Abstract
Unusual proliferative intravascular lesions were seen in multiple organs of a 2-year-old Corriente steer presumed to be persistently infected with bovine viral diarrhea virus (BVDV), based on widespread immunohistochemical detection of BVDV antigen. Proliferations of spindle cells, which were immunohistochemically positive for von Willebrand factor-related antigen, partially-to-completely occluded vessel lumens and were supported by cells that were immunohistochemically positive for smooth muscle actin. Distribution and character of the intraluminal proliferations are strikingly similar to those described in feline systemic reactive angioendotheliomatosis, a rare entity of unknown cause. The presence of occasional intravascular thrombi suggests that the proliferative vasculopathy was associated with an underlying thrombotic process with immunohistochemical similarities to thrombotic thrombocytopenic purpura of humans. Death of the steer was due to hemorrhage from a castration wound, which may indicate thrombocytopenia or platelet dysfunction. The role of persistent BVDV infection in the formation of the intravascular lesions is unknown.
Keywords
Several intravascular proliferative disorders in humans have been classified as angioendotheliomatosis, which includes benign and malignant variants. 19, 26 Of the malignant variants, intravascular angiotropic lymphoma is the disease entity most commonly referred to as angioendotheliomatosis. 4 Rare cases of malignant angioendotheliomatosis in humans have been substantiated to be disseminated, intraluminal proliferations of neoplastic endothelial cells. 13 Benign reactive angioendotheliomatosis, in which lesions are restricted to the skin, is characterized by proliferation of intravascular endothelial cells and pericytes, is often associated with systemic infections, and is suggested to represent a hypersensitivity reaction to foreign antigens. 11
Systemic reactive angioendotheliomatosis, a rare entity of unknown cause, has recently been described in cats. 10 This disease is characterized by the intravascular proliferation of plump endothelial cells and occasionally surrounding pericytes within multiple organs. Arterioles within the myocardium are the most commonly and severely affected, with lesser but fairly consistent involvement of small vessels within kidneys, spleen, lymph nodes, and intestine. 9, 10, 20, 22 Hematologic evaluation of one of the cats with vascular lesions led to the diagnosis of thrombotic thrombocytopenic purpura, a chronic form of which was proposed to have caused the advanced vascular lesions in this cat. 7 Vascular lesions similar to those reported in cats with systemic reactive angioendotheliomatosis have not been previously documented in other domestic or laboratory animal species. 10 In this report, we describe unusual intraluminal proliferations involving arterioles in multiple organs of an approximately 2-year-old Corriente steer that was persistently infected (PI) with bovine viral diarrhea virus (BVDV). Distribution and character of the vascular lesions in this steer were strikingly similar to those reported in cats with feline systemic reactive angioendotheliomatosis (FSRA).
An approximately 2-year-old Corriente steer that died shortly after postpurchase vaccination, deworming, and castration was presented for necropsy. The steer was thin and had a recent castration wound with large amounts of caked blood on hair of the medial aspects of both hind limbs. Blood was thin and watery; lungs and kidneys were markedly pale. No gross lesions were seen in the mucosa of the rumen, abomasum, small intestine, or colon. No gross lesions were seen in other organs or tissues. At the time of necropsy, death was attributed to marked blood loss associated with uncontrolled hemorrhage from the castration wound.
Representative tissue samples from various organs were fixed in 10% neutral-buffered formalin, processed routinely, and embedded in paraffin for histopathologic and immunohistochemical examination. Immunohistochemistry (IHC) was performed on a Dako autoimmunostainer using the streptavidin-biotin complex method with the chromogens DAB (Dako) or Nova RED (Vector Laboratories). The primary antibodies included anti–smooth muscle actin (anti-SMA) antibody (monoclonal mouse anti-human smooth muscle actin, clone 1A4, Dako) at a dilution of 1 : 150, anti–von Willebrand factor antibody (polyclonal rabbit anti-human vWF, Dako) at a dilution of 1 : 1200 and anti-bovine viral diarrhea virus-1 (BVDV1) antibody (monoclonal mouse-anti-BVDV1, clone Mab 3.12 F1, Oklahoma State University) at a dilution of 1 : 30,000. Secondary antibodies (biotinylated rabbit anti-mouse IgG or biotinylated goat anti-rabbit IgG) were used at a dilution of 1 : 500. Negative controls used nonimmune mouse IgG or rabbit IgG in place of the primary antibody.
Histopathologic examination included heart, liver, lung, lymph nodes, kidney, adrenal gland, and brain; lesions in these tissues were limited to blood vessels. Microscopically, lesions were most severe in the heart in which numerous arterioles were partially-to-completely occluded by plump spindle cells that often formed glomerulus-like arrangements within their lumens (Fig. 1). Variably sized vascular channels were present within and adjacent to the glomeruloid structures within affected arteriolar lumens. Myocardium in the region of affected vessels was characterized by cardiomyocyte loss with replacement by coalescing bands of fibrosis that contain a moderate infiltrate of lymphocytes, plasma cells, neutrophils, and macrophages. In a few scattered foci, cardiomyocytes were acutely necrotic. Intravascular lesions similar to those in the heart were also present in other organs, especially lymph node and spleen, and less frequently in adrenal glands and kidneys.
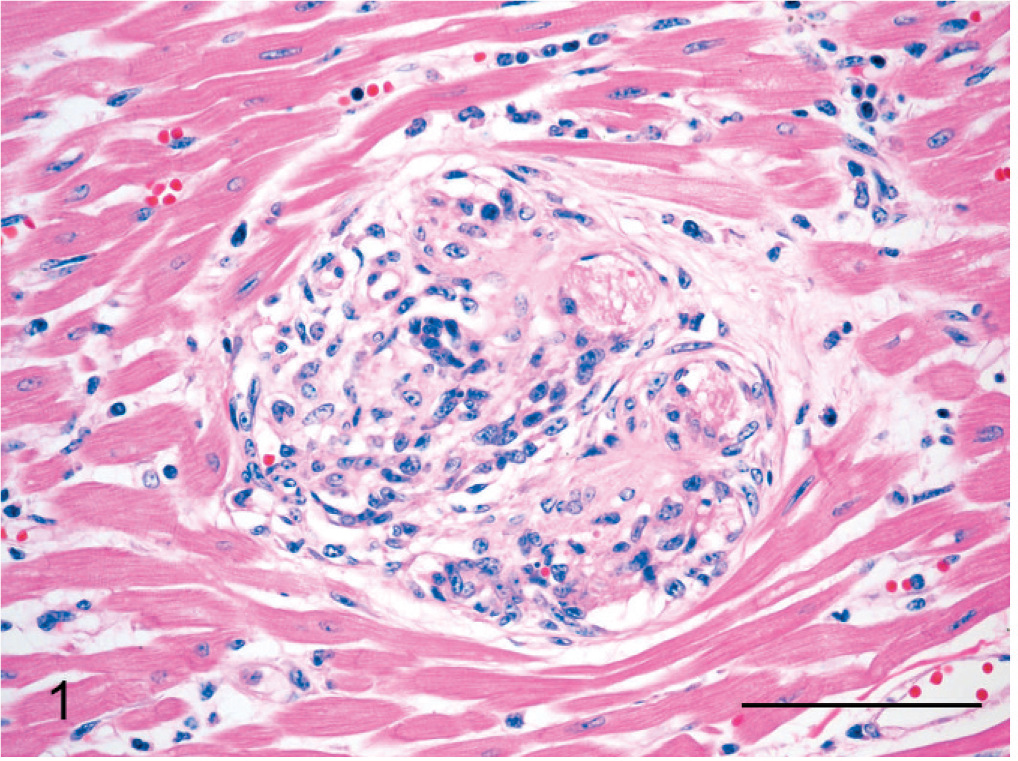
Heart; steer. Arteriole that is expanded and partially occluded by a glomerus-like intraluminal proliferation of spindle cells. HE. Bar = 75 μm.
Many of the plump spindle cells in vessel lumens, especially those lining the vascular spaces remaining within the intraluminal proliferations, were IHC-positive for vWF (Fig. 2a). Prominent populations of spindle cells, particularly toward the periphery of intraluminal proliferations and in the center of glomeruloid “tufts” supporting vWF-positive cells, were strongly positive for SMA (Fig. 2b). A moderate number of the spindle cells in glomeruloid intravascular proliferations were positive for BVDV antigen via IHC stains (Fig. 3); smooth muscle cells within the wall of unaffected vessels were also BVDV positive. Scattered cardiomyocytes had cytoplasmic positivity for BVDV antigen, especially in regions of myocardial necrosis and fibrosis. No BVDV immunoreactivity was seen in blood vessels of tissues from a calf without BVDV infection. All intravascular cells were IHC negative for CD3 and CD79a. In addition to organized glomeruloid structures, occasional vessels (especially within the adrenal gland) contained granular to hyalinized eosinophilic accumulations that were often attached to and continuous with vascular walls (Fig. 4). These accumulations are most consistent with platelet thrombi, given their strong IHC positivity for vWF (Fig. 5) and tinctorial qualities that are inconsistent with fibrin in phosphotungstic acid hematoxylin (PTAH) and Masson's trichrome-stained sections.
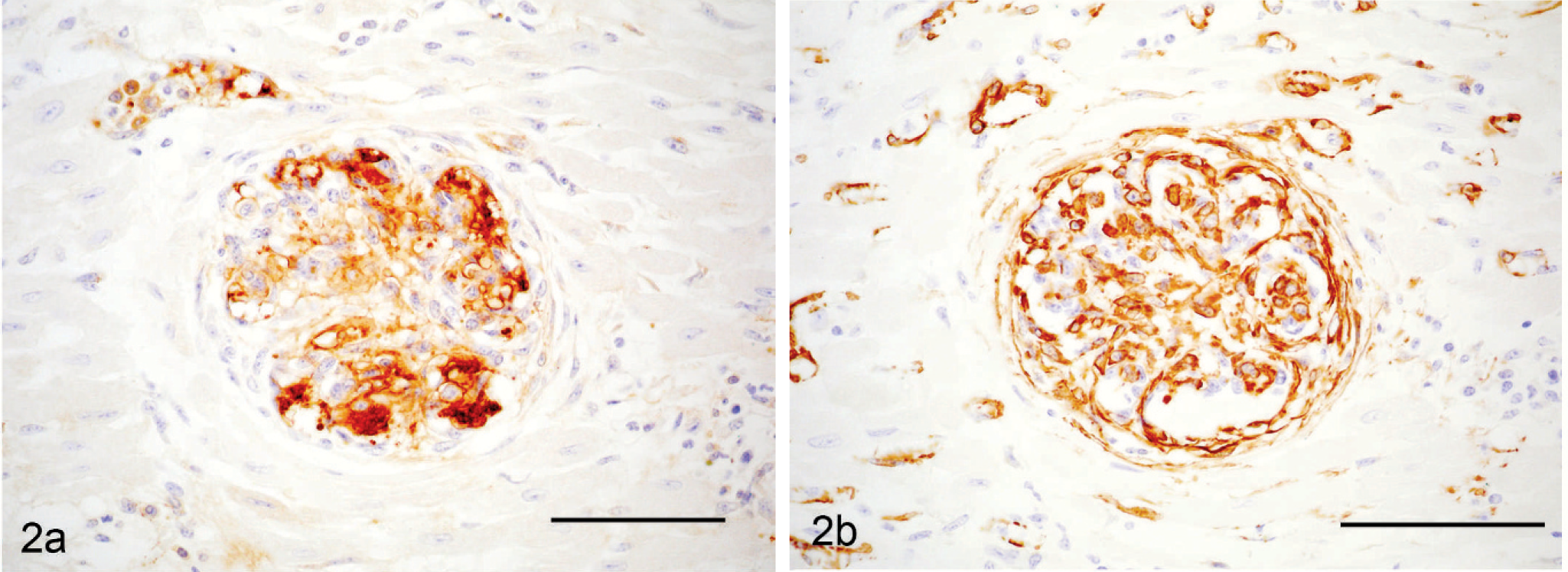
Heart; steer. Intraluminal spindle cells within an arteriole exhibiting changes similar to the vessel in Fig. 1. Endothelial origin of the proliferating spindle cells is indicated by positive vWF antigen staining. Immunohistochemical staining for vWF antigen. Bar = 75 μm. Fig. 2b. Heart; steer. Serial section of arteriole in Fig. 2a. Intraluminal smooth muscle and pericytes, which stain positive for smooth muscle actin (SMA), support proliferating endothelial cells that form glomeruloid expansions within vessels lumens. Immunohistochemical staining for SMA. Bar = 75 μm.
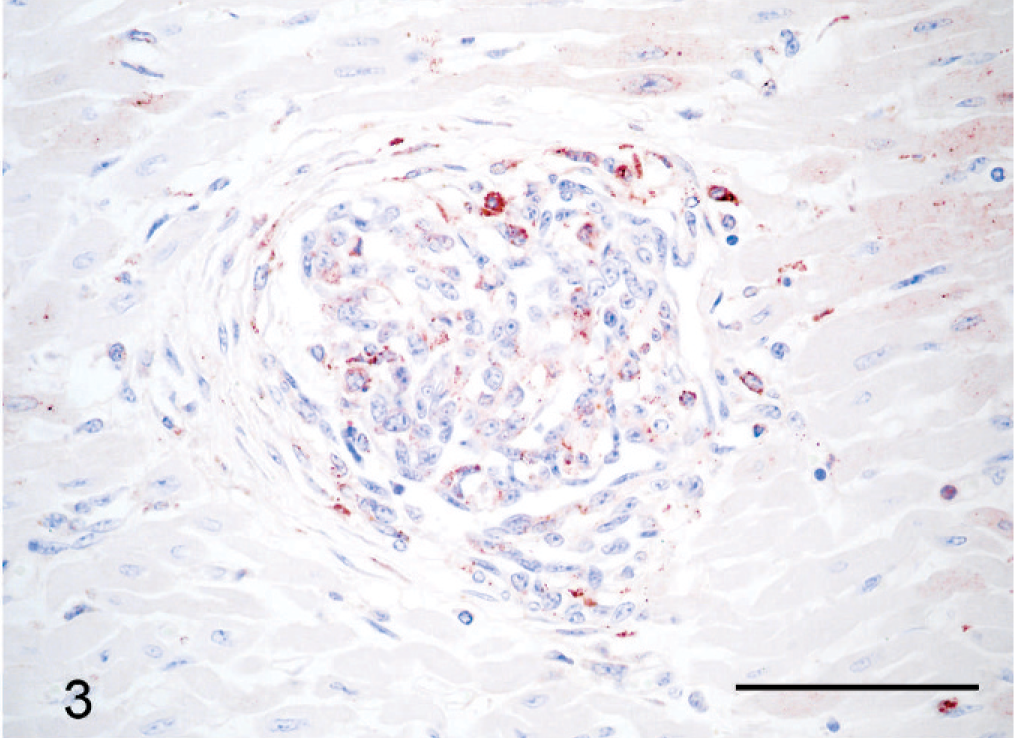
Heart; steer. Arteriole similar to that in Figs. 1 and 2. Scattered cells within the intraluminal glomeruloid proliferation have moderate granular intracytoplasmic positivity for BVDV antigen. Immunohistochemical staining for BVDV antigen. Bar = 75 μm.
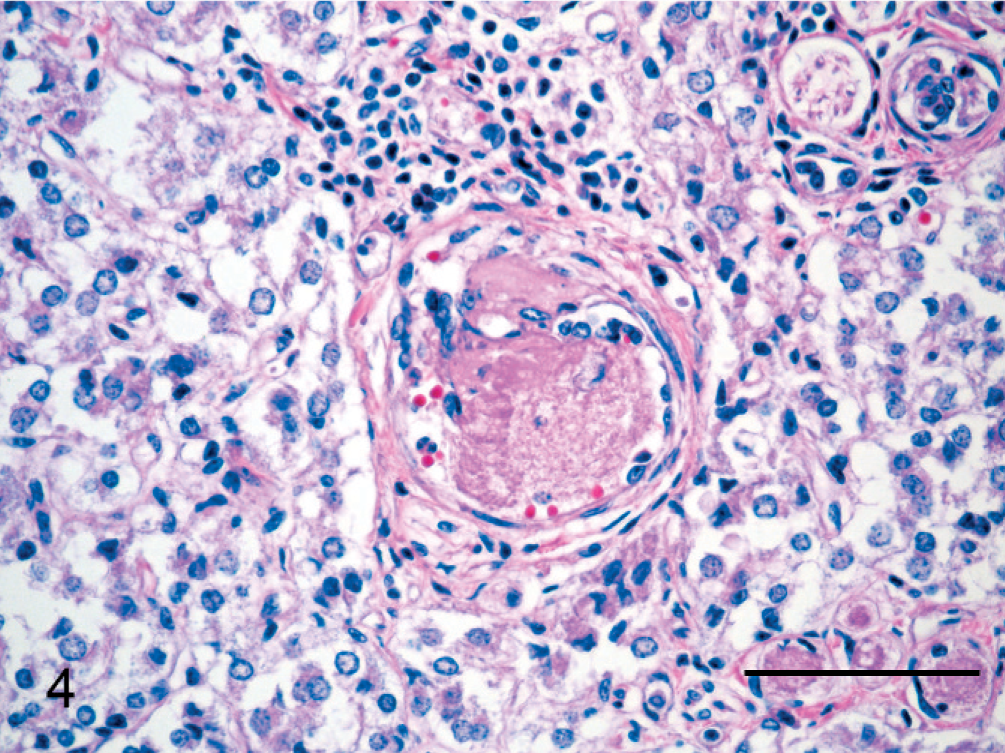
Adrenal gland; steer. Arteriole with focal intraluminal spindle-cell proliferation that is partially occluded by a granular, eosinophilic thrombus. HE. Bar = 75 μm.
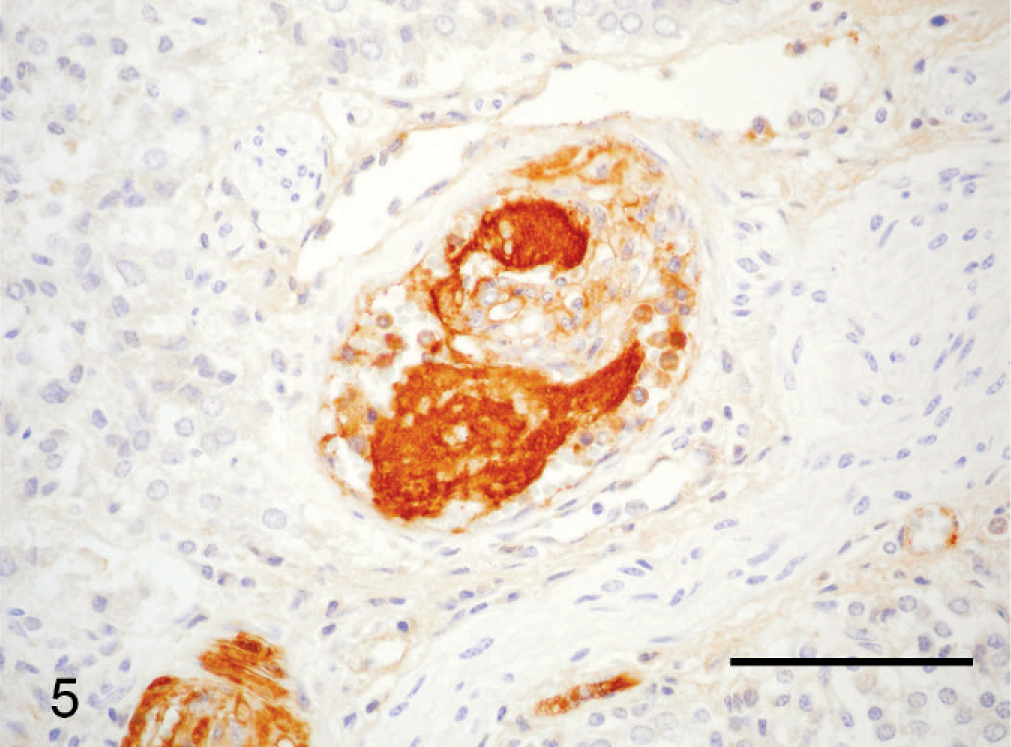
Adrenal gland; steer. Arteriole with thrombus similar to that in Fig. 4. Intense positivity for von Willebrand factor (vWF) antigen within the thrombus is consistent with a platelet thrombus rather than a fibrin thrombus. Immunohistochemical staining for vWF. Bar = 75 μm.
Histologically, the glomeruloid spindle cell proliferations within arteriolar lumens are virtually identical to the lesions of the rare intravascular disease of cats, feline systemic reactive angioendotheliomatosis (FSRA). 7, 9, 10, 20, 22 IHC staining of the intraluminal spindle cells are consistent with proliferation of both endothelial cells and pericytes, as demonstrated for FSRA. 7, 9, 10, 20, 22 In addition, the distribution of affected vessels in this steer is similar to that reported in most cases of FSRA, with the heart being most consistently and severely affected. 9, 10, 20 In all but 2 of the reported cases of FSRA, no cause of the vascular lesions could be determined; however, a reactive or hyperplastic process, possibly in response to thrombosis, vasculitis, or an infectious agent, was believed to be more likely than a neoplastic process. 9, 10, 20, 22 One cat with FSRA had hematologic evidence of thrombotic thrombocytopenic purpura (TTP), to which the widespread and advanced vascular lesions were presumably attributed. 7
TTP, a thrombotic microangiopathy of humans, involves the formation of platelet thrombi due to the accumulation of unusually large multimers of vWF as a result of either an acquired or a genetic defect in vWF metalloprotease. 3 Secondary TTP can be precipitated by drugs, viral infections, pregnancy, and systemic lupus erythematosus and can occur as a single episode, may relapse in intermittent episodes, or persist in a chronic, unremitting form. 1 Glomeruloid structures within arterioles have been associated with TTP, especially in patients with recurrent or chronic manifestations of this syndrome. 23 Proliferative vascular lesions having some features comparable to those in the steer described in this report but lacking the overt formation of glomeruloid structures have recently been reported in Göttingen minipigs with an immune-mediated thrombocytopenic purpura syndrome. 5, 14
The formation of intra-arteriolar glomeruloid proliferations in TTP is hypothesized to be associated with the formation of intravascular thrombi, especially those formed from aggregates of platelets, which are subsequently incorporated into the vessel wall and covered by proliferating endothelial cells. 2, 23 We suspect a similar mechanism for the proliferative vasculopathy in the steer described in this report. The presence of recognizable thrombi within vascular lumens and within vascular spaces of intraluminal glomeruloid structures suggests that endothelial and pericyte proliferation in numerous vessels may be the result of thrombosis with subsequent organization and recanalization. In addition, endothelial-lined spaces within intraluminal proliferations often contain eosinophilic, granular material that is strongly positive for vWF antigen. These granular accumulations do not have tinctorial properties consistent with fibrin thrombi on PTAH or trichrome stains and thus are suspected to be aggregations of platelets rather than fibrinogen or fibrin. The distribution of vascular lesions within arterioles is similar to that in TTP. 2 Neither capillary involvement, which would be expected in disseminated intravascular coagulation (DIC), nor arterial involvement, which is typical of polyarteritis nodosa, was seen in this steer. Lack of venule and capillary involvement to accompany the arteriolar lesions indicates that immune-complex deposition is not a likely cause of vascular lesions.
Due to the widespread distribution of BVDV antigen detected via IHC in the epidermis, hair follicular epithelium, and adnexa, this steer was presumed to be PI with BVDV. 15 Isolation of BVDV from a lymph node of this steer further confirmed BVDV infection. The significance of persistent BVDV infection as it relates to the proliferative vasculopathy in this steer is not known. BVDV antigen has been demonstrated in many tissues of PI cattle including smooth muscle cells, vascular endothelium, and macrophages within vessels walls. 6, 8, 16, 21 IHC detection of BVDV antigen in vessel endothelium and/or walls was occasionally associated with detectable vessel damage; however, the presence of BVDV antigen and vessel lesions was not always colocalized. Segmental vascular necrosis with lymphohistiocytic perivasculitis has been reported in small arteries and arterioles of the intestinal submucosa of cattle that were PI with BVDV and subsequently developed late onset mucosal disease, 12 but intraluminal proliferative lesions have not been previously reported in association with BVDV infection. Affected vessels in the present case are occasionally surrounded by a few mixed inflammatory cells, but intraluminal proliferations are not accompanied by significant inflammation within the vascular tunics, thus thrombosis is likely not secondary to vasculitis. Thrombocytopenia and altered platelet function have been reported in cattle acutely infected with type-II BVDV, but neither decreased platelet numbers nor altered platelet function has been demonstrated in cattle that are PI with BVDV. 18, 24, 25 One study reported a significant increase in platelet counts in BVDV-PI cattle. 17 If the persistent BVDV infection in this steer was associated with the concurrent proliferative vasculopathy, it is possible that BVDV infection altered platelet function and thereby initiated intravascular thrombosis and subsequent recanalization with intraluminal spindle-cell proliferation. Alternatively, persistent BVDV infection may have resulted in widespread endothelial or vessel damage that led to reactive proliferation of intraluminal endothelial cells and pericytes. In any case, the morphology and distribution of the intravascular lesions suggest a reactive process for which the specific initiating factor or factors remain to be defined.
Footnotes
Acknowledgements
We wish to thank Dr. Roger Panciera for critical review of the manuscript.
