Abstract
A rare, multisystemic intravascular proliferative disorder was identified postmortem in eight cats. The majority of these cats died or were euthanized following episodes of dyspnea, lethargy, and anorexia. Microscopic examination revealed occlusive, intraluminal proliferations of spindle cells within small vessels. The heart was consistently involved, and myocardial dysfunction was the probable cause of illness in all cats. Immunohistochemically, the majority of intravascular cells expressed von Willebrand factor, and a smaller number expressed smooth muscle actin, compatible with a dual population of endothelial cells and pericytes, suggesting a reactive rather than a neoplastic process. Four cases of a similar feline vascular disorder from the veterinary literature are reviewed. The histopathology resembles reactive angioendotheliomatosis in humans, a benign cutaneous intravascular endothelial and pericytic proliferative condition. However, in contrast, this feline disease is multisystemic and fatal. We propose the name “feline systemic reactive angioendotheliomatosis” for this unique, idiopathic disorder of domestic cats.
The term angioendotheliomatosis has been used to describe several intravascular proliferative disorders in humans. While originally separated clinicopathologically into benign and malignant variants, during the past 15 years, it has become clear that angioendotheliomatosis actually represents three distinct diseases. 9, 26, 36 Antigenic phenotyping by immunohistochemistry and limited karyotypic analysis have shown almost all examples of the fatal and multisystemic malignant variant to be an intravascular, angiotropic lymphoma (predominantly of B-cell type). 5, 13, 30, 35 Malignant angioendotheliomatosis was originally thought to be a disseminated intraluminal proliferation of neoplastic endothelial cells (angiosarcoma), but only rare examples of this second neoplasm have been substantiated. 22 The third variant is benign and reactive, with lesions limited to the skin, and represents a proliferation of intravascular endothelial cells admixed with pericytes. 20
Feline intravascular proliferative disorders are extremely rare, and they fall into one of two categories: intravascular angiotropic lymphoma and a variant of reactive angioendotheliomatosis (RAE). A single case of malignant intravascular lymphoma has been reported in a Siamese cat that affected the vessels of the brain and kidney. 19 The intravascular cells consisted of noncohesive, pleomorphic, round cells that had the immunohistochemical properties of T cells. In contrast, four cases of an unnamed, multisystemic, and fatal disease characterized by plump endothelial cells and fewer pericytes proliferating in the vascular lumina have been documented by light and electron microscopy and immunohistochemistry in cats. 7, 29, 31 We describe eight additional cats with this rare entity, review the four previously documented cases, and propose the name “feline systemic reactive angioendotheliomatosis” (FSRA) for this unique disease in cats.
Materials and Methods
All tissues were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 4–6 μm, and stained with hematoxylin and eosin. In some cases, special histochemical staining, including Masson's Trichrome and a Modified Steiner silver stain, were performed.
Immunohistochemical staining for vimentin (Dako, monoclonal, microwave pretreatment, 1 : 40), von Willebrand factor (vWF) antigen (Dako, polyclonal, trypsin pretreatment, 1 : 1,000), smooth muscle actin (SMA; Dako, monoclonal, no pretreatment, 1 : 40), and cytokeratins (Keratin AE1/3, Boehring-Mannhein, monoclonal, trypsin pretreatment, 1 : 100) were performed on selected tissues from each of the eight cats. In addition, we performed immunohistochemical staining for the following leukocyte antigens: CD3 (Dako, polyclonal, trypsin pretreatment, 1 : 200), CD79a (Dako, monoclonal, microwave pretreatment, 1 : 160), CD18 (Dako, monoclonal, no pretreatment, 1:160), and CD45RA (Dako, monoclonal, microwave pretreatment, 1 : 160).
Case summaries (clinical histories, gross and microscopic lesions)
Cat No. 1. A 4-year-old, castrated male, Sphinx cat presented in acute respiratory distress. Froth was exuding from the nostrils and mouth, and the cat died during the physical examination. Six days earlier, the owner had noted that the cat had unusual splotchy red skin lesions on his neck and back. At necropsy, there was moderate fibrinous pericardial effusion, hemorrhages throughout the lungs, and abundant white foamy exudate in airways.
Histologically, approximately 70% of the subepicardial and scattered subendocardial (papillary muscle) arterioles of both ventricular free walls of the heart contained variably sized, loosely arranged intraluminal proliferations of bland spindle cells, which partially to completely filled the lumina. Within the nests of spindle cells, slit-like, narrow channels were present, occasionally containing erythrocytes. The spindle cells were characterized by plump, elliptical nuclei with clumped-to-stippled chromatin and scant eosinophilic cytoplasm. The nuclei of the proliferating cells lacked features of atypia, and mitotic figures were extremely rare. Many of these arterioles were thickened by concentric rings of proliferative adventitial fibroblasts. In a few arterioles, there was fibrinoid necrosis of the tunica media, thrombosis, and perivascular plasma exudation. Associated with affected vessels were multiple, small areas of myocardial infarction. Larger muscular arteries, veins, and capillaries were unaffected by the proliferative intravascular process.
Intravascular proliferative lesions were also present in the small arterioles within the splenic parenchyma and in the periglomerular arterioles at the corticomedullary junction of the kidney. In these organs, the proliferating spindle cells were arranged in tight whorls, often forming glomeruloid structures containing fibrin thrombi (Fig. 1). Neither fibrinoid necrosis nor parenchymal infarction was associated with these affected vessels.
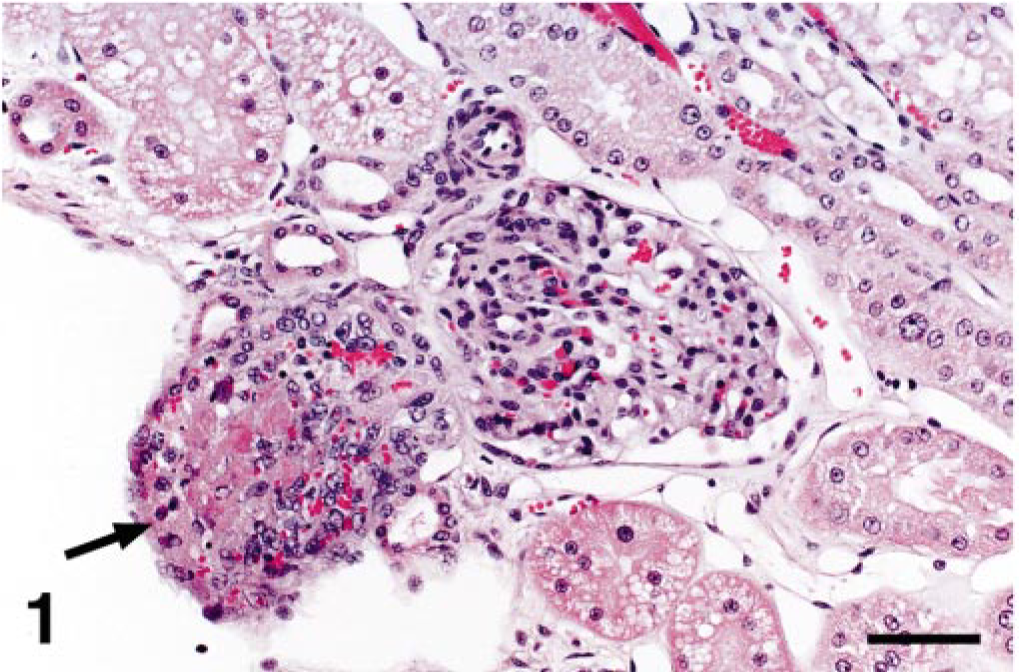
Kidney; cat No. 1. Periglomerular arteriolar expansion and occlusion by fibrin thrombus and exuberant intravascular spindle cell proliferation (arrow). HE. Bar = 60 μm.
Cat No. 2. A section each of the liver and left ventricular myocardium were submitted for histopathologic evaluation from a 4-year-old female domestic shorthaired cat. No clinical signs were reported. The referring veterinarian noted that the heart walls were thickened, and the liver appeared edematous at necropsy.
Approximately 80% of the myocardial arterioles contained intraluminal spindle cell proliferations similar to cat No. 1, but in this cat, there was marked fibrinoid necrosis of many of the subepicardial vessels with associated hemorrhage and myocardial necrosis.
Cat No. 3. A 3-year-old castrated male domestic short-haired cat presented for seizures. Abnormal nystagmus, retinal hemorrhages, and hematuria were also observed on physical examination. The cat died spontaneously. No gross necropsy findings were described.
Histologically, prominent vascular lesions were identified in arterioles of the heart, kidney, spleen, lymph node, thyroid gland, iris, and brain. The lesions were more severe than in cat Nos. 1 or 2, affecting more vessels and expanding them to a greater degree. The intravascular proliferations were similar to those described above, but in addition, many had a vasoformative appearance characterized by multiple small channels lined by spindle cells and supported by a fine interlacing collagenous network. Throughout the cerebral and cerebellar sections, vessels of the parenchyma and meninges were affected with some proliferative foci resembling renal glomeruli (Fig. 2). Many of the affected vessels contained fibrin thrombi, although no parenchymal hemorrhage or necrosis was observed. In the kidney, periglomerular proliferations at the vascular pole often extended into and compressed the glomerular tuft. Proximal tubular epithelial cells contained granular hemosiderin, and scattered tubules contained hemoglobin. Mild-to-moderate extramedullary hematopoiesis (EMH) was present in the spleen and liver. A few arterioles in the hepatic portal tracts were affected.
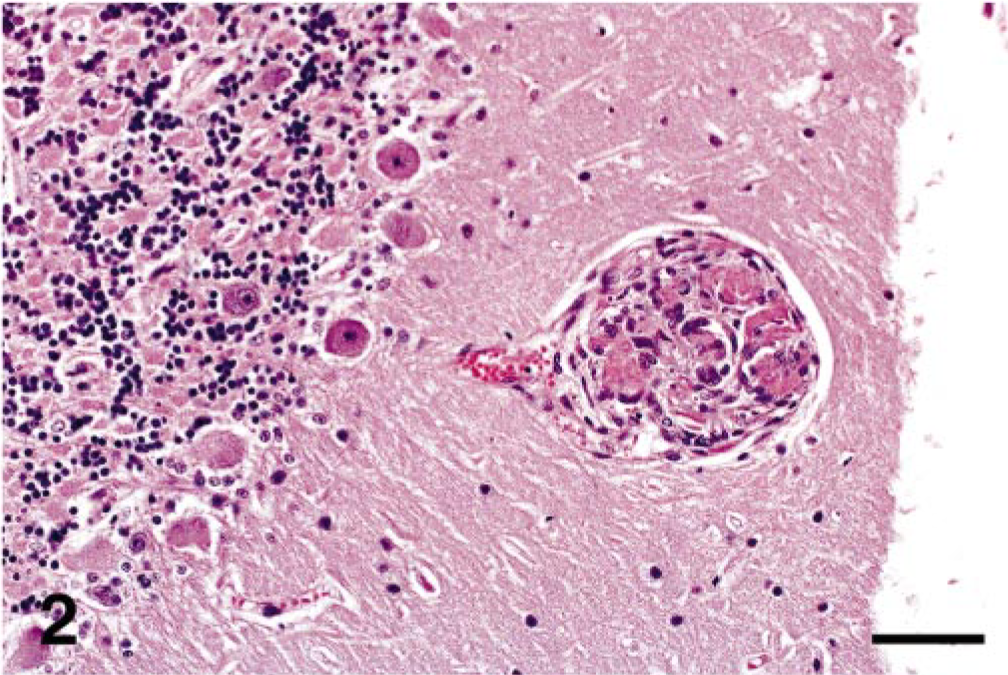
Cerebellum; cat No. 3. Proliferative focus resembling a renal glomerulus. No corresponding parenchymal hemorrhage or necrosis was observed. HE. Bar = 60 μm.
Cat No. 4. A 6-year-old spayed female domestic long-haired cat presented for an acute illness characterized by tachypnea, lethargy, anorexia, and weight loss. On physical examination, the referring veterinarian noted muffled heart sounds and cranial abdominal pain. Radiographically, the cardiac silhouette appeared slightly enlarged. Clinical hematology and chemistry showed a mild anemia, hyperbilirubinemia, hyperglycemia, elevated blood urea nitrogen, and hypokalemia. The cat died spontaneously.
Postmortem examination findings included edema of the pericardial sac with serosanguineous and fibrinous pericardial effusion, petechial hemorrhages on the epicardium, blood-tinged urine in the urinary bladder, and a pyloric trichobezoar. Aerobic cultures of the liver, lung, small intestine, spleen, pericardial effusion, and urine yielded no bacterial growth. An impression smear of the spleen was negative for feline leukemia virus (FeLV) by immunofluorescence. A feline immunodeficiency virus (FIV) enzyme-linked immunosorbent assay was negative, and a feline infectious peritonitis (FIP) viral titer was negative.
Intravascular proliferative lesions were found in the heart, brain and meninges, pancreas, and small intestine. The heart was most significantly affected, with approximately 60% of the right and left ventricular arterioles involved, many containing thrombi and necrotic cells within the proliferative cellular tuft. Hemorrhage, plasma exudation, scattered myo-fiber necrosis, and a minimal mixed inflammatory cell infiltrate were common adjacent to affected vessels. Cardiac dysfunction resulted in mild pulmonary edema with alveolar histiocytosis. The vessels of the small intestinal submucosa were affected, but there was no associated mucosal lesion.
Cat No. 5. A 1-year-old spayed female domestic short-haired cat presented with a complaint of weight loss for 1 month. The veterinarian noted a decreased packed cell volume and white blood cell count but no other clinical abnormalities. The cat died acutely at home. At necropsy, approximately 50 ml of serosanguineous fluid was found in the thoracic cavity. Again, the arterioles of the heart were most severely affected (Fig. 3). The vessels involved were located in the subepicardial one third of the left ventricular myocardium, the interventricular septum, and the right ventricular myocardium. This cat developed marked pulmonary edema and histiocytic interstitial pneumonia. Vessels of a grossly enlarged mesenteric lymph node were also affected.
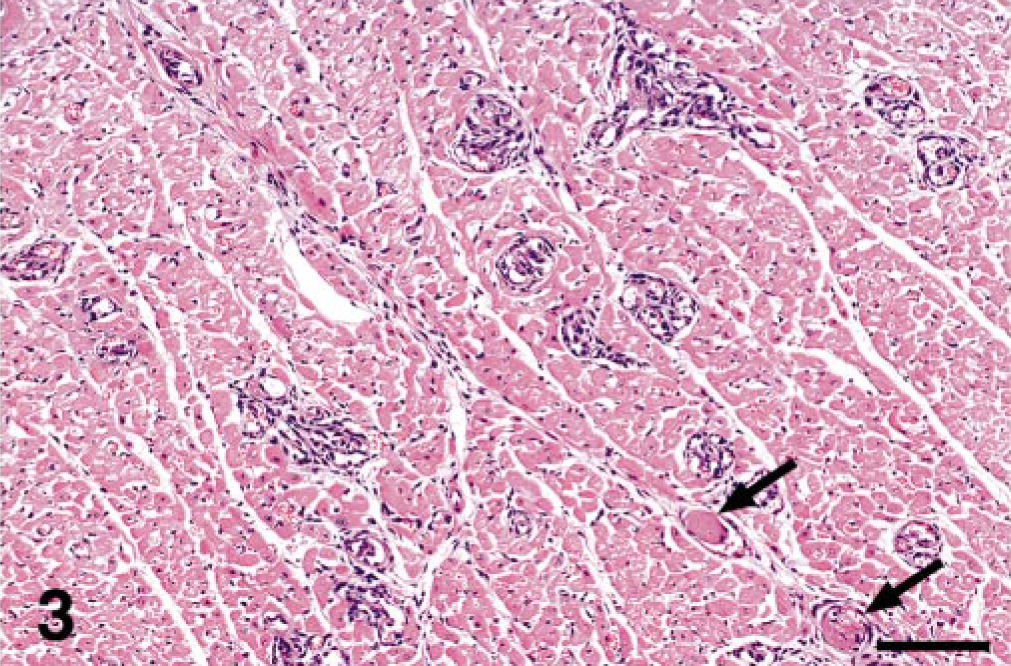
Heart; cat No. 5. Prominent enlargement of many arterioles within the myocardium by intravascular proliferation of spindle cells. A few vessels contain fibrin thrombi (arrows). HE. Bar = 120 μm.
Cat No. 6. A 5-year-old castrated male domestic short-haired cat presented with a 2-day history of lethargy and dyspnea. Thoracic X-rays showed diffuse pulmonary disease compatible with pulmonary edema and/or interstitial pneumonia. The cat died despite supportive therapy. Grossly, the lungs were firm with a pink-to-red mottled surface. As in the majority of the affected cats, this cat developed moderate interstitial pneumonia from chronic passive congestion with edema, alveolar histiocytosis, and scattered hemosiderophages. The majority of the arterioles and small arteries of the heart (right and left ventricular free walls and interventricular septum) were severely affected, and the proliferative spindle cells formed glomeruloid structures within thin, smooth muscle walls. The vascular lesions in this cat were chronic, with marked adventitial fibroplasia surrounding the affected vessels (Fig. 4). Vessels of the kidney, spleen, small intestines, and lymph nodes were also affected. The liver was characterized by a mild lymphoplasmacytic portal hepatitis but with no vascular lesions.
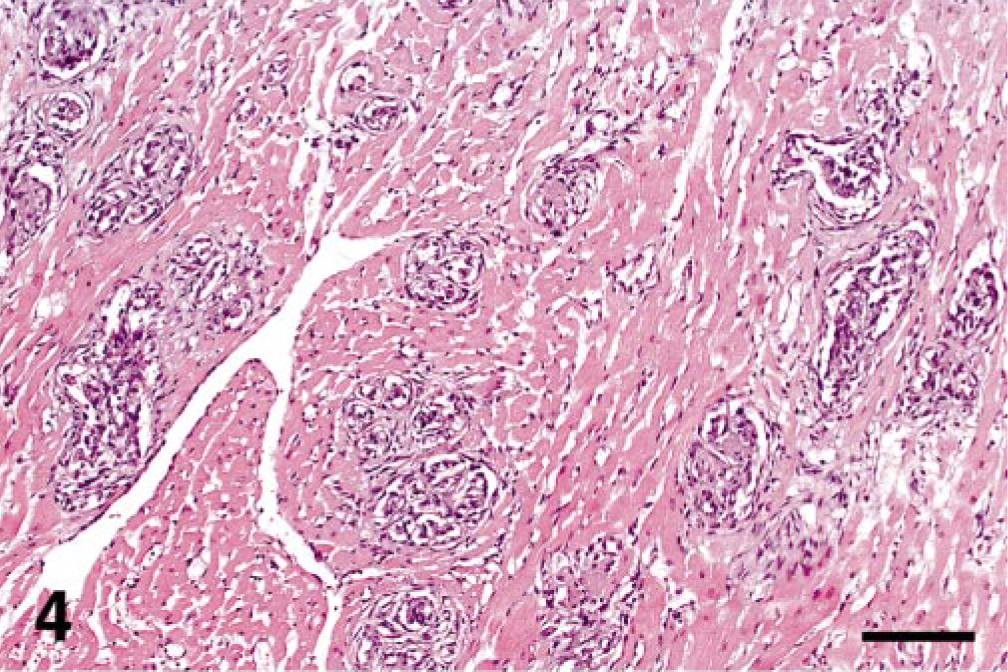
Heart; cat No. 6. Severe, chronic involvement of myocardial vessels with formation of glomeruloid structures and marked perivascular adventitial fibroplasia. HE. Bar = 120 μm.
Cat No. 7. A 7-year-old castrated male domestic short-haired cat presented with a 2-day history of anorexia, lethargy, and dyspnea. Physical examination identified increased bronchovesicular sounds bilaterally with intermittent panting, proprioceptive deficits in the pelvic limbs, and negative menace and pupillary light response on the left. A complete blood cell count and serum chemistry showed a regenerative anemia, neutropenia, thrombocytopenia, and mildly elevated total bilirubin and alanine transferase. The next day, the cat was demented and minimally responsive; the owners elected euthanasia.
The carcass was diffusely icteric. The entire gastrointestinal tract contained a large amount of digested blood and mucus, and a 2-cm linear ulcer was found near the gastric pylorus. The lungs were edematous, and the thorax contained 30 ml of serosanguineous effusion. There was dark red urine within the urinary bladder and petechiae on the mucosal surface. The bone marrow in multiple sites was bright red.
The small arterial and arteriolar lesions were most numerous in the heart, but they were also present in the spleen, kidneys, brain, spinal cord, sciatic nerve, stomach, small and large intestine, and liver. In addition, the liver contained centrolobular degeneration and necrosis with sinusoidal congestion. The spleen had marked EMH, and there were multiple submucosal hemorrhages in the urinary bladder.
Cat No. 8. Sections of heart, liver, and kidney were submitted for histopathologic evaluation from a 14-year-old castrated male domestic shorthaired cat with a 2-year history of chronic hepatitis, persistent eosinophilia, and congestive heart failure. The referring veterinarian described no terminal illness and no gross necropsy findings. The heart contained numerous arteriolar intravascular proliferations, with fewer in the liver and kidney. The lesions were similar to those of all cats previously described. However, in the heart, there was diffuse neutrophilic myocarditis with myocyte degeneration and necrosis and fibrin deposition associated with the vascular lesions. The liver had severe lymphoplasmacytic portal hepatitis.
Results
In all cats, the majority of intravascular spindle cells (85–95%) stained positively for vimentin, while the proportion of these cells positive for vWF antigen ranged from 30 to 85% among cats (Fig. 5). A smaller proportion (5–50%) stained positively for SMA (Fig. 6). These results suggest that the proliferating cells were a mixture of endothelial cells and pericytes. In the most severe lesions, the pericytes often encircled the newly formed endothelial cell-lined channels in an orderly arrangement. In the heart, where thrombi were often seen in the affected vessels, vWF antibody stained these platelet-rich thrombi as well as the exuded perivascular plasma. In cat Nos. 1 and 6, the proliferative adventitial fibroblasts were positive for vimentin and negative for vWF antigen and SMA. Only minimal collagen deposition was detected by Masson's Trichrome staining within and surrounding the vascular plexus. In cat Nos. 1, 2, and 7, all intravascular cells were negative for cytokeratins and the leukocyte antigens, CD3, CD79a, CD18, and CD45RA. No organisms were seen in cat Nos. 5 and 6 by Modified Steiner silver stain.
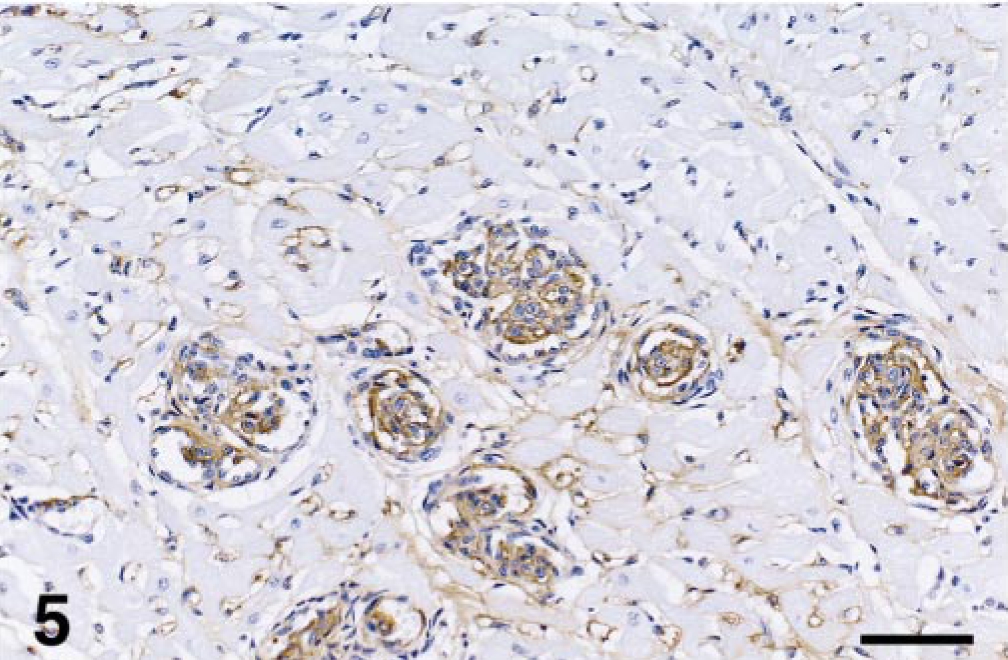
Heart; cat No. 5. Positive vWF antigen staining of many intraluminal spindle cells indicates endothelial cell histogenesis. Immunohistochemical staining for vWF antigen. Bar = 60 μm.
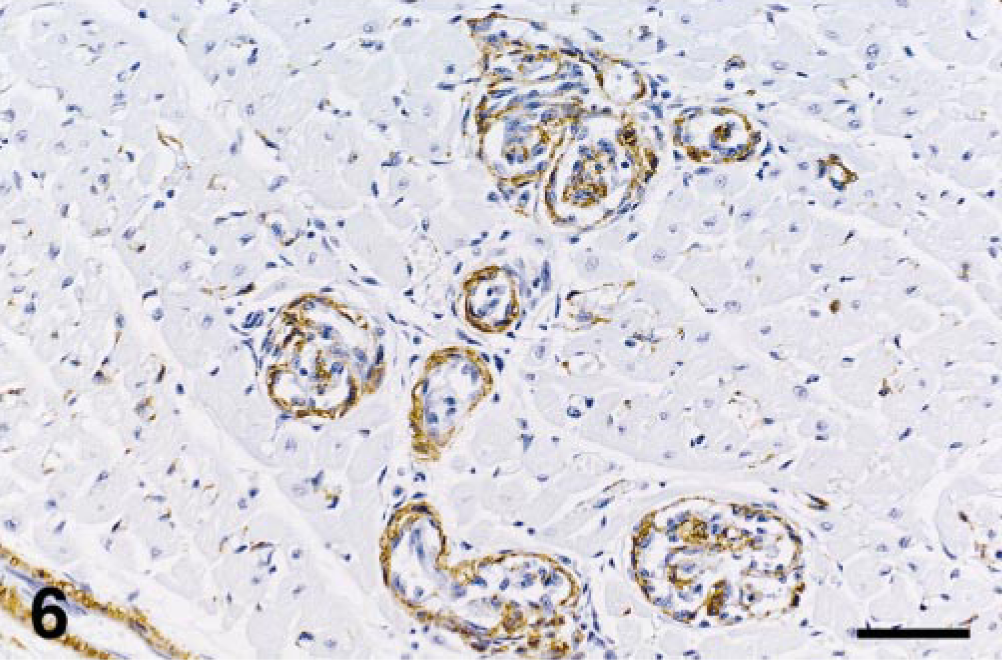
Heart; cat No. 5. Positive SMA staining of arteriolar smooth muscle as well as intraluminal cells (pericytes) admixed with endothelial cells. Immunohistochemical staining for SMA. Bar = 60 μm.
Ultrastructural examination of the affected vessels in the spinal cord of cat No. 7 revealed the intraluminal cells to be plump, irregular, and primarily spindle-shaped within a network of interlacing basal laminae (Fig. 7). Consistent with an endothelial origin, they contained small amounts of rough endoplasmic reticulum, few plasmalemmal Weibel-Palade bodies, few mitochondria, and open-faced nuclei with marginated chromatin. Admixed with these immature endothelial cells were scattered smaller spindle cells, made more electron dense by cytoplasmic bundles of microfilaments and separated from the basal lamina by disorganized deposits of collagen fibrils. Laminated clusters of fibrin, platelets, and occasional erythrocytes were also seen within vascular lumina. No infectious organisms were identified in the lesions.
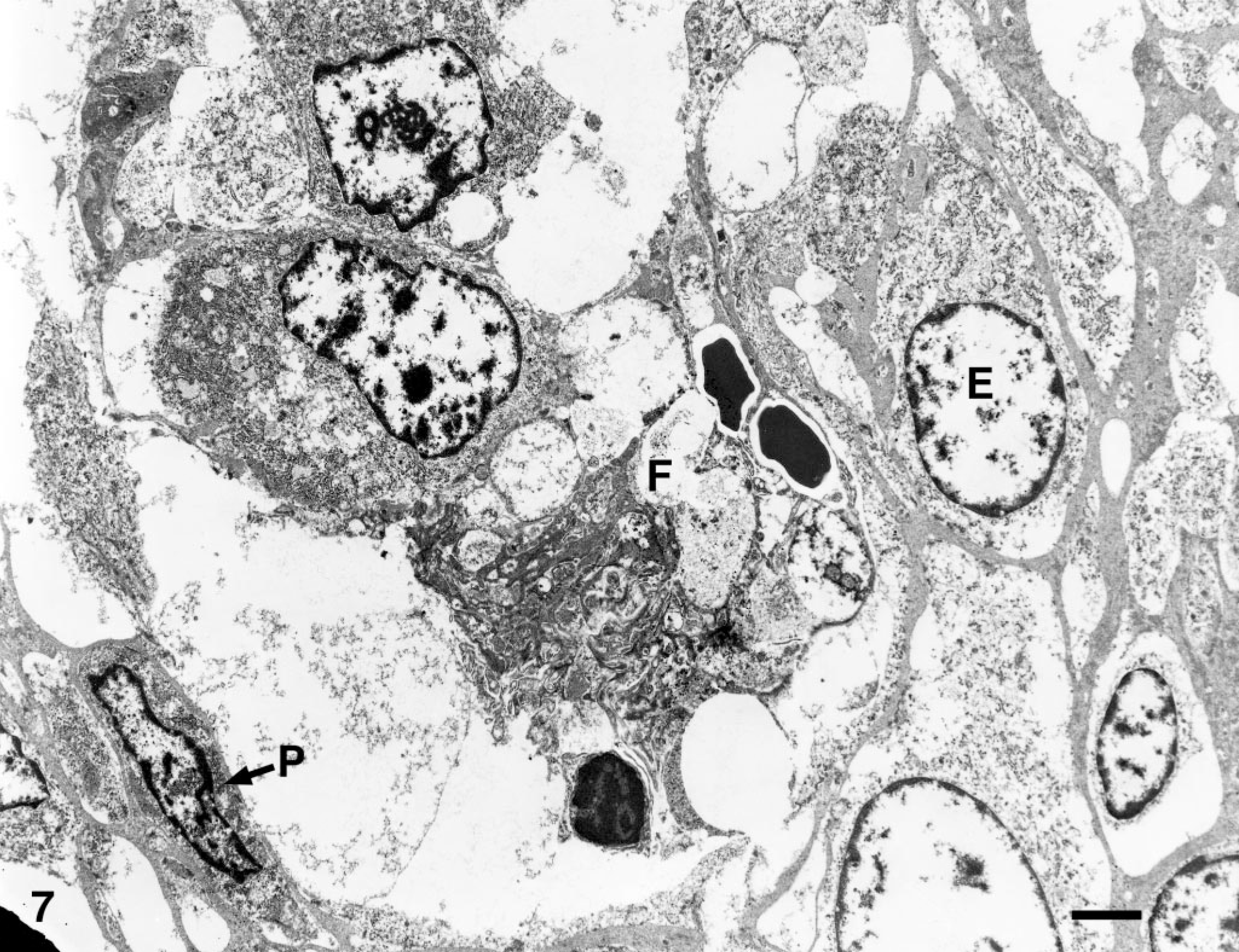
Spinal cord; cat No. 7. Electron microscopic examination shows an intravascular mixture of plump endothelial (E) and pericyte-like (P) cells within a network of interlacing basal laminae. Fibrin, platelets, and erythrocytes are also present within the vascular lumen (F). Bar = 1.8 μm.
Discussion
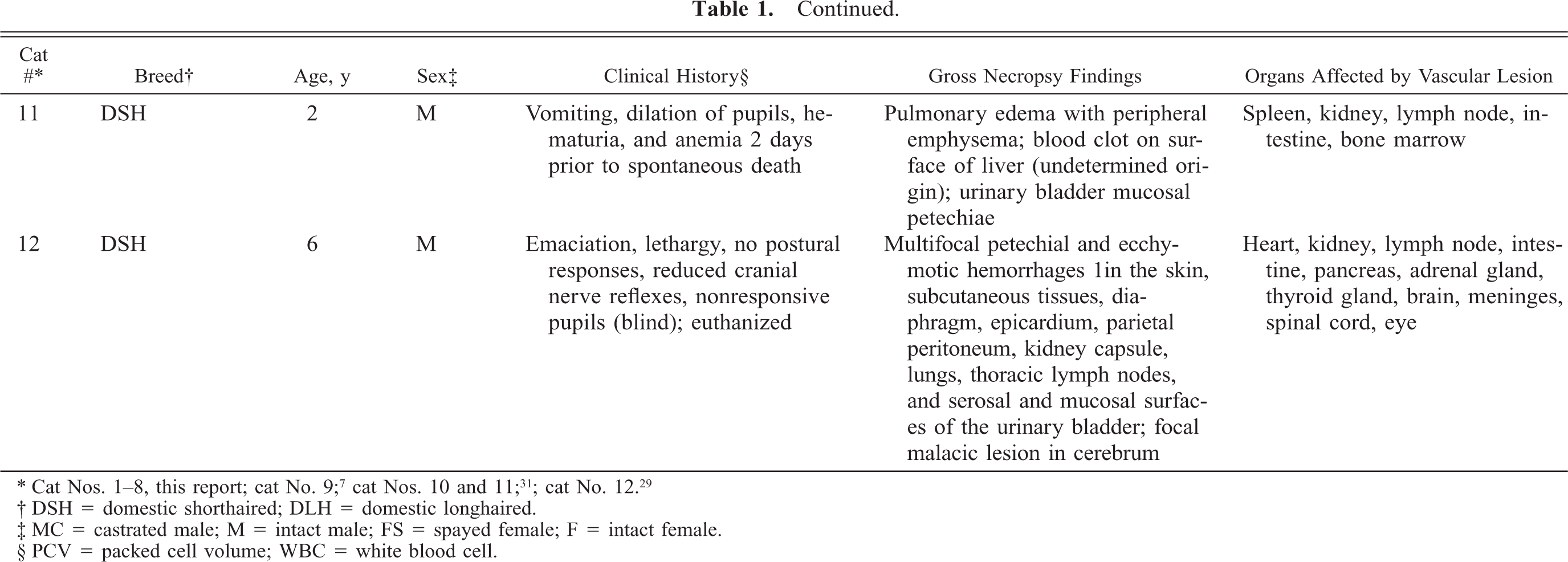
Since 1985, an unusual multisystemic vascular disease characterized by intraluminal endothelial and pericyte proliferation has been documented in four cats (cat Nos. 9–12, Table 1): one from Australia, two from Switzerland, and one from the UK. 7, 29, 31 To our knowledge, similar cases have not been previously reported in the United States. We believe that the identification of only three additional cases cataloged at Cornell University since 1973 and five submitted in consultation since 1998 attests to the rarity of this condition.
Systemic reactive angioendotheliomatosis in 12 cats.
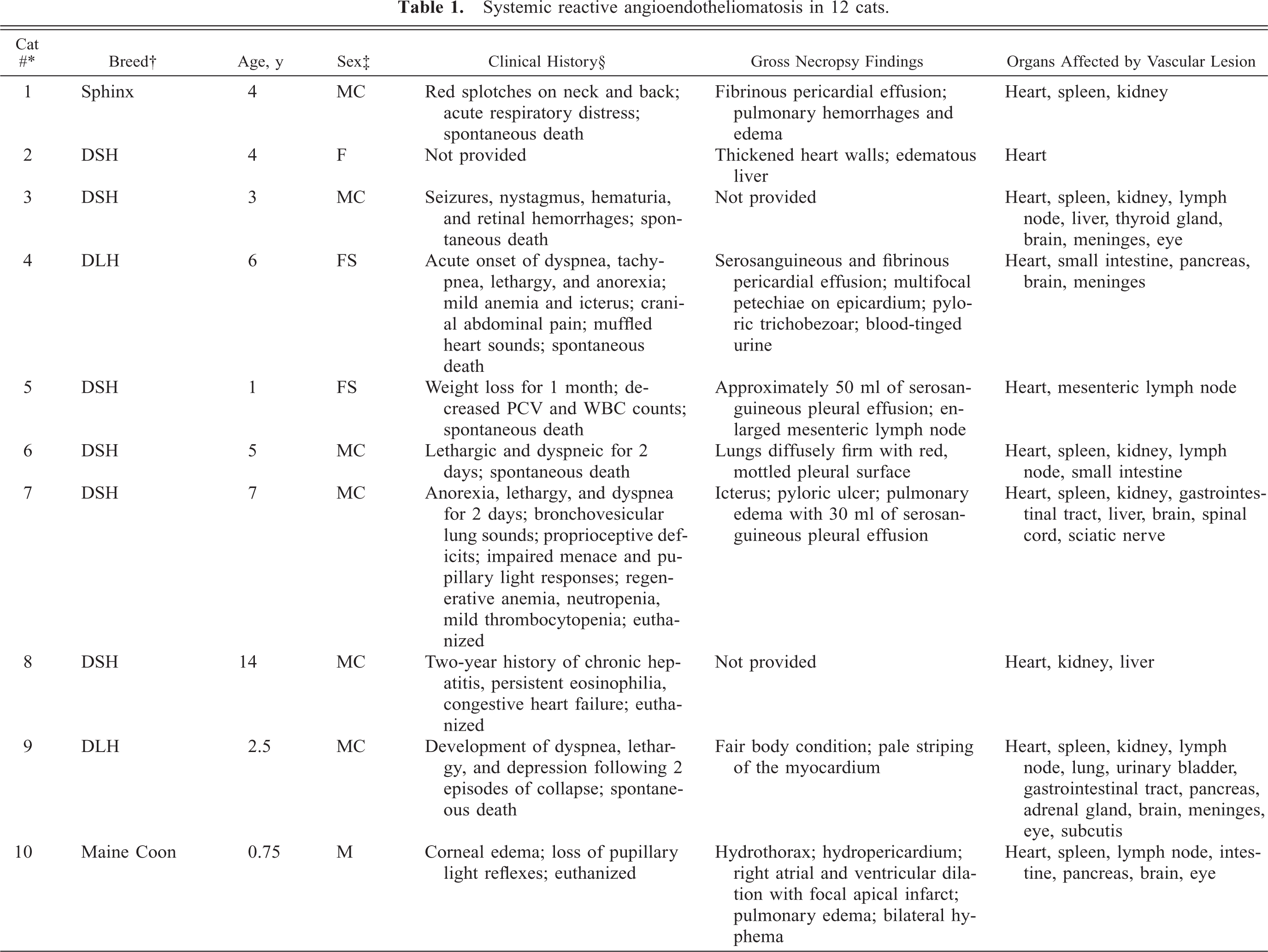
† DSH = domestic shorthaired; DLH = domestic longhaired.
‡ MC = castrated male; M = intact male; FS = spayed female; F = intact female.
§ PCV = packed cell volume; WBC = white blood cell.
Ages of the 12 affected cats ranged from 0.75 to 14 years (median, 4 years), although most were juvenile to young adults (7/12 cats were 4 years or younger). No specific breed appeared to be predisposed; affected animals included eight domestic shorthaired cats, two domestic longhaired cats, a Sphinx, and a Maine Coon cat. Nine of the 12 cats were intact or castrated males; a larger series would be needed to suggest a predilection in the male cat. The heart was the most commonly and severely affected organ, with 11/11 cases (heart not examined in one cat) involving the myocardial vessels. Other organs commonly involved were the kidneys (8/10), spleen (7/8), lymph nodes (7/8), gastrointestinal tract (7/8), brain and meninges (6/6), eyes (4/4), and pancreas (4/4). Affected in only a few cats were the liver (3/11), spinal cord (2/2), adrenal glands (2/3), and thyroid glands (2/3). Involvement of the sciatic nerve (1/1), subcutis (1/3), lung (1/9), bone marrow (1/3), and urinary bladder (1/4) was each identified in one cat. Table 1 summarizes the clinical and pathologic findings from the eight cats of our study and the four previously reported.
In cat Nos. 1, 2, 4, 8, and 10, the vascular lesions were associated with multifocal fibrin thrombosis, hemorrhage, and/or myocardial necrosis, which was the presumed cause of acute myocardial failure and death. The pulmonary edema with alveolar histiocytosis and interstitial pneumonia present in the majority of cats suggest cardiac insufficiency of variable severity and duration. A focal malacic lesion in the cerebrum was associated with the vascular lesions in cat No. 12 and was presumably the cause of the cat's neurologic signs. Petechiae were observed in the organs of cat Nos. 1, 4, 11, and 12, and hyphema was reported to be associated with the vascular eye lesions in cat No. 10. In all other cats, the vascular lesions in organs other than the heart were associated with no significant hemorrhage or parenchymal damage. Minimal protein-rich fluid and red blood cells were present in the proximal tubules of the kidney in cat No. 3. EMH in the spleen in cat Nos. 3 and 7 may suggest regenerative anemia.
Histologically, the vascular lesions of the four reported cats were similar to the eight cats of our study. In the cats of our study, the lesions varied in both chronicity and severity of the intraluminal proliferation. The majority of involved vessels appeared to be small arterioles, each composed of a thin (1–2 cells thick) tunica media surrounding an endothelial cell–lined lumen and lacking an internal elastic lamina. The location of the affected vessels in the kidney is consistent with the periglomerular arterioles. Immunostaining for vWF antigen performed on tissues from the previous cases confirmed that a variable number of the intravascular proliferative spindle cells were of endothelial origin. Immunostaining on the cats of our study was similar. To determine the identity of the remaining vWF-negative spindle cells, Rothwell et al. 29 performed electron microscopic studies that showed pericyte-like cells admixed with endothelial cells. Our ultrastructural studies yielded similar findings, and the SMA staining of a variable proportion of the proliferative cells in the cats of our study further supports a pericytic origin.
No similar multisystemic syndrome has been identified in humans; however, several cutaneous disorders characterized by proliferative, mixed endothelial cell and pericyte, intravascular lesions have been described. These include intravascular papillary endothelial hyperplasia (IPEH), RAE, acroangiodermatitis (pseudo-Kaposi's sarcoma), and glomeruloid hemangioma (POEMS syndrome). 28
IPEH was first described by Masson in 1923, named hemangioendothelioma vegetant intravasculaire, and was suspected to be a neoplasm. Now believed to be an unusual form of organizing thrombus, Levere et al. 21 demonstrated its association with elevations in basic fibroblast growth factor (bFGF). They hypothesized that increased bFGF results in an autocrine feedback loop, stimulating exuberant endothelial cell proliferation. Clinically, IPEH manifests as a solitary cutaneous nodule that histologically consists of a dilated blood vessel expanded by numerous papillary ingrowths of endothelia covering delicate collagenous cores. Ultrastructurally, endothelial cells, pericytes, and undifferentiated cells have been identified. 8 Recently, a case of IPEH has been reported in the eye of a horse, with the lesion arising from a presumed conjunctival vascular malformation. 11 In the feline disorder, the lesions are multisystemic rather than focal and nodular, but the common presence of thrombi within proliferative spindle cell tufts suggests that this lesion also may represent an exaggerated response to thrombosis. In several cats, the distention of the vessels by an intravascular arrangement of small, capillary-sized channels supported by pericytes and interlacing fine collagen fibers is reminiscent of a recanalized vessel.
RAE is the disorder in people that has historically been confused with intravascular angiotropic lymphoma (malignant angioendotheliomatosis), although RAE has always been clinically distinct in that it is confined to the skin and is self-limiting. The cytologic features of RAE differ from lymphoma, with the former characterized by glomeruloid whorls of bland spindle cells often obliterating the lumina of the small dermal vessels and associated with microthrombi. The overlying skin is often erythematous and occasionally ulcerates. The majority of the intravascular cells are immunohistochemically positive for vWF antigen, CD34, and Ulex europaeus, indicating that they are endothelial. The remainder are positive for muscle-specific actin. 5, 9, 20, 26, 30, 35, 36 Although the feline vascular lesions are multisystemic, morphologically and immunohistochemically, they are most similar to this condition in humans. Thus, we propose the acronym FSRA as the morphologic description of this disease.
A variety of associated conditions have been identified in about 75% of the approximately 35 reported cases of human RAE. 23 One hypothesis is that the lesion represents an unusual residuum of leukocytoclastic vasculitis, such as that due to immune complex deposition in a hypersensitivity reaction. In a 5-month-old child studied by Wick and Rocamora, 36 the suspected antigen was a cow's milk protein, as the lesions resolved following dietary change. Other immune-mediated reactions that have been suspected to cause this unusual vasculopathy include those associated with chronic infections (subacute bacterial endocarditis, Chaga's disease, pulmonary tuberculosis, and hepatitis C virus) and autoimmune diseases such as rheumatoid arthritis and systemic lupus erythematosus with anti-phospholipid syndrome. 6, 20, 26, 32 In the cats of our study, no underlying infectious or inflammatory disease was documented clinically or identified at necropsy, and minimal to no inflammatory cells were observed within the affected vessels.
Further cutaneous conditions that may share pathogenic mechanisms with RAE include acroangiodermatitis and glomeruloid hemangioma. Acroangiodermatitis features endothelial cell proliferation associated with thrombotic vessel recanalization or in response to the stimulus of endothelial growth factors induced by ischemia, while glomeruloid hemangioma is a rare cutaneous vascular proliferation (resembling renal glomeruli) in humans with POEMS syndrome (Polyneuropathy, Organomegaly, Endocrinopathy, M-protein, Skin changes) and is associated with multicentric Castleman's disease. 14, 16, 27, 33, 37
Occlusive, intra-arteriolar endothelial and pericytic proliferations (termed glomera or glomeruloid structures) have also been described in humans in unusual cases of chronic glomerulonephritis, in plexiform vessels of the lung associated with primary pulmonary hypertension, and multisystemically in cases of chronic disseminated intravascular coagulation and thrombotic thrombocytopenic purpura (TTP). 34 While the pathogeneses of the vascular lesions are distinct and complex in these various diseases, all are believed to represent a “unique form of exuberant angiogenesis,” with a focus on platelet aggregation, angiogenic cytokines, and/or dysfunctional endothelial regulation of the coagulation and fibrinolytic systems. 12 TTP is characterized by platelet thrombi within the small vessels of many organs, primarily the central nervous system and kidneys. The disease is caused by an inherited or acquired failure to degrade unusually large multimers of vWF. 2, 25 Although the vascular lesion in cats is histologically similar, the clinical presentation of TTP includes thrombocytopenia and erythrocyte fragmentation, which were not consistent findings in the cats of our study.
Proliferative endothelial cell lesions have also been reported with infectious diseases in humans with acquired immunodeficiency due to human immunodeficiency virus, particularly Kaposi's sarcoma (human herpesvirus-8) and bacillary angiomatosis (Bartonella henselae and Bartonella quintana). 3, 4, 17 We have viral serology data on only two animals (cat Nos. 4 and 10), both of which were negative for FIV, FeLV, and FIP. A Modified Steiner silver stain was performed on the cardiac tissue from cat Nos. 5 and 6, and no organisms suggestive of Bartonella spp. were seen, although PCR may be required to adequately evaluate infection by these bacteria. Furthermore, our electron microscopic examination, as well as that performed by Rothwell et al., 29 identified no infectious organisms. In general, infection and bacteremia of cats with B. henselae (also the cause of human cat scratch fever) have not been associated with any clinical disease; however, in experimental infections, lesions reportedly included lymph node and splenic lymphoid hyperplasia, cholangitis, hepatitis, nephritis, and myocarditis. 1, 10, 18 No endothelial proliferative lesions have thus far been seen in cats with experimental Bartonella infection.
That the bland, mixed intravascular proliferations in these cats represent an unusual malignant neoplastic condition such as disseminated intravascular angiosarcoma or endovascular papillary angioendothelioma (Dabska's tumor) is considered unlikely. 26 Further, the novel, mixed intravascular lesions of FRSA are readily distinguished from feline hemangioma and hemangiosarcoma and from the more recently recognized case of cerebral angiomatosis. 15, 24
To our knowledge, the vascular lesion reviewed in this study in cats has not been documented in any other domestic or laboratory animal species. The intravascular proliferations are characterized by glomeruloid whorls of cohesive spindle cells that are immunoreactive for endothelial cell and pericyte markers. Given the multisystemic nature of this disorder in cats and the morphologic similarities to the human entity, RAE, we suggest that this unusual idiopathic lesion be referred to by the acronym FSRA. What remains to be defined for this condition in these primarily young adult and often male cats is the underlying disorder (infection? vasculitis? thrombosis?) with a predilection for the myocardial vasculature that precipitates this widespread and fatal disease.
Footnotes
Acknowledgements
We thank Christina Smith and Joy Cramer for assistance with immunohistochemistry and Alexis Wenski-Roberts for assistance with photomicroscopy.
Addendum
Recently, two further cases of this feline syndrome were reported in abstract form. One cat received hematologic evaluation, leading to the diagnosis of thrombotic thrombocytopenic purpura (Cooley AJ et al. Vet Pathol
