Abstract
Since the First International Canine Leukocyte Antigen Workshop in 1994, individual laboratories have worked to expand the repertoire of monoclonal antibodies (mAbs) used in clinical testing and research in dogs. Here, we employed flow cytometry to document the specificity of mAbs submitted to the canine workshop and the animal homologue section of the human HLDA8 international workshop. We also provide an initial characterization of new mAbs derived from hybridomas developed from mice immunized with leukocytes from the blood of a healthy dog and from mice immunized with cells from a dog with B-cell leukemia. These mAbs enhance the tools available for characterizing leukemias, lymphomas, and other hematologic disorders in dogs, as well as for researching the canine immune response to pathogens. Importantly, some of the mAbs submitted to the canine and HLDA8 workshop recognize highly conserved epitopes expressed on orthologues of cluster of differentiation molecules first identified in humans and present an opportunity to develop a cross-species panel for clinical testing and research.
Keywords
Following the demonstration that B cells producing monoclonal antibodies (mAbs) of known specificity could be immortalized by fusion with a myeloma cell adapted for culture, 9 an extensive effort was initiated to use this technology to develop mAbs specific for leukocyte differentiation molecules (LDMs) for use in humans. The first international workshop was convened in 1982 to summarize the characteristics of mAbs developed by individual laboratories. 2 An alphanumeric nomenclature was established to provide a common method for naming LDMs in humans, whereby each molecule identified by ≥2 mAbs from different laboratories was given a numeric cluster of differentiation (CD) designation.
Efforts to develop mAbs for use in veterinary species were initiated at about the same time, mainly focusing on livestock. The first international workshop for veterinary species was convened in 1989. 8 The collective decision was made to use the same numeric CD nomenclature to name mAbs that recognized orthologues of molecules first identified in humans. Two additional successful workshops were convened for studies of mAbs for use in ruminants, 12 3 for pigs, 7 and 2 for horses. 10 One workshop was convened to summarize efforts to develop mAbs for use in dogs. 3
The last major effort to identify mAbs for use in veterinary species was the animal homologues section of the human HLDA8 workshop in 2004. 13 This section focused on screening mAbs developed against human CD molecules for those that recognized epitopes conserved on orthologous CD molecules in one or more veterinary species. We submitted a set of mAbs we developed that recognized epitopes conserved on human CD molecules and CD orthologues in veterinary species, including dogs.13,14 Subsequent efforts to develop reagents for use in different species have been undertaken by individual laboratories.
As reported here, we have continued our efforts to develop mAb reagents for use in veterinary species. We adopted 2 approaches to identify mAbs for use in clinical flow cytometry and research: 1) the identification and documentation of mAbs that recognize epitopes conserved on orthologous CD molecules in humans, dogs, and other species, and 2) the direct development of mAbs specifically for use in dogs.
Materials and methods
Animals
Multiple donor dogs were used as a source of blood over the past several years to support our ongoing research involving canine immunology. Formal consent was obtained from all owners who volunteered their dogs for blood donation. Blood samples collected for clinical testing through the Clinical Pathology Laboratory at the Washington State University (WSU) College of Veterinary Medicine (Pullman, WA, USA) were submitted to our laboratory for the diagnosis of leukemias, lymphomas, and other hematologic disorders. These blood samples were obtained opportunistically and used when available to screen sets of mAbs developed from mice immunized with leukocytes from various species. 5 One blood preparation served as a source of peripheral blood mononuclear cells for mouse immunization, leading to the development of a set of mAbs described herein, named CADO. Additionally, a B-cell leukemia sample submitted to the clinic for diagnosis was used as a source of cells for the development of mAbs targeting molecules expressed on B-cell leukemias, named DLL. Finally, a dog from a private owner provided a blood sample for the final studies described here. Consent was obtained from the owner, who was fully informed of the objectives of the study and the intended use of the blood. Approval for animal use was obtained from the WSU Institutional Animal Care and Use Committee (ASAF 6948).
Antibodies used in our study
Multiple sets of hybridomas were developed using published methods. 6 Each set of hybridomas was given a distinct name for identification. Abbreviations of the names were used as a prefix with a number to name each cloned hybridoma cell line producing an antibody of interest. We conducted our studies with mAbs obtained from the following sets of hybridomas: BAG, BAQ, CADO, CAM, CL, DH, DLL, F, H, HUH, LND, LT, and PT. The selected mAbs described here recognized an epitope conserved on orthologous molecules including dogs, as well as mAbs that recognized epitopes only expressed on canine leukocytes. Information on the immunization strategies used to develop the sets of the hybridomas is provided elsewhere. 4
The sets of hybridomas were developed during the past 40 y. Flow cytometry was used as the primary method for identifying and clustering mAbs that yielded an identical pattern of labeling on cells from multiple species. The CD21 (CA2.1 D6) and CD45 (CA12.10C12) mAbs that we used were a gift from Dr. Peter Moore (UCDavis; University of California–Davis, Davis, CA, USA). The hybridoma cell lines LSM12.125 and LSM8.53, specific for CD4, and LSM4.78 and LSM1.140, specific for CD8, were donated to the WSU Monoclonal Antibody Center (Pullman, WA, USA) for use and preservation.
Preparation of leukocytes for flow cytometry
Previously described methods were used to label preparations of leukocytes for flow cytometric (FC) analysis. 1
Data acquisition and analysis
A flow cytometer (FACS Calibur; Becton Dickenson) was used to collect the data. Software (FCS Express 7; De Novo) was used to analyze the data and prepare the scatter plots. Electronic gates were used in log side scatter (SSC) versus linear forward scatter (FSC) to distinguish granulocytes (neutrophils and eosinophils) from mononuclear cells. Additional gates were placed on mononuclear cells to distinguish large (predominantly monocytes) from small (predominantly lymphocytes) mononuclear cells. Artificial coloring was used to further distinguish granulocytes (red), from monocytes and large lymphocytes (blue), and lymphocytes (orange). The signature patterns of expression of orthologues of MHC and CD molecules on leukocytes displayed in SSC versus fluorescence were used to document that mAbs submitted to the canine international workshop and to the animal homologues section of the HLD8 human leukocyte antigen workshop recognized bona fide orthologues of canine MHC and CD molecules.
Results
Monoclonal antibodies that recognize epitopes expressed on orthologues of known MHC and CD molecules in dogs
In FC analysis of the mAbs specific for MHC I and MHC II, epitopes recognized by the mAbs were conserved on the canine orthologues of MHC I, MHC II DR, and MHC II DQ described in humans. The FC patterns of expression of MHC molecules on leukocytes from dogs were identical to the patterns of expression observed on leukocytes from humans and other species (Table 1; Figs. 1A–D).
Monoclonal antibodies reactive with canine leukocytes.
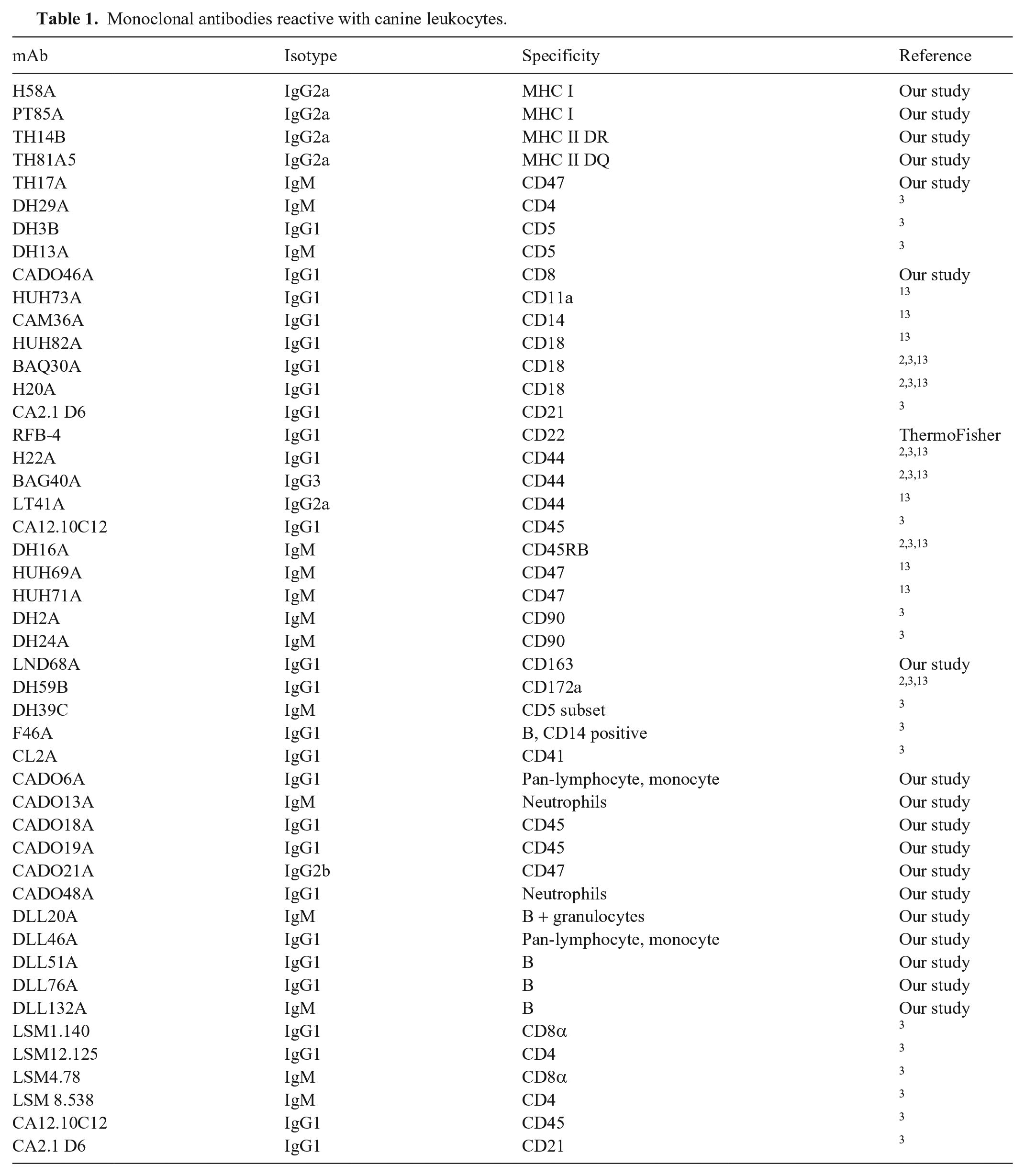
Representative flow cytometric (FC) scatter plot profiles (
In FC analysis, the patterns of labeling obtained with mAbs specific for orthologues of CD molecules recognized orthologues of CD molecules in dogs. All of the CD4-specific mAbs labeled CD4 T cells and neutrophils, a pattern of expression unique to canine CD4 (Table 1; Fig. 1E). All of the CD5- and CD8-specific mAbs recognized epitopes on the respective subsets of lymphocytes (Table 1; Figs. 1F, 1G). The FC patterns of expression were unique for each molecule. The CD4-, CD5-, and CD8-specific mAbs recognized species-restricted epitopes.
Except for DH24A, the mAbs recognized epitopes conserved on orthologues of CD molecules in dogs and other species (Table 1; Fig. 1). The FC profiles of expression in dogs are representative of the FC profiles obtained with leukocytes from other species (Table 1; Figs. 1H–M, 1O, 1P). DH24A recognized the canine orthologue of CD90 (Table 1; Fig. 1N)
Progress in expanding mAbs available for clinical flow cytometry and research in dogs
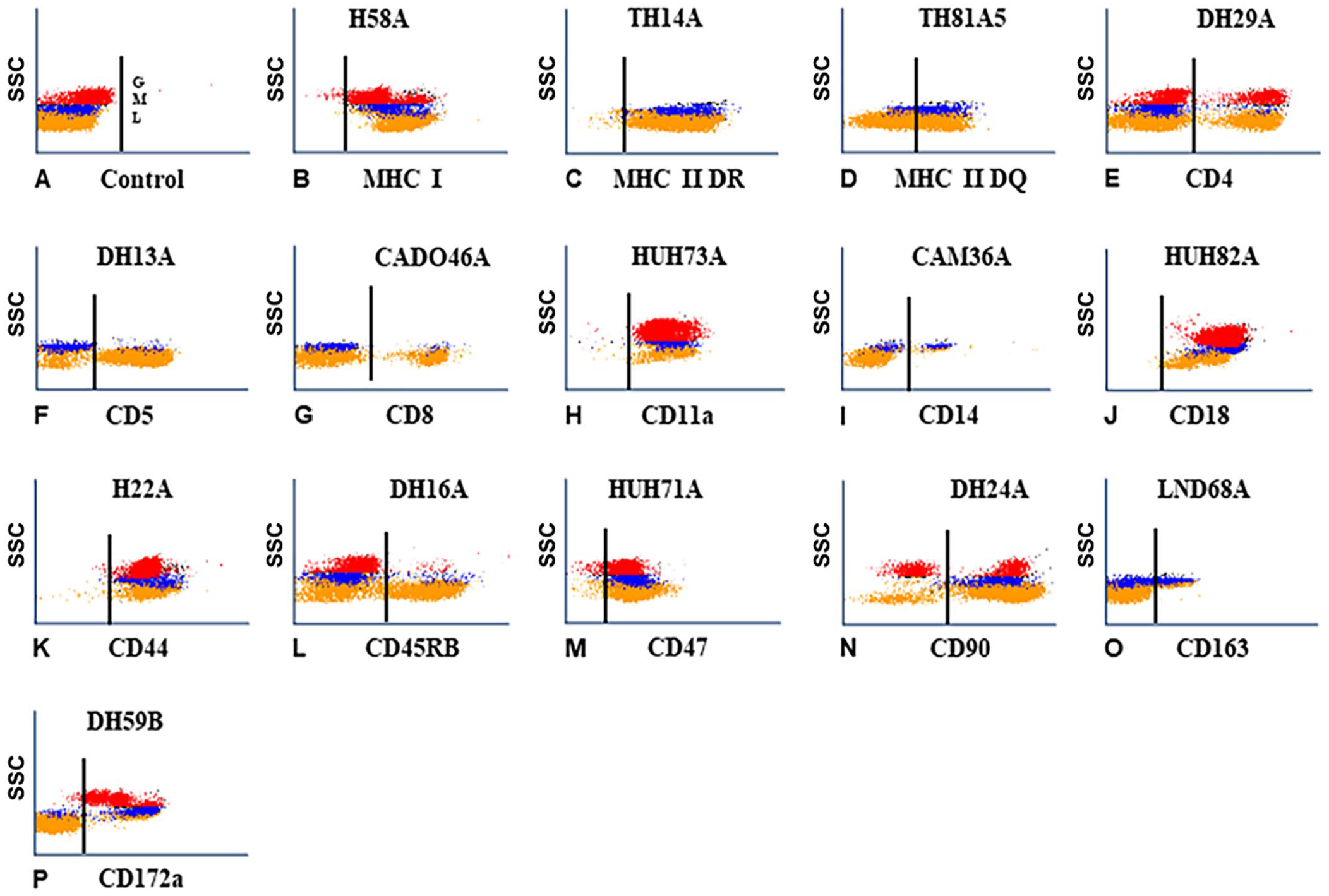
We identified 2 mAbs from the mAbs submitted to the canine workshop for further characterization, and 10 additional mAbs from sets of mAbs produced by hybridomas developed from mice immunized with canine leukocytes (CADO), and mice immunized with cells from a dog with B-cell leukemia (DLL). One mAb, DH39A, submitted to the canine workshop yielded a pattern of labeling similar to the pattern of labeling obtained with mAbs specific for CD5 (Table 1; Fig. 2A), but the molecular weight reported in the canine workshop suggested that it identified a different molecule. 3 In 2-parameter co-labeling of DH39C with CD4 and CD8 mAbs, the mAb identified a T-cell subset that includes subsets of CD4 and CD8 (Table 1; Figs. 2B, 2C). In workshop studies with the second mAb (F46A), the mAb recognized a molecule expressed on a subset of B cells in the canine workshop. Our further FC analysis reported here showed that the molecule is expressed on B cells and a subset of non-B cells (Table 1; Fig. 2D).
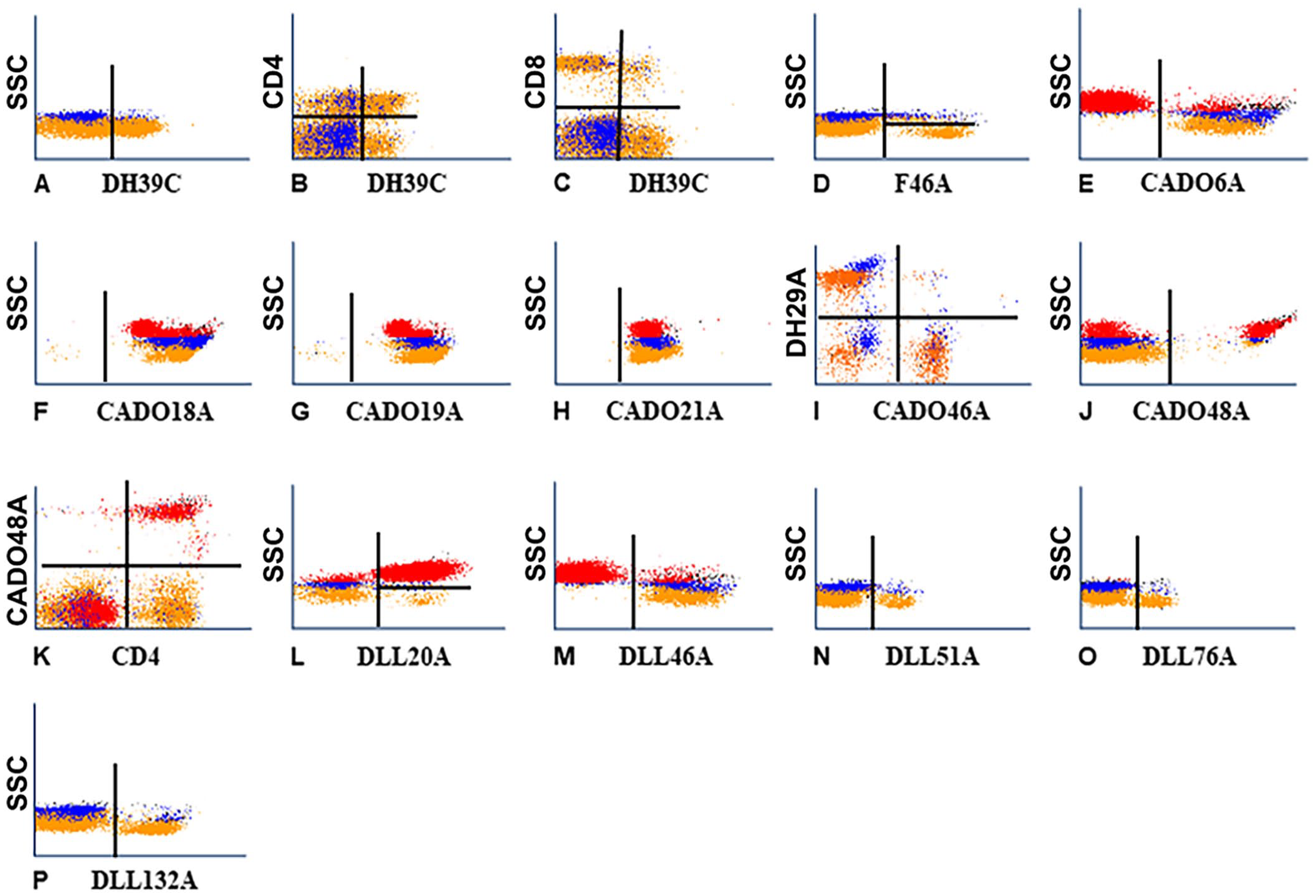
Representative flow cytometric (FC) profiles of leukocytes labeled with mAbs of known specificity and new mAbs reported herein.
Comparison of the FC profiles of the CADO set of mAbs yielded a range of findings. The molecule identified with CADO6A was expressed on all lymphocytes and monocytes (Table 1; Fig. 2E). Both CADO18A and CADO19A mAbs recognized the same molecule, and the molecule detected was expressed on all leukocytes (Table 1; Figs. 2F, 2G). Comparison of the FC profile of CADO21A (Table 1; Fig. 2H) with the profile obtained with HUH71A, a mAb specific for CD47, showed that CADO21A (Table 1; Fig. 2H) recognized an epitope expressed on CD47 (not shown). In the FC profile obtained with CADO46A, the mAb recognized a subset of lymphocytes. In the FC analysis of CADO46A with a mAb specific for CD4, CADO46A recognized CD8 (Table 1; Fig. 2I). The FC profile obtained with CADO48A recognized a subset of granulocytes (Fig. 2J). In 2-parameter co-labeling with CD4, the molecule recognized by CD48A is expressed on neutrophils (Table 1; Fig. 2K). Canine CD4 is expressed on CD4 T cells and neutrophils. 11 Both CADO13A and CADO48A mAbs recognized neutrophils (not shown).
Comparing the FC profiles obtained with the DLL set of mAbs, 2 mAbs recognized molecules with different patterns of expression, and 3 recognized molecules with a similar pattern of expression. DLL20A recognized a molecule expressed on granulocytes and a subset of lymphocytes (Table 1; Fig. 2L). DLL46A recognized a molecule expressed on all lymphocytes and monocytes (Table 1; Fig. 2M). The pattern of expression was similar to the pattern of expression obtained with CADO6A (compare Fig. 2E with Fig. 2M). DLL51A, DLL76A, and DLL132A recognized a subset of lymphocytes with a similar pattern of expression, suggesting that the mAbs might recognize the same subset of lymphocytes (compare Figs. 2N–P).
Further analysis of the specificity of selected mAbs
We selected the mAbs DLL20A, DLL51A, DLL76A, F46A, DLL132A, CADO18A, and CADO19A for further analysis. Initial studies indicated that DLL20A recognized an epitope expressed on granulocytes and a subset of lymphocytes. In 2-parameter co-labeling with a mAb specific for CD21, the lymphocyte subset recognized by DLL20A was B cells. Co-labeling of CD21 with DLL132A showed that DLL132A also recognized B cells (Fig. 3B). Diagonal-pattern labeling occurs when mAbs recognize different epitopes on the same molecule; DLL132A recognized CD21. Co-labeling with mAbs specific for different molecules on the same lymphocyte population yield a diffuse pattern of labeling, owing to a difference in the concentration of the molecules expressed in the membrane (as illustrated with co-labeling by DL132A with a mAb specific for CD22; Fig. 3C). In labeling of DLL132A with DLL51A and DLL76A, both mAbs recognized different non-CD21 molecules on all B cells (Figs. 3D, 3E). In co-labeling of DLL132A with F46A, the molecule detected was expressed on all B cells and a small subset of non-B cells (Fig. 3F). In co-labeling of F46A with a mAb specific for CD14, the small non-B subset of cells was positive for CD14 (Fig. 3G). Co-labeling of CD45 (CA12.10C12) with CADO18A and CADO19A yielded a diagonal pattern of labeling indicating that both mAbs recognized canine CD45 (Fig. 3H, not shown for CADO19).
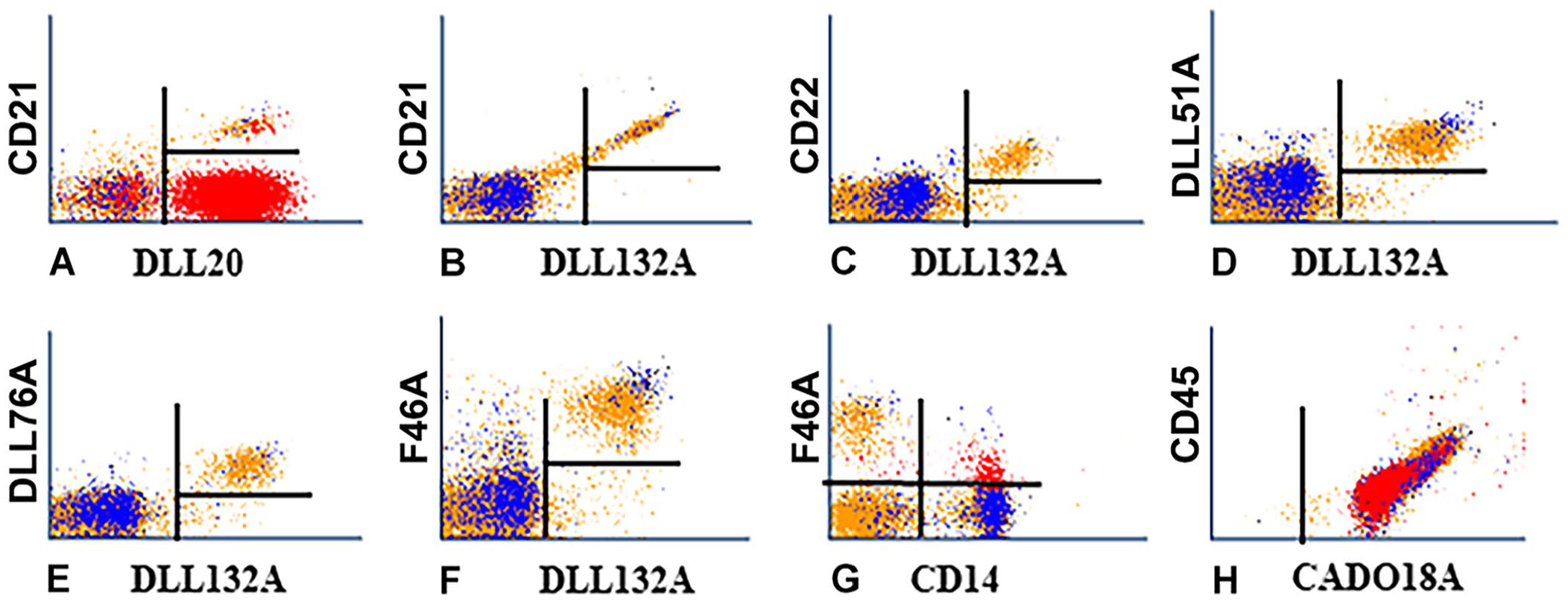
We used 2-color flow cytometry with mAbs known to be expressed on all B cells (CD21 and CD22) to determine the specificity of 5 new mAbs that recognized subsets of lymphocytes.
Discussion
We used flow cytometry to assess the reactivity of previously characterized mAbs with canine orthologues of CD molecules. These mAbs recognized the corresponding CD molecules in dogs, as evidenced by consistent expression patterns identical to those observed for their human counterparts. Our work extends the availability of reagents for both clinical and research applications in canine immunology, providing essential tools for immunophenotyping and diagnostic advancements.
Further characterization of the molecule identified with mAb DH39C revealed that it differs from CD5 and is expressed on subsets of CD4 and CD8 T cells. This novel expression profile indicates the potential utility of DH39C for defining specific T-cell subsets in dogs. The molecule recognized by mAb F46A was expressed on all B cells and a subset of CD14-positive cells, which suggests the relevance of mAb F46A in both B cell and myeloid cell biology. In clinical FC screening of dogs with leukemia (unpublished), mAb F46A distinguished 2 types of B-cell leukemias: 1 positive for the molecule recognized by F46A and 1 negative. Furthermore, mAb F46A was highly expressed on CD14-positive myeloid leukemias (unpublished), underscoring its potential utility as a diagnostic marker for these conditions.
One of the CADO set of mAbs recognized a molecule highly expressed on neutrophils. Initial clinical FC results indicated that this molecule was also expressed on myeloid leukemia cells (unpublished), suggesting its relevance in diagnosing and understanding myeloid disorders. Additionally, 4 mAbs from the DLL set recognized molecules expressed on B cells. Specifically, DLL20A bound to a molecule expressed on both B cells and granulocytes; DLL32A recognized CD21, a well-known B-cell marker. In co-labeling experiments with mAbs DLL132A, DLL51A, and DLL76A, DLL51A and DLL76A targeted different molecules expressed on B cells, as evidenced by differences in their levels of expression. Our findings suggest that DLL51A and DLL76A may help to differentiate specific B-cell subpopulations, although further studies are needed to evaluate their differential expression in B-cell lymphomas or leukemias.
Although additional studies may further elucidate the specific roles of these mAbs, our data provide robust evidence for their utility in clinical flow cytometry and research applications in canine immunology, demonstrating their immediate relevance and effectiveness in advancing the field. These mAbs expand the toolkit available for immunophenotyping, diagnostics, and the study of immune responses in dogs.
Moreover, these mAbs provide a valuable opportunity to include dogs in comparative studies of immune responses to pathogens affecting multiple species. The highly conserved epitopes present on MHC I, MHC II DR, and MHC II DQ are also expressed on canine orthologues of these molecules. Therefore, mAbs specific for MHC I, MHC II, and canine CD molecule orthologues can be employed in both clinical flow cytometry and cross-species immune response research, thereby facilitating broader insights into immune mechanisms across species.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was funded by the Washington State University Monoclonal Antibody Center, Pullman, WA, USA.
