Abstract
Investigations were carried out on 8 specific pathogen-free cats (5 male and 3 female) from a colony experiencing “outbreaks” of progressive hind limb ataxia in 190 of 540 at-risk animals ranging from 3 months to 3 years old. These studies identified moderate to severe bilateral axonal degeneration within white matter regions of the cervical, thoracic, and lumbar spinal cord and in the white matter of the cerebral internal capsule and peduncle, in the roof of the fourth ventricle and inferior cerebellar peduncle, and in the external arcuate and pyramidal fibres of the medulla. There were varying degrees of accompanying microgliosis, astrocytosis, and capillary hyperplasia. Such a clinicopathologic syndrome, termed feline leukoencephalomyelopathy, has previously been described in cat colonies in Britain and New Zealand, although its etiology has not been determined. The degenerative nature of the lesions and their bilateral distribution suggest possible nutritional, metabolic, or toxic causes. Although these findings provide circumstantial evidence that the exclusive feeding of a gamma-irradiated diet of reduced vitamin A content is associated with the development of the neuronal lesions, further tissue micronutrient and antioxidant analysis will be required to support this hypothesis.
The presentation of numbers of cats with progressive hind limb ataxia associated with leukoencephalomyelopathy has been described within colonies of specific pathogen-free (SPF) cats in Britain 17 and New Zealand. 9 A clinically and pathologically similar syndrome has been described in captive-bred cheetahs, 16 in a snow leopard, 20 and in a black-maned lion. 13 Although various infectious and nutritional etiologies have been suggested in the case of each of these species, no cause has been established. Between 1998 and 2001, three “outbreaks” of a syndrome matching the previously reported clinical and pathologic criteria occurred in an SPF cat colony. Both male and female animals were affected, and the age range was from 3 months to 3 years. This report describes the investigation of 8 of these cases and provides initial circumstantial evidence that this disease is associated with the long-term exclusive feeding of cats on a gamma-irradiated dry diet.
Over a 4-year period, a total of 190 group-housed domestic short-hair breed cats aged 3 months to 3 years that were from an SPF breeding colony held at a research laboratory developed progressive hind limb ataxia and proprioceptive defects. The colony was established in February 1997, and the first cases appeared over a 2-month period in the autumn of 1998, with 30 animals out of a total of 120 exhibiting neurologic defects. A second “outbreak” of cases occurred over a similar time span in the autumn of 1999, with 70 cats out of a colony total of 200 SPF animals affected. A third cluster of cases presented in the autumn of 2001, when 90 animals out of a colony total of 220 cats presented with ataxia. Throughout this time, kittens of between 8 and 12 weeks of age that were transferred from the SPF to a conventional disease status colony on the same site did not develop clinical signs. Affected animals were both male and female and did not share common ancestry. Both SPF and conventional status cats had been fed to appetite on the same commercial formula ration (Gilbertson and Page Ltd., Welwyn Garden City, UK), except that the ration fed to the SPF cats had been irradiated by a single-exposure gamma-radiation treatment between 36.3 and 47.3 kGy (Cobalt 60 irradiator; Isotron Ireland, Tullamore, Ireland). The irradiated diet was consumed to the same extent as the nonirradiated diet, and affected animals did not lose weight until the developing ataxia hindered their access to food. Dietary constituents were determined 1, 2, 8, 11 prior to and after gamma-irradiation treatment. Following supplementation of the irradiated diet with pasteurized proprietary tinned cat food in the winter of 2001 and, ultimately, the replacement of the irradiated diet with an equivalent pasteurized diet, no further cases occurred.
During the second cluster of cases in the autumn of 1999, 8 cats (cat Nos. 1–8) were submitted for investigation to The University Veterinary Hospital, University College Dublin. These animals comprised 5 males and 3 females between 12 and 18 months old that had exhibited progressive hind limb ataxia for between 4 and 8 weeks. All were subject to full neurologic assessment. Prior to euthanasia, each cat was anesthetized, and cerebrospinal fluid (CSF), blood, and urine samples were collected. Serologic examinations for antibodies to the following pathogens were carried out on all animals: feline infectious peritonitis virus, feline corona virus, feline viral rhinotracheitis virus, feline panleukopenia virus (FPLV), and chlamydiae (European Veterinary Laboratory test kits, Worden, Germany); feline immunodeficiency virus, feline leukemia virus, Bordetella bronchiseptica, Salmonella spp., Campylobacter spp., Pseudomonas aeruginosa, and Streptococcus pneumonia (Surrey Diagnostics, University of Surrey, Guildford, UK); and Toxoplasma gondii (MAST Diagnostics, Bootle, UK). Hematologic and biochemical analyses of blood samples were carried out in addition to urinalysis and cytologic examination of CSF samples. Detailed postmortem examinations were performed on all 8 cats. Brain, spinal cord (including cervical, thoracic, and lumbar segments), and samples of sciatic nerve, vastus lateralis muscle, liver, kidney, myocardium, and lungs were fixed by immersion in 10% neutral buffered formalin. Following fixation, tissues were dehydrated and embedded in paraffin wax. All sections were stained with hematoxylin and eosin for histopathologic examination; to identify astrocytes, selected sections of brain and spinal cord were stained using rabbit antiserum to glial fibrillary acidic protein (GFAP) using a peroxidase-antiperoxidase (PAP) technique (DAKO PAP kit; Dako Corp., Carpinteria, CA). Unfixed samples of liver from all 8 cats were stored at −70°C for subsequent measurement of copper, zinc, and lead concentrations. This analysis involved thawing 1 g of liver sample and digesting it in 5 ml of a mixture of nitric, perchloric, and sulphuric acids (7 : 2 : 1 by volume) on a heated rack for 30 to 45 minutes at 100°C. 7, 25 An atomic absorption spectrophotometer (Varian SpectrAA 640; Varian, Palo Alto, CA) was used to assay acid tissue digests for copper and zinc in accordance with the spectrophotometer instruction manual and onboard PC software. Lead in tri-acid digests was assayed with the same instrument using a hydride-generating accessory.
The neurologic findings for all 8 cats were similar. Animals exhibited hind limb ataxia with impaired postural reactions of both hind limbs. The cats were unable to wheelbarrow, and hopping reactions were attenuated. Proprioception was reduced. Both patellar and flexor reflexes were exaggerated. Crossed extensor reflexes were present in the hind limbs. Peripheral and deep pain sensation was present in both hind legs. Cranial nerve examinations, forelimb spinal reflexes, and sensation to the forelimbs were normal. The panniculus reflex was present, and the cats were continent of feces and urine. All cats were serologically negative for antibodies to the previously listed pathogens. All hematologic and clinical chemistry values were within laboratory reference ranges. Urinalyses were normal, and no significant findings were noted on cytological examination of cerebrospinal fluid. Gross postmortem examination revealed bilateral loss of gluteal muscle mass. Histopathologic examination of transverse cross-sections of cervical, thoracic, and lumbar spinal cord from each cat revealed bilateral vacuolation of ventral, lateral, and dorsal white matter funiculi (Fig. 1). Vacuole density was greater in peripheral cord regions, particularly in dorsolateral and ventral funiculi, with relative sparing of dorsal tracts. The vacuoles variously contained swollen axons, axonal debris, and myelinophages (Fig. 2). These lesions were consistent with moderate to severe axonal degeneration. There were varying degrees of associated microgliosis, astrocytosis, and capillary hyperplasia in the 8 animals. In longitudinal sections of spinal cord segments there were chains of myelin ellipsoids as well as swollen axons and myelinophages (Fig. 3). Similar, bilateral lesions of varying intensity occurred in the white matter of the cerebral hemispheres (internal capsule; Fig. 4), cerebral peduncle, roof of the fourth ventricle, inferior cerebellar peduncle, and in the external arcuate and pyramidal fibers of the medulla. No significant abnormalities were observed in the other tissues sampled. Dietary analysis indicated reduced concentrations of vitamins A, B1, and B6 in the feed following gamma-irradiation treatment (Table 1). Analysis of liver tissue indicated 5 cats (cat Nos. 3, 4, 5, 6, and 8) had lower than normal copper concentrations, 5 whereas zinc concentrations were raised in only 1 animal (cat No. 6) and were lower than the reference range in 4 cats (cat Nos. 2, 3, 4, and 5) 6 (Table 2).
Constituents of cat ration prior to and after a gamma-irradiation treatment of between 3.6 and 4.7 mRad.
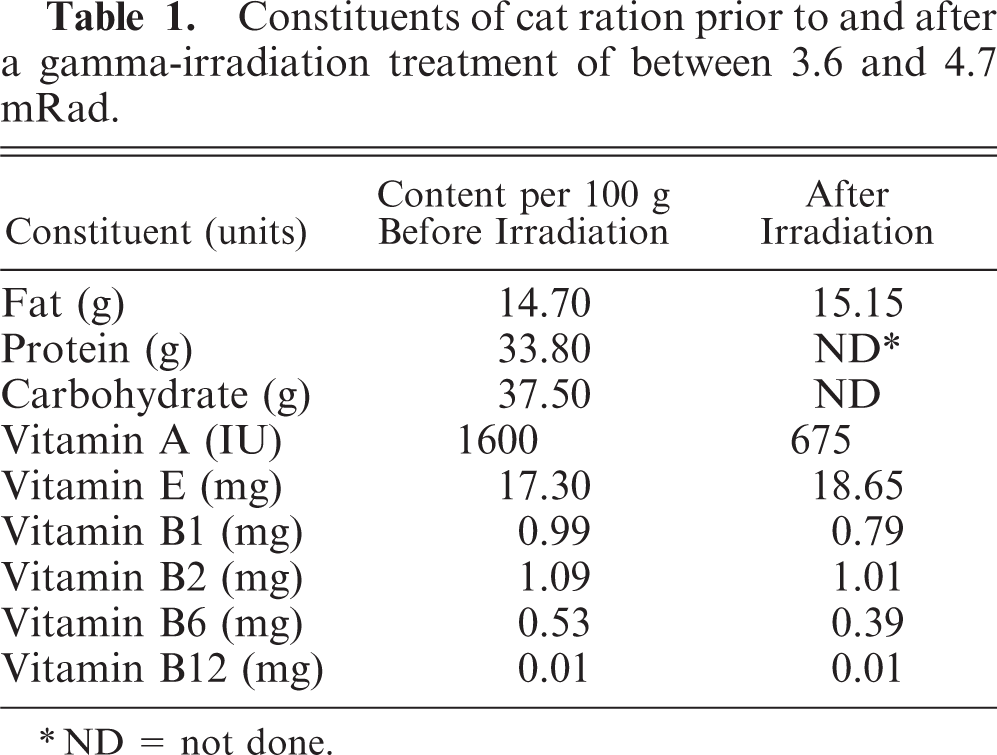
∗ND = not done.
Liver copper, zinc, and lead concentrations from 8 cats (cat Nos. 1–8) presenting with leukoencephalomyelopathy.
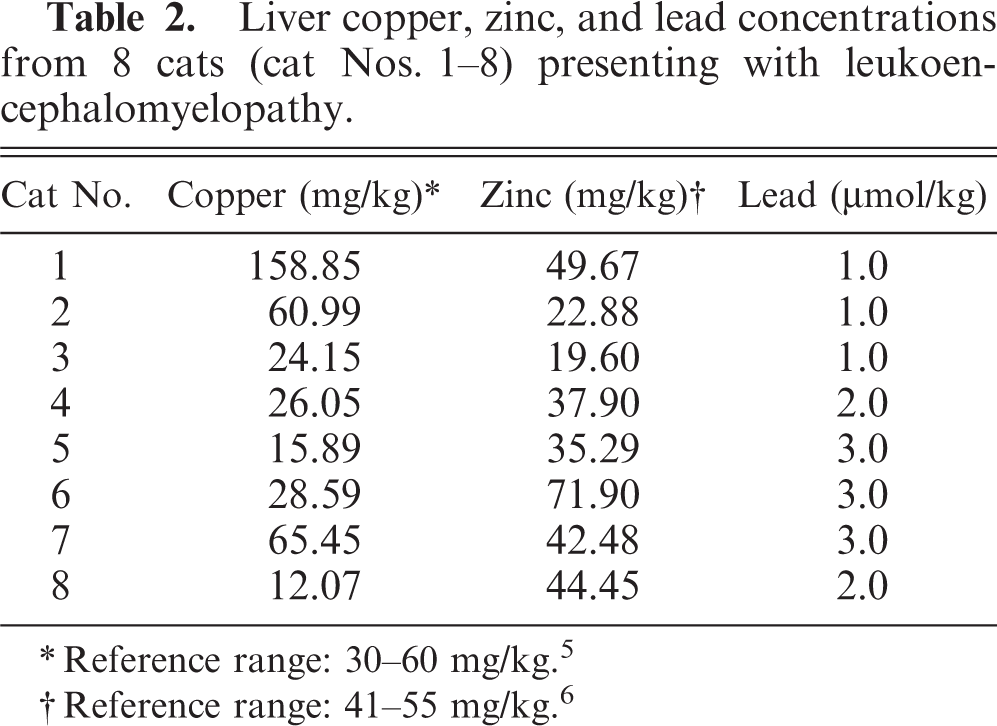
∗Reference range: 30–60 mg/kg. 5
†Reference range: 41–55 mg/kg. 6
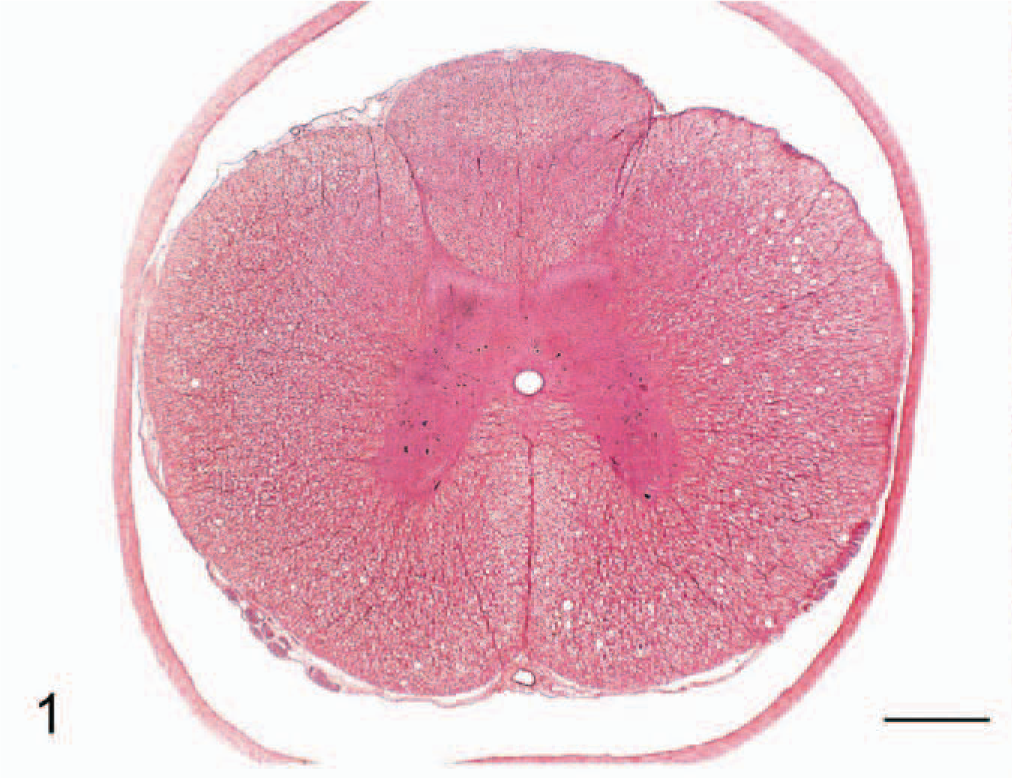
Transverse section of thoracic spinal cord; cat No. 3. Bilateral vacuolation of ventral and lateral white matter funiculi. Hematoxylin and eosin. Bar = 500 μm.
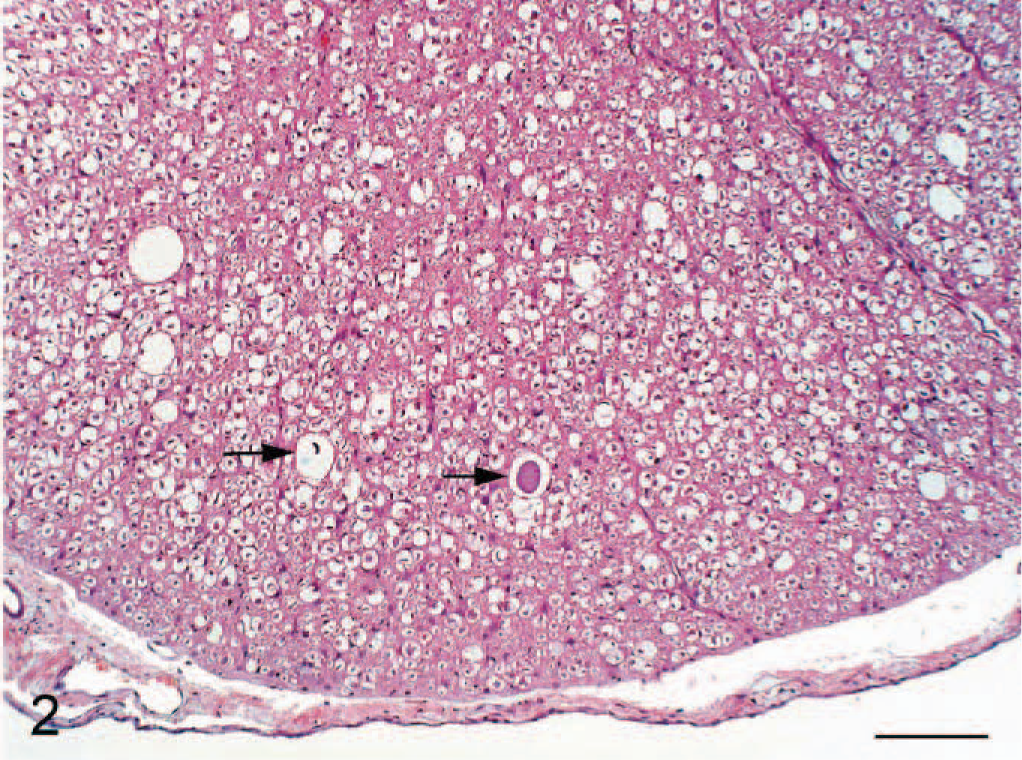
Higher magnification of ventral funiculus in Figure 1; cat No. 3. Vacuoles variously contain a swollen axon and cell debris (arrows). Hematoxylin and eosin. Bar = 100 μm.
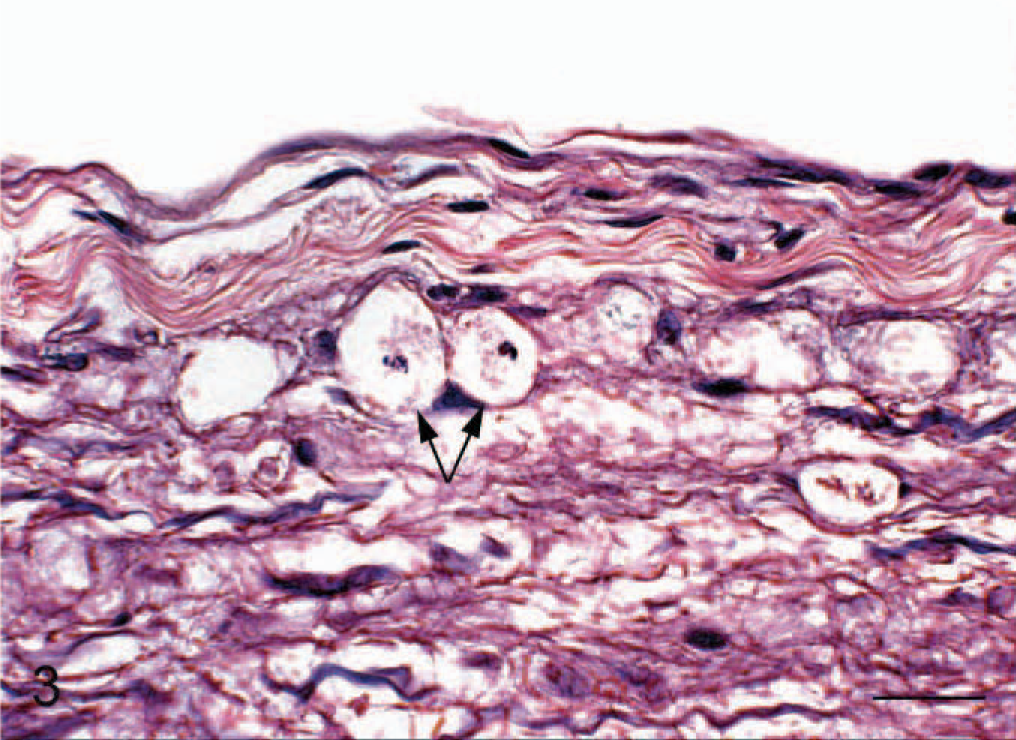
Longitudinal section of lumbar spinal cord; cat No. 5. Vacuoles containing cell debris (arrows) within ventral white matter funiculi. Hematoxylin and eosin. Bar = 17 μm.
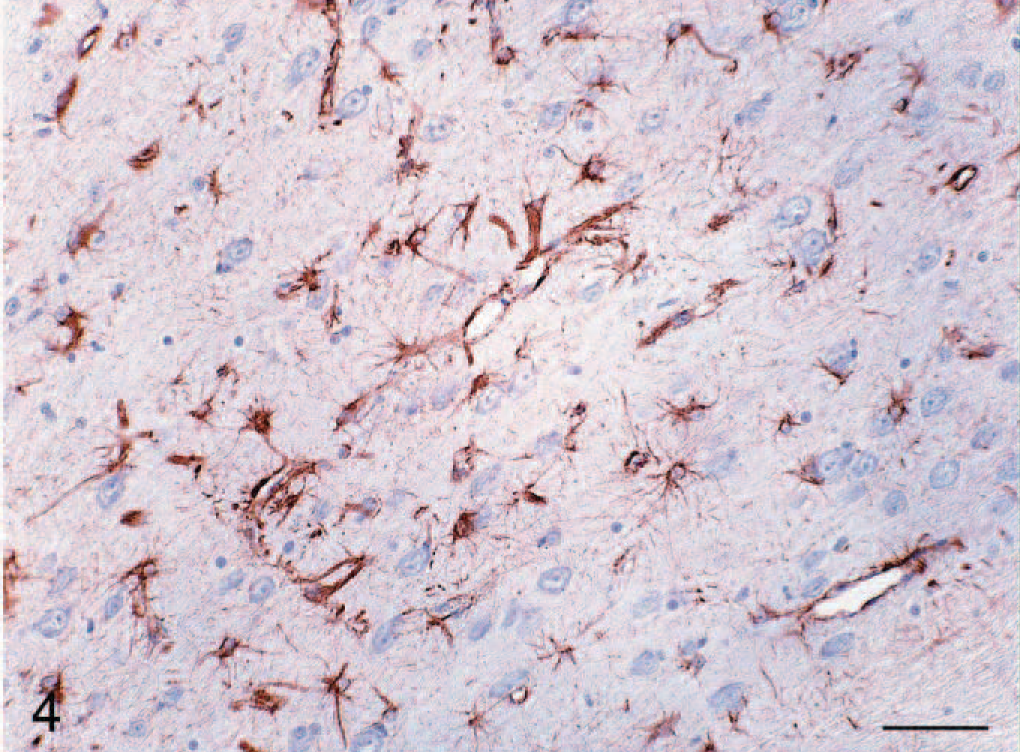
Cerebrum; cat No. 3. Diffuse astrocytosis of white matter. Glial Fibrillary Acidic Protein (GFAP) stain. Bar = 100 μm.
The occurrence of “clusters” of cats with progressive hind limb ataxia and bilateral CNS white matter tract degeneration has previously been described in cat colonies in Britain 17 and New Zealand, 9 and a clinicopathologically similar syndrome has been described in captive-bred cheetahs, 16 in a snow leopard, 20 and in a black-maned lion. 13 The etiology of the syndrome, termed a leukoencephalomyelopathy, has not been determined, although the primarily degenerative nature of the lesions and their bilateral distribution suggest possible nutritional, metabolic, or toxic causes. The fact that the only significant difference between the affected cats in the SPF colony and their conventional status counterparts, moved out of the SPF colony at weaning, was the exclusive feeding of a gamma-irradiated diet and that no further cases of the condition were reported once supplemental or replacement pasteurized diets were introduced, provides circumstantial evidence that the ataxia may have a nutritional basis. In particular, the reduced vitamin A content of the diet following irradiation may be significant, given that the recommended vitamin A concentration in feline diets is 1,000 IU per 100 g. 21 However, pending micronutrient and antioxidant analysis of cat tissue, the role of vitamin A in the pathogenesis of this disease process remains speculative. The apparent seasonality of the disease episodes, with “outbreaks” occurring in the autumn, might also implicate additional husbandry-related factors in the pathogenesis. However, given that no significant differences in animal management were in place in 2000, when no cases of the disease occurred, such potential contributory factors remain unidentified.
Gamma-radiation is used to sterilize food fed to SPF animals through the generation of free radicals and the subsequent fragmentation of the genome of contained microorganisms. 18 Such free radical formation has the potential to alter the concentration of essential micronutrients within food, resulting in potential deficiencies in animals fed exclusively on such diets over prolonged periods. Irradiation is known to reduce the vitamin content of food, 18 the effect of which may be indirect, in that inadequate amounts of these compounds may be available to counteract the effects of free radicals generated by normal cell metabolism. A previous study 3 found that irradiation of a feline diet containing 9.8% fat with a 2- to 5-Mrad dose totally destroyed its vitamin A and β-carotene content, whereas thiamine, vitamin B6 (pyridoxine), and folic acid were depleted to a lesser extent, and vitamin E concentrations appeared to be unaffected by this dose of radiation. The relatively high dietary fat requirement of cats 12 may be significant in this context in that irradiation of this fat component could potentially generate higher concentrations of micronutrient-damaging free radicals than would be generated on irradiating laboratory animal diets of lower fat content. 22
Vitamin A deficiency has previously been implicated in ataxic syndromes in lions 13, 14 and cheetahs. 17 In these reports, axonal degeneration was variously attributed to thickening of the cranial bones 14 or to elevations in CSF pressure, 13 leading to CNS compression. Although neither meningeal thickening nor osseous metaplasia was observed in the current study, the more peripheral distribution of the axonal degeneration within dorsolateral and ventral funiculi might reflect increased intravertebral fluid pressure. White matter tract degeneration in the brain and spinal cord has also been described in English Foxhounds deficient in vitamin B12, 19 as well as in Landrace-cross pigs in which there was suboptimal nicotinamide activity. 14 The dry nature of the diet fed to the cats in this study may also be of significance, as this can favor the persistence of contained free radicals and increase the antioxidant requirements. 18 The possibility of radiation injury to tissues from the irradiated diet is highly unlikely, given that radioactivity is not detectable in irradiated food 24 hours after treatment. 25 Zinc-induced copper deficiency was previously implicated in cases of encephalomyelopathy in a cat colony in New Zealand, 9 and the significance of the low hepatic copper and zinc status of 5 and 4 of the 8 cats, respectively, and the elevated liver zinc concentration in 1 animal in the current study remains unclear. Axonal degeneration within spinal dorsolateral and ventromedial tracts is reported in lambs with the delayed form of swayback, a disease associated with copper deficiency. 10 This proposed axonopathy is thought to result from failure of copper-utilizing enzymes, such as superoxide dismutase, to counter locally generated free radicals in brain and spinal cord regions undergoing rapid phases of development. 10, 23 Lead poisoning in the cat can result in cortical neuronal and Purkinje cell necrosis and in subtle peripheral neuronal segmental demyelination. 24 However, insignificant concentrations of this metal were detected in liver samples.
Although axonal degeneration was observed in ventral, lateral, and dorsal white matter cord funiculi at all levels, there was relative sparing of dorsal tracts. This sparing of dorsal spinal cord tracts was also reported in a clinicopathologically similar condition of captive-bred cheetahs 16 and may result from the extra-CNS location of the neurons supplying the dorsal tracts. These neurons reside in the dorsal root ganglia, whereas those supplying the lateral and ventral tracts are located within the CNS. 16 While largely similar in terms of the nature and distribution of the CNS white matter injury, differences were also noted in the lesions described in the current study and those previously reported. 9, 17 Both of these groups additionally describe loss of Purkinje cells from the cerebellar cortex and chromatolysis and necrosis in the motor neurons in the intermediate columns and ventral regions of the spinal gray matter. Despite detailed microscopic examinations of multiple sections, such changes were not found in the current study.
The occurrence of multiple cases of this condition in groups of cats within relatively short time periods has previously led to suspicions of an infectious cause, such as FPLV infection. 4 Given that the cats maintained their SPF status throughout the disease “outbreak,” an infectious cause appears unlikely. Furthermore, if an agent such as FPLV had been responsible for the disease, it is surprising that other clinical and pathologic manifestations of infection were not observed.
This report describes the investigation of multiple cases of leukoencephalomyelopathy in a, SPF cat colony and suggests a possible association with the long-term feeding of cats exclusively on a gamma-irradiated dry diet deficient in vitamin A. Further studies assessing the free radical content of the irradiated diet and the micronutrient status of the animal tissues are currently in progress.
Footnotes
Acknowledgements
We would like to acknowledge the assistance and advice of the following: J. McLoughlin, E. O'Neill, E. McGauley, C. Brady, B. Cloak, Prof. B. Sheahan, Prof. S. Callanan, and M. O'Mahoney.
