Abstract
A presumptive case of metronidazole toxicity in a 3.4-kg adult cat is described. The cat had been treated for suspected inflammatory bowel disease with an anti-inflammatory dose of prednisone and metronidazole (73.5-147 mg/kg PO q24h) for approximately 40 days prior to presentation. Clinical signs were primarily related to the central nervous system, including acute tetraparesis, unresponsiveness, tremors, and vocalization. The patient was euthanatized after 12 days of supportive care. Necropsy revealed no significant macroscopic lesions. Histologic evaluation revealed multifocal, fairly well-demarcated foci of necrosis in the brainstem, extending from the diencephalon to the medulla oblongata. To our knowledge, this is the first report to document histologic lesions associated with metronidazole administration in a cat.
Metronidazole (Flagyl, G. D. Searle & Co., Chicago, IL) is a nitroimidazole antibiotic that is commonly used in veterinary medicine to treat a wide variety of conditions, including anaerobic bacterial infections,
10,
17
protozoal infections (e.g., giardiasis),
10,
17
A 3.4-kg, 14-year-old, spayed female domestic short-haired cat was presented to the University of Minnesota, Veterinary Medical Center (U of MN VMC) for an acute onset of weakness, ataxia, and tetraparesis. The cat had been treated with prednisone (1.5 mg/kg PO q24h) and metronidazole hydrochloride (G. D. Searle) (73.5–147 mg/kg PO q24h), for presumptive IBD, for approximately 40 days prior to presentation. Physical examination revealed hypothermia (97.7°F) and tachypnea (60 breaths·min). The patient was comatose and tetraparetic. Cranial nerve examination revealed bilaterally miotic pupils, positional nystagmus, slow direct and consensual pupillary light responses, and lack of a menace response; the function of all other cranial nerves was normal. Peripheral reflexes were decreased in all limbs, with the presence of a cross-extensor reflex in the hind limbs. The complete blood cell count and coagulation profile showed no abnormalities. Venous blood gas showed the presence of severe respiratory alkalosis with compensatory metabolic acidosis.
The metronidazole was discontinued upon hospitalization. Following stabilization in the intensive care unit, the cat was treated with IV fluids and dexamethasone (0.4 mg/kg SQ q24h) for 5 days. Clinical improvement was not seen during hospitalization. At home, the cat received approximately 10 days of supportive care, including syringe feedings, physical therapy, and urinary bladder expression as needed. The cat became anorexic, vomited, and was again admitted to the U of MN VMC. Following IV fluid therapy (physiologic saline), feedings via nasoesophageal tube, metoclopramide (0.3 mg/kg IM q8h), and prednisone (0.7 mg/kg PO q12h), the owners took the cat home for supportive care. The cat became stuporous overnight, and the owners elected to have the cat euthanatized.
At necropsy, no significant macroscopic lesions were observed in any organ, including the brain and cranial cervical spinal cord. Tissue samples of the lung, heart, liver, kidney, urinary bladder, spleen, thyroid glands, adrenal glands, small and large intestine, eyes, brain, and cervical spinal cord were fixed in 10% neutral-buffered formalin and embedded in paraffin. Sections cut at 4 μm were stained with hematoxylin and eosin (HE); additional sections of the brain were stained with Luxol fast blue, periodic acid–Schiff (PAS), and Bielschowsky silver. Aerobic bacterial cultures of the lung, liver, and spleen yielded no growth.
The clinically significant microscopic findings were restricted to the CNS. In sections taken at multiple sites in the brainstem, there were several fairly well-demarcated areas of necrosis and increased cellularity, affecting central brainstem regions, beginning at the level of the diencephalon and extending caudally throughout the medulla oblongata (Fig. 1). The areas of necrosis did not appear to be symmetric. The reticular formation of the central brainstem was predominantly affected, and the adjacent major tracts and nuclei of the brainstem were generally spared. The necrotic foci affected the medullary and pontine reticulospinal tracts and the rubrospinal tract. Within the necrotic foci, there was no apparent preservation of neurons, astrocytes, or oligodendrocytes. The normal, uniform, eosinophilic-staining neuropil was replaced by pale-staining, necrotic neuropil with fine basophilic granularity. Luxol fast blue highlighted the necrosis as fairly well-demarcated, pale gray, poorly staining foci (Fig. 2). These areas contained a marked increase in the number of macrophages and microglial cells (especially gitter cells) (Fig. 3); many contained phagocytosed, PAS-positive granular material and some golden brown pigment. There were occasional eosinophilic ellipsoids and small, round, dark eosinophilic globules of degenerative myelin scattered throughout the affected neuropil and increased prominence of the capillaries, often with slightly swollen endothelial cells. Moderate numbers of variably sized, clear vacuoles were present in the neuropil, especially at the interface of areas with increased gitter cells and the normal neuropil. Many large-to-swollen phagocytic cells contained variable amounts of blue, phagocytosed myelin at the margins of the lesions (Fig. 4). Bielschowsky silver stain detected scant axonal preservation in the necrotic foci, suggesting a demyelinative component in addition to the more destructive nature of the overall pathologic process (Figs. 5, 6).
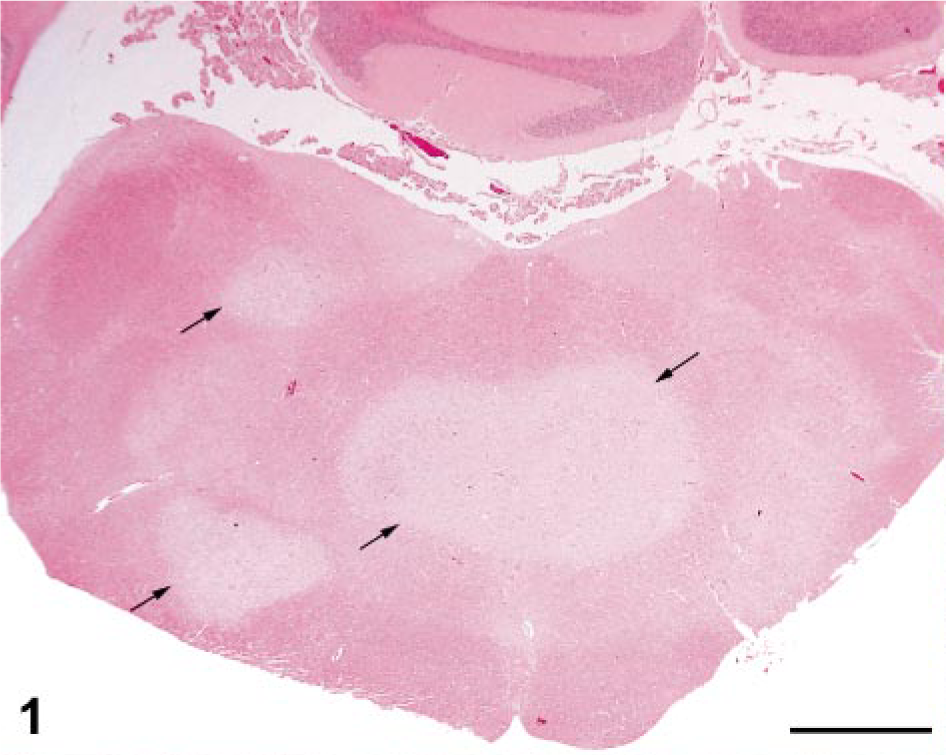
Brainstem (caudal medulla oblongata); cat. Note well-demarcated foci of necrosis (arrows). HE. Bar = 2 mm.
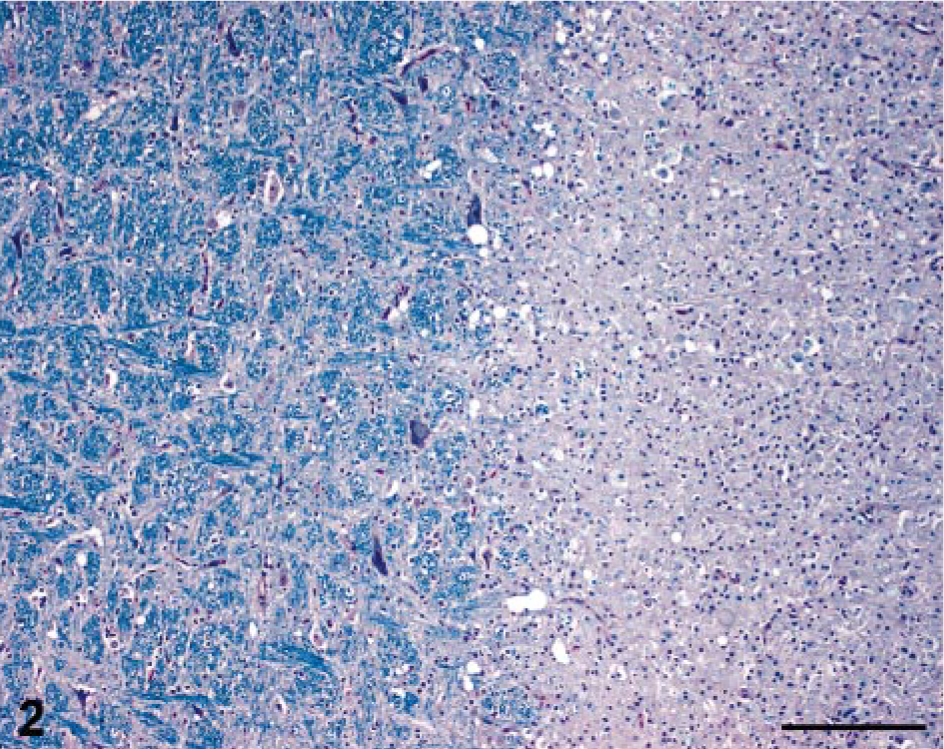
Brainstem; cat. Note well-delineated separation between the normal myelin (left) and the loss of myelin staining in the affected areas (right). Luxol fast blue. Bar = 200 μm.
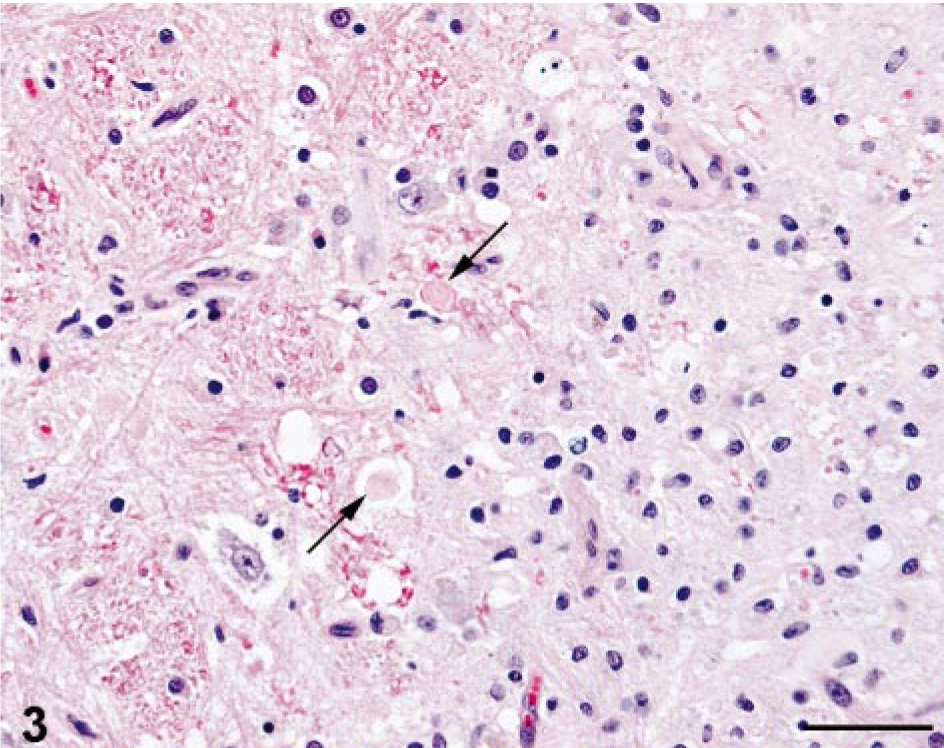
Brainstem; cat. Typical lesions at interface of normal and affected areas. Occasional swollen axons (arrows) admixed with focal increase in glial cells and the loss of neuropil uniformity. Note increased prominence of capillaries. HE. Bar = 50 μm.
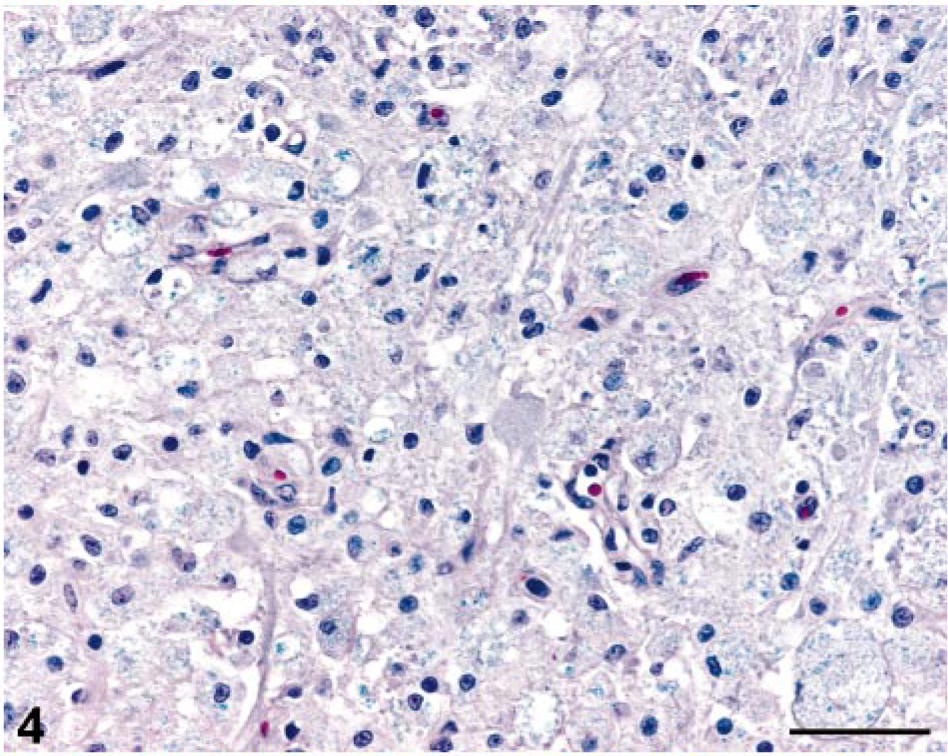
Brainstem; cat. Large macrophages with phagocytosed myelin. Luxol fast blue. Bar = 50 μm.

Brainstem; cat. Note well-delineated separation between the normal density of axons (top) and the relative paucity of axons in the affected areas (bottom). Bielschowsky silver. Bar = 200 μm.
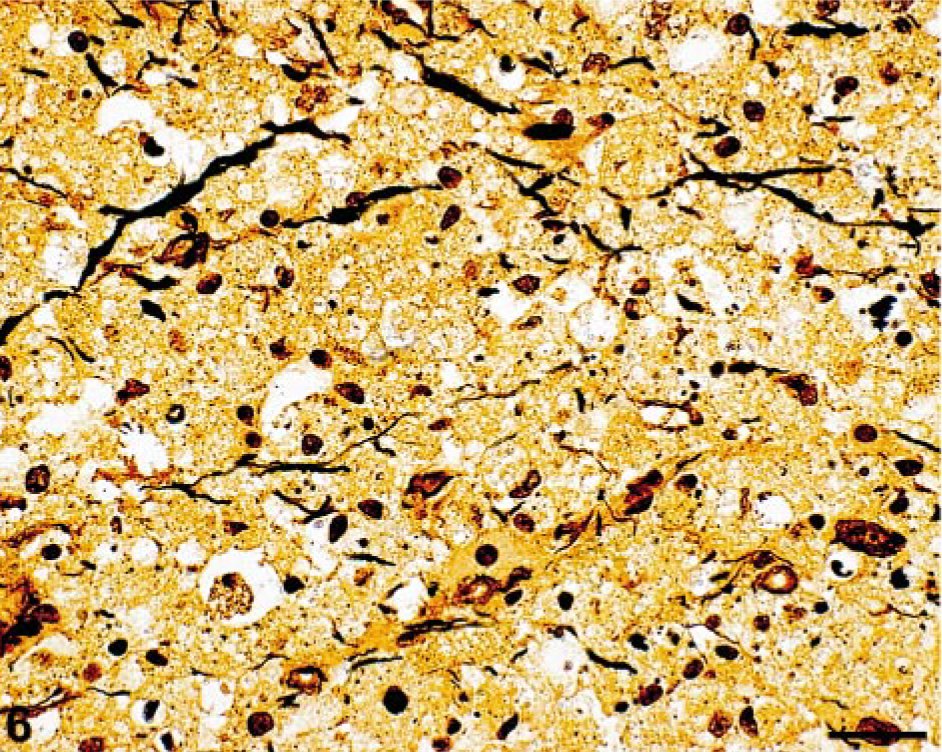
Brainstem; cat. Some axons are preserved in the affected areas. Bielschowsky silver. Bar = 50 μm.
In veterinary medicine, the most commonly reported side effects of oral administration of metronidazole include lethargy, anorexia, vomiting, and diarrhea, 5 in addition to salivation/ptyalism in cats. 10, 17 However, undesirable side effects on the CNS have been reported in both natural cases and experimental studies of metronidazole toxicosis in rodents, 19 dogs, 5, 6, 8, 21, 25 and cats. 4, 20
In 5 dogs with metronidazole toxicosis, drug doses ranged from 67 to 129 mg/kg/d administered for 3–14 days. 5 These dogs showed acute signs of CNS dysfunction such as generalized ataxia and vertical or positional nystagmus. The neurologic signs often followed anorexia and intermittent vomiting. Two of these dogs were euthanatized because of severe neurologic dysfunction, whereas the remaining 3 dogs recovered slowly after several months. 5 Metronidazole toxicosis has also been reported in a Plotthound-cross dog that received 89.5 mg/kg/d for approximately 5 weeks. 8 This dog presented with progressive ataxia, nystagmus, knuckling, and intermittent periods of paddling and disorientation; the neurologic signs resolved within 2 weeks of discontinuing the drug. 8
Metronidazole neurotoxicity has been previously reported in 5 cats. 4, 20 In one report of reversible CNS toxicosis associated with metronidazole therapy in three cats, one cat received 48 mg/kg/d for 10 months prior to presentation. The metronidazole dosage for the second cat was 62.5 mg/kg/d for 1 month, and the third cat received 62 mg/kg/d for 5 days. In another report of feline metronidazole toxicity in two cats, one cat received 111 mg/kg/d for approximately 8 weeks, followed by an increase to 222 mg/kg/d for 4 days, and the other cat had been given a metronidazole dose of 58 mg/kg/d for 6 months. The neurologic abnormalities included ataxia, blindness, disorientation, hyperactivity, vertical nystagmus, altered mentation, and seizures. In all five cats with metronidazole toxicosis, the neurologic signs resolved within several days to 1 week of withdrawing the drug and with supportive care.
In the present report, the cat had been on metronidazole therapy (73.5–147 mg/kg/d) for 40 days with a total cumulative dose of metronidazole ranging from 10.0 to 20.0 g. Metronidazole drug dosage and duration were within the range of previous reports of metronidazole toxicosis in dogs and cats.
Toxicologic studies conducted in the early 1970s described the effects of high doses of metronidazole on the brains of rats. 19 Histologically, these studies showed well-demarcated, often symmetric lesions in the vestibular and cochlear nuclei, rostral colliculus, and cerebellar and olivary nuclei after administration of high doses (800 mg/kg) of metronidazole. The CNS lesions in the rats were characterized by spongiform changes (cytoplasmic vacuolation) within the neurons.
In an experimental toxicity study, Schärer 21 found clinical signs associated with CNS dysfunction in dogs given 4–6 doses of metronidazole at 250 mg/kg/d. These dogs showed muscle spasms of the hind limbs and lumbar muscles, weakness, dorsiflexion of the tail, and opisthotonus, with eventual death after prolonged, high-dose administration. Histologic examination of brain specimens showed degeneration (swelling, vacuolation, and clumping of protoplasm) and selective loss of Purkinje cells. These dogs all had clinical neurologic abnormalities within 4–6 days and prior to the development of histologic lesions in the brain; most of the dogs died within 1 week after the onset of clinical signs. 21
Similar histopathologic changes were noted in a retrospective study of five dogs treated with metronidazole. 5 Two of the five dogs were euthanatized because of severe neurologic dysfunction. One dog had mild swelling of axon sheaths in the vestibular-cerebellar white matter tracts, and another dog had bilateral, but asymmetric, areas of leukomalacia near the radix of the vestibular nerve. 5
Histologic lesions in the CNS of the cat of this case report are similar to those reported in rodents and dogs. 5, 19, 21 On the basis of numerous sections that were studied, the anatomic distribution and characteristics of the lesions are consistent with those associated with metronidazole neurotoxicosis. To our knowledge, none of the previous case reports of metronidazole toxicosis in cats have included histopathology, as all of these feline patients recovered after discontinuing the drug.
There are relatively few publications addressing the mechanism of metronidazole neurotoxicity. It is thought that the intermediate metabolites of metronidazole are able to bind to and disrupt cellular deoxyribonucleic acid (DNA) and induce cell death in anaerobic microorganisms. 3, 25 In mammalian cells, it has been proposed that metronidazole and/or its metabolites may bind to ribonucleic acid (RNA) instead of DNA. 3, 4, 14 This RNA binding may inhibit RNA and/or protein synthesis, potentially leading to axonal degeneration. 3, 4, 14, 25 Using a rodent auditory model, investigators have also demonstrated that the severity of the lesions associated with metronidazole toxicity may be modulated by the level of functional brain activity (neurons). 18 In humans, reversible changes have been observed in the brains of patients with metronidazole-induced encephalopathy by magnetic resonance imaging. Ahmed et al. 1 suggested that the reversible changes associated with the acute toxic effects of metronidazole are most likely due to axonal swelling with increased water content rather than due to a demyelinating process. Another proposed mechanism involves vascular spasm that could produce mild reversible localized ischemia. 1 The mechanism of action of metronidazole neurotoxicosis also may involve the modulation of the inhibitory neurotransmitter γ-aminobutyric acid (GABA) receptor within the cerebellar and vestibular systems. 6 In support of this hypothesis, it has recently been demonstrated that diazepam, a benzo-diazepine with major effects on GABA neurotransmitters in the brain, dramatically improved the recovery times for dogs with metronidazole toxicosis. 6
The majority of reported cases of metronidazole neurotoxicosis in cats and dogs have resolved following the discontinuation of drug administration. All five cats of the previous case reports recovered completely within 1 week after metronidazole therapy had been discontinued. 4, 20 The slow clinical improvement after the cessation of metronidazole administration in the cat of this article is not typical of previous reports of feline metronidazole toxicosis. Although most veterinary metronidazole toxicosis patients improved within 5–7 days after discontinuation of the drug, weeks and sometimes months may be required to completely resolve the neurologic effects. 10 Thus, the time course to resolution of neurologic clinical signs is variable and likely multifactorial.
Previous case definitions of metronidazole toxicosis in veterinary species have been based on a history of having received metronidazole, showing compatible clinical signs, having an absence of other clinical disease, and experiencing the eventual recovery of the animal upon discontinuation of the drug. 6 It is not known if this cat's neurologic dysfunctions would have resolved as time progressed, because the cat was euthanatized within 10 days of discontinuing the drug. Clinically, hypothermia, tachypnea, respiratory alkalosis, and cranial nerve involvement were compatible with the distribution of brainstem lesions in this cat.
In the cat of this study, there was no evidence of neoplasia, inflammation, or vascular disease in any of the brain sections examined. Nutritional deficiencies are not likely, as the cat of this case report had been fed a commercial diet. The history of metronidazole administration, the clinical signs, and the absence of other clinical disease are supportive of the presumptive diagnosis of metronidazole neurotoxicosis. Furthermore, the type and distribution of the CNS lesions are consistent with both natural and experimental cases of metronidazole toxicosis in other veterinary species. The pathogenesis of metronidazole neurotoxicity is not currently known, and the cat of this study may represent an idiosyncratic reaction to the drug. Thus, to our knowledge, this is the first case report documenting histologic lesions in the brain of a cat in association with metronidazole therapy.
Footnotes
Acknowledgements
We thank Drs. Deb Nielsen, Nancy Scott, and Michelle Wells for submission of this clinical case and for providing clinical information. We are grateful to Dr. Brent Clark (Department of Laboratory Medicine and Pathology, University of Minnesota, Minneapolis, MN) and to Dr. Tom Fletcher (Department of Veterinary and Biomedical Sciences, University of Minnesota, St. Paul, MN) for technical support. We also thank Dr. Arno Wünschmann (Minnesota Veterinary Diagnostic Laboratory) for his translations of the German references.
