Abstract
Chelonian myxozoanosis is rarely reported and has previously not been documented to cause disease. This report describes myxozoanosis associated with significant renal disease in two Crowned River turtles (Hardella thurjii). One turtle presented with emaciation and died. The cage mate presented with emaciation and was euthanized. Histologically, renal intratubular myxozoan spores were associated with renal tubular necrosis, tubular mineralization, and chronic interstitial nephritis, with membranoproliferative and mes-angioproliferative glomerulopathy. Both turtles also had disseminated metastatic mineralization. On the basis of these findings, chronic renal insufficiency from myxozoanosis and subsequent metastatic mineralization were considered the primary problems. By light and electron microscopy, the myxozoan spores had features of the genus Myxidium. Maximum parsimony analysis of small-subunit rDNA sequences placed the turtle myxozoan basal to a clade containing Myxidium truttae and a Myxidium sp. with strong bootstrap support. This myxozoan agent appears to be a significant pathogen in H. thurjii on the basis of morphologic changes in the kidneys of in the infected turtles.
Myxozoa have recently been reclassified from protozoan to metazoan organisms. 7, 15 Almost all representatives of this phylum are parasites of fish, and although a majority of these infections are benign, the pathogenic species are well known. 10 Myxozoan infections are occasionally seen in other vertebrate hosts, particularly amphibians. 12 For example, a Chloromyxum sp. was reported as a significant renal pathogen in Asian horned frogs. 1 Myxozoan infections are also rarely documented in mammals, 3 birds, 11 and reptiles. 4 This report describes renal myxozoanosis associated with significant renal disease in two Crown River turtles (Hardella thurjii).
The Crown River turtle is a large (up to 54 cm) aquatic turtle native to India and Pakistan. Individuals of this monotypic genus prefer bodies of water with a slow current and high concentration of aquatic vegetation. These turtles are considered strict herbivores. 2
The first case (No. 1), a privately owned adult male Crowned River turtle with a carapace length of 13.6 cm and weighing 233 g was presented for necropsy. This animal was part of a larger group of assorted turtles imported from Pakistan for the pet trade. The turtle was housed with a conspecific female from the same shipment. The pair had been in captivity for approximately 2 months before presentation. The owner noted that at the time of acquisition, both animals were dehydrated and underweight. The turtles were kept in shallow water (12 cm) and maintained at an ambient temperature of 23–27°C. The turtles initially consumed small amounts of assorted greens (dandelion, romaine lettuce, endive, etc.). The owner had administered enrofloxacin (Baytril, Bayer, Shawnee Mission, KS) at 10 mg/kg q48h IM for seven treatments, ceftazidime (Fortaz, Glaxo Wellcome, Research Triangle Park, NC) at 20 mg/kg q72h IM for six treatments, fenbendazole (Panacur, Hoechst Roussel Vet, Warren, NJ) at 100 mg/kg PO and repeated 14 days later, metronidazole (Metronidazole, Martec, East Hanover, NJ) at 100 mg/kg PO and repeated 14 days later, and praziquantel (Droncit, Eayer, Shawnee Mission, KS) at 8 mg/kg IM and repeated 14 days later to each turtle. One week before presentation, the male turtle became anorexic, constantly floated, sloughed parts of its skin, and acquired bilateral diffuse corneal opacities. Death occurred approximately 12 hours before examination.
Two weeks later, the second turtle (No. 2), a sub-adult female with a carapace length of 15.5 cm and weighing 215 g, was presented for advanced cachexia, and euthanasia was performed for humane and diagnostic purposes with sodium pentobarbital (Euthasol, Delmarva, Midlothian, VA) administered into the coelomic cavity.
Materials and Methods
For turtle No. 1, samples of skin, skeletal muscle, liver, kidney, spleen, lung, stomach, small intestine, colon, pancreas, and heart were fixed in 10% neutral buffered formalin. For turtle No. 2, urine was collected in a sterile tube at necropsy, and direct and Wright-Giemsa–stained smears were examined microscopically. Samples of heart, lung, liver, stomach, small intestine, colon, pancreas, kidneys, urinary bladder, bone, spleen, skeletal muscle, and head disarticulated at the cervical junction were fixed in 10% neutral buffered formalin. Remaining tissue was frozen. Sections containing bone were decalcified in EDF decalcifying solution (Statlab, Lewisville, TX). All tissues were processed routinely, embedded in paraffin blocks, sectioned at 5 μm and stained with hematoxylin and eosin. Samples of frozen gall bladder and kidney from turtle No. 2 were thawed, and smears were prepared and examined under a light microscope. Thirty spores were photographed, and measurements were made digitally (Diagnostic Instruments Inc., Spot RT Software 3.0, Sterling Heights, MI).
Electron microscopy
Turtle kidney received for electron microscopy in 10% neutral buffered formalin was subsequently transferred to modified (½ strength) Karnovsky's fixative 6 before further postfixation in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. 14 After ossification, tissue was rinsed in 0.2 M sodium cacodylate, dehydrated through a graded ethanol series, and finally infiltrated and embedded in Spurr's epoxy resin formulation. 16 Thick sections, 0.5 μm, were cut, mounted on glass slides, stained with Toluidine blue O, and examined by light microscopy. Thin sections were mounted on 150-mesh copper grids, stained briefly with 6% methanolic uranyl acetate and Reynold's lead citrate, 13 and examined at 60 kV accelerating voltage in a Zeiss 10C transmission electron microscope (LEO Electron Microscopy, Carl Zeiss electron microscopes, Thornwood, NY).
DNA amplification and sequencing
The QIAgen DNeasy Tissue Kit (QIAGEN Inc., Valencia, CA) was used to extract DNA from the thawed infected gall bladder. Methods and myxozoan general primers of Kent et al. 8 were used in a polymerase chain reaction (PCR) to amplify overlapping fragments of the small-subunit (SSU) rRNA gene. Amplification products were sequenced in both directions with PCR primers and AP Biotech® DYEnamic ET Terminator cycle sequencing chemistry with Thermo Sequenase II (Amersham Biosciences, Piscataway, NJ) on an ABI PRISM® 377 DNA Sequencer (Applied Biosystems, Foster City, CA). The resulting sequence fragments were manually aligned, yielding a 2,007-bp partial sequence of SSU rDNA.
Phylogenetic inference
The SSU rDNA sequence of Myxidium hardella n. sp. was aligned to representative sequences of the major myxozoan classes and orders with the Clustal X sequence alignment software. 18 The following sequences from GenBank (accession numbers in parentheses) were used: Myxidium truttae (AF201374), Myxidium sp. from Cottus bairdii (U13829), Myxidium lieberkuehni 1 (X76638), M. lieberkuehni 2 (X76639), Sphaerospora onchorynchi (AF201373), Enteromyxum leei (AF411334), Enteromyxum scophthalmi (AF411335), Zschokkella mugilis (AF411336), Kudoa thyrsites (AF031412), Myxobolus cerebralis (U96493), Henneguya salmonicola (AF031411), and Tetracapsuloides bryosalmonae (U70623). Parsimony analysis was conducted in PAUP∗4.0b10 17 with a heuristic search algorithm with 50 random additions of sequences and tree bisection–reconnection (TBR) branch swapping. Bootstrap values were calculated with 1,000 replicates and a heuristic search algorithm with simple sequence addition and TBR branch swapping. Maximum likelihood analysis employed a heuristic search algorithm with 50 random sequence additions, TBR branch swapping, and the HKY85 substitution model. A heuristic search with simple sequence addition was used to calculate bootstrap values with 100 replicates.
Results
Gross lesions
Turtle No. 1 had multifocal red discoloration interpreted as hemorrhage on the coelomic surfaces of both shells, and the bony plates in some areas were separated at the serrated sutures. The lungs were thickened with no visible adipose tissue. Necropsy of turtle No. 2 revealed no abnormalities other than complete absence of adipose tissue.
Microscopic lesions
In both cases, histologic lesions were most severe in the kidney. Affected tubules were variably dilated and sometimes had necrosis of the epithelial lining, exfoliation of necrotic cells, and accumulations of protein casts or mineralized debris within tubular lumina. Foci of mild lymphocytic peritubular inflammation and fibrosis were also noted. Many tubules contained spore stages of a myxozoan parasite (Fig. 1). Glomeruli had mild to moderate globoid hyaline thickening of the mesangium and mildly increased mesangial cellularity. Basement membranes had moderate to marked hyaline thickening with occasional mineralization and with associated reduction in diameter of capillary lumina. Bowman's capsules were often thickened, and the urinary space was slightly dilated for some glomeruli. Few glomeruli had synechiae of the tuft to the parietal surface of Bowman's capsule (Fig. 2). Turtle No. 1 had myxozoans in bile ducts of identical size and morphology as those in the kidney, but with no associated tissue response (Fig. 3). Myxozoan spores were also detected microscopically in the lumina of the urinary bladder and ureter in histologic sections and in cytologic preparations of urine sediment from turtle No. 2 (Fig. 4).
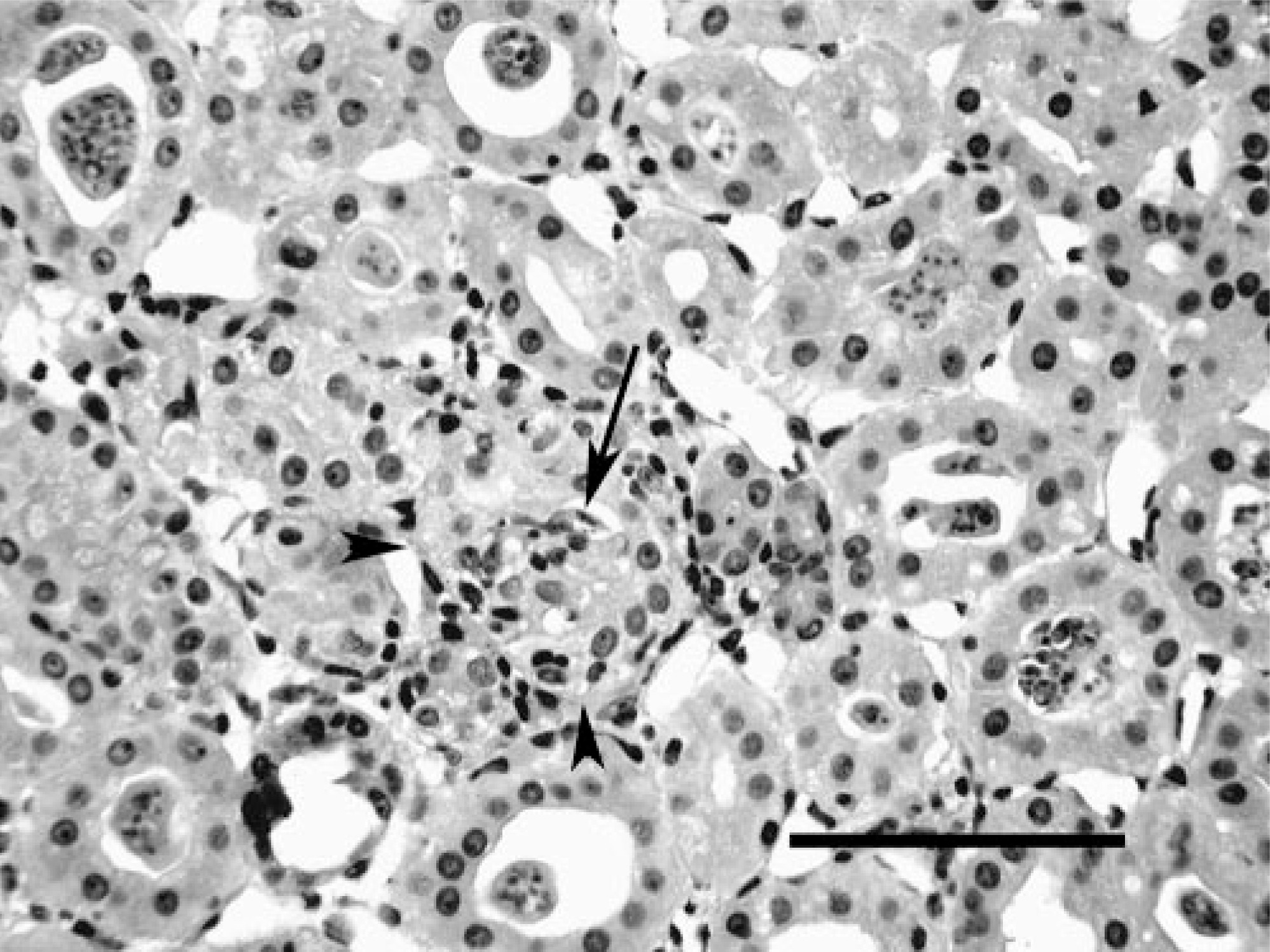
Kidney; Crowned River turtle No. 1. Note lymphocytic inflammation within and around a partially abrogated tubule (arrowheads) containing a myxozoan spore (arrow). Numerous tubules in the field contain luminal plasmodia. HE. Bar = 100 μm.
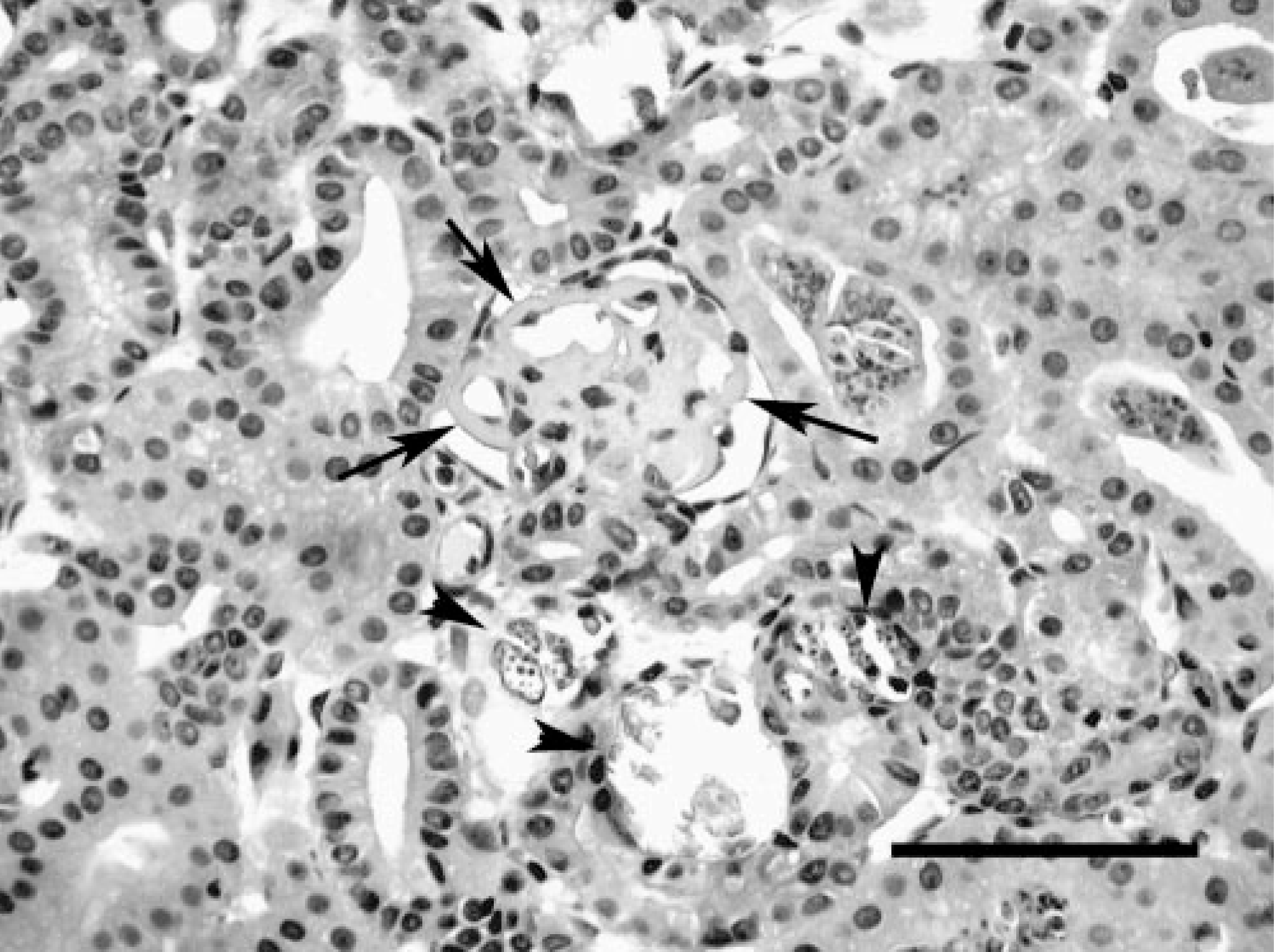
Kidney; Crowned River turtle No. 1. Note thickening of glomerular capillary basement membranes (arrows) and mineralization of adjacent tubules (arrowheads). HE. Bar = 150 μm.
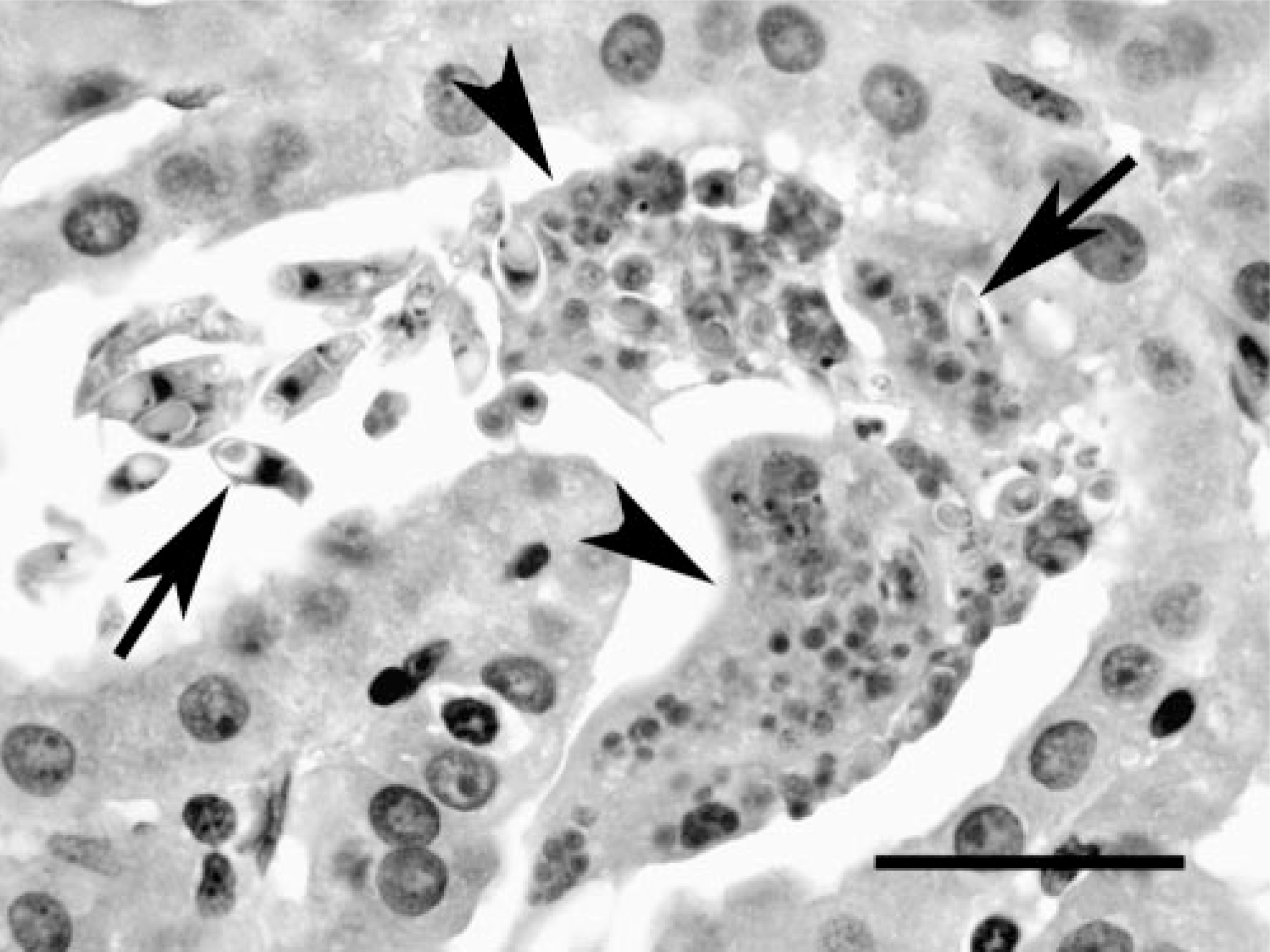
Liver; Crowned River turtle No. 1. Sporogonic plasmodia (arrowheads) and spore stages (arrows) of the myxozoan in bile ducts of Hardella thurjii. HE. Bar = 45 μm.
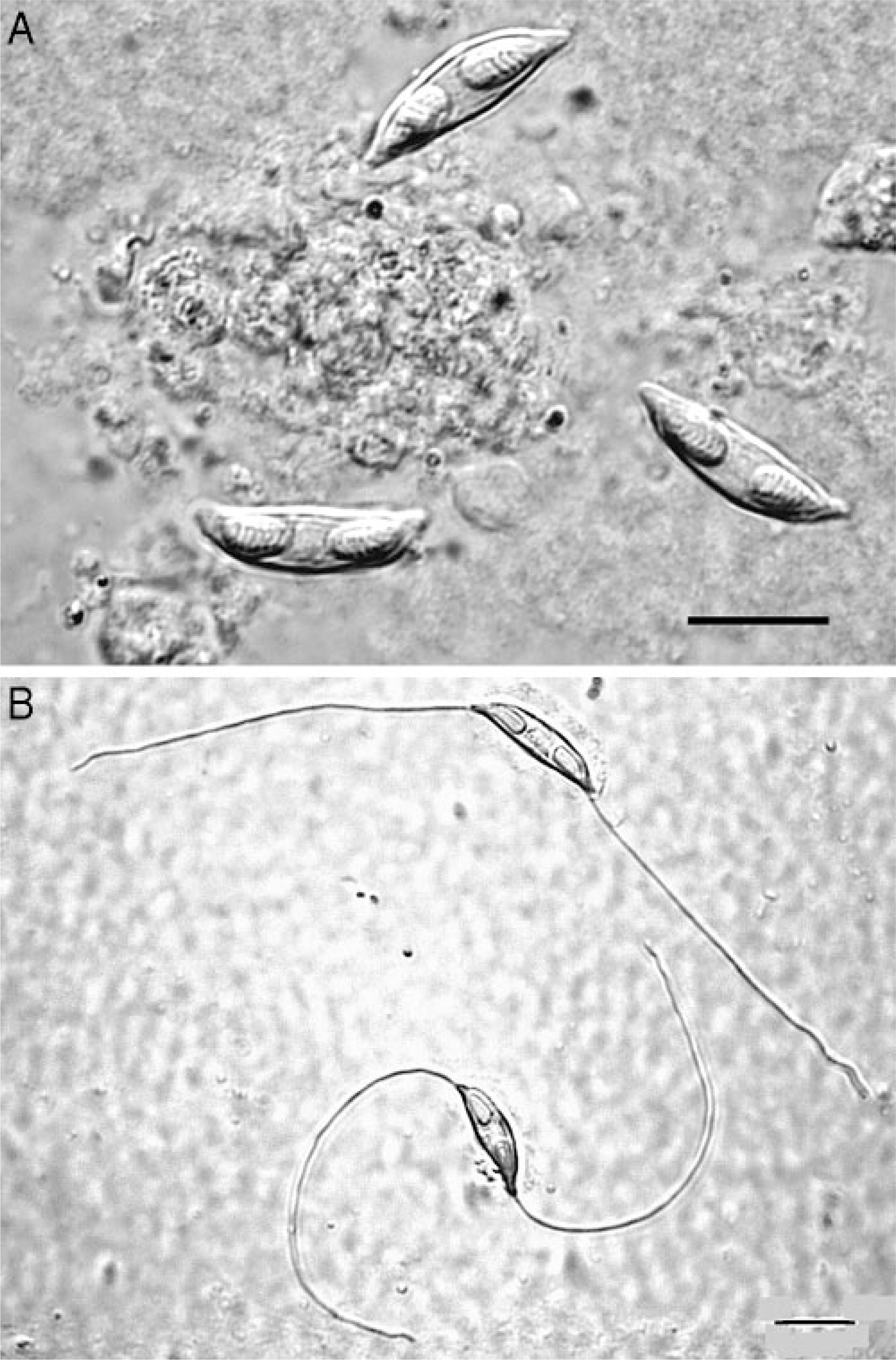
Kidney; Crowned River turtle No. 2. Spores of Myxidium hardella n. sp. from wet mount preparations of (
Multiple additional histologic lesions were noted in both turtles: Extensive, generally severe mineralization involving parenchyma and blood vessels of most coelomic viscera was noted in both turtles. Disseminated but mild visceral trematode ova microgranulomatosis was also present in both cases. Necrotizing to granulomatous vasculitis with intralesional septate fungal elements was seen in the aorta of turtle No. 1, and focal granulomatous inflammation and bone necrosis with septate branching fungal elements was seen in the skin, underlying soft tissues, and dorsal cranial bone of turtle No. 2, although no reactive bone response was noted adjacent to the affected bone. Both turtles had atrophy of fat in the fat bodies adjacent to the kidneys, and atrophy of hepatic cords in centrilobular regions. Case 2 also had granulomatous steatitis and few Sarcocystis-like cysts in skeletal muscle.
Parasite Description
Slide material deposited in U.S. National Parasite Laboratory, Beltsville, MD.
Host. Hardella thurjii
Location in host. Coelozoic in kidney tubules, bile duct, and gall bladder.
Type material. GenBank (AY688957).
Spores. Fusiform and straight to slightly arched with pointed ends. From measurement of 30 spores, average spore length was 17.7 μm (range 14.9–20.0 μm) and width 4.9 μm (range 4.5–5.7 μm). A sutural line bisected each spore. Polar capsules pyriform, equal in size, and situated at both ends of the spore. Average polar capsule length was 6.2 μm (range 4.9–7.1 μm) and width 2.9 μm (range 2.7–3.3 μm). Polar filament with 7–8 coils. Shell valves longitudinally striated (Fig. 5).
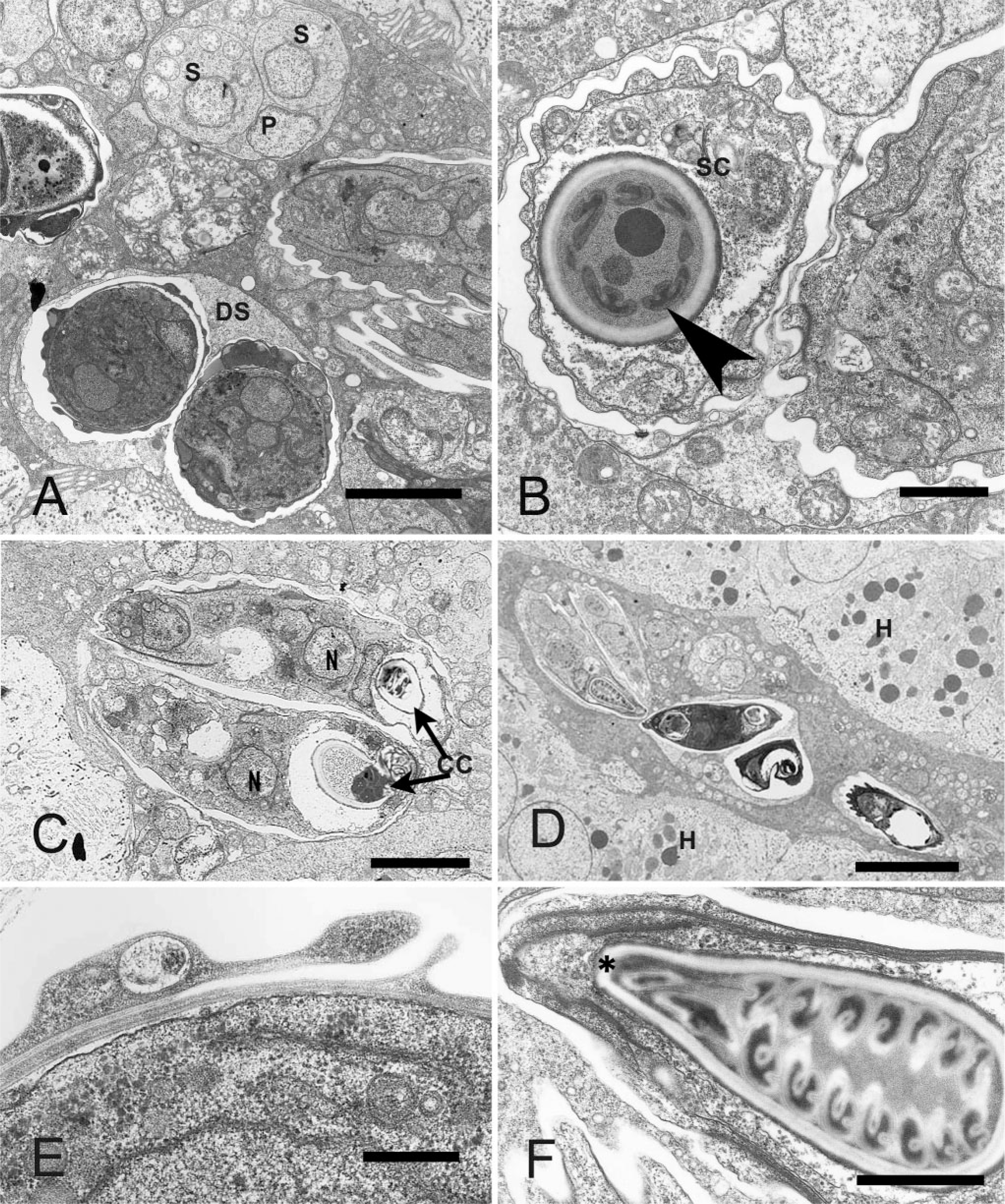
Kidney; Crowned River turtle No. 2. Transmission electron microscopy images of Myxidium hardella n. sp.
Developmental stages. Sporogenesis did not appear to be synchronous because immature stages and mature spores were seen in the lumen of the bile ducts and kidney tubules (Figs. 1, 3).
Ultrastructural observations. Developmental stages included primary cells harboring one or more secondary cells (Fig. 5A), which matured into disporic pan-sporoblasts (Fig. 5A–D). A binucleate sporoplasm was located between the capsulogenic cells, which lie at opposing ends of the spore (Fig. 5B–D). The sutural borders of the two spore valves overlapped and were connected by desmosome-like junctions (Fig. 5E). In more mature spores, the striations of the spore valves (Fig. 5B) and the coils of the polar filament (Fig. 5B, F) were discernible.
DNA sequence. Approximately 2,000 bp of M. hardella n. sp. SSU rDNA sequence (GenBank AY688957) was obtained from infected tissue with myxozoan-specific PCR primers. The most closely related sequences, determined by BLAST searches, were those of M. truttae and an undescribed Myxidium sp. Phylogenetic analysis was conducted with a maximum parsimony algorithm resulting in a single most parsimonious tree (Fig. 6). The resulting tree placed the turtle myxozoan at the base of a group containing M. truttae and Myxidium sp. with strong bootstrap support. Maximum likelihood analysis yielded a tree with identical topology and equally high bootstrap support for the branch nodes.
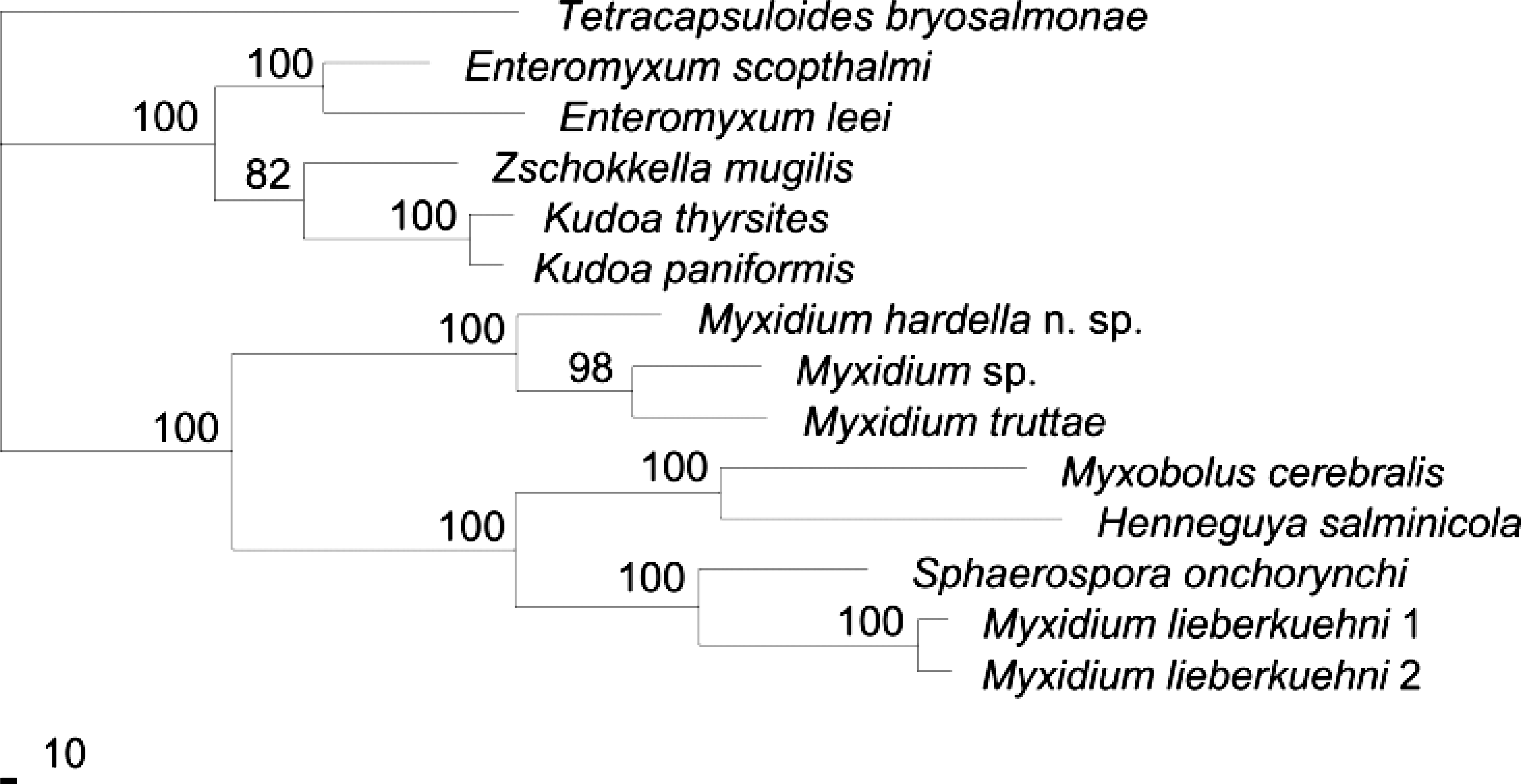
Single most parsimonious tree from phylogenetic analysis of CLUSTAL-aligned 18S rDNA data for Myxidium hardella n. sp. and closely related and representative myxozoan sequences.
Discussion
This is the first description of a myxozoan from a nonfish vertebrate host as a result of both morphological and DNA sequence analysis. Both analyses placed the organism within the genus Myxidium, providing support for classification of species on the basis of DNA sequences data. On the basis of the origin of the host, likely from India or Pakistan, and the distinct morphology of the spores, a new species name was proposed. Although described specimens of Myxidium from turtles share characteristics of the genus, comparison of M. hardella with Myxidium sp. described from North American turtles indicates the spore and polar capsules of M. chelonarum were less elongate 4 and the polar filaments of M. americanum 9 were less coiled. However, definitive comparison with these species would require DNA sequence analysis.
Histologic lesions in the kidney were largely attributed to the presence of myxozoan spores in the renal tubules. Spores often were present in large numbers and appeared to partially obstruct the tubules. Interstitial inflammation and fibrosis could have in part been a result of abrogation of tubules and leakage of urine into the interstitium. Chronic glomerular lesions were also attributed in part to the myxozoan infection, possibly because of increased pressures and gradient alterations associated with the tubular damage. Immune-mediated glomerulonephritis associated with the myxozoan-induced renal inflammation is also considered a possible contributing factor for the glomerular lesion. Age-related scarring or immune-mediated processes associated with other inflammatory events might also have contributed to the glomerular lesions.
Myxozoan spores in which microscopic features were identical to those in the kidney were also detected in the bile ducts of turtle No. 1, although in this location, the spores were not associated with recognizable lesions. Although spores were not seen in the bile ducts of turtle No. 2, spores were found in the freshly thawed gall bladder secretions of turtle No. 2. It is possible that this parasite has a tropism for the biliary system similar to that of the kidney, but with less severe or no associated lesion development. In other species, biliary myxozoanosis can be associated with biliary lesions but can also occur in the absence of recognizable lesions. 10, 11 The presence of spores in the biliary tree indicates potential for shedding of spores via the alimentary tract. Additionally, the presence of spores in the urinary bladder and urine of turtle No. 2 indicates that excretion through the urine is also an important means of shedding the organism. On the basis of these observations, microscopic examination of feces and urine can be useful means of screening for infected turtles.
Mineralization was observed in a variety of tissues from both turtles. Evidence of pre-existing disease in these regions was not apparent; thus, this mineralization was considered to be metastatic mineralization rather than dystrophic mineralization. The turtles had not received vitamin supplements and were on a diet that was not known to cause metastatic mineralization in other species of turtles in the collection. Although serum chemistry analytes were not available for either turtle, it seems probable on the basis of histologic changes in the kidneys that both turtles had renal in-sufficiency that might have contributed significantly to the metastatic mineralization.
Both turtles were emaciated on the basis of clinical findings during physical examination and of the histologic findings of atrophy of fat and liver. The emaciated condition could have resulted from multiple factors, including chronic renal disease, suboptimal nutrition during transport, or the stress associated with maladaptation to a captive environment.
Evidence of acute to subacute mycotic infection was apparent in both animals: one involving skin and underlying soft tissues and the other representing an acute septic event involving the aorta. Although cultures were not obtained, these were likely opportunistic pathogens associated with immunosuppression because of concurrent chronic disease processes such as renal disease, stress, and emaciation.
A variety of tissues from both turtles contained trematode ova granulomas. This condition is common in aquatic chelonians and is sometimes referred to as spirorchidiasis, although often the adult trematodes are not found and the true identity of the trematode ova is not known. In most cases, these organisms are considered incidental findings, although a few reports of significant disease associated with their presence have been documented. 5
Although myxozoanosis has been described in turtles, 4 infection could be a rare event. A retrospective search of the >11,000 reptile cases in the files at Northwest ZooPath revealed no other cases of myxozoanosis in chelonians or other reptiles. This appears to be the first formally documented report of myxozoan infection in chelonians that was associated with significant disease.
Footnotes
Acknowledgements
We acknowledge the expertise of Leroy Brown at Histology Consulting Services for excellent slide preparation and of Jamie Kinion at Northwest ZooPath for data retrieval.
