Abstract
Cytauxzoonosis is a tick-borne disease of felids caused by the protozoan Cytauxzoon felis. This article characterizes the neuropathologic changes in 8 cases of natural C. felis infection in domestic cats with typical clinical signs and pathologic changes in multiple organs. Histologic changes in the brain included intravascular schizont-laden macrophages in leptomeningeal and parenchymal arterioles and venules. Small capillaries throughout the gray and white matter and choroid plexus also appeared occluded. Affected vessels were often surrounded by small to moderate numbers of lymphocytes and plasma cells and multiple areas of hemorrhage. Vasculitis and fibrin thrombi were occasionally present. Parenchymal changes consisted of variable degrees of vacuolation of the gray and white matter, with microgliosis, astrocytosis, astrogliosis, and multiple microhemorrhages. Multiple well-demarcated areas of necrosis observed in 4 cases were characterized by marked vacuolation of the gray or white matter, with necrotic neurons containing shrunken hypereosinophilic cytoplasm and pyknotic nuclei, nuclear debris, swollen axons, and scattered foamy macrophages. The changes in the brains of affected cats were attributed to vascular occlusion and secondary ischemia caused by the protozoal infection.
Cytauxzoonosis is an important tick-borne disease of domestic and wild felids caused by the protozoan Cytauxzoon felis of the family Theileriidae. 4,6,7,13,18,19 C. felis organisms have an erythrocytic (piroplasm) and a macrophagic (schizont) phase within the mammalian host. 4 The disease has no age, sex, or breed predisposition, and organisms are primarily transmitted to domestic cats by the tick Dermacentor variabilis. 4 The reservoir host for C. felis in North America is the bobcat (Lynx rufus), and clinical disease in domestic cats is usually detected in rural or suburban areas. 4 Infection in bobcats is usually subclinical with long-lasting parasitemia. 17 Cytauxzoonosis was originally reported in Missouri in 1976 and is endemic in the southeastern, south central, midwestern, and mid-Atlantic United States. 2,3,8,9,11,12,16,17,23
While the erythrocytic phase of infection is typically associated with anemia, the macrophagic phase leads to systemic circulatory compromise due to partial or complete vascular obstruction by schizont-laden macrophages. 17 Affected cats may have subclinical infection or develop acute, systemic, frequently fatal disease characterized by a range of clinical signs, including anorexia, dehydration, depression, fever, icterus, anemia, and dyspnea. 1 Common gross pathologic changes consist of icterus, pulmonary hemorrhages, splenomegaly, lymphadenomegaly, and disseminated hemorrhages in multiple organs. 2,4,11,17 Major histologic changes include the presence of swollen schizont-laden macrophages that adhere to the vascular endothelial surface and partially or completely occlude the lumen of blood vessels. * The resulting vascular occlusion and occasional vasculitis are responsible for tissue ischemia and have been regarded as one of the major pathogenic mechanisms of disease in cases of C. felis infection in domestic cats. 17 The antemortem diagnosis of cytauxzoonosis can be suspected on the basis of clinical signs and confirmed by the detection of parasitized erythrocytes on peripheral blood smears, but this method has not been regarded as sensitive, since piroplasms may be absent in up to 50% of samples. 4,17 Thus, infection may be also confirmed by detection of circulating schizont-laden macrophages on blood smears or by polymerase chain reaction. 4 Postmortem diagnosis is usually based on the pathologic changes and identification of intravascular schizont-laden macrophages. 2,4,10,11,17
With the exception of rare descriptions of vascular occlusion by schizont-laden macrophages and cerebral edema, no study has fully described the neuropathologic changes in the brain of cats that died due to C. felis infection. 1,12 –14,18 This report describes the neuropathologic changes in 8 cases of feline cytauxzoonosis in domestic cats. Cases of C. felis infection were identified retrospectively in domestic cats that presented for necropsy at the Athens Veterinary Diagnostic Laboratory at The University of Georgia College of Veterinary Medicine between January 2000 and July 2014. All retrieved cases were examined, but only those that had brain tissue available for examination were included in this study. Information regarding age, sex, breed, clinical signs, pathologic findings, and final diagnosis were collected from these cases. All archived histology slides from selected cases were reviewed independently by both pathologists (L.L.C. and D.R.R.), and neuropathologic changes were classified as vascular (meningeal, parenchymal, choroid plexus), gray matter, and white matter. Changes were then collectively evaluated and standardized by both pathologists.
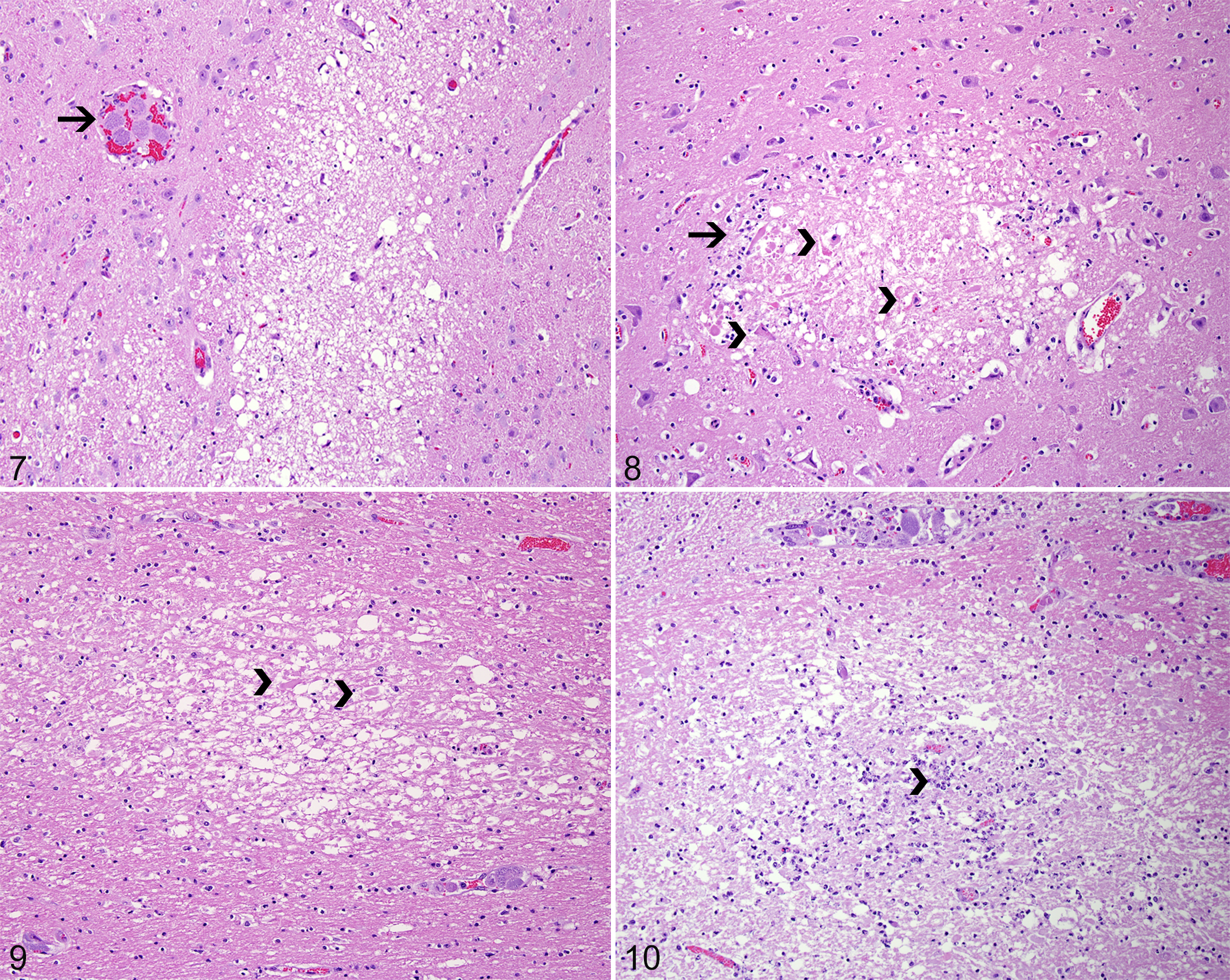
Twenty cases of C. felis infection in domestic cats were identified, but only 8 (Nos. 1–8) met the criterion for inclusion in this study. The signalment, major clinical signs, and clinical outcome in the affected cats are summarized in Supplemental Table 1. Gross changes in all cats were similar to those previously reported in cases of C. felis infection and included generalized icterus (8 of 8), pulmonary edema and hemorrhage (7 of 8), lymphadenomegaly (5 of 8), splenomegaly (5 of 8), hepatomegaly (5 of 8), hydrothorax (4 of 8), petechial epicardial hemorrhages (3 of 8), hydropericardium (2 of 8), and petechial renal hemorrhages (1 of 8). All 8 cases of cytauxzoonosis had histologic lesions in the brain. These changes are detailed in Supplemental Table 2. Vascular changes consisted of intraluminal schizont-laden macrophages within arterioles and venules throughout the leptomeninges, gray, and white matter (Fig. 1), as well as within small capillaries throughout the gray and white matter and choroid plexus (Fig. 2). Areas of perivascular hemorrhage affected leptomeningeal or parenchymal venules (Figs. 3, 4). In addition, arteriolar and venular walls were often surrounded by small to moderate numbers of lymphocytes and plasma cells. Lymphoplasmacytic vasculitis was observed in 1 case (Fig. 5). Although schizont-laden macrophages were typically observed within blood vessels, they were also seen infiltrating the surrounding brain tissue (Fig. 6). Parenchymal changes (Figs. 7–10) consisted of areas of vacuolation (edema) of the gray and white matter, with microgliosis, astrocytosis (astrocytic hypertrophy), astrogliosis (astrocytic hyperplasia), and multiple microhemorrhages. These changes were often accompanied by the presence of luminal schizont-laden macrophages within adjacent venules and capillaries. Necrotic foci contained multiple necrotic neurons and swollen neuronal processes (spheroids) that were embedded in a highly vacuolated and loosened parenchyma and were in turn surrounded by a thin rim of glial cells. The final diagnosis in all cases was based on the characteristic clinical signs and pathologic changes, including the visualization of intravascular schizont-laden macrophages in multiple organs. †
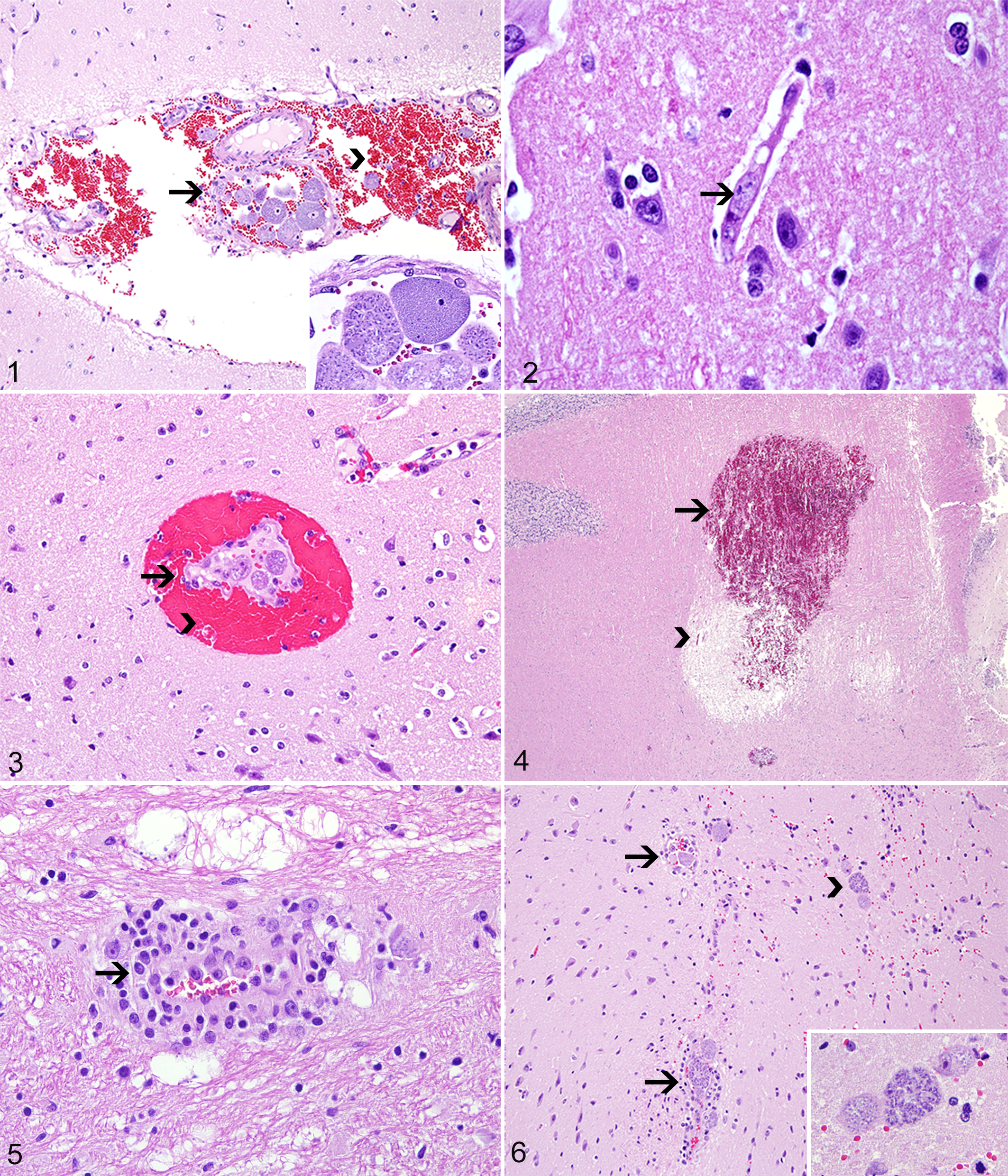
Cytauxzoonosis. Hematoxylin and eosin.
Cytauxzoonosis. Hematoxylin and eosin.
All cats in this study presented with clinical signs and pathologic changes classically attributed to C. felis infection. 1,2,4,8,9,11,12,17,21 Five had a suspected diagnosis of C. felis infection before death that was based on the clinical signs. Swollen schizont-laden macrophages have been regarded as a substantial mechanism of vascular occlusion and impaired blood flow in multiple organs during C. felis infection in cats. 17,23 Death has been attributed to shock with respiratory failure due to decreased pulmonary perfusion and gas exchange from vascular obstruction and interstitial pneumonia. 4,21 Furthermore, the marked systemic inflammatory response in these cases is thought to be important in the pathogenesis of the disease. 5 The findings of this study support previous speculations and indicate that a major neurologic component may be also important in the course and development of clinical disease and death due to C. felis infection. Cerebral edema and disseminated intravascular coagulation, with possible cerebral or cardiac thrombosis, have been only briefly reported in early experimental studies with cats, 14,24 and incoordination, nystagmus, and deafness were attributed to occlusion of vessels in the brain of a lion with C. felis infection. 19 However, despite these descriptions, previously detailed examination of the brain has not been conducted to characterize the changes associated with vascular compromise in C. felis infection. While C. felis infection has not been classically referred to as a neurologic disease in cats, the findings in this study suggest that some of the clinical signs, such as lethargy, disorientation, and tremors, could be due to brain damage rather than systemic compromise caused by the protozoal infection.
Given the retrospective nature of this study, brain sampling was not standardized for the cases examined; however, lesions appeared to be random, affecting the gray and white matter equally, with no evidence of selective neuronal injury. These findings are consistent with an ischemic mechanism, and the distribution of lesions coincides with the occlusion of arterioles, venules, and capillaries by protozoal organisms. 22 Intravascular schizont-laden macrophages were observed in all cases of this study, and it is important that cytauxzoonosis is differentiated from other degenerative diseases of cats, especially thiamine deficiency and feline ischemic encephalopathy (FIE). 15,20,25 In contrast with the brain lesions associated with C. felis infection, changes classically attributed to thiamine deficiency in carnivores are characterized by bilateral and symmetrical vacuolation, hemorrhage, and necrosis of the neuropil with targeting of the periventricular gray matter in the caudal colliculi, medial vestibular nuclei, and lateral geniculate bodies. 15 The random areas of necrosis observed in 4 of the cases of this study (Nos. 3, 6, 7, 8) were similar in distribution to the lesions described in cases of FIE caused by migration of Cuterebra spp larvae. 20,25 The distribution of the necrotic lesions in cases of FIE may be associated with active larval migration or vascular compromise and ischemia. 20 However, in addition to cerebral infarcts, other hallmarks of FIE were not observed in the current cases—such as extensive superficial laminar cerebrocortical necrosis, subependymal rarefaction and astrogliosis, subpial astrogliosis, and eosinophilic meningoencephalitis. 20,25
In summary, this report details the neuropathologic changes of vascular compromise and secondary ischemia due to cytauxzoonosis in domestic cats. The vascular changes characteristic of C. felis infection consisted of vascular occlusion by circulating schizont-laden macrophages with occasional perivascular lymphocytes and plasma cells and areas of hemorrhage. Parenchymal changes consisted of areas of edema in the gray and white matter, with microgliosis, astrogliosis, microhemorrhages, and well-demarcated areas of necrosis in 4 cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
