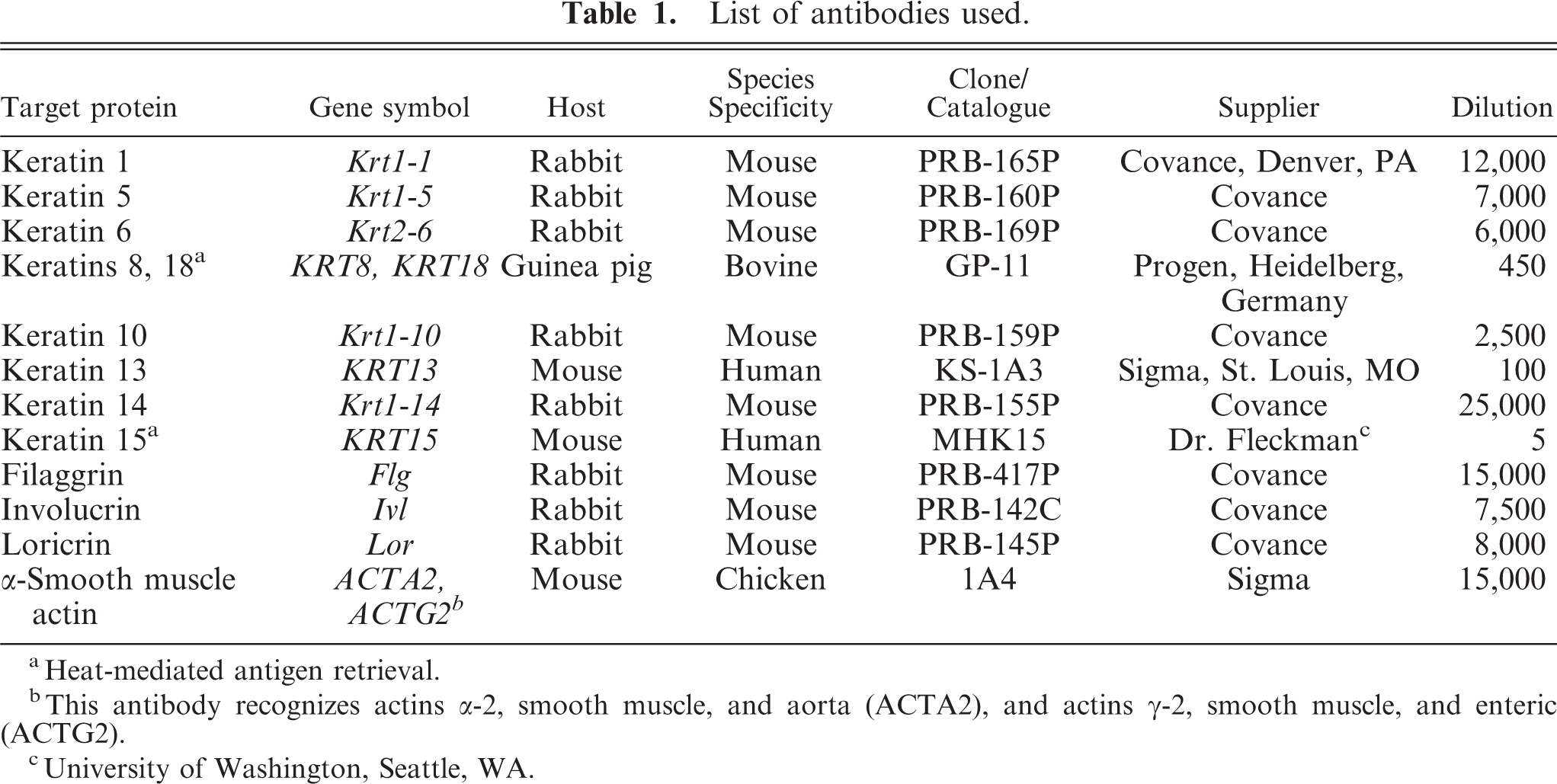
aHeat-mediated antigen retrieval.
bThis antibody recognizes actins α-2, smooth muscle, and aorta (ACTA2), and actins γ-2, smooth muscle, and enteric (ACTG2).
cUniversity of Washington, Seattle, WA.
Keratins 5, K6, and K14 have previously been identified in the mouse mammary gland by indirect immunofluorescence, 57 and immunofluorescence and immunohistochemical analysis were compared for all antibodies used in this study except K8/18, vimentin, and α-smooth muscle actin (SMA). 60 Expression of these markers was re-evaluated because the antibodies used in this study can be used in paraffin-embedded tissues, which allows better evaluation of the architecture of the mammary gland. We also evaluated the mammary gland for expression of K1, K10, filaggrin, loricrin, and involucrin because these markers may be important in categorizing some types of mammary neoplasms with squamous differentiation, although these proteins are not expected to be expressed in the normal mammary gland. 57
Immunohistochemical scoring
The intensity of immunolabeling was assessed by use of a scoring system of 0–3, where 0 = no labeling; 1 = weak labeling; 2 = moderate labeling; 3 = intense labeling. Outcome was measured as the average labeling intensity for each anatomic division, and cell layer location of labeling was noted. Intermediate grades (e.g., 2–3) were given to take into account that the intensity of immunolabeling differed among mammary lobules.
The location of the cells was categorized as “luminal” or “basal” because mammary luminal cells are composed of stem cells, transient amplifying cells of the luminal lineage, and fully differentiated luminal cells, whereas basal cells include transient amplifying cells of the myoepithelial lineage and myoepithelial cells. 10, 42, 67 The mammary tree was divided for evaluation into five anatomic divisions: main galactophorous duct, interlobular ducts, intralobular ducts, acini, and terminal end buds.
Western blot analysis
Western blot analysis was performed with the anti-K8/18 polyclonal Guinea pig antibody (GP11, Progen, Heidelberg, Germany) because it is the only antibody that labels structures that are not reported to express the proteins at which it is targeted. This antibody is reported to react with human K8 and K18. 18
Total cell lysates of the postpubertal mammary gland, colon, haired skin, and skeletal muscles from an 8-week-old FVB/NJ mouse and a culture of TM-β1 hybridoma 64 as negative controls were prepared by placing the cell samples in cold lysis buffer (50 mM Tris, pH 8.2, 150 mM NaCl, complete protease inhibitor cocktail Roche No. 1 697 498). The cell lysates were clarified by centrifugation, and Western blot analysis was performed as described. 53 Briefly, 10 µg of total supernatant protein/lane was loaded on a 4–12% polyacrylamide differential gel, and a constant voltage of 150 V was applied for 2 hours. The proteins were transferred to a Millipore PVDF membrane (Millipore, Billerica, MA) using a constant voltage of 125 V for 2 hours The membrane was incubated with the primary antibody at dilutions of 1 : 2,000 and 1 : 5,000 for 1 hour, followed by hybridization with a horseradish peroxidase-conjugated rabbit antiguinea pig antibody for 1 hour. The reaction was developed using an enhanced chemiluminescence kit (ECLplus, Amersham, Piscataway, NJ), and was radiographed using Kodak X-Omat film (Kodak, Rochester, NY).
Reverse transcriptase-polymerase chain reaction analysis
Absence of immunolabeling of the mammary gland for K6 and K13 in this study contradicted reports in current literature. 43, 57 Therefore, K6 and K13 expression in the mammary gland throughout its development was evaluated by use of RT-PCR analysis (supplementary data) because this method is more sensitive and more specific than indirect immunohistochemical analysis. The RNA was prepared using the FastPrep FP120 (Qbiogene, Carlsbad, CA) centrifuge and the FastRNA-Pro Green Kit (Qbiogene) according to the manufacturer's instructions. Quality of the RNA was verified using the 2100 Bioanalyzer (Agilent, Palo Alto, CA). Complimentary DNA (cDNA) was synthesized using the First-Strand cDNA Synthesis Kit (Amersham Biosciences, Little Chalfont, England, UK) according to the manufacturer's instructions. The primers (Ransom Hill Bioscience, Ramona, CA) were generated to target two exons of a conserved region common to K6a and K6b and two exons of K13. The primers were 5′-GCAGACAGCTGGACAGCATC-3′ and 3′-TCCAGGCTACGGTTGTTGTC-5′ for K6, and 5′-GCAGAGAAGAATCGGAGGGATG-3′ and 3′-TTCCAGCCTTGTTTTGATGTCC-5′ for K13. For PCR analysis, Advantage cDNA Polymerase Mix (BD Biosciences, Palo Alto, CA) was used, and the thermal cycler was programmed as follows: 1 minute at 94°C; 30 cycles of 30 seconds at 94°C and 3 minutes at 68°C; and 3 minutes at 68°C, followed by refrigeration at 4°C. The PCR analysis yielded products of 322 and 345 bp for K6 and K13, respectively; the identity was confirmed by sequencing.
Results
Fixatives
Cellular preservation of mouse tissues was qualitatively best using Fekete's solution, Bouin's solution, and neutral-buffered 10% formalin. Preservation was acceptable using 4% paraformaldehyde and was suboptimal using IHC Zinc fixative™. The results presented here (Table 2) are for the fixatives that yielded the strongest labeling. Fekete's and Bouin's solutions yielded similar results for K1, K5, K6, K10, K13, K14, filaggrin, involucrin, and loricrin. Only Fekete's solution results are presented here, since Bouin's fixative was often associated with nonspecific intranuclear labeling. The IHC Zinc fixative™ was the best fixative for anti-K8/18 labeling.
Expression scores of terminal differentiation markers throughout mammary gland development in the mouse. a
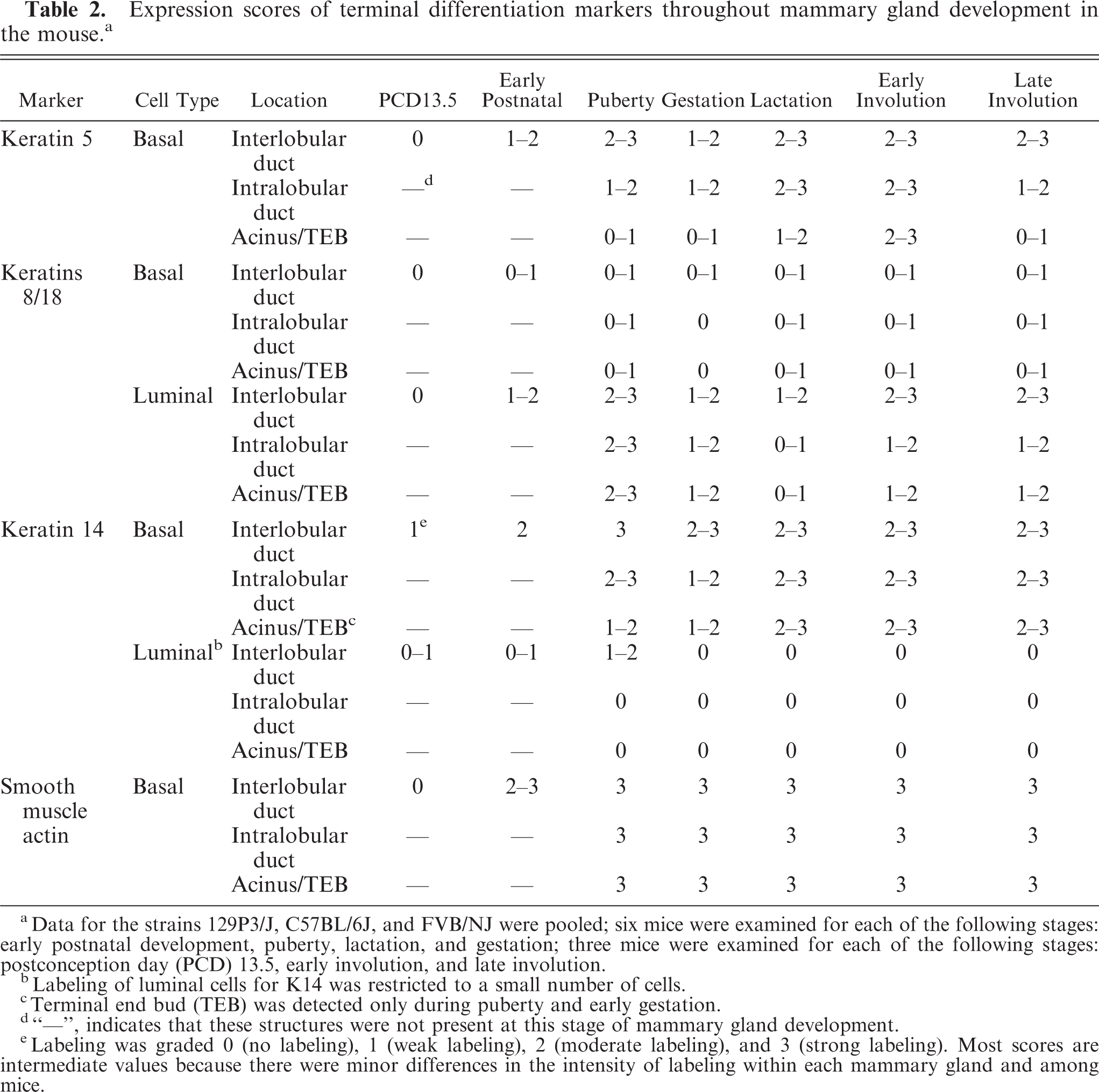
aData for the strains 129P3/J, C57BL/6J, and FVB/NJ were pooled; six mice were examined for each of the following stages: early postnatal development, puberty, lactation, and gestation; three mice were examined for each of the following stages: postconception day (PCD) 13.5, early involution, and late involution.
bLabeling of luminal cells for K14 was restricted to a small number of cells.
cTerminal end bud (TEB) was detected only during puberty and early gestation.
d“—”, indicates that these structures were not present at this stage of mammary gland development.
eLabeling was graded 0 (no labeling), 1 (weak labeling), 2 (moderate labeling), and 3 (strong labeling). Most scores are intermediate values because there were minor differences in the intensity of labeling within each mammary gland and among mice.
Mammary morphogenesis and data pooling
There were no apparent expression differences among the three strains; therefore, the results for the three strains were pooled and are discussed as one. Mammary gland morphogenesis was initially divided into 11 stages; however, on the basis of immunohistochemical results, some stages were pooled as follows: samples taken from adult mice at 7.5 and 13.5 days' gestation were pooled into the category “gestation”; the stages “mammary gland at birth” and “mammary gland at 10 days” were pooled into the stage “early postnatal development”; the stages “early puberty” and “late puberty” were pooled into the stage “puberty”; and samples taken at parturition and 14 days of lactation were pooled into the category “lactation”. All antibodies yielded positive labeling in control tissues (appropriate for each antibody) present in the same tissue sample (data not shown).
Mammary gland morphogenesis followed the pattern described in other studies. 12, 13, 34, 58, 71 Briefly, the mammary bud was identifiable at PCD 13.5, after which it slowly developed into ducts until puberty. The pubertal mammary gland (21 and 42 days of life) was characterized by the formation of terminal end buds and acini by 21 days of age, and were most prominent and more numerous at 42 days of age. Gestational development of the mammary gland was characterized by the presence of acini, with gradual secretion of milk and accumulation of lipid droplets in the cytoplasm of acinar and ductal luminal cells. A few terminal end buds could be detected at this stage, especially in the early stages of gestational development. In adult females, mammary ducts and acini were markedly expanded by milk during lactation. The cytoplasm of all luminal cells was pale and contained numerous lipid droplets. Involution was complete by 3 days after weaning, and was characterized by collapse of mammary acini, presence of macrophages in the interstitium and within the lumen of ducts and acini, and mild-to-moderate dilatation of interlobular ducts by inspissated milk. A small amount of pale-yellow, granular pigment was present in a moderate number of luminal cells. These changes were associated with a drastic reduction in the number of luminal cells. Plump luminal cells had repopulated the mammary gland 3 weeks after weaning.
Immunohistochemical markers
Keratin 14
Keratin 14 was the earliest keratin detected in the epidermis and in the mammary gland and was consistent with results of in situ hybridization studies 8 ; K14 was detected in the apical portions of the cells of the basal layers of the epidermis and of the hair follicle anlagen at PCD13.5. Labeling for K14 in the mammary gland was also detected at PCD 13.5, when labeling was weak and was detected only in basal cells and a few randomly distributed suprabasal cells (Fig. 1a). Basal cells in the mammary ducts were weakly positive at birth, and labeling intensity increased with duct cavitation to become moderately to strongly positive at puberty. Labeling persisted throughout other developmental and involutional stages. Labeling of acinar basal cells was absent or weak (Fig. 1b) until gestation, and became moderate to strong only during lactation, when the intensity became similar to that of ductal basal cells. In eight of nine mice, a small-to-moderate number of luminal cells of interlobular ducts had mild (PCD 13.5)-to-strong (from birth to puberty; Fig. 1b) labeling for K14. These cells were scattered among nonlabeled luminal cells but, on occasion, formed a continuous row. The ducts containing these K14-positive luminal cells were located at some distance from the epidermis and, thus, were not interpreted as a transition between the epidermal cells and the luminal cells of the developing ducts. Positive luminal cells were not detected after puberty.
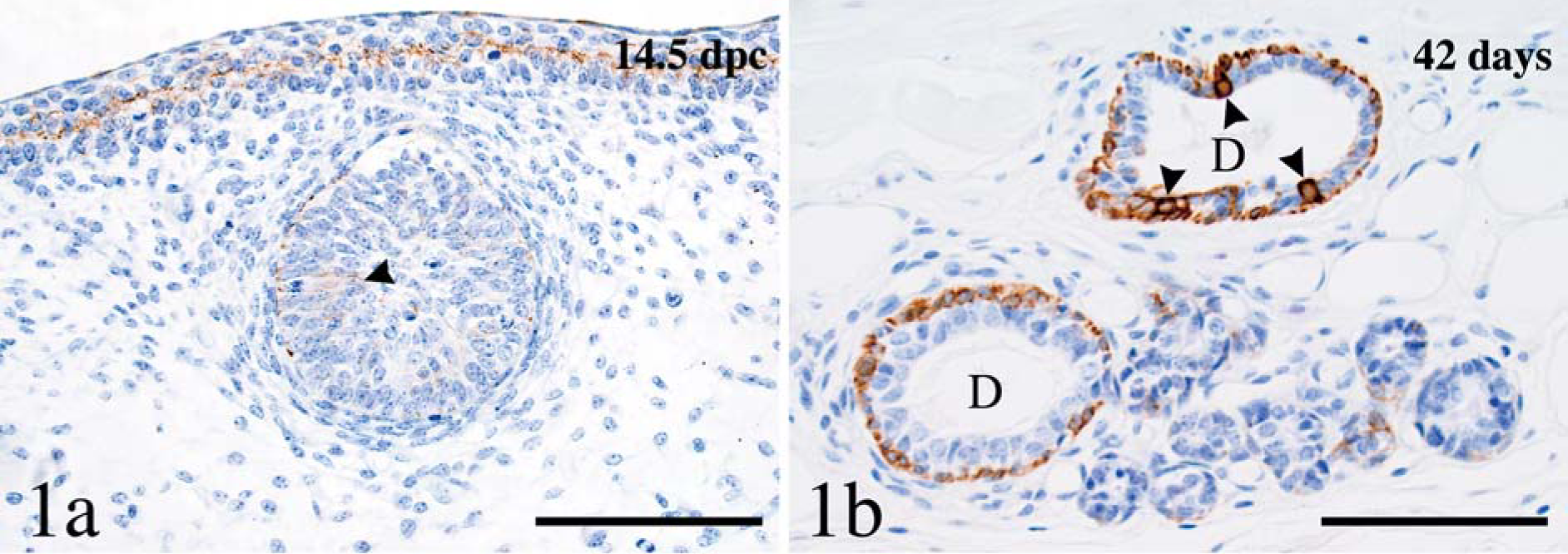
Mammary gland; mouse. Keratin 14 is predominantly expressed in basal cells of the mammary gland in the mammary gland anlagen (
Keratin 5
Basic keratin 5 and its acidic pair, K14, are well-defined proteins in basal keratinocytes of the epidermis and mucous membranes. Keratin 5 was first detected at low amounts in the apical regions of the cytoplasm of basal cells of the epidermis at PCD13.5. The protein was restricted to basal cells in the mammary gland and was not detected until 10 days of age in basal ductal cells. Keratin 5 was subsequently detected at 42 days in acinar basal cells. Labeling was strongest in interlobular ducts and was absent to weak in acini and terminal end buds (Fig. 2a, b). Keratin 5 expression was moderate at puberty, decreased with gestation, and was again moderate during lactation and involution.
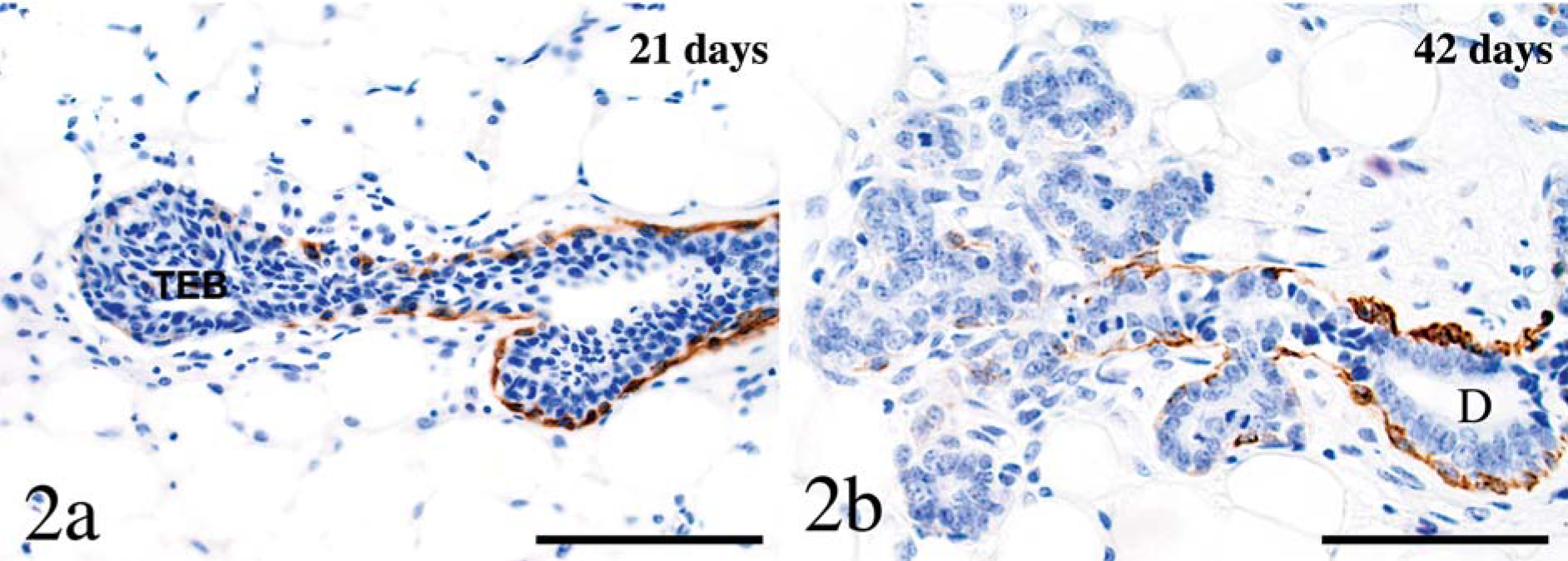
Mammary gland; mouse. Keratin 5 labeling is restricted to basal cells. Labeling is stronger in ducts than in cap cells of the terminal end bud (TEB;
Keratin 8/18
Keratins 8 and 18 should be expressed only in simple epithelia.
40
Nine commercially available antibodies, (complete list, the Mouse Tumor Biology Database, is available on line at
On Western blots, GP11 recognized 47- and 52-kd proteins, corresponding to the molecular masses of K18 and K8, respectively (supplementary data). These proteins were recognized in the mammary gland and the colon, but not in the skin, TM-β1 hybridoma cells, or skeletal muscle. These bands were largest in the colon, which corresponds to the tissue for which immunolabeling was the most intense. This antibody also recognized a variety of other proteins, especially in the colon, but also in the skin and the mammary gland. In addition, a band located at approximately 70 kd was identified in all specimens, including skeletal muscle and TM-β1 hybridoma cells. This band was interpreted as a Western blot artifact because the skeletal muscles and lymphoid organs were not immunolabeled with this antibody.
Lumen formation in the ducts of the postnatal mammary gland was associated with mild-to-moderate labeling of luminal cells for K8/18. Labeling was then detected at all stages of mammary morphogenesis except during lactation when it was weak or absent. Labeling intensity returned to moderate to strong during mammary involution. Labeling for K8/18 was strongest in ducts (Fig. 3a) and in terminal end buds (Fig. 3b), and was generally weaker in mammary acini. Scattered cells in terminal end buds and ducts were not labeled for K8/18 (Fig. 3b, c). Scattered ductal and acinar luminal cells showed strong immunolabeling from day 10 onward (Fig. 3a), most prominently after mammary involution (Fig. 3d). These strongly K8/18-positive cells were most prominent in ductal areas and were occasionally arranged in pairs (Fig. 3a). Some had processes extending between basal cells (Fig. 3c) whereas others contained a few, minute, clear vacuoles interpreted to be lipid vacuoles (Fig. 3e). Mild-to-moderate labeling of basal cells (Fig. 3a, c) was detected throughout mammary morphogenesis and was most easily visualized during lactation (Fig. 3f), at a time when mammary luminal cells almost entirely lost labeling for K8/18.
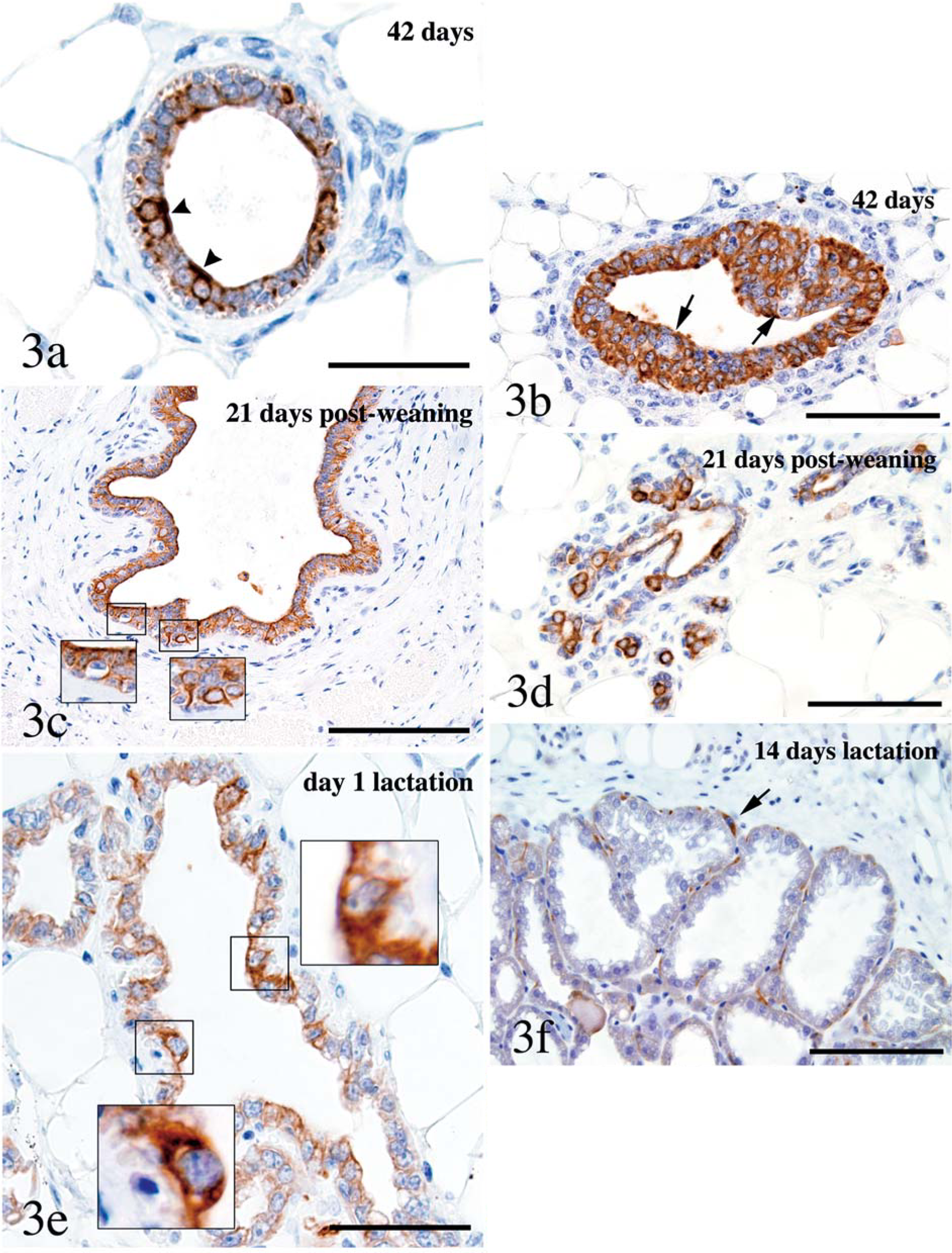
Mammary gland; mouse. Most luminal cells were labeled for K8/18 throughout the postnatal mammary tree (
α-Smooth muscle actin
α-Smooth muscle actin is a microfilament expressed in myoepithelial cells in a variety of glands and in smooth muscle fibers. 39 Labeling for α-SMA was strong in basal cells of interlobular ducts, intralobular ducts, and acini. Labeling was first noticed at birth, was moderate at 10 days of age, and had increased at puberty, after which appreciable changes were not evident (Fig. 4). The basal cells formed a continuous layer in the ducts, but were thin and stretched in the acini.
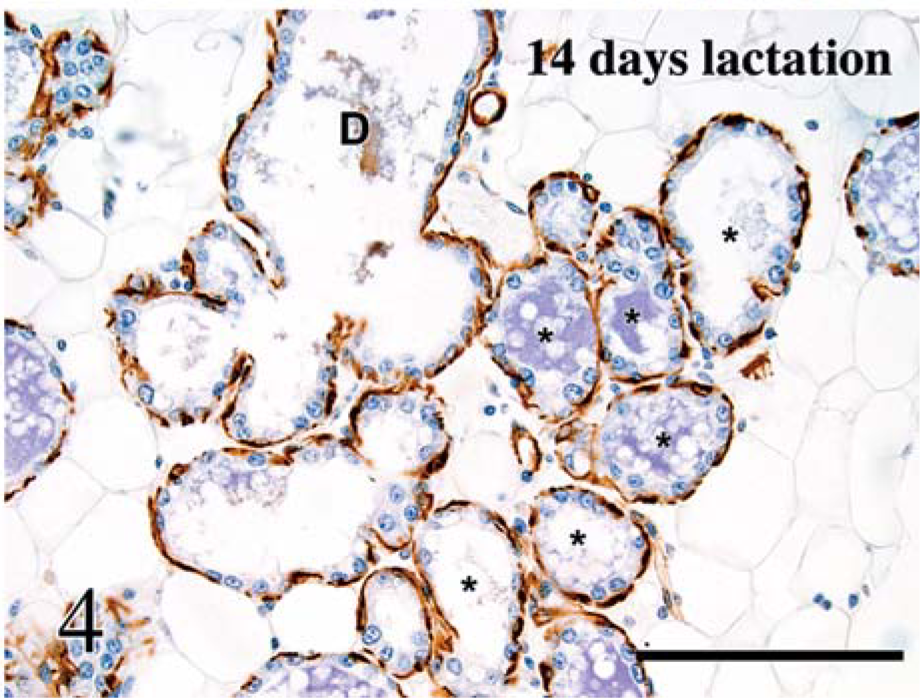
Mammary gland; mouse. Labeling for α-smooth muscle actin is restricted to basal cells and is similar in ducts (D) and alveoli (∗). Avidin-biotin peroxidase method, Mayer's hematoxylin counterstain. Bar = 100 µm.
K1, K6, K10, K13, K15, filaggrin, involucrin, loricrin
Keratin 6 (nonspecific hyperproliferation marker, and marker of the inner layer of the outer root sheath and mucous membranes), 63 K13 (nonspecific proliferation marker), 31 K15 (follicular bulge and epidermal stem cell marker), 70 K1, K10 (suprabasilar terminal differentiation proteins), 39 and filaggrin, involucrin, and loricrin (markers of the stratum granulosum) 22 were absent in the glandular portions of the mammary gland at all stages of development.
Reverse transcriptase-polymerase chain reaction analysis
Expression of K6 and K13 was not detected at any stage of mammary gland development (supplementary data). Expression of these keratins was detected in the skin and the esophagus (positive controls), but not in the heart (negative control). Sequencing of the PCR products confirmed that the PCR analysis was specific.
Discussion
In the study reported here, interpretation of immunohistochemical results indicated that: 1) the luminal compartment of the mouse mammary gland is composed of heterogeneous populations of cells; 2) mouse mammary luminal and basal cells can be differentiated on the basis of protein expression in paraffin-embedded tissues; 3) the degree of labeling of K5, K8/18, K14, and SMA depends on the developmental stage of the mammary gland; and 4) the nature of keratins expressed by basal and luminal cells is conserved throughout the mammary tree, although expression levels are dependant on location in the mammary tree.
Heterogeneity of mammary luminal cells
Heterogeneous labeling of luminal cells for keratins was striking at two distinct periods of mammary gland development: at puberty, when clusters of mammary luminal cells were labeled for K14; and in the postnatal gland, when some luminal cells were labeled more intensely for K8/18 than were most luminal cells.
Scattered individual cells and small clusters of ductal luminal cells were labeled for K14 in the prepubertal and pubertal mammary gland of the mouse. Labeling of ductal luminal cells for K14 45, 65 or K5, 6 both of which are considered basal cell markers, 45 has been reported in scattered cells of the large ducts of the human infant and adult breast. Smith, Mehrel, and Roop 57 also identified labeling of luminal cells for K14, but this pattern of labeling was most prominent during early pregnancy, whereas this pattern of labeling was limited to the prepubertal and pubertal mammary glands of this study. These cells have been interpreted as committed stem cells, 6 or as cells with a phenotype intermediate between that of cells of the stratum spinosum of the epidermis and of the mammary luminal ductal cells. 2 The location and distribution of these cells, individually or in small clusters, are consistent with those of “light cells,” as defined ultrastructurally 10, 15, 16, 55 and on semithin sections. 15, 56 However, these cells are unlikely to represent all “light cells” because these cells were few in number and were detected only during prepuberty and puberty. Immunoelectron microscopic evaluation of these cells is needed to characterize them morphologically, but attempts were unsuccessful using the anti-K14 antibody in this study.
Individual luminal cells expressed higher levels of K8/18 than did other nearby luminal cells after ductal cavitation. Similar observations in the human breast and in mammary cell culture have been made. 2, 35, 59 It is currently accepted that differentiation is associated with gradual expression of terminal differentiation markers. Hence, cells that express high levels of K8/18 should be more differentiated than neighboring cells that express lower levels of K8/18. However, three lines of evidence suggest that, in the mammary gland, the hypothesis that the most differentiated cells express higher levels of terminal differentiation markers is invalid. First, some of these strongly K8/18-positive luminal cells contained optically empty vacuoles, consistent with lipid droplets, which were smaller and less numerous than vacuoles in neighboring cells, an indication that these cells are less differentiated than are fully differentiated secretory mammary cells. Second, these strongly K8/18-positive luminal cells also were detected during involution of the mammary gland, whereas the current theory is that fully differentiated glandular mammary cells are lost during involution. 6 Third, some of these cells had a process extending between myoepithelial cells, a feature of large light cells. 10, 55, 56 The luminal location of these cells, the relative abundance of their cytoplasm, and their overall scarcity was reminiscent of large light cells, a cellular population intermediate between committed mammary stem cells and fully differentiated mammary glandular cells. 55 These cells are unlikely to represent a pure population of committed mammary stem cells because they were not identified before ductal cavitation (at 10 days of age), they had relatively abundant cytoplasm, some of them contained lipid droplets, and cells undergoing mitosis were not labeled for K8/18. A few small suprabasal cells failed to be labeled for K8/18. The suprabasal location of these cells and their small size suggests that they are less differentiated than are other suprabasal mammary cells. Because the antibody used to detect K8/18 was developed against bovine proteins, these observations need to be confirmed by labeling paraffin-embedded mouse tissues with antibodies specific to mouse K8 and K18, when such antibodies become available.
Differentiation of luminal and basal cells on the basis of terminal differentiation markers
Pubertal and postpubertal mouse mammary myoepithelial cells can be differentiated from luminal cells in that they differentially express K5 and SMA, a feature that had been previously identified in the human breast 5 and in the mouse mammary gland 57 using less-specific antibodies or frozen sections. None of the antibodies tested specifically labeled luminal cells, although K8/K18 generally is considered a specific marker of luminal cells of simple epithelia in humans. Some of the basal cells labeled by the K8/18 antibody may represent myoepithelial precursor cells that have retained some features of less-differentiated cells. 59 Alternatively, mouse and human mammary myoepithelial cells may differ with respect to their K8/18 expression status or, as suggested by the Western blot results, the anti-K8/18 antibody used for this study may recognize proteins other than K8/18.
Sequential expression of differentiation markers during mammary gland morphogenesis
The number of differentiation markers expressed by mammary luminal and basal cells gradually increased during mammary morphogenesis. Puberty was associated with the onset of differential expression of K5 and SMA in basal cells, which represents a major step toward differentiation of myoepithelial cells. Interestingly, basal cells acquire their contractile phenotype (SMA expression) before their keratinization pattern (keratin expression) is established, indicating that features typical of basal cells are acquired in a multi-step process rather than a switch-type event. In the human eccrine gland, acquisition of the basal phenotype is also a multi-step process, although keratinization precedes establishment of the contractile phenotype. 38
In contrast to that in the epidermis, 8 expression of K14 and K5 was asynchronous in the developing mammary gland, with K14 expressed as early as PCD 13.5, and K5 first detected at 21 days of age. Differential expression patterns of K5 and K14 were not totally unexpected because, although K5 is the main partner of K14, 39, 45 K5 can bind to other keratins. 23 In addition, mice carrying transgenes with K5 and K14 promoters can be phenotypically different. 33 The absence of immunolabeling for K5 in the prepubertal mammary gland is not the result of failure of the antibody to detect this keratin because K5 and K14 strongly labeled the periderm and moderately labeled the basal cells of the epidermis at PCD 13.5. Thus, K14 may be paired with a yet undetermined keratin other than K5 during early mammary morphogenesis.
Results of gene array experiments have indicated that expression of K14 increases, from the prepubertal mammary gland through puberty and gestation. 32 Results of our study confirmed this observation and determined, at the cellular level, the distribution of K14 in the mammary gland.
Labeling for K8/18 was reduced during lactation. This observation contradicted results of studies in humans that K18 expression is slightly increased during lactation. 35 Decreased K8/18 labeling may be the result of severe expansion of the cytoplasm of secretory luminal cells during lactation, with subsequent dilution of the antigen, rather than of an actual decrease of K8/18 expression. The pattern of K8 and K18 expression in the mammary gland could be monitored by engineering mice transgenic for the green fluorescent protein under control of the promoter of one of these keratins. The morphology and pattern of K8/18 expression were unique in the involuted mammary tissue, compared with those at all other stages of mammary development. This observation indicates that the stage of involution is distinct from all other stages of mammary development, an observation that has been confirmed at the RNA level. 47
The intensity of SMA labeling in myoepithelial cells was not influenced by lactation. This result was consistent with results of studies in women that indicated that SMA expression is not up-regulated in the lactating, relative to the resting mammary gland. 35 Thus SMA, in conjunction with K5, is a reliable marker of mouse mammary basal cells. Unlike those in the human breast, 74 myoepithelial cells in the mouse mammary gland are not heterogeneous with respect to SMA expression.
Conservation of ductal and acinar phenotypes throughout the mammary tree
The antibodies used in this study could not differentiate luminal cells of the ductal and secretory portions of the mammary gland, whereas ductal and secretory portions of the human eccrine glands 38 and mouse salivary glands 61 can be differentiated on the basis of keratin expression. The extent of remodeling associated with cyclic lobulo-alveolar development of the mammary gland may account for the absence of specific keratin markers for luminal cells of the ductal and secretory portions of this gland. In the absence of specific markers for the ductal and secretory portions of the mouse mammary gland, the ductal or acinar origin of mammary neoplasms probably cannot be differentiated by immunohistochemical analysis at this stage.
Other immunohistochemical markers
Keratin 15 was not expressed at any stage of development of the mammary gland, but was detected in the suprabasal and basal cells of the epidermis and in hair follicles at birth. It has been suggested that K15 is expressed at an early stage of keratinocyte differentiation, before cells are committed to the epidermal or the hair follicle differentiation pathways. 70 Absence of K15 in the mammary epithelium suggests that the mammary cell lineage diverges from the epidermal and follicular lineages before the follicular lineage diverges from the epidermal lineage. Consistent with this observation, mammary anlagens develop from PCD 10.5 to PCD 11.5, 34 whereas rudimentary hair follicles in the abdominal area are first detected at PCD13.5–14.5. 41 However, mammary stem cells, which also are the putative origin of mammary tumors, 26, 44, 50, 54 retain some features of follicular stem cells, as indicated by their ability to form tumors with pilar 36, 37 and sebaceous (personal observation) differentiation.
Keratin 6 was not detected at any stage of development of the mammary gland, which is in contrast with the observations of Smith, Mehrel, and Roop, 57 who identified K6 expression by immunofluorescence in a small number of cells in the growing end buds and in a few luminal cells in intralobular ducts and acini of the mouse mammary gland, and who induced others to consider K6 a marker of mammary stem cells. 30, 68 Keratin 6 is typically associated with epidermal proliferation and is expressed in the inner layer of the outer root sheath, hyperplasic epidermis, conjunctiva, oral mucosa, and some preneoplastic and neoplastic mouse mammary lesions. 19, 27, 28, 49, 57, 60, 72 On the basis of the pattern of distribution of K6 in stratified epithelia, this keratin should not be expressed in the normal mammary gland, which we confirmed at the RNA level. Similarly, our results indicated that K13 is not expressed in the mouse mammary gland, whereas this keratin has been reported to be expressed in the human mammary gland. 43
Keratins 1 and 10 are suprabasal terminal differentiation proteins of squamous epithelia, whereas filaggrin, loricrin, and involucrin are epidermal differentiation markers in the stratum granulosum of the normal epidermis. 20, 22, 29, 60 As expected, none of these markers was expressed in normal mouse mammary gland, but they were evaluated because some are expressed in specific mouse models of transgenic mammary carcinogenesis 36, 37 and in human breast carcinomas. 46, 66 Hence, as suggested by others, 36, 37 expression of these markers in some models of mammary adenocarcinomas is consistent with follicular and epidermal transdifferentiation.
Conclusion
Terminal differentiation markers are acquired sequentially throughout mammary gland development. α-Smooth muscle actin and K5 specifically identify basal cells of the mammary gland, but none of the markers tested specifically identified mammary gland luminal cells. In addition, on the basis of their expression of terminal differentiation markers, mammary gland luminal cells are heterogeneous, with some of them not expressing any marker. These observations, when applied to the analysis of mammary neoplasms in mice, will be critical for defining tumor cell types.
Footnotes
Acknowledgements
This work was supported in part by grants from The National Institutes of Health (CA89713, CA34196, RR173), The National Science Foundation, The Clark Foundation, and The Goldsmith Foundation for support of The Jackson Laboratory Summer Student Program. We thank Dr. W. G. Beamer for his support of the polymerase chain reaction experiments, J. A. Budzik for technical assistance, and S. L. Williamson for artwork. We are grateful to T. V. Golovkina, T. P. O'Brien, and B. A. Richards-Smith for critical review of the manuscript.
