Abstract
We examined the expression of CD20 in normal canine peripheral blood mononuclear cells, normal canine spleen, and canine non-Hodgkin lymphoma (NHL) to determine the feasibility of using this antigen as a diagnostic aid and as a possible target for therapy. An antibody generated against a C-terminal (intracytoplasmic) epitope of human CD20 recognized proteins of 32-36 kd in normal and malignant canine lymphocytes. This antibody showed restricted membrane binding in a subset of lymphocytes in peripheral blood, in the B-cell regions from a normal canine spleen and lymph node, and in malignant cells from 19 dogs with B-cell NHL, but not from 15 dogs with T-cell NHL. The patterns of CD20 reactivity in these samples overlapped those seen using an antibody that recognizes canine CD79a. This anti-CD20 antibody is therefore suitable as an aid to phenotype canine NHL. In contrast, normal canine B cells were not recognized by any of 28 antibodies directed against the extracellular domains of human CD20 (including the chimeric mouse-human antibody Rituximab) or by any of 12 antibodies directed against the extracellular domains of mouse CD20. Thus, the use of CD20 as a therapeutic target will require the generation of specific antibodies against the extracellular domains of canine CD20.
CD20 is a tetraspanning transmembrane phospho-protein that is expressed predominantly in pre-B cells and in mature peripheral B cells in humans and mice, 25, 26, 28, 29, 32 although it is also expressed at very low levels in a heterogeneous subset of human T cells (predominantly those bearing γδ T-cell receptors). 11 In humans, CD20 is also strongly and homogeneously expressed on most mature B-cell malignancies, except for chronic lymphocytic leukemia cells, where expression is more variable; 32 CD20 expression also has been described in rare cases of peripheral T-cell lymphoma and in the malignant epithelial cells of thymomas. 5, 16 The function of CD20 remains incompletely understood, although it appears to be important for receptor-induced calcium signals. Engagement of CD20 using antibodies leads to increased intracellular calcium in human B cells, 2, 22 and CD20-deficient mice show compromised calcium mobilization upon engagement of CD19. 29
CD20 is among the first molecules that have been successful as immunotherapeutic targets. 32 This molecule was an attractive target because its expression can be determined in both fresh and archival tissues and because it is stable in cell membranes (i.e., it is not readily internalized or shed upon antibody binding). The humanized monoclonal antibody Rituximab (Rtx), which consists of the variable (antigen binding) region of the murine monoclonal antibody C2B8 linked to a human constant region, is now part of the standard-of-care for low-grade and indolent non-Hodgkin lymphoma (NHL) in humans. 18, 23, 32 Recent data show that, in combination with multiagent chemotherapy protocols, Rtx also improves the outcomes of patients with diffuse large B-cell lymphoma (DLBCL), and it may be useful as an adjuvant to treat a variety of B-cell–dependent immune-mediated diseases. 12, 18, 32 Since DLBCL and B-cell–dependent autoimmune conditions are commonly diagnosed in dogs, these latter results warranted investigation of CD20 expression in canine B cells.
For this study, we used immunoblotting, immuno-histochemistry, and flow cytometry to validate the cross-reactivity of anti-human or anti-mouse CD20 antibodies against canine B cells. Our results show that the antibody that recognizes the intracellular domains binds a canine CD20 homolog and is suitable for use as part of a panel to identify normal and malignant canine B cells in routine diagnostic samples. However, since none of the antibodies directed against extracellular domains bind canine CD20, specific antibodies directed against canine CD20 extracellular domains will be required before the benefit of therapies targeting this molecule can be explored in dogs.
Materials and Methods
Chemicals and reagents
Tissue culture media was purchased from Gibco BRL (Grand Island, NY), fetal bovine serum was obtained from Hyclone (Logan, UT), and antibiotics for tissue culture (Primocin) were from Invivogen (San Diego, CA). Antibodies for flow cytometry directed against canine CD3, CD4, CD5, CD8, CD14, CD45, and a pan–B-cell marker (which is probably CD21) were obtained from Serotec (Raleigh, NC). Antibodies for immunohistochemistry directed against canine CD79a and canine CD3 were obtained from Dako (Carpinteria, CA) and Sigma Chemical Company (St Louis, MO), respectively. Anti-human CD3 (Orthoclone OKT3, Ortho Biotech, Raritan, NJ) and Rtx (Genentech, South San Francisco, CA) were purchased at the University of Colorado Hospital. The HB20 and MB20 series of antibodies that bind extracellular epitopes of human (n = 27) and mouse (n = 12) CD20, respectively, were a kind gift from Dr Thomas Tedder (Duke University, Durham, NC). Rabbit anti-human CD20 was purchased from Lab Vision Corp (Fremont, CA). This antibody is referred to as LV-anti-CD20 throughout the manuscript for clarity.
Tissues and cells
Blood from healthy pet dogs and tumors from dogs with NHL were obtained through a protocol reviewed and approved by the Institutional Animal Care and Use Committee and the Institutional Review Board of AMC Cancer Center; all samples were obtained with owner consent. The signalment for the dogs with NHL is shown in Table 1; the breed bias reflects one of the principal goals of the study for which dogs were recruited, which was to identify heritable factors that contribute to lymphomagenesis. Two samples came from one dog (dog No. 2, the first from a peripheral lymph node at the time of initial diagnosis and the second from a mesenteric lymph node and from the small intestine at the time of euthanasia approximately 24 months later). Control tissues from normal dogs were obtained as described.
13
Blood from healthy human volunteers was obtained through a protocol reviewed and approved by the Colorado Multiple Institutional Review Board. C57Bl/6 mice were maintained under a protocol approved by the Institutional Animal Care and Use Committee of the University of Colorado Health Sciences Center. Mice were not subjected to experimental manipulations in vivo. They were humanely sacrificed using CO2 asphyxiation and cervical dislocation, per the Guidelines for Euthanasia of the American Veterinary Medical Association. Spleens were removed aseptically, and single-cell suspensions were obtained using gentle mechanic disruption. Human and canine peripheral blood mononuclear cells (PBMC) and mouse splenic lymphocytes were purified over a discontinuous gradient of Ficoll-Hypaque (Histopaque 1.077, Sigma) as described.
1,
7
Sterile biopsy samples from dogs having a clinical diagnosis of NHL were submitted from veterinary clinics throughout the USA and shipped at 4°C in sterile transport media provided by the investigators (RPMI 1640 supplemented with 10% fetal bovine serum and Primocin at a concentration of 100 μg/ml) using a priority overnight courier. Representative sections from the samples were fixed in 10% neutral-buffered formalin and processed for histology using routine methods. The remaining biopsy sample was mechanically disrupted and passed through 80-μm mesh filters to obtain a single-cell suspension. The viability of these samples, as determined by trypan blue dye exclusion, was between 60 and 99%. The single-cell suspensions were resuspended in a solution of 90% fetal bovine serum and 10% dimethyl sulfoxide, separated into aliquots of 1 × 107 to 1 × 109, and cryopreserved in liquid nitrogen. Human Raji B-lymphoma cells and Jurkat T-cell lymphoma cells were obtained from the American Type Culture Collection (Manassas, VA) and were maintained in culture at 37°C in a humidified atmosphere of 5% CO2 in RPMI 1640 supplemented with 10% fetal bovine serum, 2 mM
Signalment of dogs with NHL.
Immunophenotyping and classification of canine NHL samples
Immunohistochemistry stains for CD79a and CD3 were performed as previously described, 15 and clonality was confirmed using a polymerase chain reaction–based antigen receptor rearrangement assay (PARR, Immunopathology Laboratory, Colorado State Diagnostic Laboratory, Fort Collins, CO). 3 Cells from samples that did not show amplification of a clonal rearrangement probably had rearrangements that used V/J regions distinct from those used for primer design. 3 Staining for CD20 was accomplished using the LV-anti-CD20 antibody at a concentration of 2 μg/ml and incubated at room temperature for 60 minutes. Paraffin-embedded sections were pretreated for antigen retrieval by heating in a microwave for 6 minutes in 0.1 M sodium citrate, pH 6.0, and allowed to cool for 20 minutes. No pretreatment was required for fresh, unfixed samples. Histologic diagnoses were made according to World Health Organization (WHO) criteria on the basis of morphology and immunophenotype. 31 Photomicrographs were obtained using an Olympus BX50 microscope with an Olympus Microfire S79809 cooled digital camera (Scientific Instrument Company, Aurora, CO) or an Olympus BX40 microscope with a Retiga digital camera model 1300. Transmitted light images under phase-contrast microscopy were captured using a gain of 1.0 and exposures ranging from 14 to 26 ms.
Flow cytometry
PBMC or lymphoma cells were divided into aliquots of 100 μl containing 5 × 105 cells/well in 96-well U-bottom plates. Nonspecific binding sites were blocked using an irrelevant antibody (mouse immunoglobulin G [IgG]2a, 10 μg/well, BD Biosciences, Franklin Lakes, NJ) diluted in staining buffer (phosphate-buffered saline, 0.1% fetal bovine serum, 0.1% NaN3) and incubated at 4°C for 10 minutes. Primary antibodies were used at 5 μl/well for directly conjugated antibodies, at 2 μl/well for negative controls, and at 25 μl of undiluted culture supernatant for unconjugated HB-20 and MB-20 antibodies. The primary antibody was added to the cells to obtain a total volume of 25 μl in staining buffer. Rtx was premixed with Zenon Human IgG Alexa Fluor 488 Labeling kit (Molecular Probes, Eugene, OR) according to the manufacturer's directions. The samples were incubated at 4°C for 30 minutes and washed once with staining buffer. Goat anti-mouse antibody conjugated to fluores-cein-isothiocyanate was used at a 1 : 250 dilution to detect binding of unconjugated antibodies by a secondary incubation step lasting 20 minutes at 4°C, followed by washing. Stained cells were resuspended in staining buffer and fixed in 2.5% buffered formalin. Samples were kept at 4°C protected from light until analysis using a Beckman Coulter Epics XL flow cytometer (Beckman Coulter, Hialeah, FL) running the Expo 32 software package or a FACSCalibur flow cytometer (Becton Dickinson, Mountain View, CA) running the CellQuest software package.
Immunoblotting
Raji cells, PBMC, and lymphoma cells were lysed in a buffer consisting of 150 mM NaCl, 10 mM Tris, pH 7.4, 0.1% sodium dodecyl sulfate (SDS), 1.0% Triton X-100, 1.0% sodium deoxycholate, 5 mM EDTA, 1 mM dithiothreitol, and a Proteinase Inhibitor Cocktail containing 1 mM phenylmethylsulfonyl fluoride, pepstatin at a concentration of 2μg/ml, leupeptin at a concentration of 2μg/ml, 1 mM aminocaproic acid, and aprotinin at a concentration of 10 μg/ml. Insoluble material was removed by centrifugation, and protein concentrations of the cell lysates were determined using the BioRad Protein Assay kit (BioRad, Hercules, CA). Cellular proteins (50 μg) were separated by SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membranes (Hybond, Amersham, Arlington Heights, IL). Rabbit anti-human CD20 was diluted 100-fold and incubated overnight at 4°C, followed by secondary anti-rabbit antibody conjugated to horseradish per-oxidase (Amersham). Detection was performed using the enhanced chemiluminescence system (Amersham) according to the manufacturer's instructions.
Results
CD20 expressed in normal canine B cells
The amino acid sequence in the transmembrane and long N- and C-terminal cytoplasmic domains of CD20 is evolutionarily conserved, 19, 25, 28, 32 suggesting that antibodies directed against epitopes in these regions are likely to show wide cross-species reactivity. To test this notion, we obtained a polyclonal rabbit antibody raised using a synthetic peptide comprising the C-terminal region of human CD20 (LV-anti-CD20) and tested its reactivity against normal canine peripheral B cells using immunocytochemistry and immunohisto-chemistry. The antibody bound approximately 3–15% of peripheral blood leukocytes and showed homogeneous staining of the corpuscular regions of normal canine spleen, including cells in the marginal and mantle zones (data not shown). Moreover, staining with this antibody in normal canine lymph nodes generally overlapped that seen with anti-CD79a (Fig. 1). LV-anti-CD20 stained cells in the B-cell areas (follicles, germinal centers, and subcapsular zone) but not CD3+ cells in the paracortical zone. At higher magnifications, it can be appreciated that CD20 expression is largely confined to the cell membrane (Fig. 1). To validate the target of this antibody in dogs as canine CD20, we examined the electrophoretic mobility of its protein targets by immunoblotting. The antibody recognized proteins of ∼34–36 kd in human Raji B cells and proteins of ∼32–36 kd in canine PBMC or canine B-cell NHL but not in canine T-cell NHL (Fig. 2).
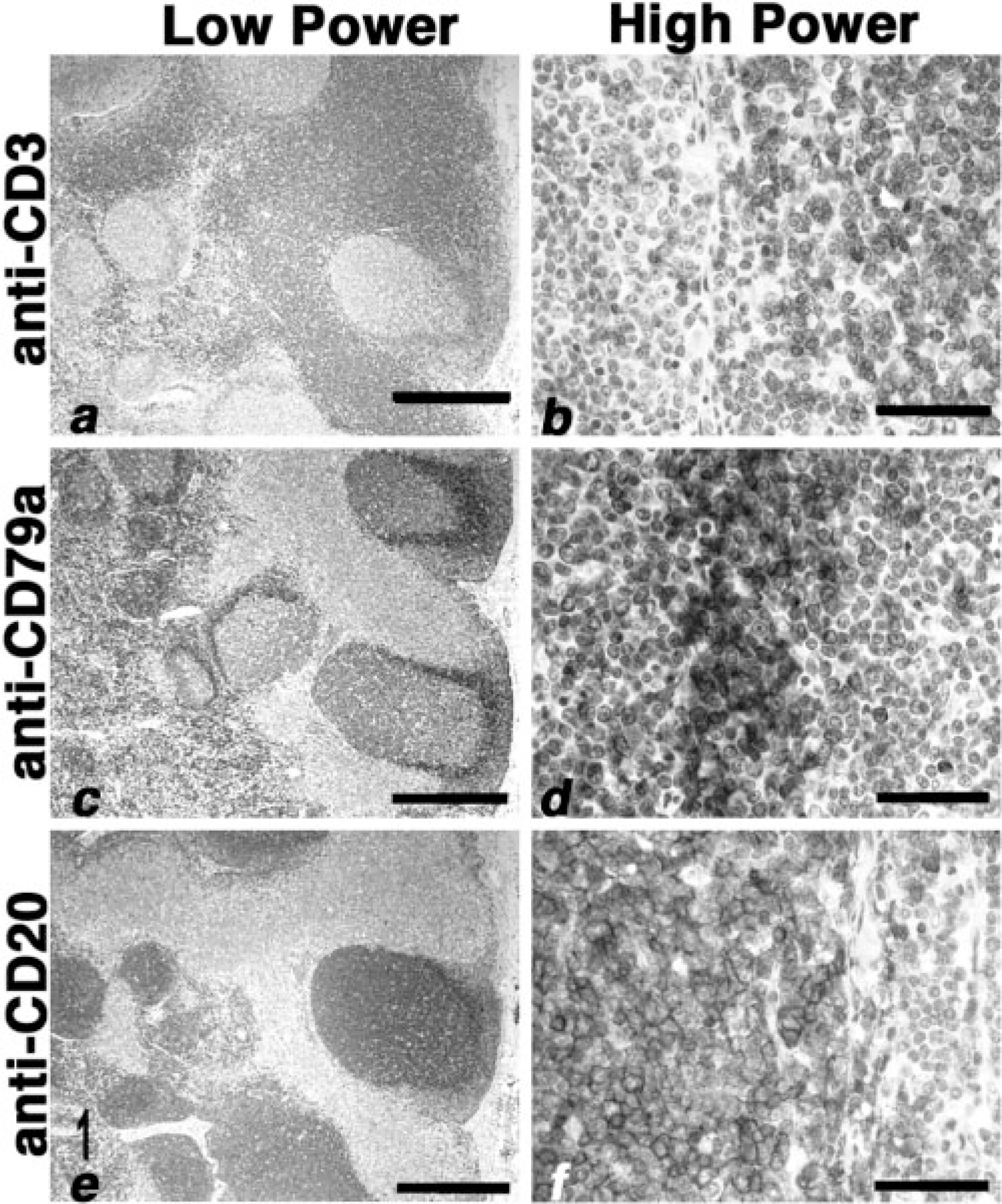
Lymph node; normal dog. Low-power (panels
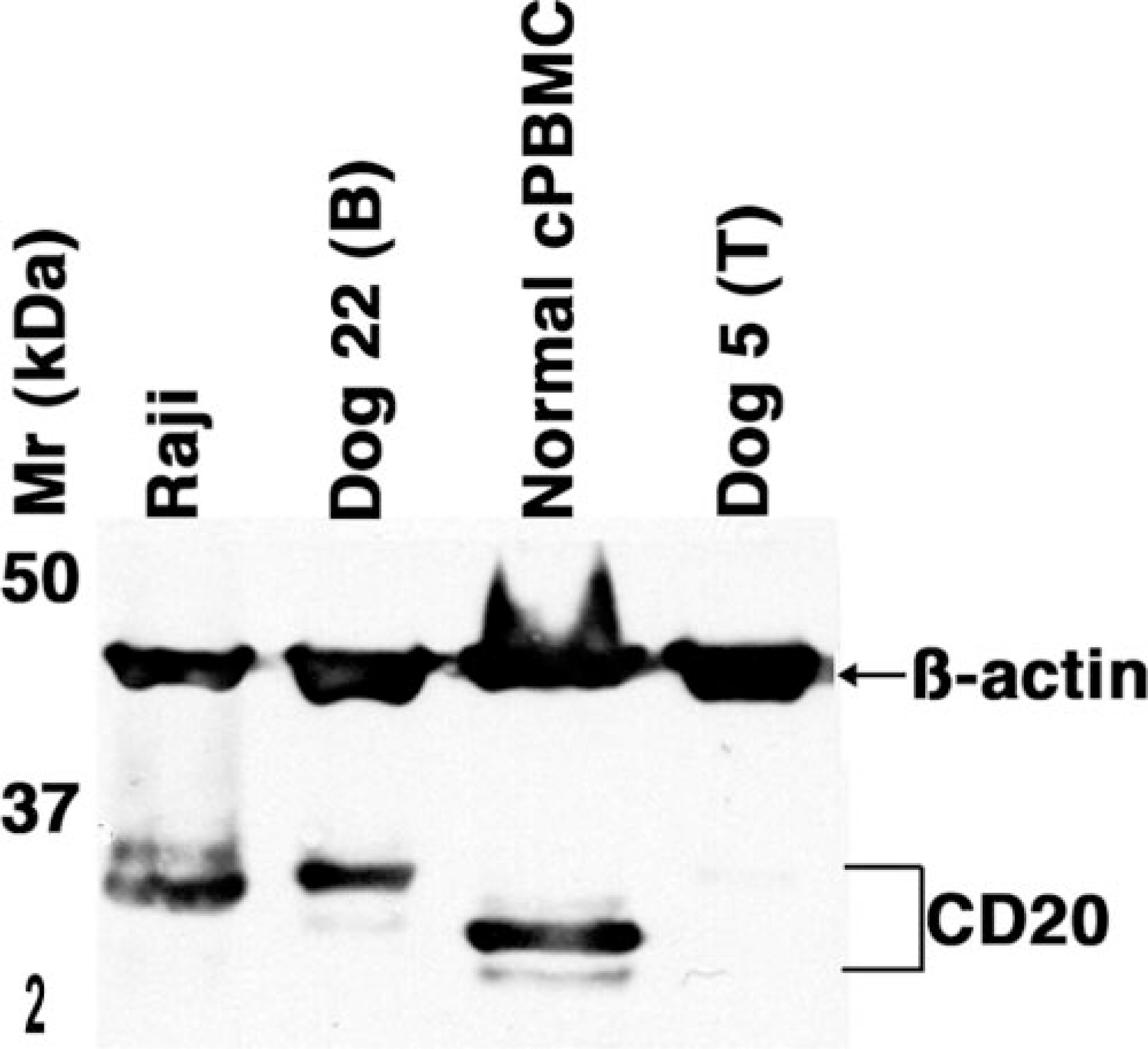
Lymphocyte immunoblots; normal dog, Raji human lymphoma, dog No. 5 and dog No. 22. Cell lysates prepared from canine PBMC, human Raji B lymphoma cells, and canine lymphoma samples (dog Nos. 5 and 22) were separated on one-dimensional SDS-PAGE and subjected to immunoblotting using the LV-anti-CD20 antibody and an anti-actin antibody to verify sample integrity. Immunoblots were developed using enhanced chemiluminescence. The bands with different electrophoretic mobility most likely represent differentially phosphorylated forms of CD20. B = B Cell Lymphoma; T = T Cell Lymphoma.
We next examined the capacity of antibodies directed against the extracellular domains of human or mouse CD20 to bind canine B cells. B cells made up 9.68 ± 7.3% of canine PBMC (range, 3.2–26%; n = 5), 2–8.9% of human PBMC (n = 2), and 17–40% of mouse spleen cells (n = 3). Rtx bound virtually 100% of human peripheral blood B cells or Raji B cells Fig 3). Predictably, Rtx did not bind human Jurkat T cells, but it also did not bind normal canine B cells (Fig. 3). Among the other anti-human CD20 antibodies tested by flow cytometry, HB20-1 through HB20-6, HB20-10 through HB20-23, and HB20-27 through HB20-30 also bound human Raji B cells (see, for example, Fig. 3). The reactivity of these antibodies showed unimodal distributions, although each had different fluorescence intensities that might reflect diverse affinity for CD20. Antibodies HB20-9, HB20-25, and HB20-26 did not bind to Raji B cells above the controls. None of these HB20 series anti-human CD20 antibodies bound canine peripheral blood B cells (Fig. 3). Similarly, anti-mouse CD20 antibodies MB20-1, MB20-2, MB20-3, MB20-6, MB20-7, MB20-8, MB20-10, MB20-11, MB20-13, MB20-14, MB20-16, and MB20-18 each bound mouse splenic B cells (see, for example, Fig. 3), albeit with different fluorescence intensities. As was true for the HB-20 series of anti-human CD20 antibodies, none of the MB-20 series anti-mouse CD20 antibodies bound canine peripheral blood B cells (Fig. 3).
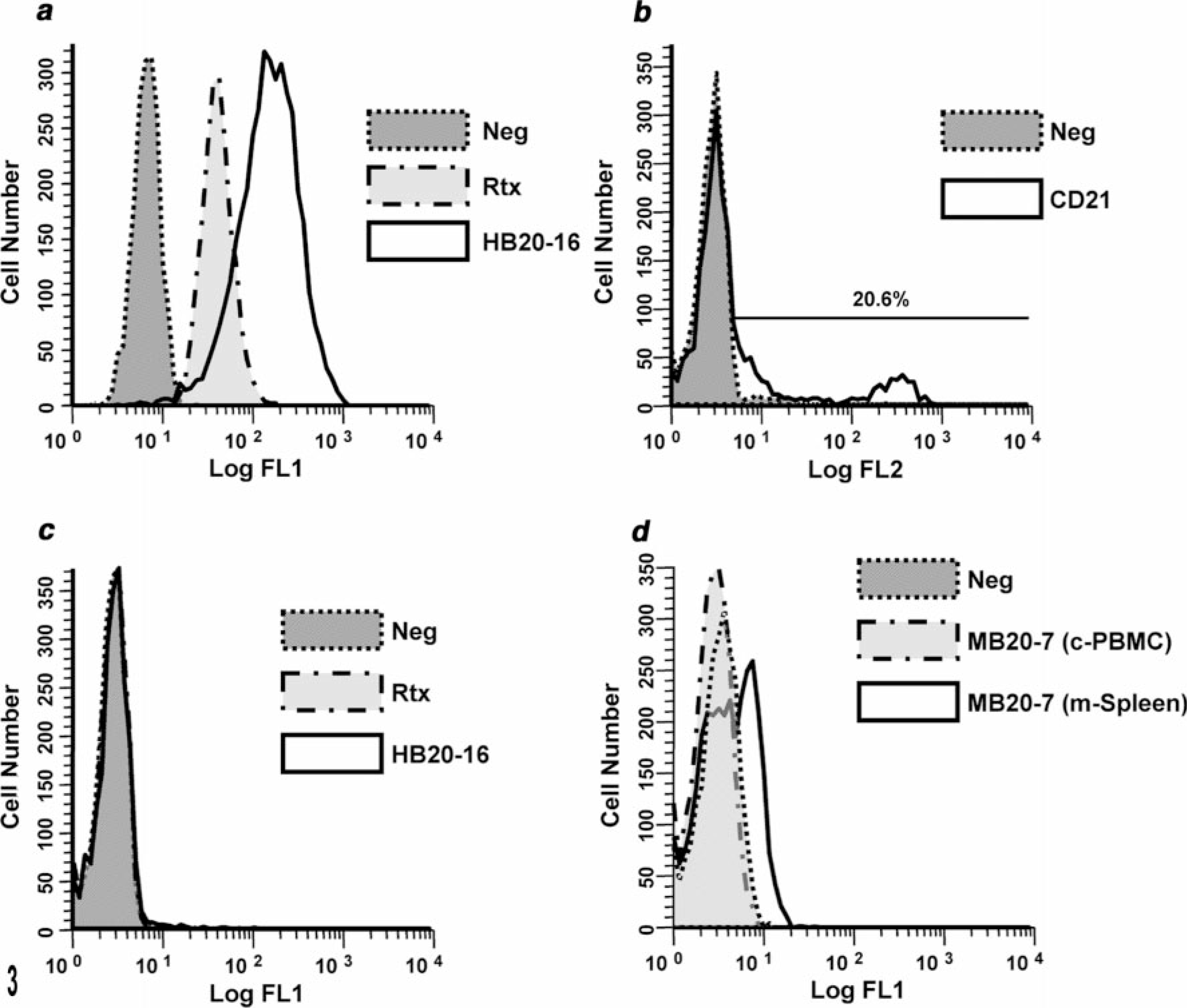
Lymphocyte flow cytometry; humans, dogs, mice. Overlay histograms show human, canine, and murine cells stained with the indicated antibodies and analyzed by flow cytometry. Panel
CD20 expressed in canine B-cell lymphomas
We used the LV-anti-CD20 antibody to examine the expression of CD20 in 34 dogs with canine NHL using immunohistochemistry. The WHO classification of the tumors and immunostaining results are shown in Table 2. Malignant cells from each B-cell tumor examined (n = 19) showed consistent membrane binding in patterns that overlapped staining with CD79a (for example, see Fig. 4). In contrast, the LV-anti-CD20 antibody stained only sporadic cells, consistent with residual normal B cells in every T-cell tumor tested (n = 15; for example, see Fig. 4). Although the staining patterns obtained with the LV-anti-CD20 antibody differed in predictable fashion on the basis of the morphologic appearance of the tumors, no differences were apparent in the staining intensity among the malignant B cells in different tumor types. Finally, we used flow cytometry to verify the lack of Rtx reactivity with malignant canine B cells. Cells from a canine B-cell NHL (dog No. 27) stained using the anti-canine B-cell antibody and Rtx were analyzed using two-color immunofluorescence. Our results showed that Rtx did not bind the canine B lymphoma cells that stained positive using the anti-canine B-cell antibody (data not shown).
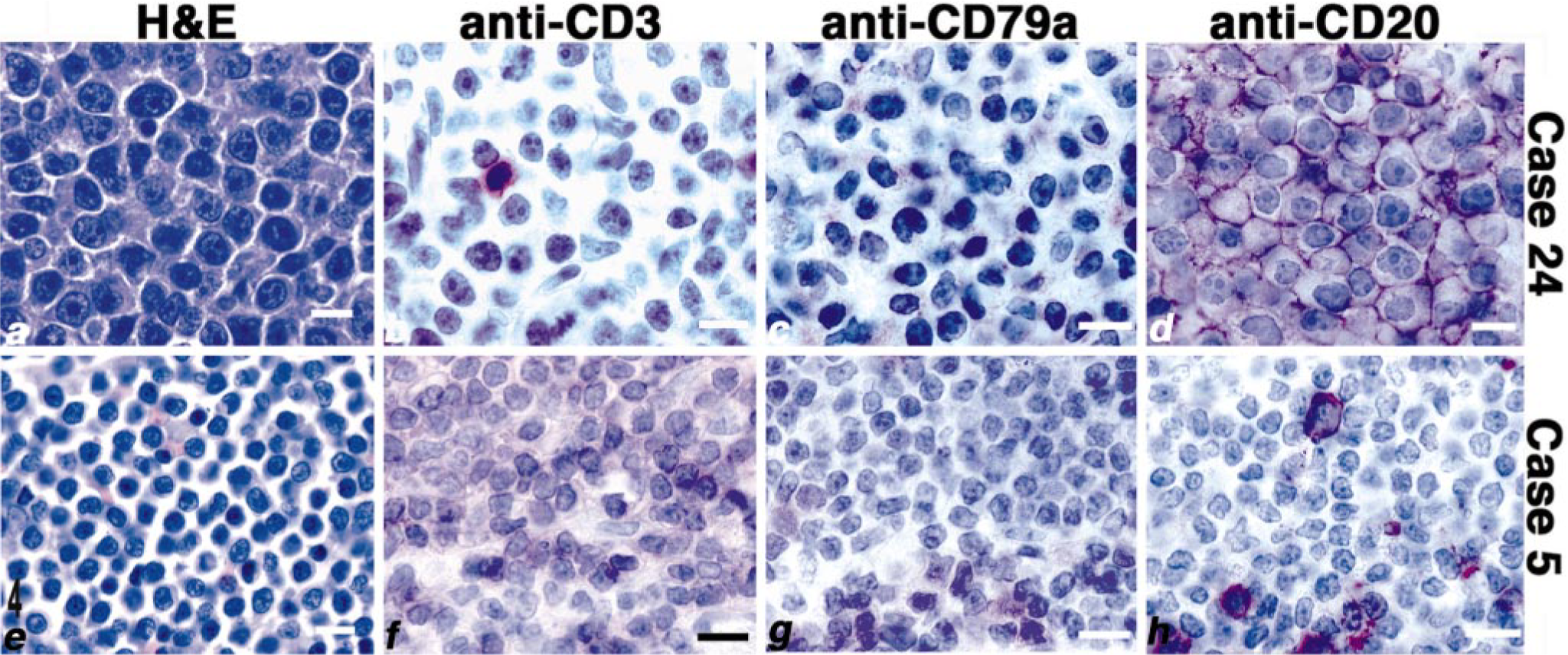
Lymphoma; dog Nos. 5 and 24. High-power photomicrographs from the lymph node samples of dog No. 24 (diffuse large B-cell lymphoma, panels
WHO classification and immunophenotyping results for canine NHL.
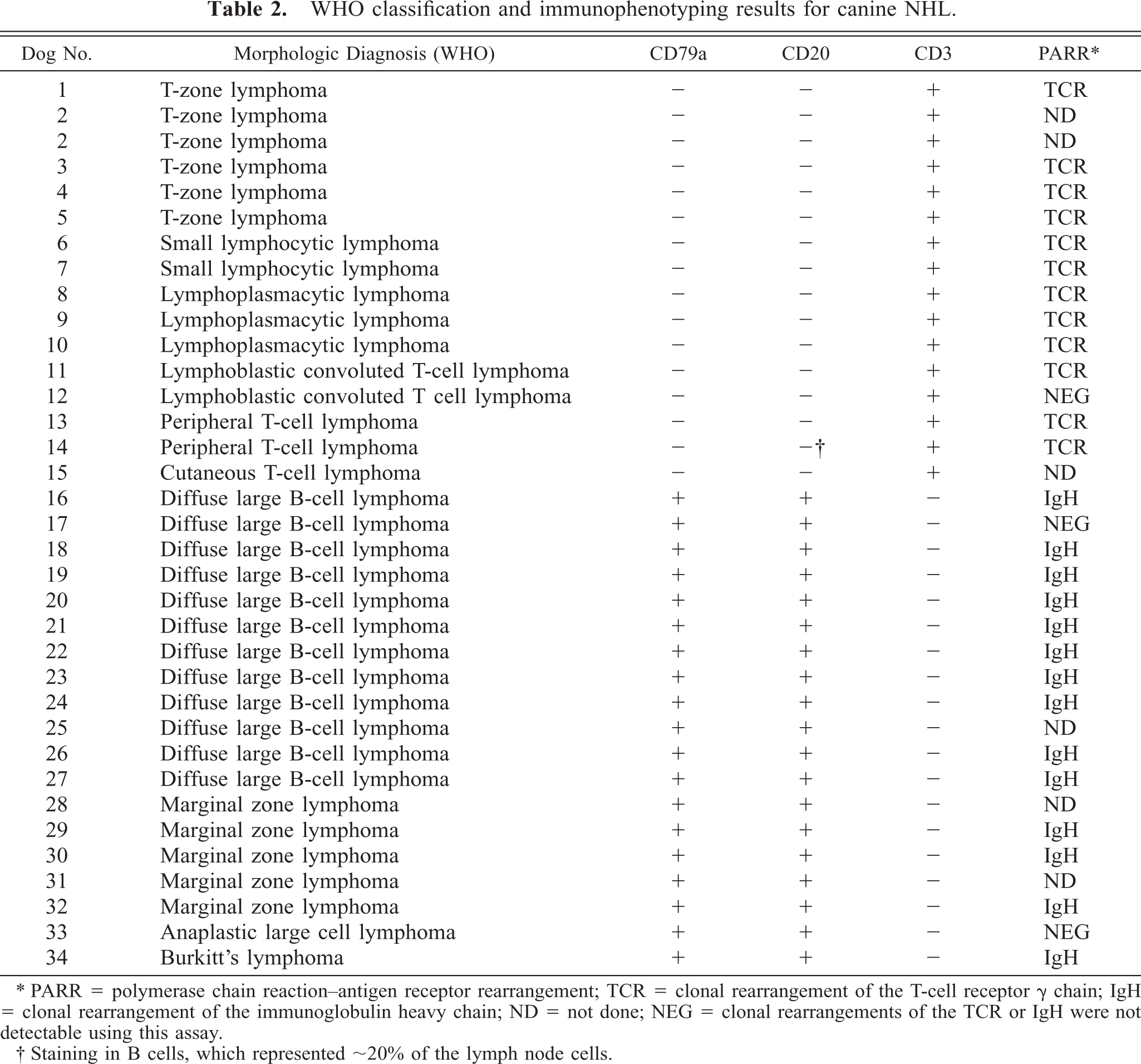
∗ PARR = polymerase chain reaction–antigen receptor rearrangement; TCR = clonal rearrangement of the T-cell receptor g chain; IgH = clonal rearrangement of the immunoglobulin heavy chain; ND = not done; NEG = clonal rearrangements of the TCR or IgH were not detectable using this assay.
† Staining in B cells, which represented ∼20% of the lymph node cells.
Discussion
CD20, a transmembrane protein whose expression is generally restricted to B cells, has been shown to be useful in the diagnosis and treatment of human NHL and other B-cell diseases. 12, 32 To assess the utility of this antigen as a similar aid for the diagnosis and treatment of canine B-cell diseases, we sought to establish the expression patterns of CD20 in normal and malignant canine B cells. Our results show that LV-anti-CD20, a rabbit polyclonal antibody raised against intracellular epitopes of human CD20, bound canine B cells and recognized proteins of ∼32–36 kd. The cytoplasmic domains of human CD20 contain multiple consensus sequences for serine and threonine phosphorylation, and differential phosphorylation of a single CD20 gene product accounts for different electrophoretic mobility species. 27 The slower migrating hyperphosphorylated species (those with higher apparent molecular weight) accumulate upon activation and are the predominant forms seen in malignant B-cell lines. 10, 27, 30 Therefore, it is not surprising that the predominant forms of CD20 in malignant Raji B cells and in malignant canine B-cell NHL were the 34- to 36-kd species, but the presumptive hypophosphorylated 32- to 33-kd species were most abundant in normal unstimulated canine peripheral B cells. Although we cannot completely rule out the possibility that differences in electrophoretic mobility between CD20 in normal and malignant canine B cells are due to differential glycosylation, previous studies using endoglycosidase digestion and metabolic labeling in the presence of tunicamycin (a glycosylation inhibitor) indicated that none of the 33-, 34-, or 36-kd forms of human CD20 is glycosylated. 26
A recent study by Coyle and Steinberg 6 showed that an antibody from Biogenex also might be suitable to identify canine B-cell lymphomas. In that study, 3 of 44 intestinal lymphomas stained positive with the Bio-genex antibody, but not with anti-CD3, and two additional tumors stained positive with both antibodies. In contrast, 33 of 44 tumors stained positive using anti-CD3, but not with the Biogenex antibody, supporting other observations that indicate canine extranodal lymphoma is predominantly of T-cell origin. 8, 9, 20, 31 The specific Biogenex antibody used or its reactivity with a protein that had the predicted electrophoretic mobility of canine CD20 was not reported in this study. Nevertheless, our results indicate that an antibody directed against conserved intracytoplasmic domains of CD20 can identify canine B cells in dogs representing major breeds 17 and age groups, is thus suitable for immunophenotyping NHL, and is complementary to anti-CD79a antibodies that have been thus far included in antibody panels for this purpose. Preliminary results from one of our laboratories (VEOV) suggest that the LV-anti-CD20 antibody also recognizes B cells in histologic sections from other domestic animal species.
An appealing property of many antibodies that bind the extracellular domains of CD20 is their capacity to deplete malignant and autoreactive B cells. 14, 21, 22 Two antibodies (Rtx and Ibritumomab tiuxetan) are approved by the US Food and Drug Administration for treatment of human NHL. In particular, because Rtx is “humanized” (that is, the murine constant region has been replaced by a human constant region), it is more effective at engaging antibody-dependent cellular cytotoxicity and less subject to inducing the production of anti-mouse neutralizing antibodies. 4, 18, 32 Rtx is now part of the standard-of-care for the treatment of low-grade or indolent NHL. 18, 23, 24, 32 Recent reports indicate that Rtx is also a useful adjuvant to treat extranodal marginal zone lymphoma, nodal DLBCL, posttrans-plant lymphoproliferative disorders, and some auto-immune conditions such as rheumatoid arthritis, autoimmune hemolytic anemia and other immune-mediated cytopenias, and systemic lupus erythematosus in human patients. 12, 32 Together, marginal zone lymphoma and DLBCL may account for as many as 60% of canine NHL, and the aforementioned autoimmune conditions are becoming more commonly diagnosed in dogs, significantly increasing the attractiveness and rationale to investigate applications of anti-CD20 therapy for these patients. For this purpose, we examined the binding of 40 antibodies directed against the extracellular domains of human (n = 28) or mouse (n = 12) CD20. None of these antibodies, including Rtx, bound canine B cells. These results were somewhat predictable, given that these antibodies show no cross-reactivity between humans and mice, although the lack of cross-reactivity between human and mouse antigens may sometimes be a result of the means of antibody generation and selection. The data are also consistent with those obtained when we have evaluated other antibodies that recognize extracellular domains of lymphocyte receptors 7 and suggest that the extracellular epitopes of CD20 are subject to selective pressures that are different from those in the intracellular domains involved in signaling. Since the physiologic ligand(s) of CD20 is not known, it is difficult to assess the possible conservation of ligand binding domains. Our data indicate that an evaluation of any of the antibodies we tested for therapeutic purposes in dogs would be unrewarding and underscore the need to generate antibodies that specifically bind the extracellular domains of canine CD20 for this purpose.
Footnotes
Acknowledgements
The authors wish to thank owners and veterinarians who contributed cases, and especially Drs. Michelle Ritt and Frank Coons for diligent follow-up information, Dr. Shairaz Baksh for assistance with construct design, Drs. Baksh, Robert Strange, Robert Sclafani, Michelle Ritt, and Robert Weiss for review of the manuscript and helpful discussions. This work was supported in part by grants 1626, 2025, and 2254 from the AKC Canine Health Foundation (to JFM, SCH, MB), 1R55CA86432 from the NCI (to JFM), and a grant from the Monfort Family Foundation (to the University of Colorado Cancer Center).
