Abstract
Leukoencephalomyelopathy of undetermined etiology has been described in specific pathogen-free cats. A study was established to assess if the long-term feeding of a gamma-irradiated diet could induce this disease. Cats fed exclusively on diet irradiated at 25.7–38.1 kGy (“typical” dose) and 38.1–53.6 kGy (high-end dose), respectively, developed typical lesions with attendant, progressively severe ataxia between study days 140 and 174. The onset of ataxia at day 140 and the number of animals affected at this time were similar in animals fed each ration. A maximum ataxia “score” was first reached by an animal on the high-end dose diet on day 167 and by 2 cats fed the “typical-end” dose diet 21 days later. Ataxic cats and 1 animal euthanized on day 93 prior to the onset of ataxia exhibited varying degrees of Wallerian degeneration in the spinal cord and brain, similar to the spontaneous disease. The elevated total antioxidant status of spinal cord segments and hepatic superoxide dismutase concentration of cats fed typical and high-end treated diets suggested free-radical involvement in the pathogenesis. The significantly elevated peroxide concentrations of the irradiated diets (1,040% and 6,440% of untreated values) may have resulted in increased oxidative insult, a factor possibly exacerbated by the treated diets' reduced vitamin A content. This study has reproduced leukoencephalomyelopathy in cats similar to spontaneous outbreaks by feeding a gamma-irradiated dry diet with elevated peroxide and reduced vitamin A concentrations.
Introduction
Progressive hindlimb ataxia associated with leukoencephalomyelopathy has been described in colonies of specific pathogen-free (SPF) cats in Ireland5, Britain,34 and New Zealand.19 Although the etiology of this disease remains undetermined, circumstantial evidence suggests an association with the long-term exclusive feeding of gamma-irradiated dry diet. This is based on the fact that irradiation of the diet represented a significant difference in the management of affected and nonaffected cats.5
Ataxia with white matter degeneration is reported in lions,28, 32 cheetahs,33 cats,20, 26 English Foxhounds,41 Landrace-cross pigs,31 rats,11 and nonhuman primates11, 36, 47 where deficiencies in vitamins A, B12 (cobalamin),41 B3 (nicotinamide),31 B6 (pyridoxine),47 and B1 (thiamine)11, 20, 26, 36 have been implicated. In affected cats26 and rats,11 the thiamine content of the diet was between 0.1 and 0.3 and 0.1 and 0.5 mg/kg,11 respectively, whereas no thiamine was present in the diet fed to nonhuman primates.36
Ionizing gamma-radiation has been used safely for over 40 years to prolong the shelf life, to improve the microbiologic safety, and to reduce the use of chemical fumigants and additives in human and animal feedstuffs.25, 40, 42 The effect of the irradiation on the food is dependant on the dose of radiation applied measured in grays (Gy). Low doses of up to 1 kGy are used to delay the ripening of fresh foodstuffs, whereas medium doses of up to 10 kGy prolong the shelf life of food.40 At doses greater than 10 kGy, this technology has been widely used to sterilize diets for SPF and germ-free animals,25, 42, 46 although such treatment can reduce the diet's vitamin content.8, 13, 25, 39, 40, 42 In pure solution the B vitamin thiamine (B1) is the most sensitive to the effects of radiation followed by vitamin C, pyridoxine (B6), riboflavin (B2), and niacin. Vitamin E is the most radiosensitive of the fat-soluble vitamins, followed by vitamins A and K. However, these sensitivities vary significantly when vitamins are incorporated within food products.25, 40 Irradiation of a feline diet containing 9.8% fat with a 20–50 kGy dose totally destroyed its vitamin A and β-carotene content while thiamine, pyridoxine, and folic acid were depleted to a lesser extent and the vitamin E concentration was unaffected.8 The fat component of the diet is particularly sensitive to ionizing radiation16 that can induce auto-oxidizing and hydrolytic reactions,48 leading to loss of essential fatty acids and vitamins.10 Where dietary fat contains high levels of polyunsaturated fatty acids, gamma-irradiation results in peroxidation of the unsaturated bonds,38 and, more specifically, the irradiation of dry cat diets significantly increases peroxides and decreases the vitamin A content.7, 13
Investigation of naturally occurring “outbreaks” of leukoencephalomyelopathy in a colony of SPF cats provided circumstantial evidence incriminating the exclusive feeding of a gamma-irradiated diet, given that the only significant difference in the management of affected and unaffected animals was the feeding of such a diet and that no further cases occurred once supplemental or replacement pasteurized diets were introduced.5 Therefore, a controlled feeding trial was designed to determine the potential toxicologic effects of the long-term feeding of gamma-irradiated dry cat food at 2 different gamma-irradiation doses, a “typical” dose of 25.7–38.1 kGy and a “high-end” dose of 38.1–53.6 kGy.7 These irradiation doses were chosen as 20–30 kGy is the dose most frequently used to treat diets intended for SPF colonies, whereas the higher dose of 40–50 kGy is typically used to treat diets intended for germ-free animals, where absolute sterility is essential.8, 25, 46 The neurologic status of the cats was carefully monitored throughout the study, and, on euthanasia, necropsy and biochemical assessment of the antioxidant status of tissues was performed.
Materials and Methods
Animal selection and study design
Thirty-one clinically and neurologically normal, male SPF cats of between 6 and 14 months of age and of between 3.1 to 6.6 kg bodyweight were randomized into 4 treatment groups. Male animals were used as investigation of naturally occurring cases did not indicate any effect of sex in disease development.5 Cats were individually identified by a subcutaneous electronic implant (Avid PLC., East Sussex, UK), and prior to vaccination and study commencement, 8 animals were serologically screened for antibodies to the following pathogens: Feline Infectious Peritonitis virus, Feline Corona virus, Feline Viral Rhinotracheitis virus, Feline Panleukopaenia virus, and chlamydiae (European Veterinary Laboratory test kits, Worden, Germany); Feline Immunodeficiency virus, Feline Leukaemia virus, Bordetella bronchiseptica, Salmonella spp., Campylobacter spp., Pseudomonas aeruginosa, and Streptococcus pneumonia (Surrey Diagnostics, University of Surrey, Guildford, Surrey, UK); and Toxoplasma gondii (MAST Diagnostics, Bootle, UK).
For the 9 months prior to the study all the cats were held in a SPF barrier facility and were fed the same dry cat diet as that used in the study but which had been pasteurized. On removal from this facility, all animals were vaccinated (Nobivac Tricat) and group-housed in a conventional animal housing unit in 4 pens, 1 containing the 7 control cats (group 1) and the other 3 each containing 8 animals (groups 2, 3, and 4). All experiments were conducted in compliance with animal experimentation legislation (Statutory Instrument Number 556 of 2002 incorporating EC Directive 86/609/EC).
In group 1, the 7 control cats were fed a complete dry diet for adult cats (Gilpa Umami) to appetite at 1 kg per pen per day. The 8 group 2 animals were fed the same diet in the same way, except the diet had previously been gamma-irradiated at 25.7–38.1 kGy (the typical irradiation treatment) (Isotron, Westport, Ireland). The 8 group 3 cats were also fed the same complete dry diet for adult cats previously gamma-irradiated at 38.1–53.6 kGy (the high-end irradiation treatment) (Isotron). Subsequent to the onset of neurologic deficits in both group 2 and 3 cats at day 140, a further 8 animals (group 4) commenced the study with a treatment regime similar to group 3. Two of these animals were euthanized at days 93 and 100 of the study, respectively, to assess if histopathologic lesions were present prior to animals becoming ataxic. The 4 remaining animals in this group were euthanized on study day 174. A single batch of the dry cat diet was used for the entire study to eliminate batch variability.
Clinical examination and neurological deficit scoring
Food consumption per pen was measured daily, and cats were weighed at the beginning of the study (study day 0), and on days 35, 84, 98, 111, 133, 166, and 193. Clinical examination and semiquantitative scoring of neurologic deficits were carried out on each cat on day 0 and subsequently at least weekly until study completion. Scoring of hindlimb neurologic deficits was as follows: score 0, normal posture and hindlimb position; score 1, posture abnormal with mild hindlimb imbalance and abduction, animal quite mobile; score 2, moderate hindlimb imbalance and abduction with swaying of hindlimbs; score 3, severe hindlimb imbalance and abduction with pronounced swaying of hindlimbs; score 4, paresis of hindlimbs, animal immobile.
Examinations were carried out without knowledge of treatment group. Once cats became ataxic they were examined daily, and animals were selected for euthanasia and necropsy at particular stages of disease development (Table 1). On reaching a neurologic deficit score of 4 (where locomotor deficit compromised welfare), animals were immediately euthanized.
Numbers of cats with varying degrees of hindlimb ataxia as assessed by semiquantitative neurologic scoring at sequential time points following the long-term exclusive consumption of typical (group 2) and high-end (groups 3 and 4) dose gamma-irradiated diets.
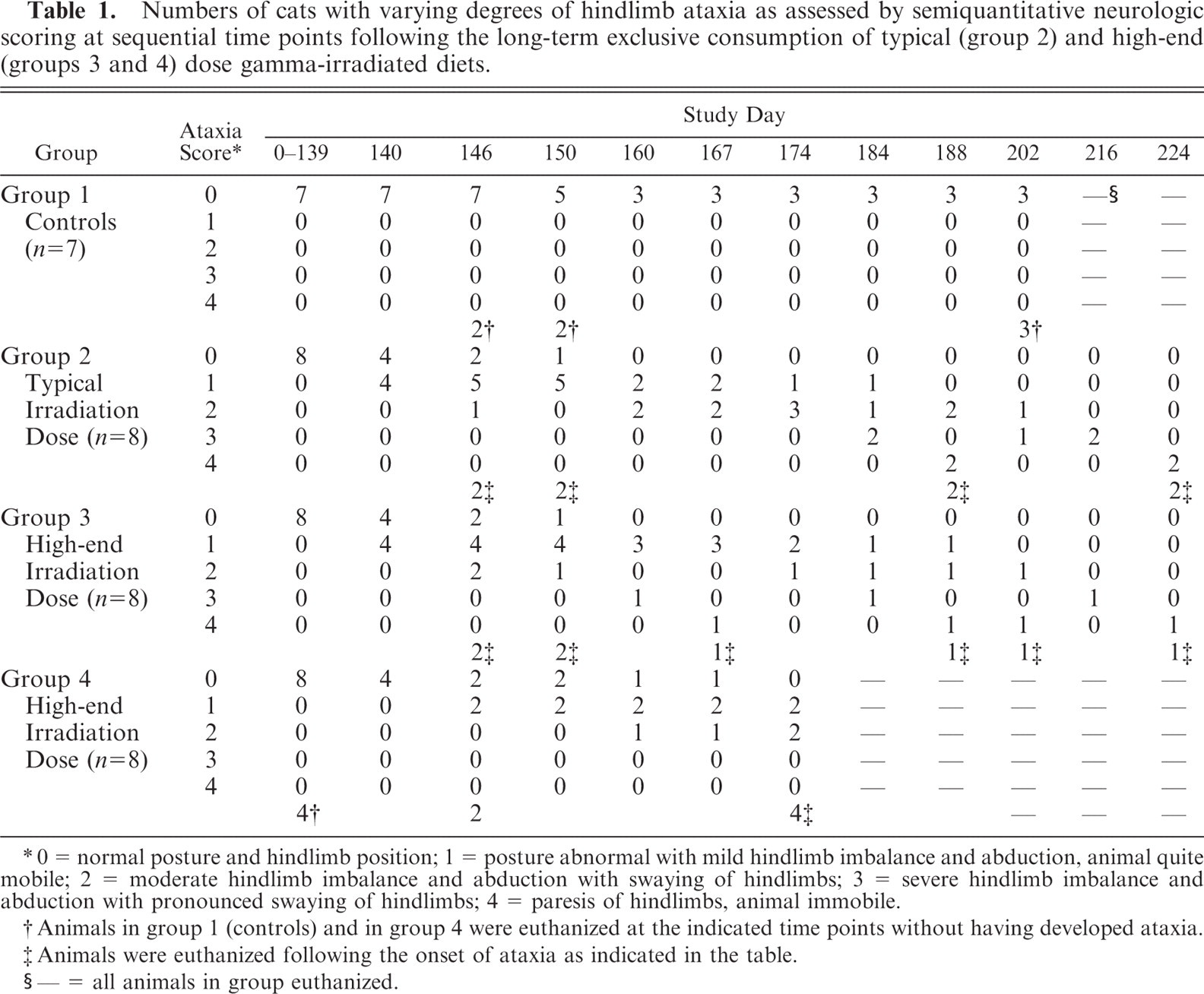
0 = normal posture and hindlimb position; 1 = posture abnormal with mild hindlimb imbalance and abduction, animal quite mobile; 2 = moderate hindlimb imbalance and abduction with swaying of hindlimbs; 3 = severe hindlimb imbalance and abduction with pronounced swaying of hindlimbs; 4 = paresis of hindlimbs, animal immobile.
Animals in group 1 (controls) and in group 4 were euthanized at the indicated time points without having developed ataxia.
Animals were euthanized following the onset of ataxia as indicated in the table.
— = all animals in group euthanized.
Blood samples for clinical chemistry and for hematology were taken prior to the start of the study and on the day of euthanasia from each cat.
Necropsy
Prior to euthanasia by intravenous sodium pentobarbitone (Chanelle Veterinary Ltd., Loughrea, Galway, Ireland), cerebrospinal fluid (CSF) and urine samples were collected, where possible, from each cat under terminal anaesthesia. Detailed postmortem examinations were performed on all animals. Brain, spinal cord (including cervical, thoracic, and lumbar segments) and samples of sciatic nerve, vastus lateralis muscle, liver, kidney, myocardium, lungs, thyroid and adrenal gland, spleen, small intestine, abdominal fat, and gonads were fixed by immersion in 10% neutral buffered formalin. Following fixation, tissues were dehydrated and embedded in paraffin wax. All sections were stained with hematoxylin and eosin for histopathologic examination. Unfixed samples of brain (cerebrum, midbrain, and hindbrain) and spinal cord (cervical, thoracic, and lumbar) regions and of liver from each animal were snap frozen in liquid nitrogen and stored at −80°C for further analysis.
Histopathologic examination of the tissue sections was carried out by 2 veterinary pathologists (JPC and ACP) independent of each other and without prior knowledge of treatment group. A semiquantitative scoring system was developed to grade the degree of injury in brain and spinal cord regions to facilitate statistical analysis of the histopathologic changes over time and between groups and to correlate the degree of injury with observed neurologic deficits. Wallerian degeneration was graded as 1 (very mild, ±), 2 (mild, +), 3 (moderate, ++), or 4 (severe, +++). If no injury was observed a 0 (−) was registered. Also, to facilitate analysis, euthanasia time points were categorized as early (study days 93–100), mid (study days 146–174), and late (study days 188–224).
Tissue total antioxidant status and superoxide dismutase analysis
Separate, 100-mg homogenized samples of thawed brain (cerebrum, midbrain, and hindbrain regions), spinal cord (cervical, thoracic, and lumbar segments), and liver from each animal were placed in Eppendorf tubes with 400 μl of phosphate buffered saline (PBS) and centrifuged at 14,170 × g for 10 minutes at 4°C (Mikro22R refrigerated centrifuge). The supernatant was transferred to a fresh Eppendorf tube and recentrifuged at 14,170 × g for 10 minutes at 4°C. The resultant homogenate was diluted in PBS. Brain and spinal cord samples were diluted 1:5 and 1:500 for the total antioxidant status (TAS) and superoxide dismutase (SOD) analyses, respectively. Liver samples were diluted 1:100 and 1:1,000 for these assays, respectively.
Commercially available kits (Total Antioxidant Status and Ransod, Randox Laboratories Ltd., Co., Antrim, UK) were used to measure tissue TAS and SOD status of the samples. Both assays were modified to suit a 96-well plate reader format, and standard curves were generated for both parameters prior to sample analysis. The TAS assay is based on the ability of antioxidants in the sample to suppress color production by the radical cation 2,2′-azino-di-(3-ethylbenzthiazoline sulphonate) relative to a standard (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid) in proportion to their concentration. Tissue sample (20 μl) as prepared above, or standard, and 100 μl of chromagen were mixed in each well of a 96-well plate, incubated at 37°C for 1 hour, and the resulting absorbance (A0 value) was read at 600 nm using a plate reader. Substrate (20 μl) was then added to each well for 5 minutes. The reaction was terminated by adding 10 μl of 50% HCl and the absorbance (A1 value) read. Standard values were plotted on a graph of log10 concentration versus change in absorbance (A1 − A0). Sample values were calculated and expressed in mmol/mg of tissue.
The SOD assay (Ransod, Randox) is based on the capacity of this enzyme to accelerate the dismutation of the toxic superoxide radical (O2 •−) to hydrogen peroxide and molecular oxygen. Xanthine and xanthine oxidase generate superoxide radicals that react with 2-(4-iodophenyl)-3-(4-nitrophenol)-5-phenyltetrazolium chloride to form red formazan dye. Superoxide dismutase activity is measured by the rate of inhibition of dye formation (1 unit [U] of SOD is that which causes 50% rate inhibition). Tissue sample (20 μl) as prepared above, or standard, and 170 μl of mixed substrate were mixed in each well of a 96-well plate, incubated at 37°C for 1 hour, and the resulting absorbance (A0 value) was read at 505 nm. Xanthine oxidase (30 μl) was added to all wells for 5 minutes. Following termination of the reaction by adding 10 μl of 50% HCl, the absorbance (A1 value) was read. Standard values were plotted on a graph of log10 concentration versus change in absorbance (A1 − A0). Sample values were calculated and expressed in U/mg of tissue.
Analysis of diet
Dietary constituents of the dry cat ration were analyzed: prior to irradiation; after 1 exposure to the typical (25.7–38.1 kGy) gamma-irradiation dose; and after 1 exposure to the high-end (38.1–53.6 kGy) gamma-irradiation dose) as previously described (Eclipse Scientific Group, Cambridge, UK).1, 2, 12, 22 A previous study indicated the relative stability of the micronutrient composition of the diet up to 36 days postirradiation.6
Statistical analysis
The significance of the histopathologic semiquantitative scores between the groups were analyzed statistically using a Kruskal-Wallis test followed, where appropriate, by the Mann-Whitney test. Significance in differences in TAS was analyzed using Dunnett's (2-sided) test.
Results
Neurologic deficit scoring
All cats in groups 2 and 3 fed exclusively on the typical and high-end gamma-irradiated diets, respectively, developed hindlimb ataxia between study days 140 and 174. Table 1 illustrates when and to what degree (score) cats presented with hindlimb deficits. The onset of ataxia on day 140 and the number of animals affected (4) were similar in both groups 2 and 3. Animals in all 3 treatment groups (2, 3, and 4), exhibited ataxia by day 174, the severity of which increased progressively over time for each cat. The first animal to present a maximum score of 4 was in group 3 on day 167. Two animals presented with this score in group 2 on day 188. No neurologic deficits were found in the control group at any time. The day on which euthanasia and necropsy was carried out was varied in order to examine the CNS at a range of time points and in animals with varying neurologic deficits. Once cats reached a neurologic deficit score of 4 they were immediately euthanized. Cats in the control and in all treatment groups gained bodyweight throughout the study, regardless of developing ataxia. No significant antibody titers to the previously listed pathogens were found in the 8 cats examined serologically.
Necropsy including histopathologic examination
No gross lesions were observed postmortem. Tables 2, 3, and 4 detail the severity of the Wallerian degeneration in brain and spinal cord regions, as scored semiquantitatively, in the cats fed exclusively on the gamma-irradiated diets. Transverse cross-sections of cervical, thoracic, and lumbar spinal cord from all ataxic cats and from 1 of the 4 cats from group 4 euthanized on day 93 prior to the onset of ataxia exhibited varying degrees of bilateral vacuolation of ventral, lateral, and dorsal white matter funiculi (Figs. 1, 2). The density of vacuolation was greater in peripheral cord regions, particularly in dorsolateral and ventral funiculi, and was somewhat less in the dorsal tracts. Vacuoles variously contained axonal debris, swollen axons, and macrophages or microglial cells containing myelin debris (Fig. 3). These lesions were consistent with Wallerian degeneration with varying degrees of associated astrocytosis and capillary hyperplasia. Longitudinal sections of spinal cord segments revealed myelin ellipsoids, swollen axons, and macrophages or microglial cells containing myelin debris. Similar, bilateral lesions, of varying severity, occurred in the white matter of the cerebrum (Fig. 4), the inferior cerebellar peduncle, and in the external arcuate and pyramidal fibers of the medulla.
Severity of Wallerian degeneration in brain and spinal cord regions scored semiquantitatively (±, very mild; +, mild; ++, moderate; +++, severe; —, no injury observed) in cats fed exclusively on typical (group 2) and high-end (groups 3 and 4) dose gamma-irradiated diets.
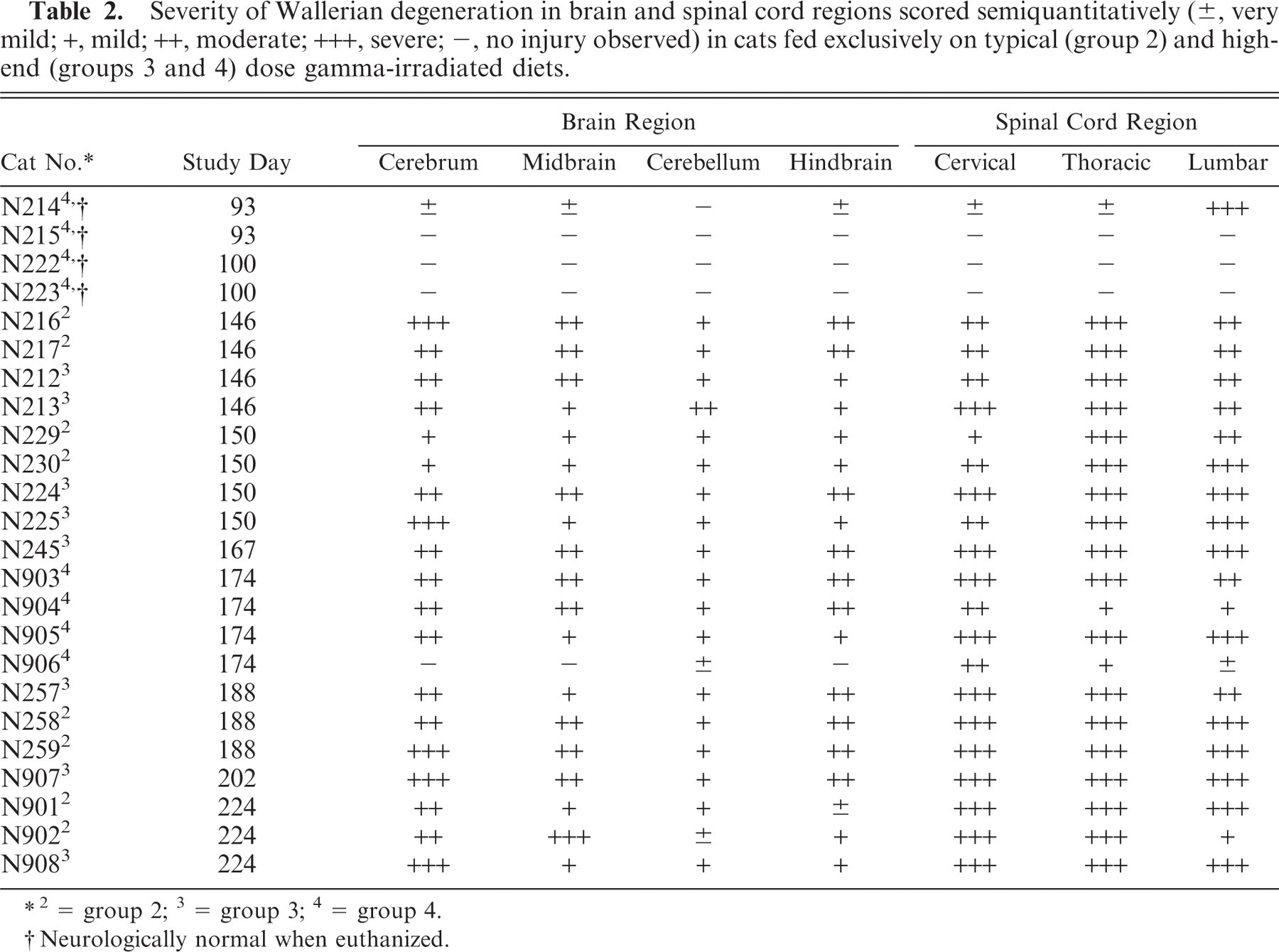
2 = group 2; 3 = group 3; 4 = group 4.
Neurologically normal when euthanized.
Severity of Wallerian degeneration in dorsal (D), lateral (L), and ventral (V) funiculi of cervical, thoracic, and lumbar spinal cord regions scored semiquantitatively (±, very mild; +, mild; ++, moderate; +++, severe; −, no injury observed) in cats fed exclusively on typical (group 2) and high-end (groups 3 and 4) dose gamma-irradiated diets.
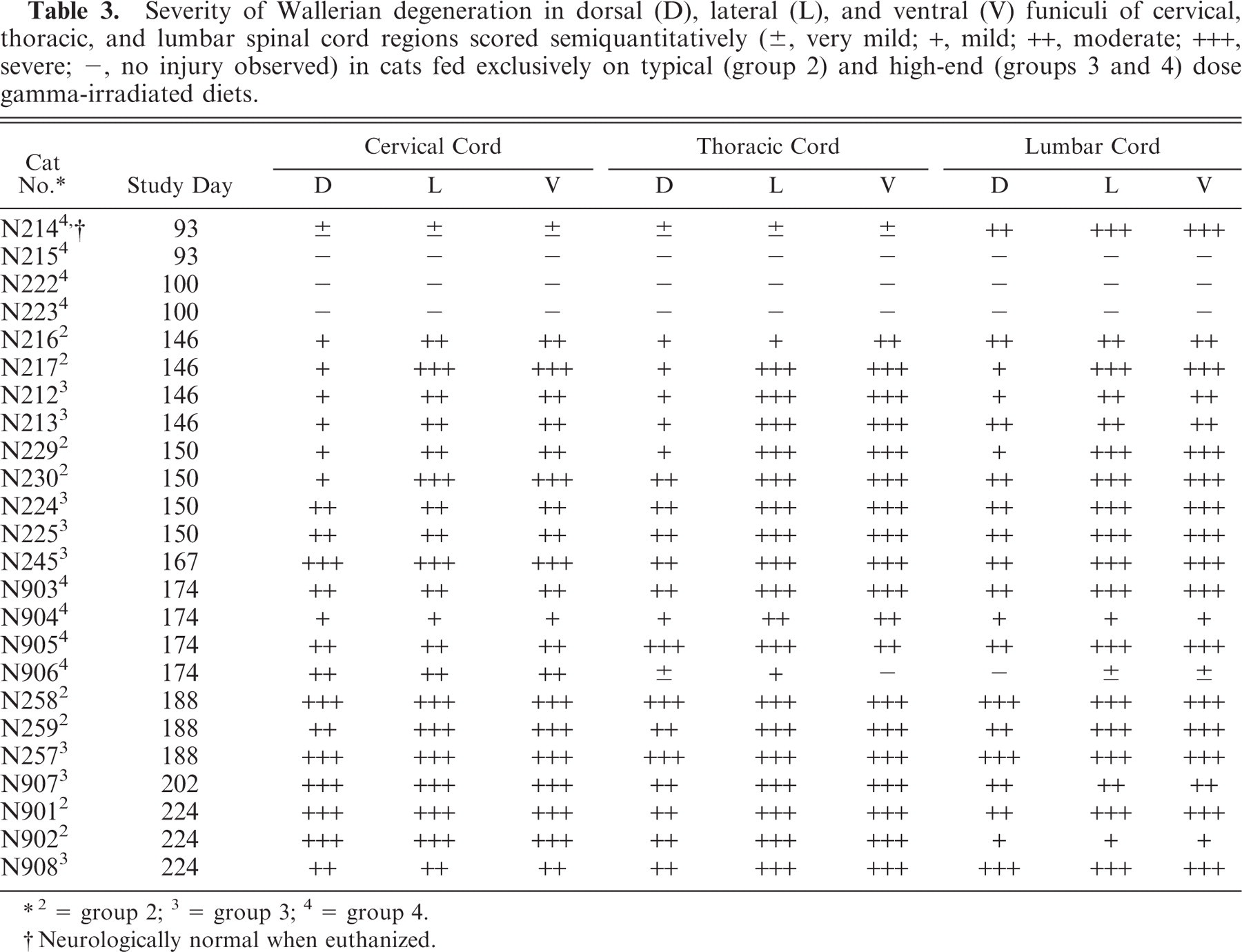
2 = group 2; 3 = group 3; 4 = group 4.
Neurologically normal when euthanized.
Median semiquantitative scores (with interquartile ranges in parenthesis) of brain and spinal cord region Wallerian degeneration in cats fed exclusively on typical (group 2) and high-end (groups 3 and 4) dose gamma-irradiated diets at early (study days 93–100), mid (study days 146–174), and late (study days 188–224) time points.
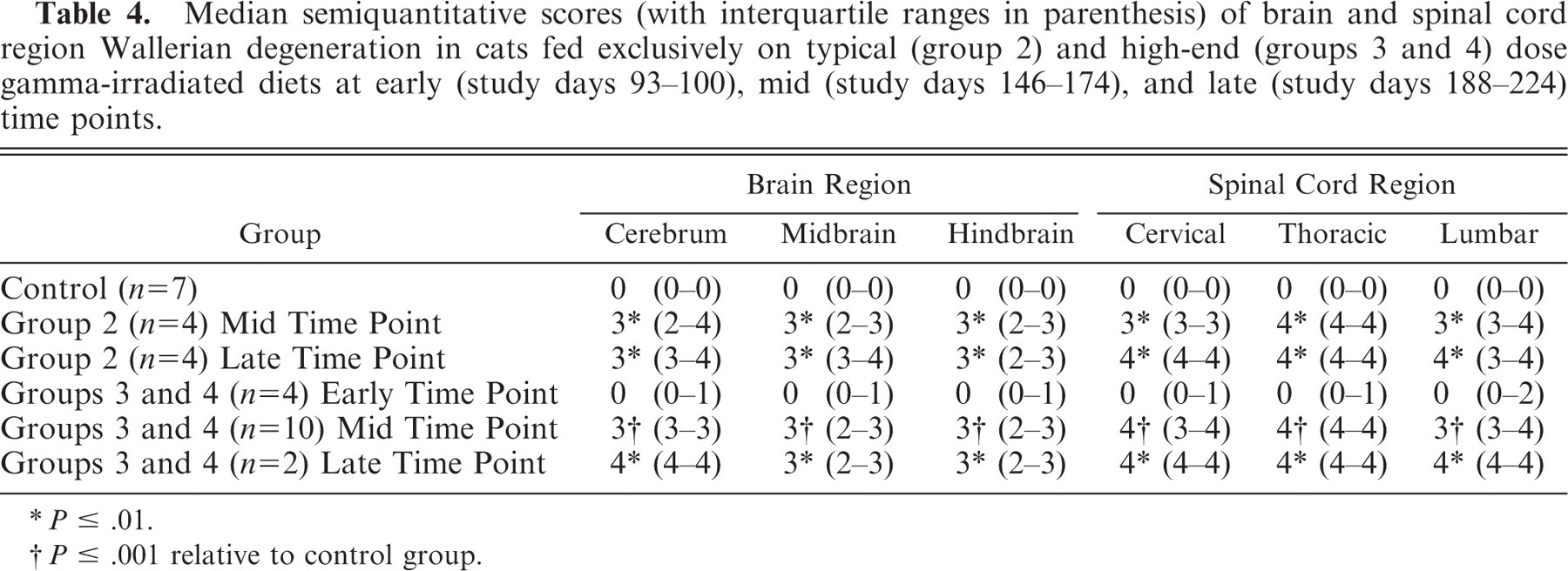
P ≤ .01.
P ≤ .001 relative to control group.
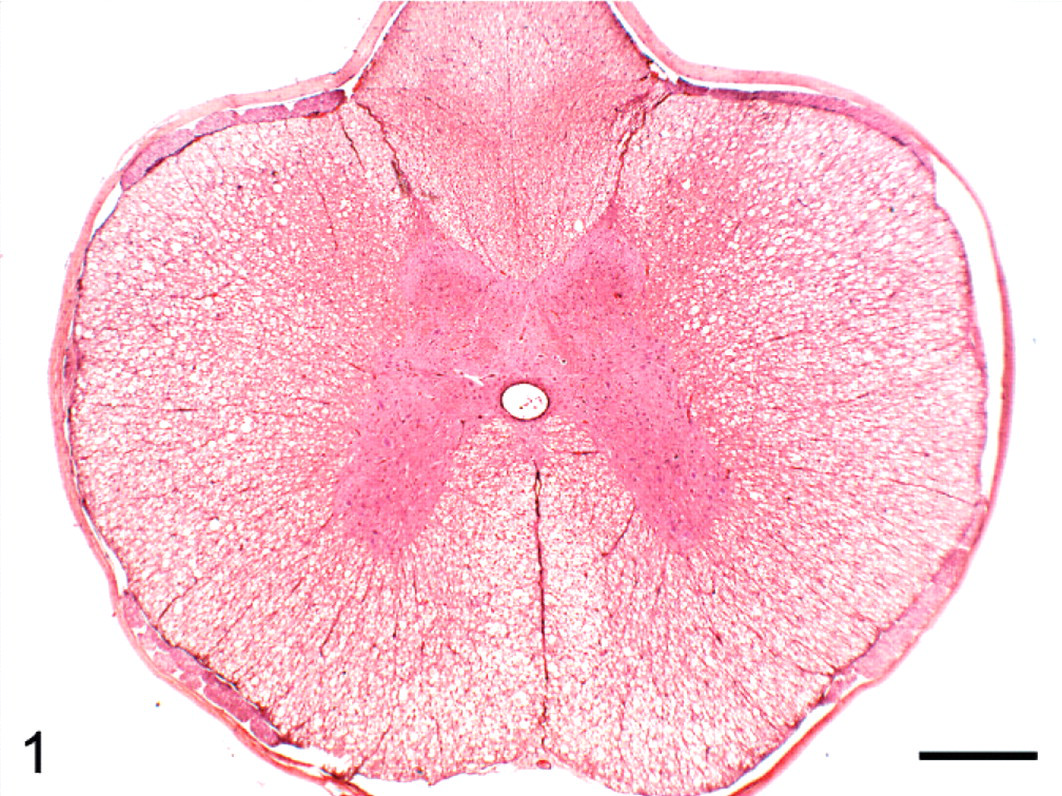
Transverse section of thoracic spinal cord; group 2 cat (typical radiation treatment of diet) euthanized on study day 154. Bilateral vacuolation of ventral and lateral white matter funiculi. HE. Bar = 500 μm.
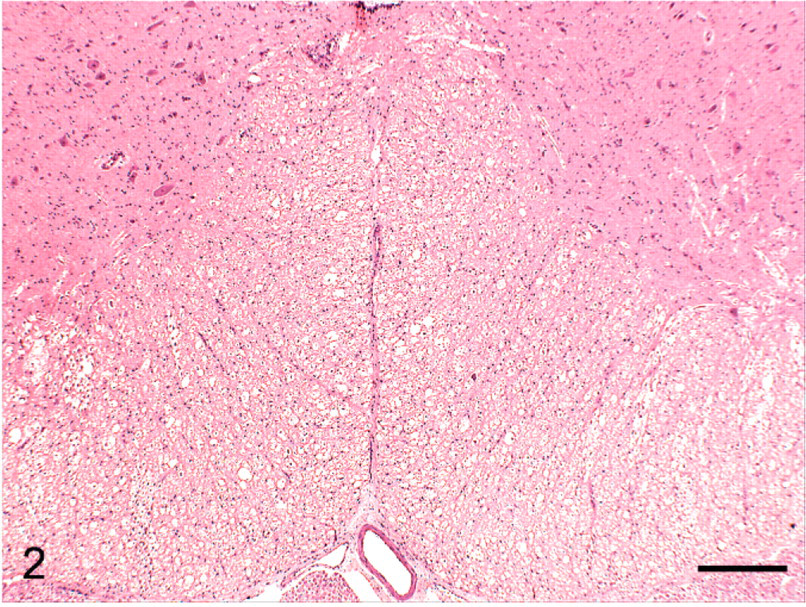
Transverse section of ventral lumbar spinal cord; group 4 cat (high-end radiation treatment of diet) euthanized on study day 93. Extensive white matter vacuolation. HE. Bar = 250 μm.
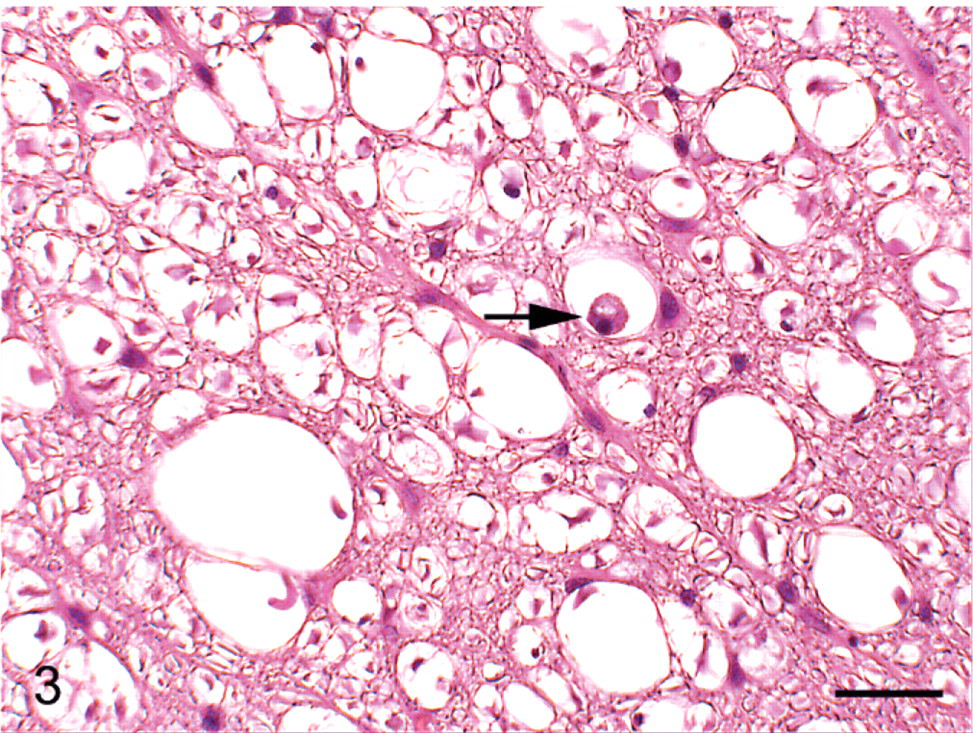
Higher magnification of dorsolateral funiculus in Fig. 1; group 2 cat (typical radiation treatment of diet) euthanized on study day 154. Vacuolation with microglial cell containing myelin debris (arrow). HE. Bar = 25 μm.
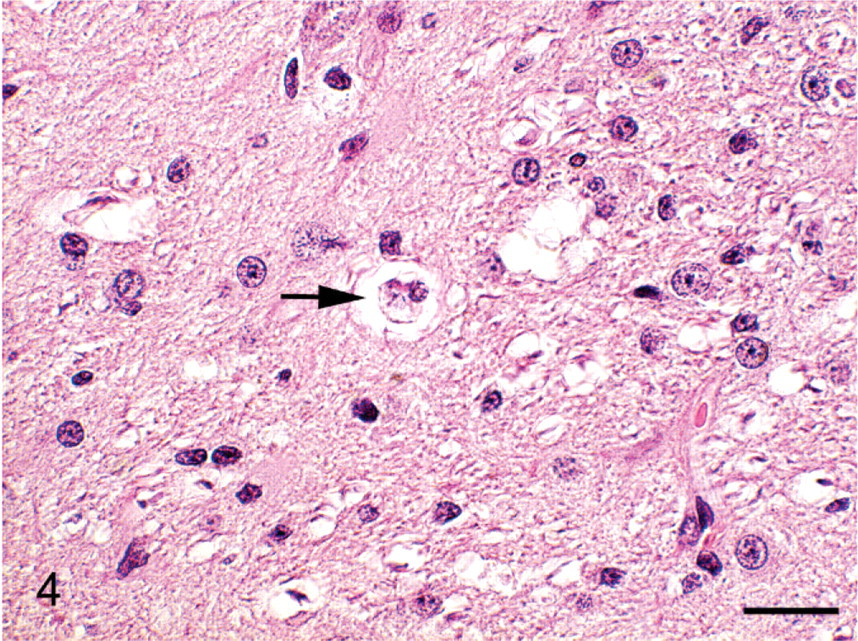
Cerebrum; group 3 cat (high-end radiation treatment of diet) euthanized on study day 170. Microglial cell containing myelin debris within a vacuole (arrow). HE. Bar = 16 μm.
One of the group 4 cats, euthanized on day 93 prior to the onset of ataxia in any animal, exhibited severe (+++) Wallerian degeneration equally affecting dorsal, lateral, and ventral funiculi of the lumbar cord. Much less severe (±) white matter injury was observed in this animal's cervical and thoracic spinal cord regions and in the cerebrum, midbrain, and hindbrain (Table 2). No abnormalities were detected in the other group 4 cats euthanized at this time point or in the 2 animals from this group euthanized at study day 100. Ataxic cats from groups 2 and 3 had relatively similar degrees of bilateral Wallerian degeneration of ventral, lateral, and dorsal white matter funiculi within the cervical, thoracic, and lumbar spinal cord segments and within cerebral, midbrain, cerebellar, and brain stem white matter tracts at each euthanasia time point (Tables 2, 3). In general, lesions were more severe in the spinal cord than in the brain, and the severity of lesions at both locations marginally increased with time in all treatment groups. No significant abnormalities were observed on histopathologic examination of other tissues from animals in groups 2, 3, and 4, and no significant abnormalities were observed in any tissues in the group 1 control cats.
There was a significant difference in the combined semiquantitative histopathologic lesion score of the brain and spinal cord regions between the control group and each of groups 2 (P < .01) and 3 (P ≤ .001) at the mid (P < .01) and late (P ≤ .001) euthanasia time points. No significant difference was found in this score between group 2 and group 3 cats.
Sufficient CSF to calculate the number of nucleated cells and measure the specific gravity could not be obtained from all animals and, where obtained, sufficient quantity was not available to perform both assessments in every case (Table 5). Two cats, 1 from each of groups 3 and 4 and euthanized on study days 154 and 100, respectively, had CSF cell counts above the normal limit (Table 5). No significant abnormalities were found on blood clinical biochemistry, hematology or on urinalysis of cats from the control or treatment groups.
Details of cerebrospinal fluid (CSF) analysis of cats euthanized at the defined times following the exclusive feeding of typical (group 2) and high-end (groups 3 and 4) dose gamma-irradiated diets. CSF could only be obtained from the listed cats and in some cases insufficient fluid was available to carry out all 3 assessments: nucleated cell count, measurement of specific gravity, and microscopic examination of a cytologic smear. ∗
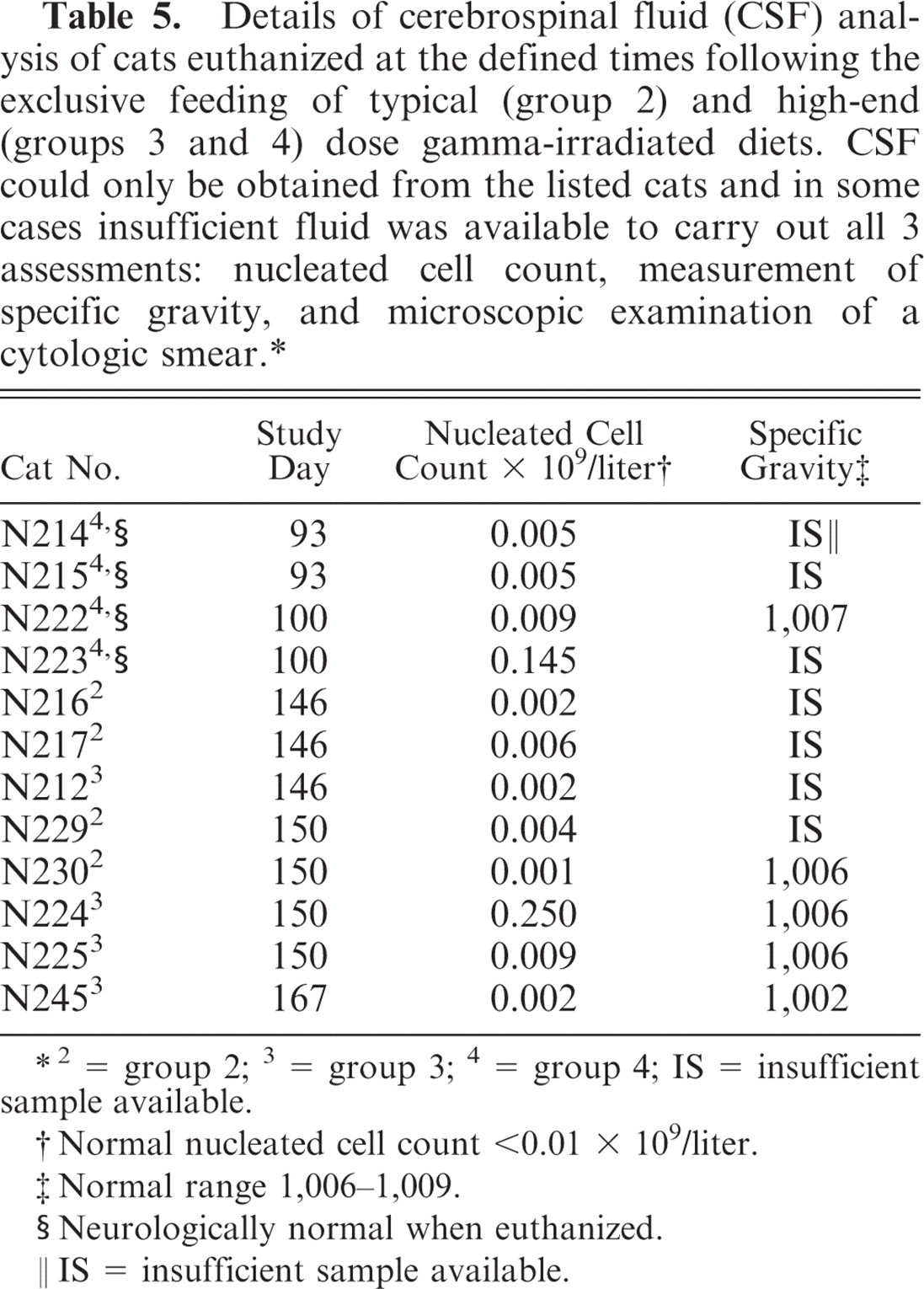
2 = group 2; 3 = group 3; 4 = group 4; IS = insufficient sample available.
Normal nucleated cell count <0.01 × 109/liter.
Normal range 1,006–1,009.
Neurologically normal when euthanized.
IS = insufficient sample available.
Tissue total antioxidant status and superoxide dismutase analysis
Results of tissue TAS and SOD analysis are detailed in Tables 6 and 7. There was a significant difference in the spinal cord segment TAS between control and group 2 cats at the mid and late euthanasia time points for lumbar (P < .05) and for cervical (P < .05) and lumbar (P ≤ .01) segments, respectively. Similarly, there was a significant difference between control and groups 3 and 4 cats at the mid euthanasia time point for thoracic (P ≤ .01) and lumbar (P ≤ .001) segments. There was no significant difference in the TAS of spinal cord segments between the control and groups 3 and 4 cats at the early euthanasia time point. Brain or spinal cord from groups 3 and 4 animals at the late euthanasia time points were not analyzed. There was no significant difference in brain region or liver TAS between control, group 2 and groups 3 and 4 animals at any of the euthanasia time points.
The total antioxidant status (in mmol/mg tissue expressed as means ± standard deviation) of brain and spinal cord regions and in the liver of cats at early (study days 93–100), mid (study days 146–174), and late (study days 188–224) euthanasia time points following the long-term exclusive consumption of typical (group 2) and high-end (groups 3 and 4) dose gamma-irradiated diets.
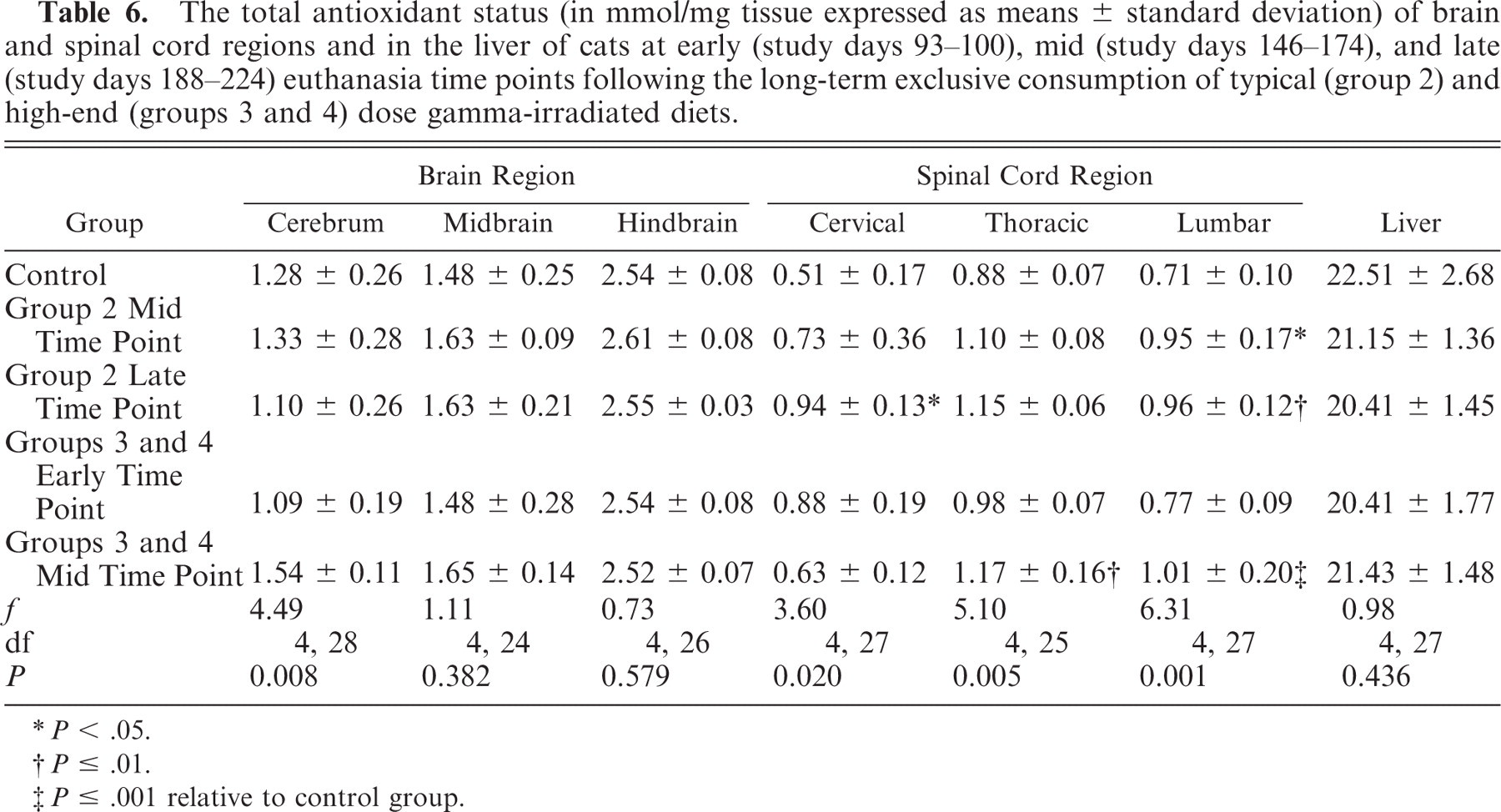
P < .05.
P ≤ .01.
P ≤ .001 relative to control group.
The concentration of superoxide dismutase (in U/mg tissue expressed as means ± standard deviation) in brain and spinal cord regions and in the liver of cats at early (study days 93–100), mid (study days 146–174), and late (study days 188–224) euthanasia time points following the long-term exclusive consumption of typical (group 2) and high-end (groups 3 and 4) dose gamma-irradiated diets.
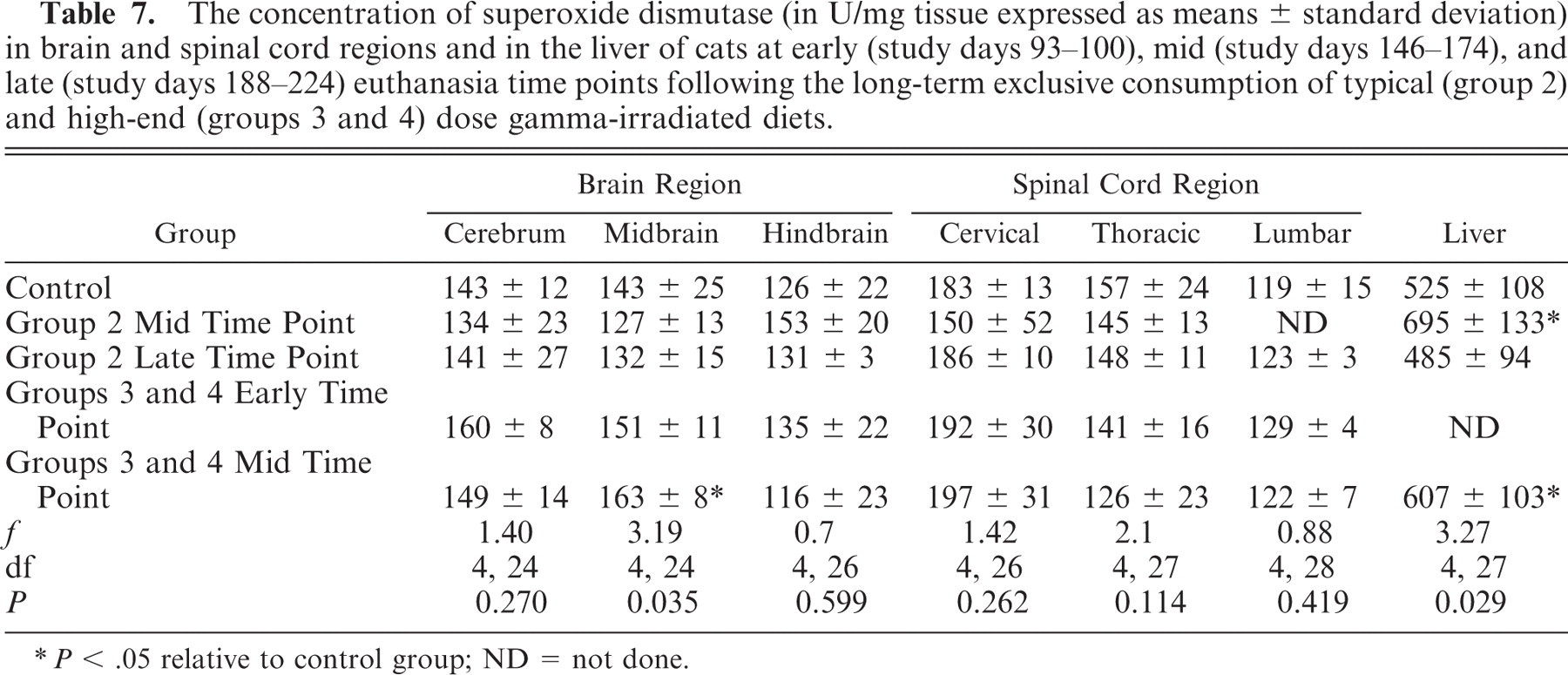
P < .05 relative to control group; ND = not done.
There was a significant difference at the mid euthanasia time point in midbrain SOD levels between control and groups 3 and 4 cats (P < .05). Also at this time point there was a significant difference in liver SOD levels between control and group 2 and between control and groups 3 and 4 combined (P < .05).
Diet analysis
Table 8 details the concentrations of particular dietary nutrients prior to and postirradiation at the typical and high-end doses. The minimum recommended concentration of vitamins and fat per kilogram dry matter content of diet for growth and reproduction in cats is included.3 The vitamin A content of the diet was reduced to 43% and 48% of its untreated value 24 hours postirradiation at the typical and high-end doses, respectively. The fat and protein content of the dry cat diet was unaffected by either radiation dose. Peroxide levels in the diet increased to approximately 1,040% and 6,440% of the untreated value (<0.5 mequiv/kg) 24 hours postirradiation at the typical and high-end doses, respectively. The concentrations of vitamins E, B1, and B6 were reduced to a minor extent by both typical and high-end radiation treatments (Table 8).
Concentrations of vitamins, fat, and peroxide in 500 g samples of dry cat diet were determined following gamma-irradiation at typical (25.7–38.1 kGy) and high-end (38.1–53.6 kGy) doses. The figures in parentheses represent what the percentage of the irradiated value is of the pre-irradiation value. The minimum recommended concentration of each nutrient per kilogram dry matter content of diet for growth and reproduction in cats is included (Association of American Feed Control Officials [AAFCO] guidelines 2007).
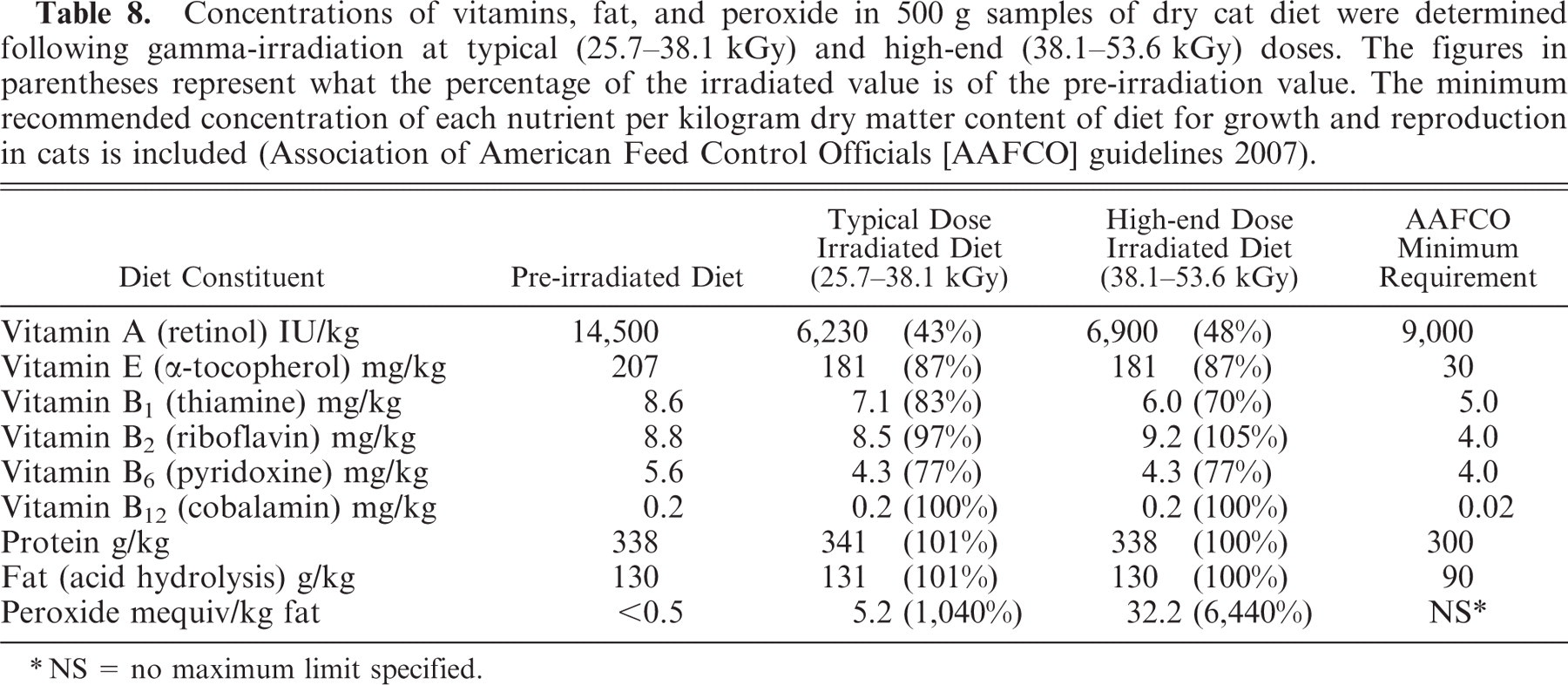
NS = no maximum limit specified.
Discussion
The findings of this study support our hypothesis that the exclusive long-term feeding of gamma-irradiated dry cat food to cats produces progressive hindlimb ataxia caused by leukoencephalomyelopathy. Increasing numbers of animals presented with severe ataxia from study day 140. Varying degrees of Wallerian degeneration were identified in fore-, mid-, and hindbrain white matter tracts and in white matter funiculi of the cervical, thoracic, and lumbar spinal cord segments of these cats, similar to that described in spontaneous cases of this disease5, 19, 33 and overall, increasing ataxia correlated with the degree of Wallerian degeneration within spinal cord and brain regions.
Although the time of initial appearance and rate of development of the induced ataxia was similar in both typical and high-end treatment groups, leukoencephalomyelopathy was detected in 1 of the group 4 high-end irradiated diet cats euthanized prior to the onset of neurologic deficits at day 93. The fact that this cat had more severe lesions in the lumbar than in the more cranial segments of its cord suggests the pathologic process may initiate at this location before progressing cranially. The finding that the TAS was significantly increased in the spinal cords and not in the brains of cats in all treatment groups would tend to support this hypothesis. However, the significant increase in the SOD concentration in the midbrain at the mid euthanasia time point in cats fed the high-end irradiation dose diet suggests increased free-radical activity was also occurring at this more cranial location.
Various types of CNS injury release the excitatory neurotransmitter glutamate, which increases the intracellular influx of calcium, impairs mitochondrial function, and generates free radicals. The damage these free radicals cause to lipid-rich membranes further increases the movement of calcium into neurons, ultimately resulting in their death.49 Locally generated free radicals have been implicated in Wallerian degeneration within spinal dorsolateral and ventromedial tracts of lambs, with the delayed form of swayback.21, 45 In this disease, free-radical damage is thought to result from failure of copper-utilizing enzymes such as SOD to negate their effects in CNS regions undergoing rapid phases of development.21, 45
Elevations in both the TAS and the SOD concentration suggest free-radical formation is a component or by-product of lesion development. Polyunsaturated fatty acids with several double bonds per molecule are found in high concentration in the CNS and are particularly vulnerable to free-radical attack.15, 37 Although short-lived, free radicals can self-propagate via membrane lipid peroxidation, resulting in cell injury through the oxidation of lipids, proteins, and nucleic acids.4, 24 This propagating property of free radicals results in the spread of their injurious effects both cranially and caudally along a segment of spinal cord. Although this study implicates these compounds in the pathogenesis of the condition, their precise role in disease development remains unclear. Furthermore, we could speculate that the reduced vitamin A content of the diet may have reduced the capacity of the cats to counteract the high levels of ingested peroxide.39 In this context assessment of the plasma glutathione peroxidase and vitamin E status of the affected animals may have provided further evidence of increased demands on tissue anti-oxidative mechanisms. Given that both elevated peroxide and reduced vitamin A concentrations were features of the irradiated diet, it is not clear which and to what degree either of these factors contributed to the pathogenesis. Further studies where cats fed the treated diet were given vitamin A supplementation would assist in elucidating if vitamin A had any role in lesion development. The possibility also remains that other undetected factors in the diet may have been contributory.
Similar typical and high-end radiation treatments of canine and laboratory rodent diets do not decrease the vitamin A content to the same extent although similar peroxide concentrations are generated.7 The fact that the feeding of such peroxide-rich diets to dogs and laboratory rodents has not resulted in disease25, 42, 46 suggests cats may be particularly sensitive to such compounds under these conditions with specific targeting of the CNS. The significant increase in the SOD concentration in the livers of groups 2, 3, and 4 cats at the mid euthanasia time points suggests increased free-radical activity at this site that did not result in hepatic injury visible by light microscopy. Elevation of the CSF nucleated cell count was found in only 2 cats, supporting the histopathologic evidence that the injurious process was primarily degenerative rather than inflammatory. Similarly the bilateral distribution of lesions within the CNS would be consistent with a nutritional or toxic etiology.
As in previous studies,5, 7 the vitamin A content of the dry cat diet was reduced to 43% and 48% of its untreated value 24 hours post gamma-irradiation by the typical and high-end irradiation treatments, respectively. These values are lower than the Association of American Feed Control Officials (AAFCO) minimum recommended vitamin A level for cats (9,000 IU/kg)3 and the National Research Council (NRC) recommended allowance (6,666 IU/kg)29 for growth and reproduction in this species. However, these reduced vitamin concentrations are higher than adult maintenance levels as recommended by the AAFCO (5,000 IU/kg)3 and NRC (3,333 IU/kg).29 Although the concentrations of vitamins E, B1 (thiamine) and B6 (pyridoxine) were also reduced by both typical and high-end radiation treatment, these reductions were relatively modest and did not result in their concentration in the ration falling below recommended levels.3 In previous studies the thiamine and pyridoxine concentrations in cat diets were depleted to a limited extent by similar doses of radiation,5, 7, 8 while vitamin E concentrations were marginally reduced7 or unaffected.5, 8 The vitamin A content of cat diets is more severely depleted by similar radiation doses than are canine or laboratory rodent rations.7 Why this is so remains unclear but may relate to diet manufacture, fat source, fat and water content, pretreatment vitamin A concentration, or the ratios of the various dietary components.
Felids have a higher dietary requirement for vitamin A than other species as they cannot convert β-carotene to vitamin A and are reliant on animal sources of preformed retinol.23, 27, 35, 44 Irradiation of a feline diet containing 9.8% fat with a 2.5 Mrad (25 kGy) dose totally destroyed its vitamin A and β-carotene content,8 and vitamin A deficiency has been implicated in ataxic syndromes in lions28, 31 and cheetahs34 where Wallerian degeneration was variously attributed to thickening of the cranial bones31 or to elevations in CSF pressure28 leading to CNS compression. Although osseous metaplasia of the cranium or vertebrae was not observed in the current study, the more peripheral and bilateral distribution of the Wallerian degeneration within dorsolateral and ventral funiculi might reflect increased intra-vertebral fluid pressure. Elevations in CSF pressure in vitamin A deficiency have been attributed to reduced absorption of the fluid from the subarachnoid space by structurally and biochemically altered arachnoid villi within the dura mater.9 Electron microscopy indicates that vitamin A deficiency results in mesenchymal tissue within the villi becoming structurally altered through the excessive accumulation of mucopolysaccharides.18 Such changes were not evident in the arachnoid villi of affected cats in the current study on light microscopy. Such a defect in CSF turnover might represent a “final common pathway” linking the development of leukoencephalomyelopathy in cats fed vitamin A–depleted diets with the development of this condition in cats and large felids presumably fed a vitamin A–sufficient ration.19, 33, 34, 43
However, the fact that other common clinical effects of vitamin A deficiency were not observed in the current study must reduce the potential significance of the reduced intake of this vitamin. Given the controlling effect of this vitamin on epithelial differentiation, deficiency has been associated with epithelial hyperkeratosis and with metaplasia of secretory epithelia. In the cat this has manifest as alopecia, cutaneous scaling, and follicular plugging.14 Furthermore, the xerophthalmia and keratomalacia described in nonhuman primates fed a vitamin A deficient diet for up to 21 months,17 and the visual defects reported in other species,30 were not evident. Differences in manifestations of deficiency may reflect differences in its severity and duration.
In conclusion, this study has successfully reproduced leukoencephalomyelopathy in cats similar to spontaneous outbreaks by feeding a gamma-irradiated dry diet with elevated peroxide and reduced vitamin A concentrations. The results suggest that free-radical formation may play a key role in the pathogenesis of the disease.
Footnotes
Acknowledgements
The authors would like to acknowledge the assistance and advice of the following: E. McGauley, C. Brady, B. Cloak, M. Murphy, E. O'Neill, and J. O'Donovan.
