Abstract
We report a tumor that developed at the root of the tail in a male, 2-year-old Shih Tzu dog. Histologically, the tumor consisted of basaloid epithelial cells, with shadow cells and neoplastic melanocyte proliferation. immunohistochemically, epithelial components were positive for keratin/cytokeratin but basaloid cells were negative. Mononuclear or multinuclear melanocytes were positive for vimentin, S-100 protein, and HMB-45. In conclusion, this dog was diagnosed with canine melanocytic matricoma on the basis of the similarity of the histologic and immunohistochemical features in humans, and it may be the first case in an animal.
Melanocytic matricoma was first diagnosed by Carlson et al. He reported two human cases of a pilomatrical tumor consisting of basaloid epithelial cells, with many shadow cells and neoplastic melanocyte proliferation. 1 In terms of neoplastic melanocyte proliferation, melanocytic matricoma can be differentiated from pigmented pilomatricoma. After their report, Rissardi et al. and Williams et al. also reported human cases that were characterized by an admixture of basaloid tumor cells and neoplastic melanocytes. 2, 5
As mentioned above, melanocytic matricoma is a relatively new diagnosis in humans and there is no report in an animal until now. Recently, we encountered a case in a dog that showed almost the same histopathologic findings as observed in the previous human cases. In this report, we describe the histopathology of this lesion.
The animal in this report is a 2-year-old male Shih Tzu dog that developed a tumor at the root of its tail. The tumor gradually enlarged and was surgically resected 9 months later. No other abnormalities were observed on general inspection and in blood tests. The tumor was solid, black colored, about 2 cm in diameter, and consisted of many nodules. Gray-white liquid flowed from the cut surface (Fig. 1).

Cut surface of formalin-fixed tumor tissue; dog. Gray-white liquid flowed from the cut surface.
The excised tumor was fixed in 10% neutral buffered formalin, embedded in paraffin, and cut into 4-μm sections. The sections were stained with hematoxylin and eosin (HE), Masson's trichrome (MT), and Fontana-Masson (FM) stains were subjected to a bleaching method for melanins using potassium permanganate–oxalic acid method (KMnO4/OA).
Immunohistochemical evaluation was performed by the streptavidin–biotin–peroxidase complex (SAB-PO) method, using Histofine SAB-PO kit (Nichirei Co., Tokyo, Japan). The primary antibodies used were vimentin (Nichirei) and HMB-45 (Signet Laboratories Inc., Dedham, MA) mouse monoclonal antibodies (mAbs) and cytokeratin (Nichirei) rabbit polyclonal antibody and S-100 protein (Nichirei) rabbit mAb. The staining of vimentin, S-100 protein, and HMB-45 was carried out after the bleaching method.
The mass was localized in the dermis and consisted of well-circumscribed nodules, which had no connection to the epidermis. Some nodules contained melanin. The superficial part of the nodules was composed of numerous basaloid cells arranged in several layers. These basaloid cells gradually underwent transition to shadow cell toward the central part of the nodule. In addition, mononuclear or multinucleated cells containing various numbers of melanin granules were also observed intermingled with shadow cells (Fig. 2A). MT-positive collagen was observed around the nodules and intercellular area of the shadow cells. In the nodules containing melanin, melanin was observed as black granules in a wide range of nodules with FM stain (Fig. 2B). The observation of granules was increased with FM stain compared with HE staining. Moreover, these granules disappeared after the bleaching method (Fig. 2C). On the other hand, in the nodules without melanin, there were no FM-positive granules. In another area, severe deposition of melanin and calcification were observed (Fig. 2D).
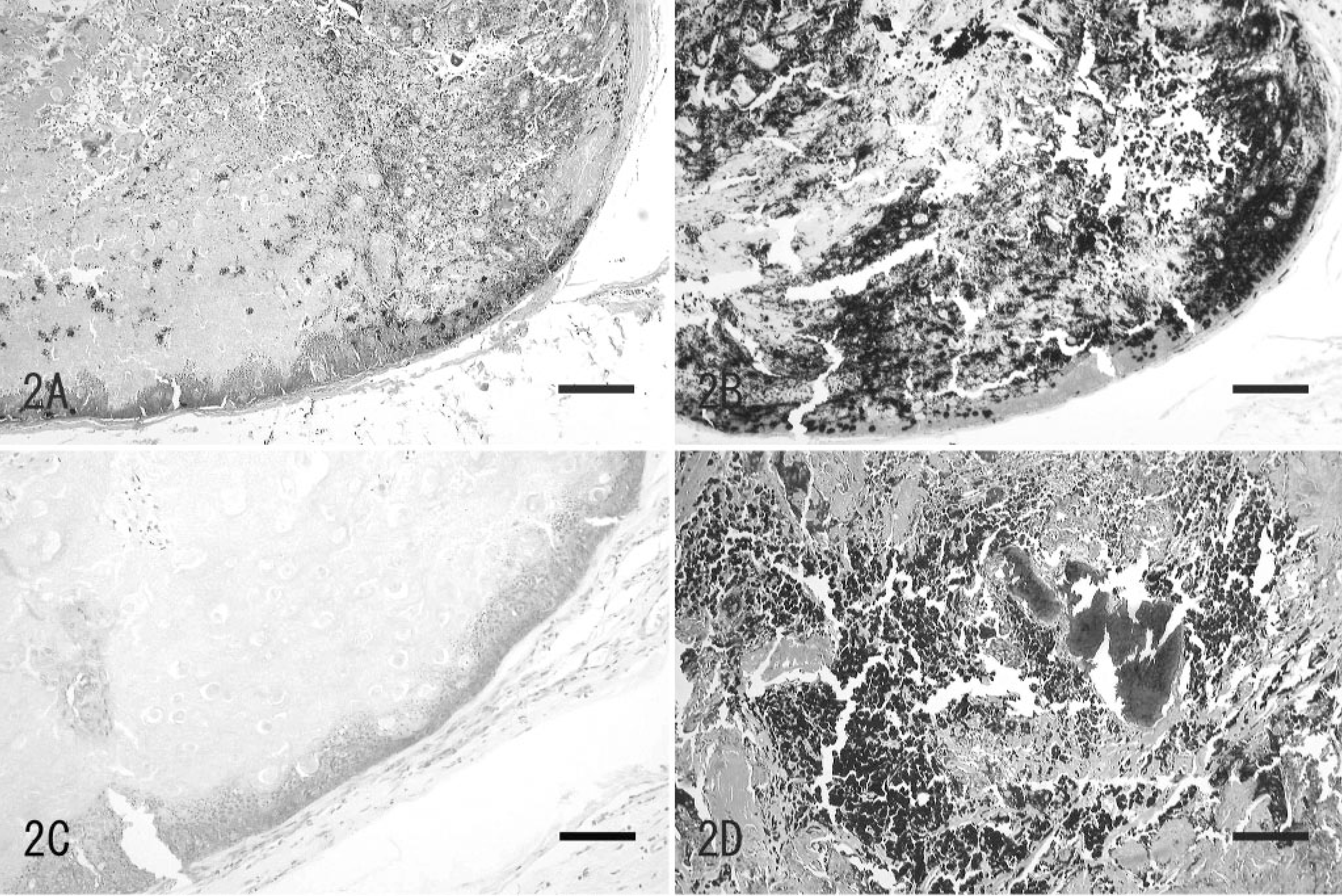
The tumor at the root of the tail; dog. The tumor consisted of a well-circumscribed nodule and had no connection to the epidermis. HE. Bar = 2.75 mm.
Immunohistochemically, shadow cells were positive for keratin/cytokeratin, whereas basaloid cells were negative (Fig. 3A). On the other hand, mononuclear or multinucleated cells containing melanin granules were positive for vimentin, S-100 protein, and HMB-45 (Fig. 3B–D).
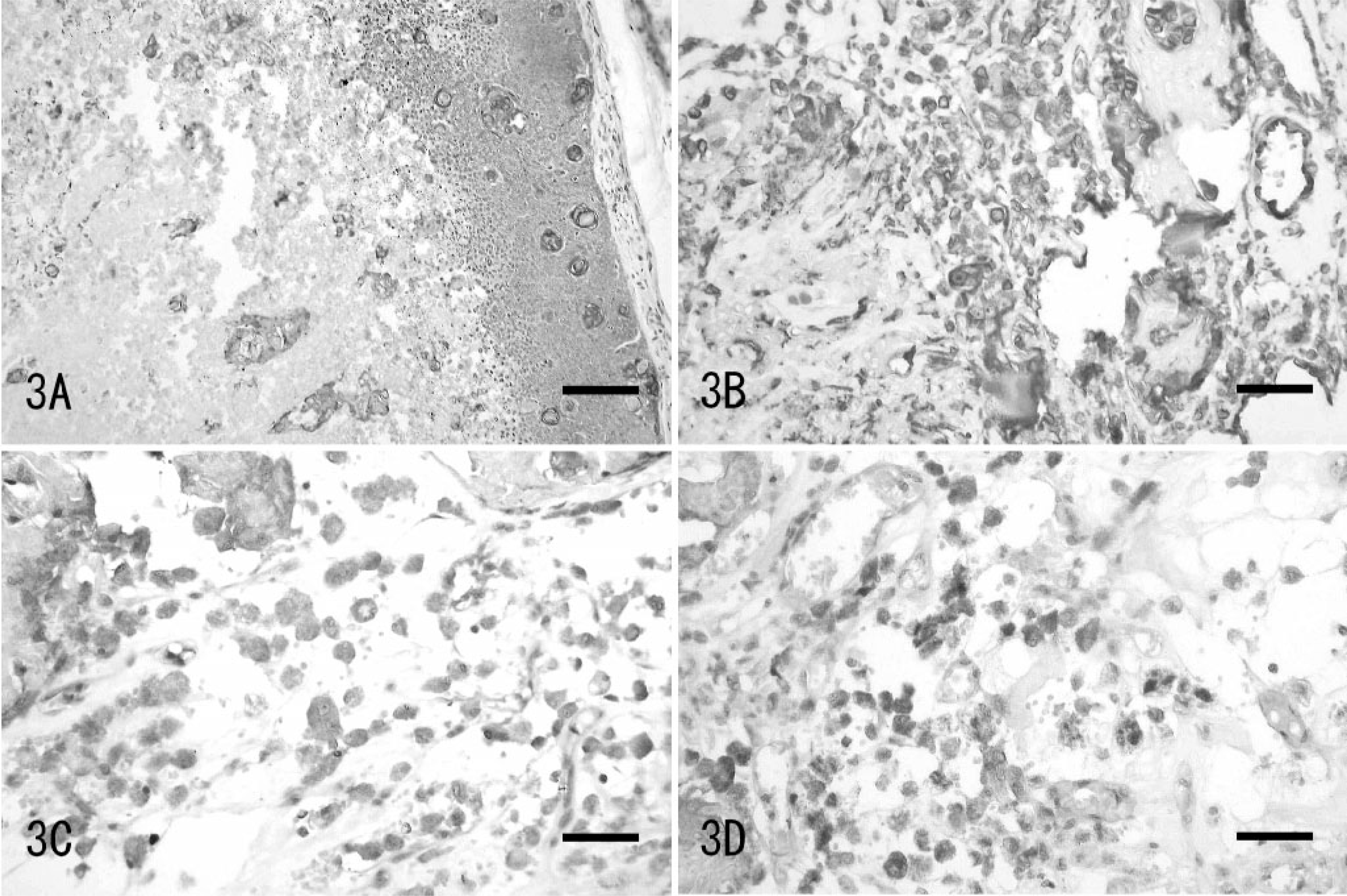
The tumor at the root of the tail; dog.
Melanocytic matricoma was originally diagnosed in humans by Carson et al. as a matrical neoplasm that recapitulates the bulb of the anagen hair follicle, composed of matrical cells and melanocytes. 1 After his report, Rissardi et al. and Williams et al. also reported similar lesions as melanocytic matricoma (Table 1). 2, 5 In the case described in this report, the mass was composed of two intermingled cell populations. One was an epithelial component consisting of shadow and basaloid cells. The other was a melanocytic component consisting of mononuclear or multinucleated cells containing various volumes of melanin. These histologic features were the same as that of the previously reported human cases. 1, 2, 5
Patient characteristics of cases of melanocytic matricoma.∗


∗ BCC = Basal cell carcinoma.
Immunohistochemically, epithelial component was positive for cytokeratin and melanocytic component was positive for vimentin, S-100 protein, and HMB-45. These immuno-histochemical results were also similar to human cases (Table 2). Originally, HMB-45 was the marker of human melanoma and embryonal melanocyte. In 2002, Sulamon et al. reported that HMB-45 can be used as the marker of canine melanoma. 4 Therefore, it was considered that the melanocytic component that increased in this case was neoplastic melanocytes.
Comparison of immunohistochemical results with those in humans.∗
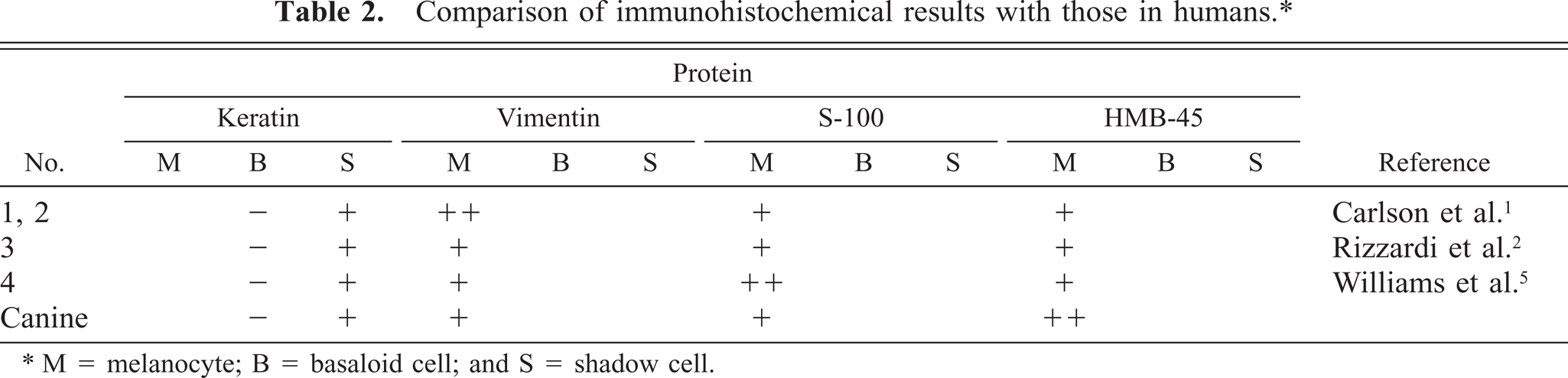
∗ M = melanocyte; B = basaloid cell; and S = shadow cell.
Histologic differential diagnosis includes pilomatricoma, matrical carcinoma, basal cell carcinoma, malignant melanoma, and proliferating pilomatricoma with matrical differentiation. 1, 5 Also in this case, malignant melanoma, pigmented pilomatricoma should be considered in the differential diagnosis. However, pigmented pilomatricoma does not contain prominent proliferation of melanocytes. 3 Instead, melanin exists in epithelial cells and nonneoplastic melanocytes. As mentioned above, this case showed a prominent proliferation of pigmented neoplastic melanocytes, so this case can be distinguished from pigmented pilomatricoma.
Granulomatous response, cyst formation, connection to the epidermis, and necrosis were not observed in reported cases of melanocytic matricoma in humans 1, 2, 5 or in the dog in this report. However, calcification and existence of multinucleated melanocytes were observed in the dog described in this study (Table 3). Therefore, it should be considered that these findings might be specific responses in canine melanocytic matricoma.
Features of melanocytic matricoma in human cases and this case.∗
Williams et al. reported that melanocytic matricoma developed in patients who had a history of basal cell carcinoma or elderly patients who had UV damage to skin (or in both). 5 However, in the case described in this study, there was no history of basal cell carcinoma or any other skin lesions. In the development of hair follicles, many factors including melanocytes and keratinocytes are related. From the histologic perspective, it is possible that melanocytic matricoma may arise as a result of damage to the developing hair follicle and that UV damage may be a stimulus that incites it. If melanocytic matricoma develops by this process, it can occur independent of age. However, the process by which this lesion develops is still not known and comparison with additional cases will be necessary.
In conclusion, this case can be diagnosed as canine melanocytic matricoma. This conclusion is based on histologic and immunohistochemical features that are similar to those described in melanocytic matricoma in humans. This may be the first case of melanocytic matricoma reported in an animal.
