Abstract
E-Cadherin and β-catenin are known for their role in tumor invasion, but both proteins also exert an influence on tumor proliferation. This study, performed on canine mammary tumors, aimed to analyze the influence of E-cadherin (E-cad) and β-catenin (β-cat), immunohistochemically assessed singly and in combination (E-cad/β-cat), on survival and their relationship with several proliferation indices (AgNOR index, MIB1 index, mitotic index). Immunohistochemistry was carried out on 60 formalin-fixed, paraffin wax-embedded specimens of canine mammary malignancies. The labeling was defined as preserved when prevalent on cell membranes of more than 75% of cells and reduced in other forms of expression (i.e., membranous less than 75%, cytoplasmic, and negative). E-cad, β-cat, and E-cad/β-cat were preserved respectively in 22, 12, and 11 out of 60 cases. Immunohistochemical expression of the two proteins in the same tumors was significantly correlated (P = 0.0001; R = 0.57). Survival analysis revealed no difference in outcome comparing the preserved versus reduced cases (E-cad, P = 0.31; β-cat, P = 0.29; E-cad/β-cat P = 0.36). Grouping cases for histologic invasiveness, the expression of E-cad or β-cat and E-cad/β-cat showed a progressive reduction that paralleled an increase in invasiveness from noninfiltrating to stage-II tumors (E-cad, P < 0.001; β-cat, P < 0.05; E-cad/β-cat, P < 0.05). No significant difference was obtained comparing mitotic index, MIB 1 index, and AgNOR index by analysis of variance between the cases grouped for preserved or reduced E-cad, β-cat, and E-cad/β-cat variables. In conclusion, reduced expression of E-cad, β-cat, or E-cad/β-cat was significantly associated with the progression from noninfiltrating to highly infiltrating tumors but not with proliferation or survival.
E-Cadherin (E-cad) is the major transmembrane component of the adherens junction in epithelial cells of all organs, such as luminal mammary epithelial cells. E-cad-mediated cell-cell adhesion requires the availability of calcium ions on the cell surface and the ordered presence of other proteins, called α-, β- and γ-catenins, on the cytoplasmic side of the plasma membrane that anchors E-cad to actin. 26
Cadherin-catenin complexes play an important role in morphogenesis and tissue architecture in embryos and in cancer progression. These complexes are thought to influence metastasis and invasion by a process involving loss of cell adhesion. 27 Recent studies on E-cad function have defined the role of this molecule in downregulation of cell proliferation. Two main mechanisms of downregulation are known: in the first, E-cad inhibits the activity of mitogenic pathways, such as that utilizing the epidermal growth factor receptor (EGFR), which in turn regulates the level of p27 in cells. 25 A second mechanism of influencing proliferation is mediated by β-catenin (β-cat). β-catenin linked to E-cad promotes cell adhesion, but it exerts transcriptional activity when free. This available β-cat quantity may originate from E-cad loss or from conditions such as adenomatous polyposis coli or β-cat gene mutation. Free β-cat links to transcription factors, such as members of the lymphoid enhancer binding factor 1 or T cell factor family, and promotes gene expression, of which c-Myc and cyclin D1 seem to be the most important. 8, 11, 12
Expression of both E-cad and β-cat is impaired in several types of human tumors (i.e., breast cancer, gastric cancer, prolactinoma) in which reduction of expression parallels the acquisition of an invasive phenotype, a poor outcome, and metastasis. 16 – 18, 24, 27 However, the influence of downregulation and cell proliferation of these proteins is controversial. Menon and Simha 13 revealed a correlation between AgNOR and loss of E-cad expression in transitional cell carcinoma of the human bladder, whereas Fricke et al. 6 failed to disclose any relationship between Ki-67 and E-cadherin in gastric carcinoma.
To the best of our knowledge, nothing is known about the correlation between E-cad and/or β-cat and mitotic index, AgNOR, and Ki-67 expression in canine neoplasms. The little information available in the dog concerns the correlation between tE-cad expression and histologic grade 19 and stage 1, 22 in mammary tumors, showing a significant decrease from benign to malignant tumors. Moreover, absent or low E-cad expression was associated with loss of β-cat expression in canine mammary lesions. 4
Considering the role of E-cad and β-cat in invasion and proliferation in a series of canine malignant mammary tumors, we aimed to investigate the relationship between E-cad and β-cat expression and invasion or proliferation. We also assessed the influence of the pattern (expression or loss) of both proteins on survival.
Materials and Methods
Animals
Sixty cases of canine malignant mammary tumors with 24-month postmastectomy follow-up data available were selected from the histopathologic database of the Department of Veterinary Public Health and Animal Pathology of the University of Bologna. For most of them, E-cad immunohistochemistry was available from a previous paper. 22 Proliferative-activity data were recorded as mitotic index (47 cases), quantification of proliferation-associated antigen recognized by the clone MIB1 antibody (MIB1 index) (39 cases), and quantification of the argyrophilic proteins associated with the antigen to nuclear organizer region (AgNOR index) (48 cases) quantified as reported in a previous investigation. 21 Briefly, quantitative analysis was performed with an image cytometer (Byk Gulden, Milano, Italy). AgNORs were evaluated by measuring the area (µm2) occupied by AgNOR dots (AgNOR area). Every image was obtained with the 40× objective. Mitotic index was calculated using a 25× objective to select 10 fields in the most mitotically active areas of the section. The total nuclear area of only the neoplastic cells was mapped and the number of mitoses was counted. For the evaluation of MIB1 index, a 25× objective was used to select 10 fields with the highest MIB1 positivity, and MIB1 index was calculated as the percentage of labeled nuclei compared with the total nuclear area.
Diagnoses were reviewed following the criteria developed by Misdorp et al. 14 and graded for invasion according to Gilbertson et al. 7 as stage 0 (noninfiltrating malignant tumor), stage I (malignant tumors with stromal invasion), and stage II (malignant tumors with neoplastic emboli in vessels and/or lymph node involvement).
Immunohistochemistry
All cases, after 12/24-hour formalin fixation, were paraffin wax embedded and underwent immunohistochemical labeling with mouse anti-human E-cad (clone 36) and mouse anti-human β-cat (clone 14) antibodies (Transduction Laboratories, Lexington, KY) as follows. The sections were dewaxed in toluene and rehydrated. Endogenous peroxidase was blocked by immersion in methanol containing 0.3% hydrogen peroxide for 30 minutes. Sections were then rinsed in Tris buffer, immersed in citrate buffer (2.1 g citric acid monohydrate/liter distilled water), pH 6.0, incubated for 10 minutes in a microwave oven at 750 W, and allowed to cool at room temperature (approximately 20 minutes). The primary anti-E-cad (2.5 µg/ml) and anti-β-cat (2.5 µg/ml) antibodies were applied overnight at 4°C and were followed by a commercial streptavidin-biotin-peroxidase kit (LSAB kit, DAKO, Amsterdam). For negative controls, an unrelated monoclonal antibody of the same isotype and similar protein concentration was used instead of the primary monoclonal antibody. Sections of canine normal mammary gland with hyperplastic change and benign tumors (two tubular adenomas and one benign mixed tumor) were used as positive controls.
Evaluation of E-cad and β-cat immunoreactivity
E-cad and β-cat immunoreactivity was classified according to its prevalent pattern of expression as membranous when the immunolabeling was localized in the cell-cell boundaries, where it appeared stronger than in the cytoplasm, whereas cytoplasmic, when the positivity was uniformly distributed throughout the cytoplasm and no distinction was recognizable between cell-cell boundaries and the cytoplasm. The positivity of β-cat was also nuclear, but, as not prevailing, it was not considered in the present classification. The cases were grouped, by semiquantitative assessment, as preserved when the positivity was higher than 75% of neoplastic epithelial cells and membranous; and reduced in all the remaining samples, including negative tumors. A third variable was obtained integrating the data from both proteins and named E-cad/β-cat. E-cad/β-cat preserved expression included cases in which both proteins had membranous expression, whereas E-cad/β-cat was classified as reduced when at least the expression of one protein was low.
Statistical analysis
Correlation between the variables E-cad, β-cat, and E-cad/β-cat was graded as a yes (preserved)/no (reduced) score and invasion was investigated by Pearson's method and tested for significance using the χ2 test. The relationship between E-cad, β-cat, and E-cad/β-cat and proliferation was investigated by one-way analysis of variance (ANOVA). The prognostic influence of E-cad, β-cat, and E-cad/β-cat expression and prognosis was investigated by survival analysis. Survival information was obtained from the referring veterinarians and/or animal owners by telephone contact. The information pertained to whether the animals had died from the tumor or were still alive on a 2-year follow-up period. For the animals that died within the 2 years, the months between surgery and death were reported. The living dogs were those that completed the 24-month follow-up period. Kaplan-Meyer survival curves were compared by the log-rank test. Analyses were performed by CSS software (Statsoft, Tulsa, OK) statistics, and a conventional 5% level was used to define statistical significance.
Results
Cases were classified as tubulopapillary carcinoma (33 cases), solid carcinoma (13 cases), squamous cell carcinoma (1 case), carcinosarcoma (1 case), and carcinoma in benign tumor (12 cases). Twelve were stage 0, 29 were stage I, and 19 were stage II.
Immunohistochemical labeling for both E-cad and β-cat disclosed membranous, cytoplasmic, or negative and, just for β-cat, nuclear positivity. The membranous pattern (Figs. 1 and 2) appeared as a brown labeling, stronger in the membrane than in the cytoplasm. The stain was instead described as cytoplasmic (Figs. 3 and 4) when weaker and confined to the cytoplasm. In two cases with reduced β-cat expression, nuclear positivity was also found. In 22 of the 60 cases (36.7%), E-cad was expressed as preserved, while in the remaining 38 (63.3%), it was reduced. As for β-cat, 12 (20%) cases were classified as preserved, whereas 48 (80%) had reduced expression (26 with a cytoplasmic stain and 10 negative). E-cad/β-cat complex was preserved (both adhesion molecules were preserved) in 11 out of 60 cases. Immunohistochemical expression of the two proteins in the same tumors was significantly correlated (P = 0.0001; R = 0.57); in fact, concordance existed in 48 cases, in 11 of which both proteins were preserved (membranous type) and in 37 both reduced (E-cad/β-cat cytoplasmic type), and dissonant in the remaining 12 cases (1 with reduced E-cad and preserved β-cat expressions and the opposite in the other 11 cases).
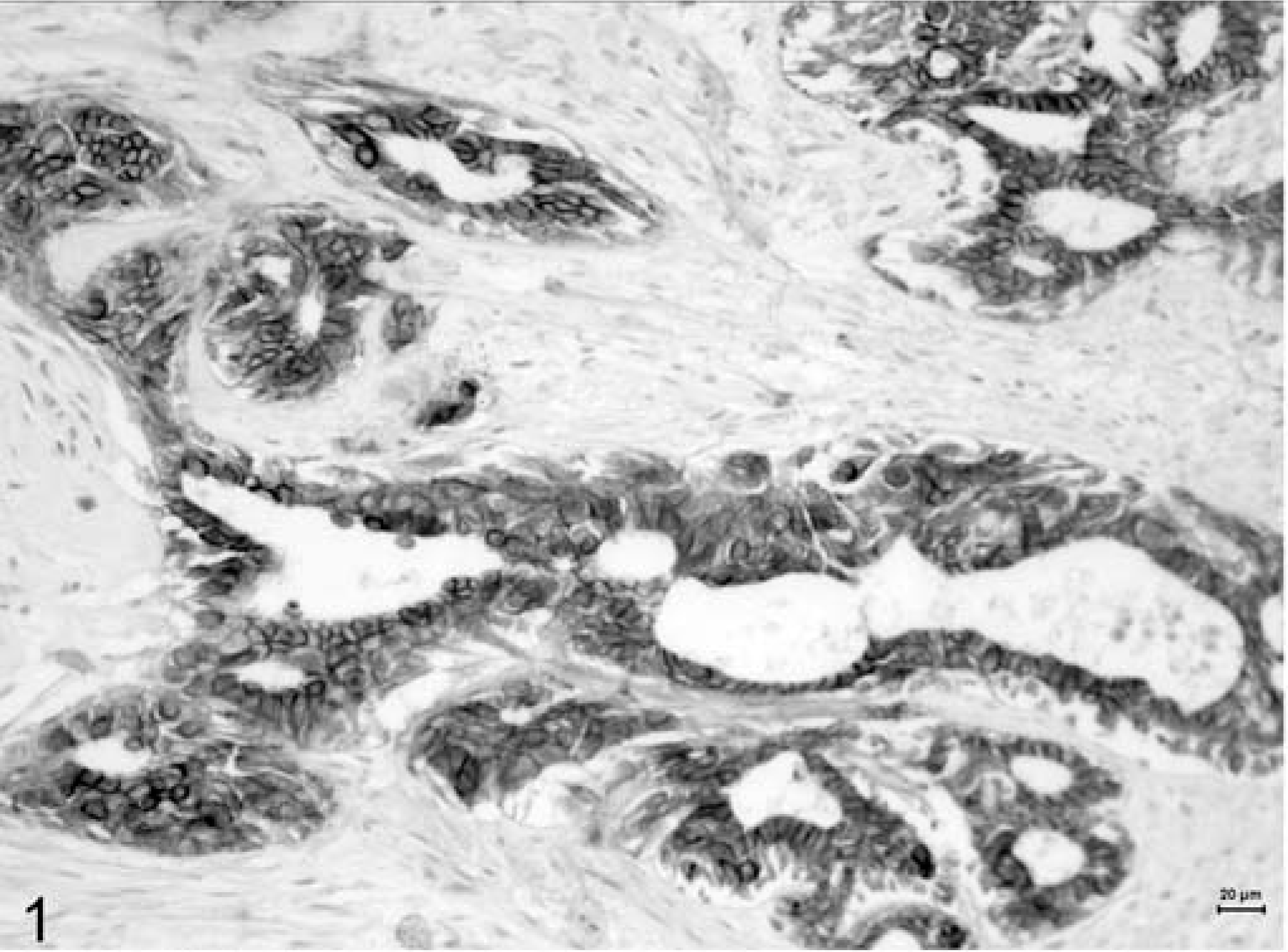
Mammary tissue; dog. Tubulopapillary carcinoma. Membranous expression of E-cadherin. Formalin-fixed section labeled with the monoclonal antibody E-cadherin. LSAB kit, Papanicolaou's hematoxylin counterstain. Bar = 20 µm.
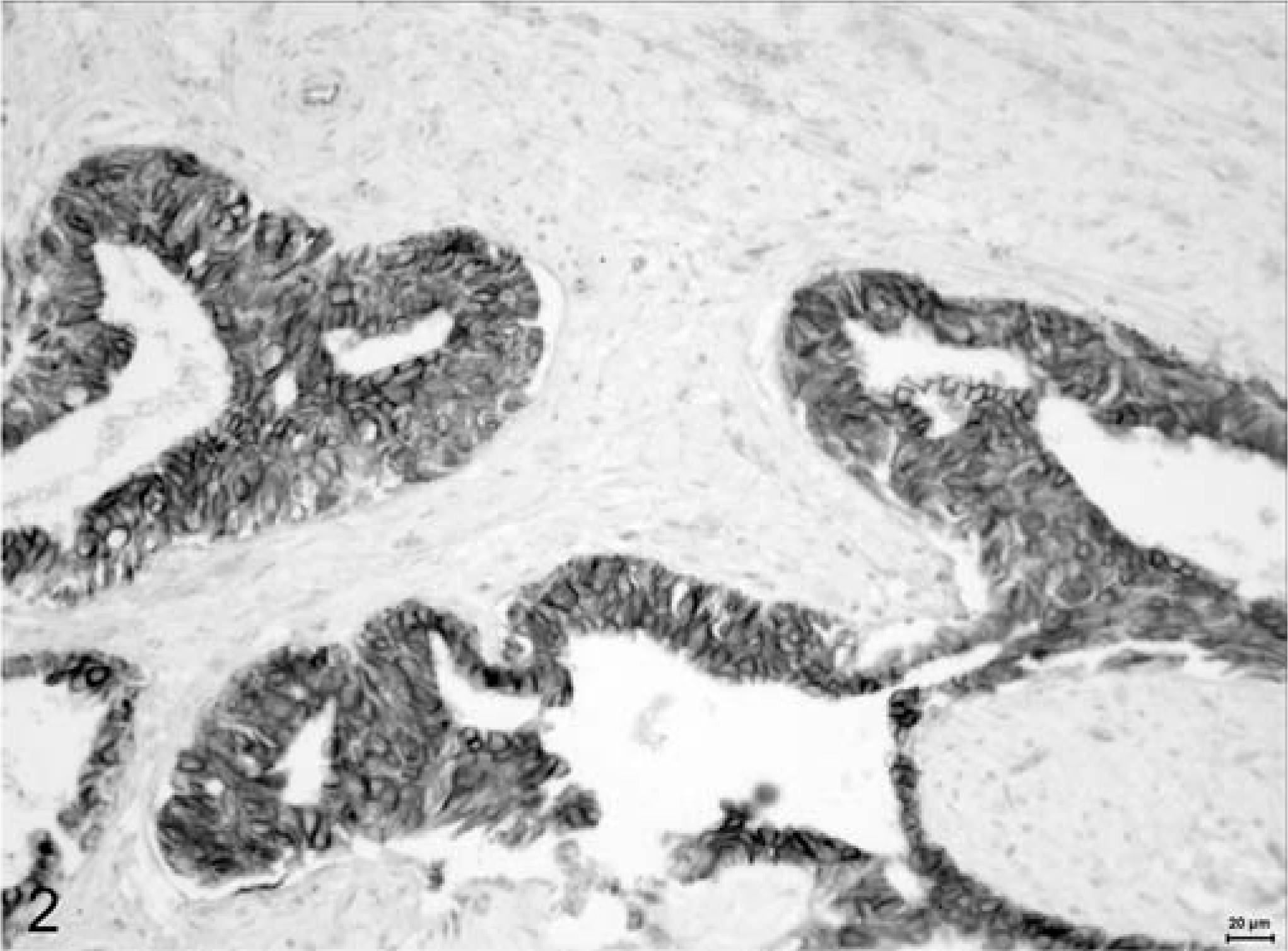
Mammary tissue; dog. Tubulopapillary carcinoma. Membranous expression of β-catenin. Formalin-fixed section labeled with the monoclonal antibody β-catenin. LSAB kit, Papanicolaou's hematoxylin counterstain. Bar = 20 µm.
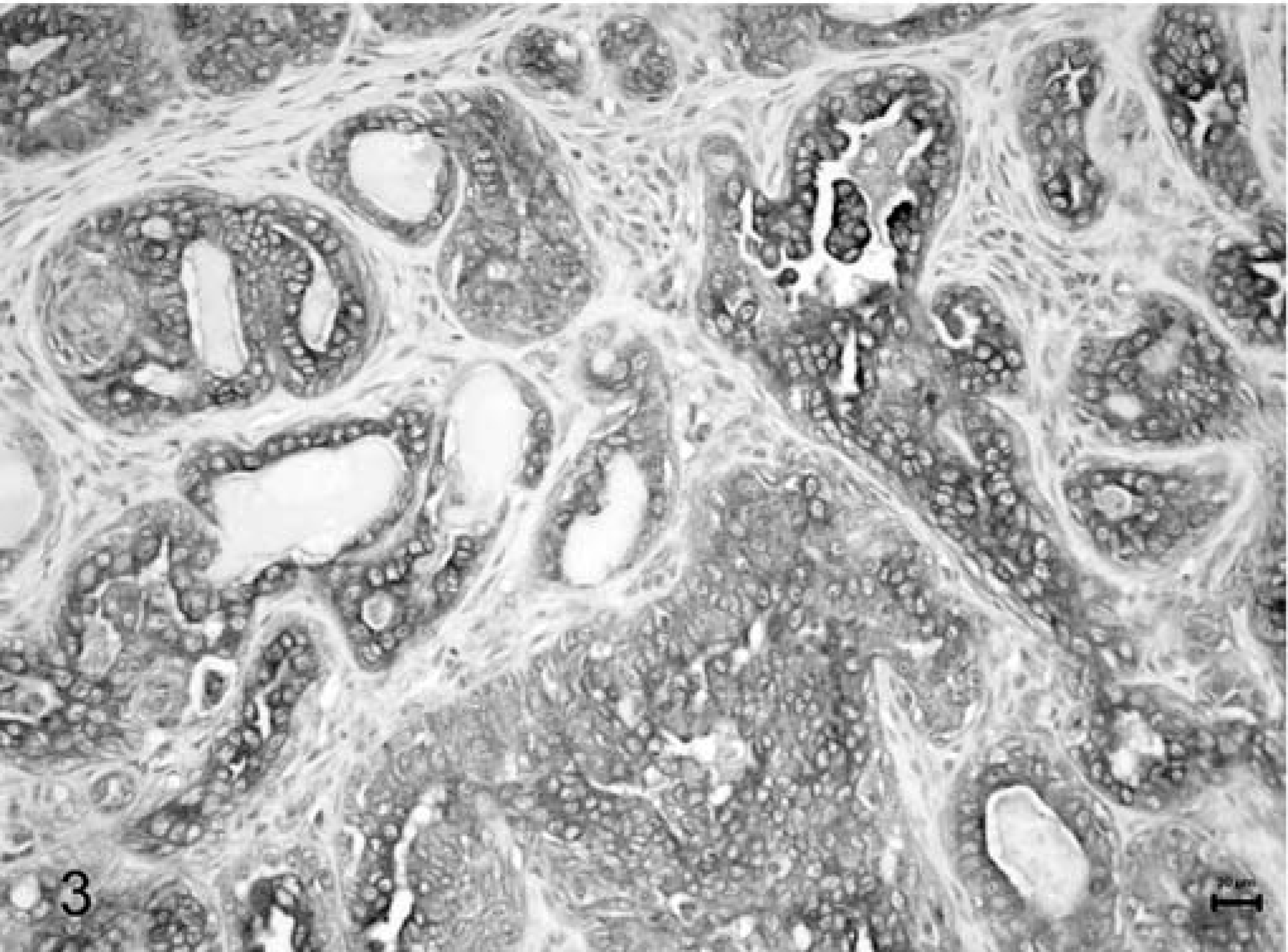
Mammary tissue; dog. Tubulopapillary carcinoma. Cytoplasmic expression of E-cadherin. Formalin-fixed section labeled with the monoclonal antibody E-cadherin. LSAB kit, Papanicolaou's hematoxylin counterstain. Bar = 20 µm.
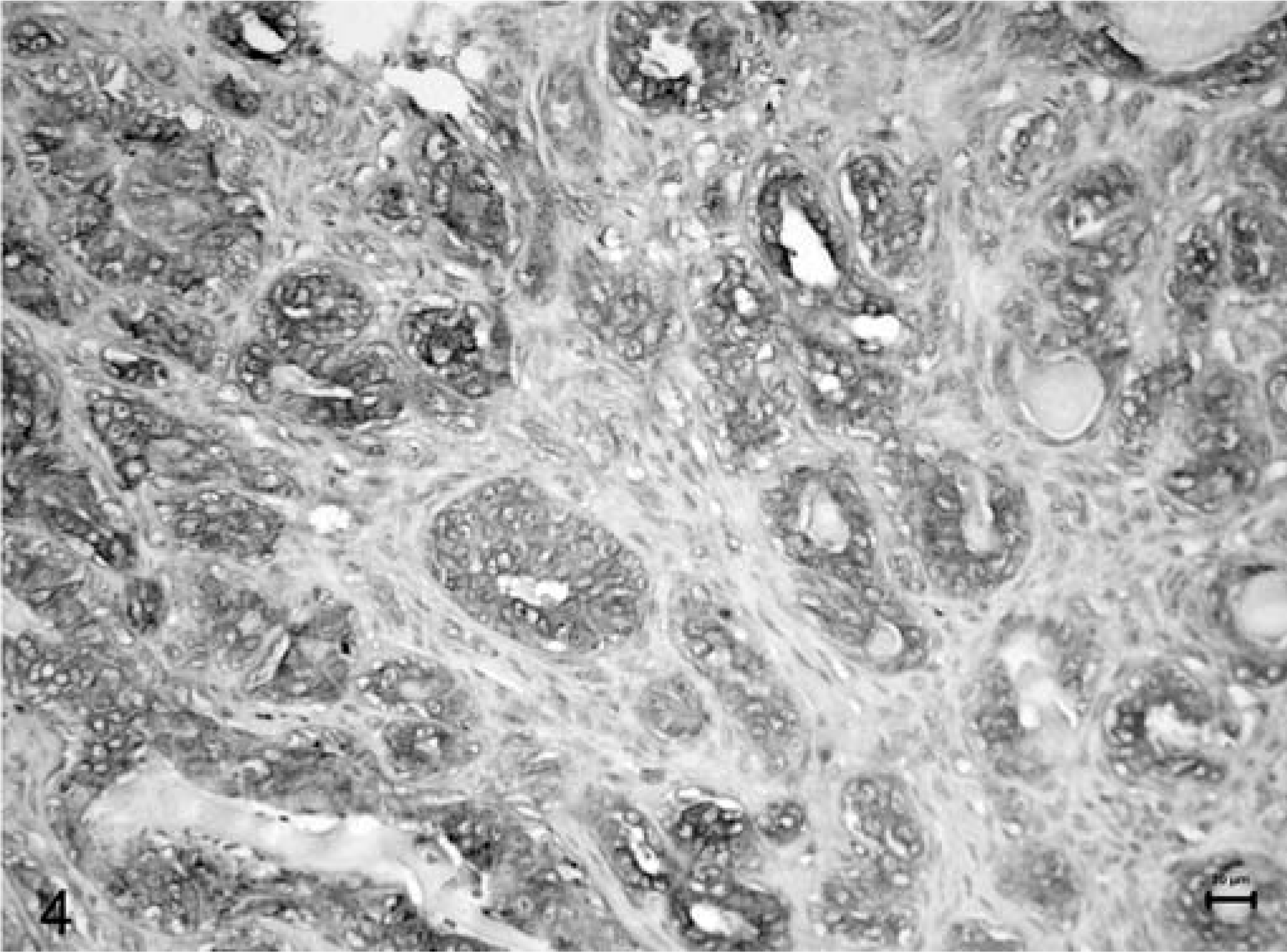
Mammary tissue; dog. Tubulopapillary carcinoma. Cytoplasmic expression of β-catenin. Formalin-fixed section labeled with the monoclonal antibody β-catenin. LSAB kit, Papanicolaou's hematoxylin counterstain. Bar = 20 µm.
Survival analysis did not reveal any difference in outcome between the preserved versus reduced expression cases (E-cad, P = 0.31, Fig. 5A; β-cat, P = 0.29, Fig. 5B; E-cad/βcat, P = 0.36, Fig. 5C). Grouping the tumors for invasiveness, survival analysis showed a significant difference in outcome only in two out of three comparisons (stage 0 versus stage II, P < 0.05; stage I versus II, P < 0.05; stage 0 versus stage I, P = 0.16; Fig. 5D).
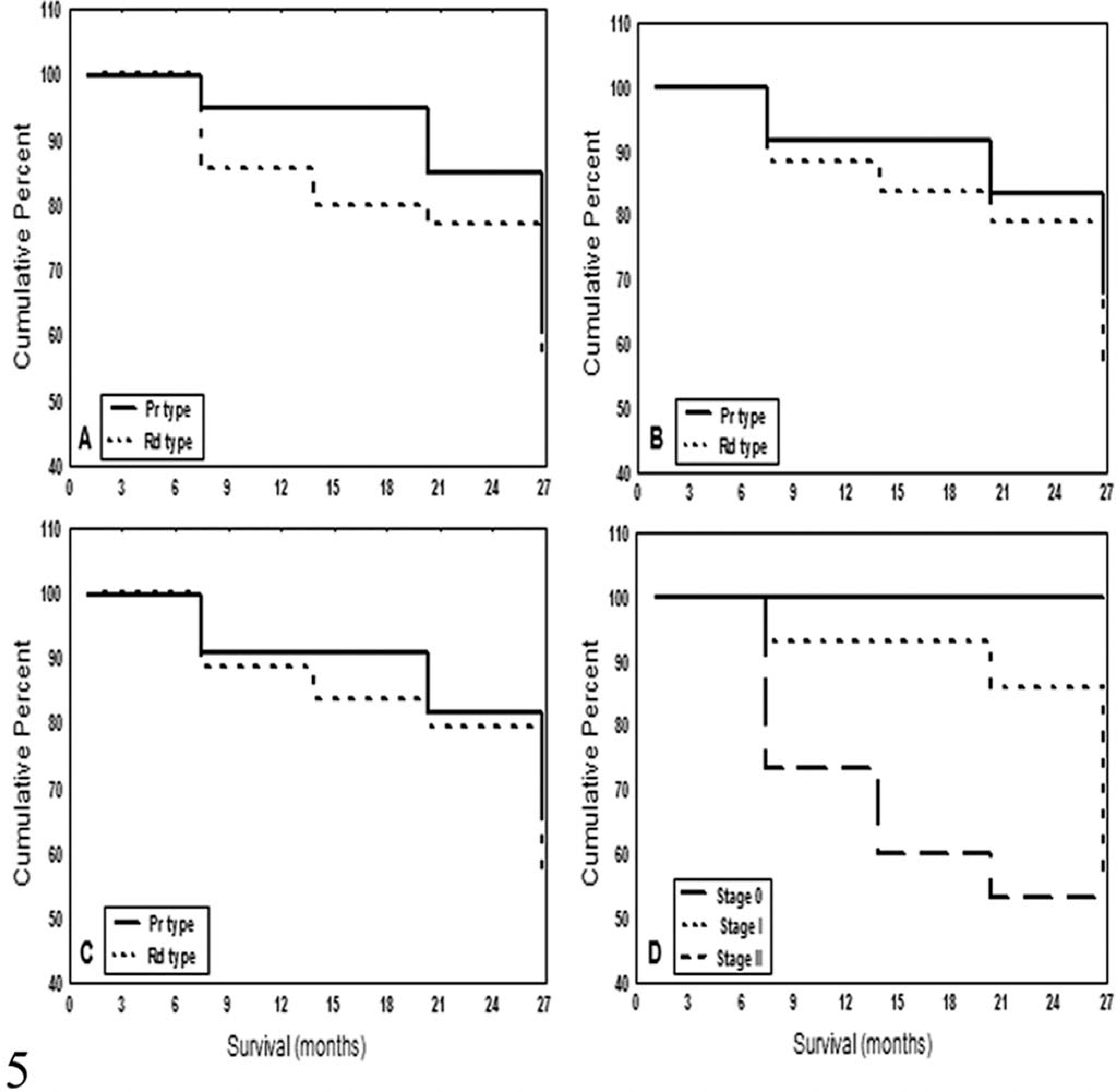
Survival curves and results of survival analysis. Graph A, E-cadherin preserved (22 cases) and reduced (38 cases) groups (P = 0.31); graph B, beta-catenin preserved (12 cases) and reduced (48 cases) groups (P = 0.29); graph C, E-cadherin/beta-catenin complex preserved (11 cases) and reduced (49 cases) groups (P = 0.36); graph D, histologic stage 0 (12 cases), stage I (20 cases), and stage II (19 cases) groups (stage 0 versus stage II, P < 0.05; stage I versus stage II, P < 0.05; stage 0 versus stage I P = 0.16).
Grouping cases for histologic invasiveness, the expression of E-cad or β-cat and E-cad/β-cat showed a progressive reduction that paralleled an increase in invasiveness from noninfiltrating, stage 0, to highly infiltrating, stage II tumors (E-cadherin, Pearson's P < 0.001, R = 0.42, Fig. 6; β-cat, Pearson's P < 0.05, R = 0.31, Fig. 7; E-cad/β-cat, Pearson's P < 0.05, R = 0.32, Fig. 8).
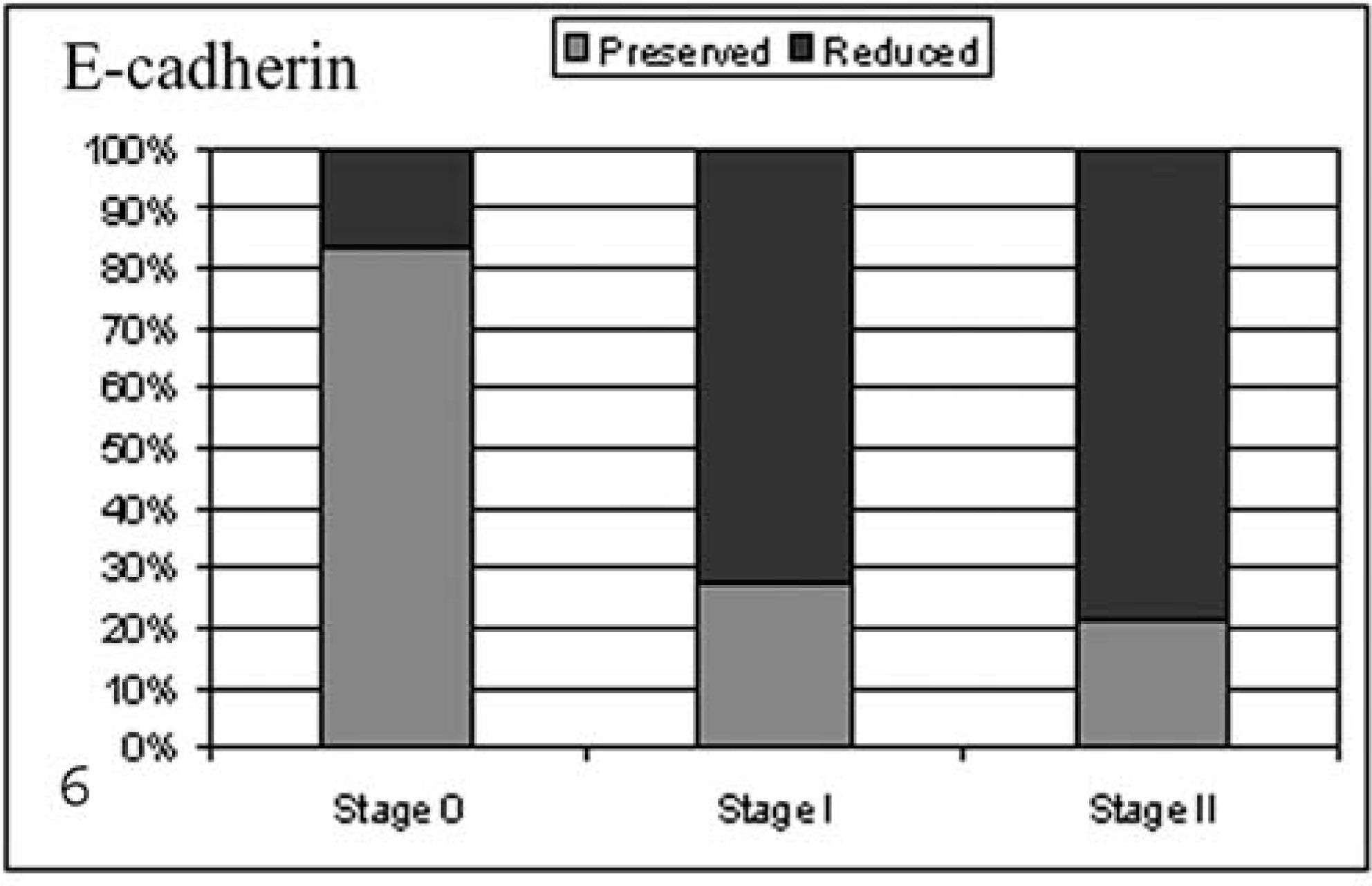
Percentage of preserved and reduced E-cadherin expression in stage 0, I, and II tumors.
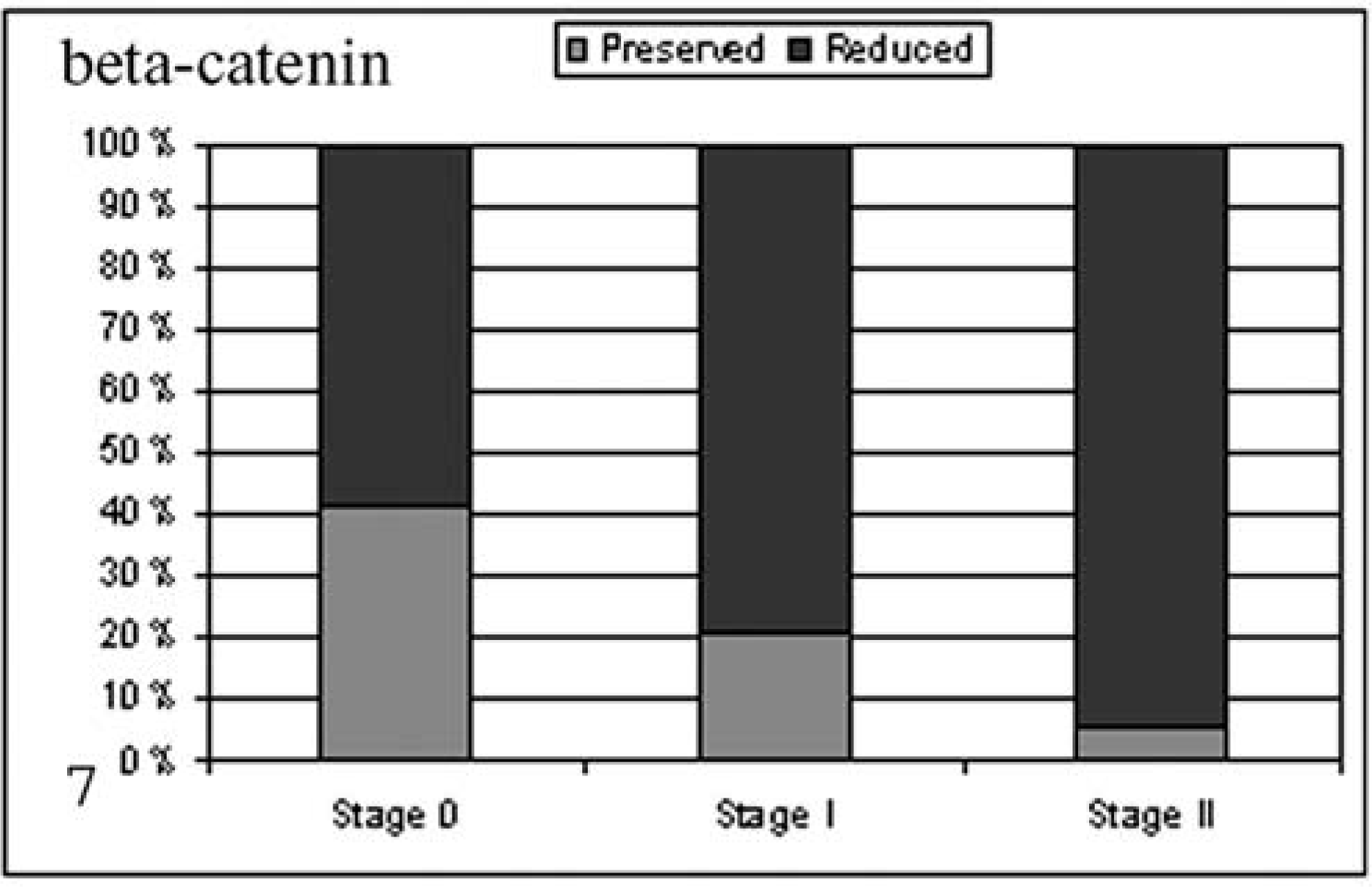
Percentage of preserved and reduced β-catenin expression in stage 0, I, and II tumors.
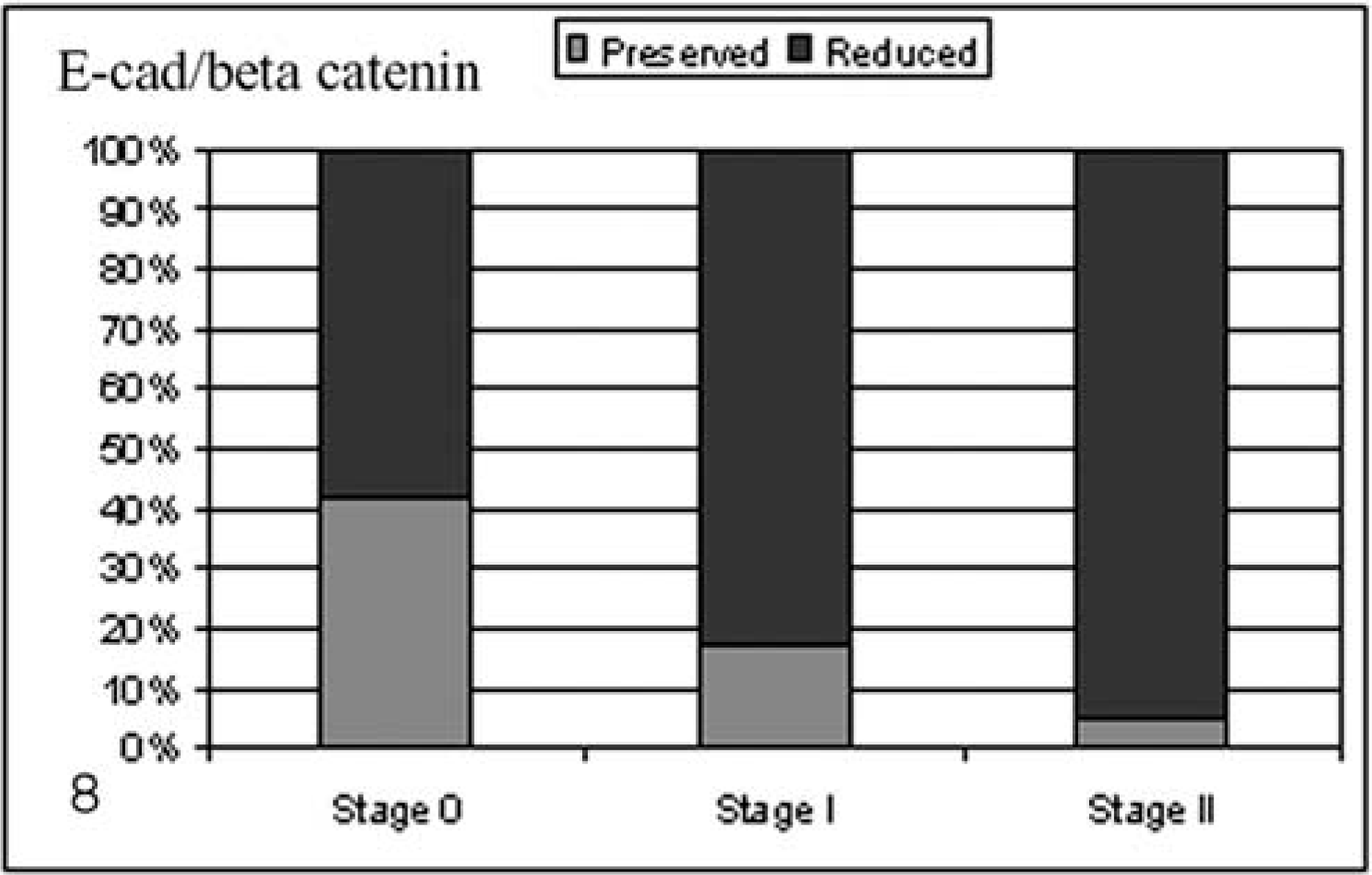
Percentage of preserved and reduced E-cad/β-catenin expression in stage 0, I, and II tumors.
No significant difference was obtained comparing the available proliferation indices (mitotic index, MIB 1 index, and AgNOR index) by one-way ANOVA between the cases grouped for preserved or reduced E-cad, β-cat, and E-cad/β-cat variables (Table 1).
Mean, standard deviation andP values (ANOVA) of the proliferation indices grouped as for preserved or reduced E-cad, β-catenin and E-cad/β-catenin expression.
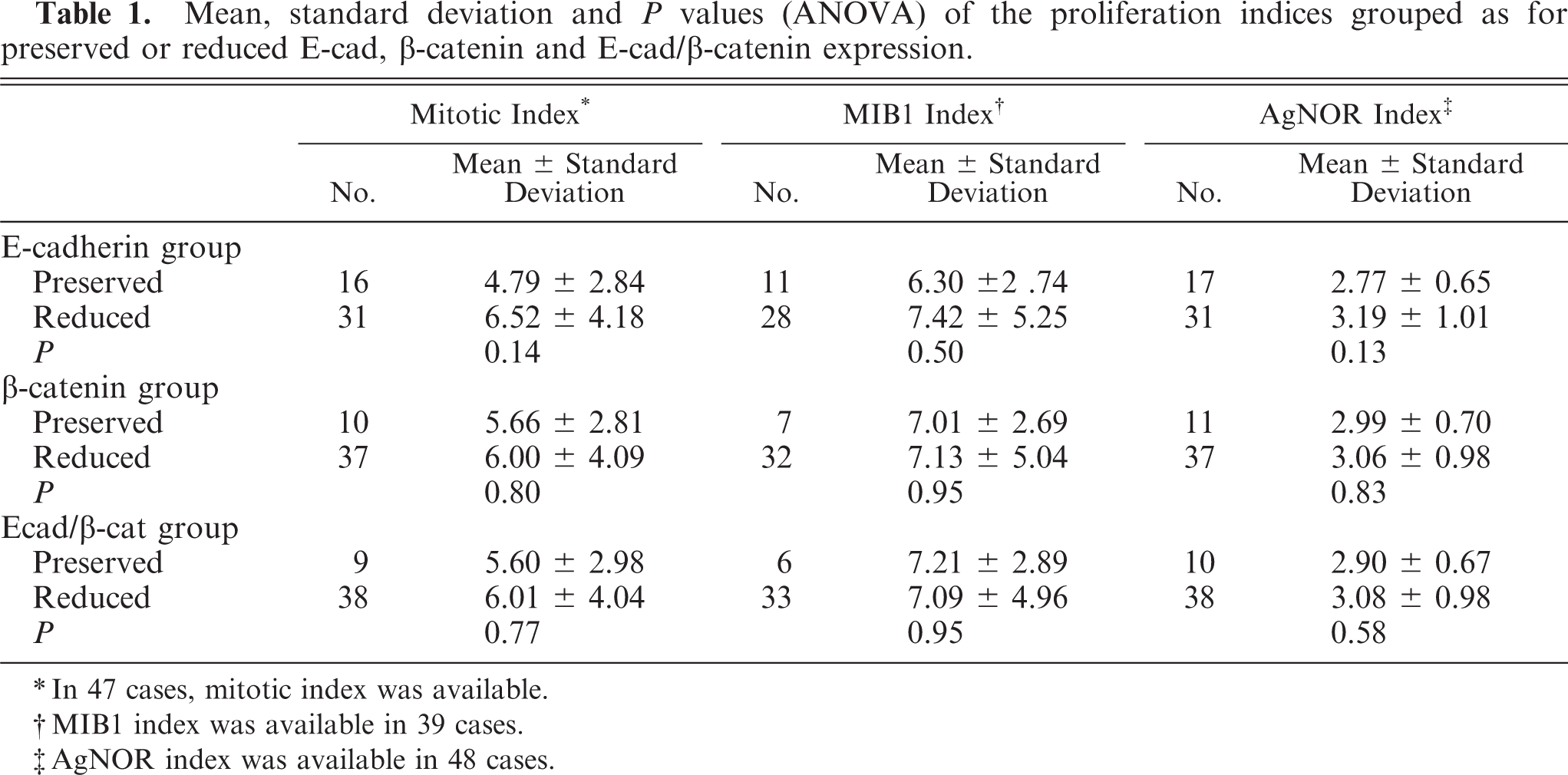
∗In 47 cases, mitotic index was available.
†MIB1 index was available in 39 cases.
‡AgNOR index was available in 48 cases.
Discussion
The process of invasion in canine mammary tumors involves the surrounding tissue or the regional lymph node and the production of distant metastases. Invasion is an important prognostic variable 21 and is included in grading systems suggested for canine mammary tumors. 7, 10
Several reports exist in canine pathology on E-cad and related molecules (catenins) and their role in influencing invasion and metastasis. 1, 2, 4, 11, 12, 19, 20, 22 Our data show that E-cad and β-cat downregulation seems to parallel the acquisition of the invasive phenotype but does not correlate with proliferation. Furthermore, they are poor prognostic variables in view of the results on survival.
In human medicine, the literature is scant and controversial. Some authors claim that E-cad and β-cat reduction is correlated with an increase in invasiveness and proliferation. In bladder lesions, a high AgNOR was correlated with decreased E-cad expression in stromal- and muscle-invading tumors, indicating a possible aggressive behavior. 13 Reduced E-cad expression is also associated with invasiveness and unfavorable prognosis in breast cancer 24 and strong immunostaining with MIB-1. 3 Shabani et al. 23 reported a direct correlation between E-cad expression and high MIB-1 index in metastatic brain tumors. In patients with hepatocellular carcinoma treated by orthotopic liver transplantation, high MIB-1 index, lower membrane expression of E-cad and β-cat, and nuclear β-cat localization were associated with disease recurrence. 5 Decreased expression of E-cad and β-cat in prolactinoma predicted high MIB-1 expression, indicating a higher tendency to invasiveness and proliferative status. 17
In contrast, other authors found that reduced E-cad and β-cat expression had no influence on invasiveness or prognosis. No correlation was found between invasivity and E-cad and β-cat expression in hepatocellular carcinomas, 15 and no relationship was observed between Ki67 staining and the E-cad mutation status in gastric carcinoma. 6 A study on gastric adenocarcinoma with lymph node metastasis tested the expression of E-cad, β-cat, Ki67, and CD44, finding that E-cad and β-cat expression is more probably related to tumor morphology than tumor progression. 9 However, a comparative analysis of the various studies is difficult because they consider different organs, lesions, and neoplasia and utilize only some common parameters.
Our previous investigation on the relationship between E-cad expression in primary canine malignant mammary tumors and their respective node metastases indicated that E-cad expression is not always parallel in both sites, showing conditions of downregulation and upregulation in lymph node metastases. Moreover, E-cad expression was unrelated to survival. 2
These findings support the contention that a reduction in E-cad or β-cat or both is mainly associated with cell adhesion loss with an obvious influence on invasion, even if the influence of these molecules on survival does not seem to be correlated to proliferative activity. On the basis of the results of the present study, E-cad and β-cat reduction is not correlated to an increase in proliferative activity, unlike human breast cancer, in which reduced E-cad correlated with a strong MIB1 immunostaining. 3 In contrast, medullary breast carcinomas in humans showed paradoxically strong E-cad and β-cat expression correlated to strong MIB1. 3
In conclusion, the reduced expression of E-cad and β-cat in canine mammary tumors seems to be related to increased invasiveness but not to proliferation and has a scant influence on survival.
Footnotes
Acknowledgements
This work was supported by grants from Alma Mater Studiorum-University of Bologna (60%, 2001 and 2002).
