Abstract
Immunohistochemical and histochemical stains are useful adjunct techniques in the diagnosis of canine cutaneous round cell tumors, which can appear histologically similar We applied a panel of monoclonal antibodies (recognizing tryptase, chymase, serotonin for mast cells; CD1a, CD18, MHC class II for histiocytes; CD3 for T lymphocytes; CD79a for B lymphocytes and plasma cells) and one histochemical stain (naphthol AS-D chloroacetate for chymase activity) to formalin-fixed, paraffin-embedded sections of canine cutaneous mast cell tumors, histiocytomas, lymphosarcomas, plasmacytomas, and unidentified round cell tumors. Of 21 tumors with a histologic diagnosis of mast cell tumor, 7/7 (100%) grade I, 6/7 (85.7%) grade II, and 3/7 (42.9%) grade III tumors were diagnosed as mast cell tumors based on positive staining for tryptase antigen and chymase activity. Mast cells were positive for both tryptase antigen and chymase activity, indicating equal efficacy of tryptase immunohistochemistry and chymase histochemistry. Chymase was detected immunohistochemically in both tumor and nontumor cells, while serotonin was not detected in most mast cell tumors, and thus, neither was useful in the diagnosis of mast cell tumors. Immunohistochemistry to detect CD18 and MHC class II was equally effective in staining histiocytomas, although lymphosarcoma must be ruled out through the use of CD3 and CD79a immunohistochemistry. Immunohistochemistry using three different monoclonal antibodies to human CD1a showed no cross-reactivity in canine histiocytomas and was not useful. A final diagnosis was obtained for 4/5 (80%) of the unidentified tumors, indicating the usefulness of multiple stains in poorly differentiated round cell tumors.
Keywords
Diagnosis of canine cutaneous round cell tumors based on histopathology alone is often challenging. Different round cell tumors may have a similar morphologic appearance, and even special stains may not be useful, particularly for poorly differentiated tumors. An accurate diagnosis is important in determining prognosis and treatment. Immunohistochemistry (IHC) has been used as an adjunct to light microscopy in the diagnosis of various neoplasms, including round cell tumors. In veterinary medicine, antibodies that have been validated for specific use in humans are often utilized for IHC; however, these antibodies have rarely been validated for animal species. 7 Because IHC is a relatively expensive diagnostic procedure, pathologists tend to use one or two antibodies to confirm diagnostic suspicions, but these results may not be reliable if the specificity of these antibodies is in question. Although certain antibodies may work in well-differentiated tumors, they are often less effective in poorly differentiated tumors that are also more difficult to diagnose histologically.
Tryptase, chymase, and serotonin are found in mast cell granules, and tryptase and chymase are mast-cell specific. 12 Serotonin is a preformed biogenic amine stored in mast cell granules, 26 but it is also present in other tissues. In the dog, there are three subtypes of mast cells, based on content of tryptase and chymase: mast cells containing tryptase only, mast cells containing chymase only, and mast cells containing both tryp-tase and chymase, the latter being the most common subtype in canine skin. 12, 27 The subtype of mast cell involved in canine cutaneous mast cell tumors has not been elucidated, although one canine mast cell tumor examined by Welle et al. 28 contained both tryptase and chymase. Immunohistochemical and enzyme-histochemical staining methods for tryptase and chymase have been described. 12, 28
Canine cutaneous histiocytoma is a tumor of epidermal Langerhans' cell origin. 17 Langerhans' cells in canine cutaneous histiocytomas express leukocyte antigens characteristic of dendritic cell differentiation, including CD1a, CD1b, CD1c, and CD11c. Expression of CD1 molecules distinguishes Langerhans' cells from macrophages. A diagnostic problem arises because these antigens are only detectable in fresh or frozen tissue, which is not often available to the veterinary pathologist. Currently, the most useful immunohistochemical criteria for identifying histiocytic cells in formalin-fixed, paraffin-embedded (FFPE) tissues are CD18 positivity, coupled with negative results for CD3 and CD79a to rule out lymphocytic origin. 1, 17 CD18, a nonspecific antigen involved in leukocyte adhesion, is present on the surface of all leukocytes. 4 Neoplastic histiocytes also consistently express MHC class II. 17 MHC class II staining is more specific for dendritic cells than is CD18, but T and B lymphocytes express MHC class II and will stain positively. 11, 14 CD1a is the most reliable marker for Langerhans' cells in the epidermis; 17 however, the only commercially available monoclonal antibodies for use in FFPE tissues were developed for use in humans and have not been investigated in the dog.
Lymphosarcoma and plasmacytoma are differential diagnoses for round cell tumors. T lymphocytes express CD3 and CD18 and, in the dog, both resting and activated T cells express MHC class II. 11, 14 B lymphocytes express CD79a, CD18, and MHC class II. 14 Macrophages and granulocytes express 10-fold more CD18 than do lymphocytes, and in FFPE tissues, lymphocytes may have undetectable CD18. 14 Plasmacytomas stain positively for CD79a approximately 80% of the time 15 and may be positive for MHC class II.
We selected a panel of monoclonal antibodies recognizing the following antigens: tryptase, chymase, and serotonin for mast cells; MHC class II, CD18, and CD1a for histiocytes; CD3 for T lymphocytes; CD79a for B lymphocytes and plasma cells; and one histochemical stain (naphthol AS-D chloroacetate for chy-mase activity). These were applied to FFPE sections of canine cutaneous tumors histologically diagnosed as mast cell tumors, histiocytomas, lymphosarcomas, and plasmacytomas, as well as unidentified round cell tumors. In addition, toluidine blue was applied to those tumors diagnosed histologically as mast cell tumors. With the exception of CD1a, these antibodies were chosen based on their previous application to FFPE tissues, the samples most often available to diagnostic pathologists. The primary objective of this study was to evaluate a panel of immunohistochemical stains and one histochemical stain in assisting the diagnosis of 72 canine cutaneous round cell tumors. Mast cell tumors and histiocytomas were the main focus, but lymphoid tumors were included to properly evaluate the stains. Of particular interest was identifying reliable markers for poorly differentiated round cell tumors.
Materials and Methods
Seventy-two archival canine cutaneous round cell tumors were selected from those diagnosed between 1995 and 2001 at the Western College of Veterinary Medicine, Saskatoon, SK, Canada. All tumors except five had been diagnosed on histopathology and, in some cases, histochemical stains or IHC. These diagnoses were not taken as the gold standard but were used as a comparison for statistical testing. Tumors selected included 21 mast cell tumors (7 each of grades I, II, and III according to a standard grading system 19 ), 19 histiocytomas (8 each of histiocytoma and regressing histiocytoma, and 3 atypical histiocytomas), 14 plasmacytomas, 9 epitheliotropic lymphosarcomas, 4 nonepitheliotropic lymphosarcomas, and 5 unidentified round cell tumors. Regressing histiocytomas were defined by the presence of numerous small lymphocytes on histology. Atypical histiocytomas did not have a typical morphologic appearance, but special stains were most consistent with histiocytoma (i.e., negative for toluidine blue and methyl green pyronine to rule out mast cell tumor and plasmacytoma, respectively). Prior IHC on one atypical histiocytoma was negative for T and B lymphocyte markers and positive for MHC class II.
A panel of monoclonal antibodies was applied to deparaffinized 4-μm sections of each skin tumor. The primary antibodies recognized tryptase (1 : 50, AA1, DAKO, Carpinteria, CA), chymase (1 : 500, CC1, NeoMarkers, Fremont, CA), MHC class II (1 : 150, TAL.1B5, DAKO), CD18 (1 : 4, CA16.3C10, P. Moore, UC-Davis, Davis, CA), and CD3 (1 : 25, CD3-12, Novocastra Laboratories Ltd., Newcastle upon Tyne, UK). Staining was done using a Benchmark staining platform (Ventana Medical Systems Inc., Tucson, AZ), with the BMK iVIEW DAB Paraffin detection kit (Ventana Medical Systems Inc.) using a streptavidin-biotin amplification system. Antigen retrieval was performed as follows for all antibodies except CD18. Heat retrieval consisted of applying cell conditioner #1 (this and other reagents are proprietary and included in the kit from Ventana Medical Systems Inc.) to skin sections and then heating to 95°C for 8 minutes, followed by 20 minutes at 100°C. I-VIEW inhibitor was added and incubated for 4 minutes at 42°C. For CD18 antigen retrieval, protease 2 was applied and incubated for 10 minutes. All slides were then incubated with primary antibody for 32 minutes, followed by I-VIEW biotin immunoglobulin for 8 minutes, I-VIEW SA-HRP for 8 minutes, I-VIEW DAB and H2O2 for 8 minutes, and I-VIEW copper for 4 minutes. Slides were counterstained with hematoxylin and bluing reagent.
Three different anti-human CD1a clones (ICA04, Neo-Markers; MTBI, Novocastra Laboratories Ltd.; JMP30, Novocastra Laboratories Ltd.) for use in FFPE tissues were tested on select canine tumors at different dilutions using both heat and protease antigen retrieval but were not applied to every tumor in the study.
Serotonin (1 : 100, YC5/45, Medicorp, Montreal, PQ, Canada) and CD79a (1 : 2, HM47/A9, NeoMarkers) staining was done with a Codon Histomatic slide stainer (Fisher Scientific Co., Edmonton, AB, Canada), using an avidin-biotin immunoperoxidase technique previously described by Haines and Chelack, 7 with the exception that antigen retrieval for CD79a was performed by heating sections to 95°C for 20 minutes in a citrate buffer, pH 6.0, instead of by proteolytic digestion. Primary antibodies were incubated overnight at 4°C. Secondary antibodies were incubated for 30 minutes at 37°C. Stains were completed with a 30-minute incubation with avidin-biotin complex at 37°C. Diaminobenzidine (Cedarlane Laboratories Ltd. Canada, Hornby, ON, Canada) was used as the chromogen substrate. Positive control slides were included in each run and examined for appropriate staining.
Each tumor was evaluated for chymase activity with an enzyme histochemical stain using naphthol AS-D chloroacetate as substrate (Procedure No. 91, Sigma-Aldrich Canada Ltd., Toronto, ON, Canada). This chymase stain has been used for normal canine mast cells in FFPE tissues. 12, 13, 27, 28 It is a modification of the original staining procedure described by Benditt and Arase, 3 who demonstrated the selectivity of chloroacyl naphthol esters for chymotryptic enzymes compared with tryptic enzymes. Tumors that were identified as mast cell tumors histologically were stained with toluidine blue (BDH Laboratory Supplies, Poole, UK) for comparison with tryptase and chymase IHC and chymase histochemistry.
A blinded examiner (NJF) reviewed all the slides for each stain consecutively to ensure consistency of evaluation. Sections were classified as positive or negative based on staining characteristics of tumor cells. Slides that were difficult to interpret due to faint staining of tumor cells were restained. Expected results for each tumor type are summarized in Table 1. Our panel was not designed to differentiate plasma-cytoma from B-cell lymphosarcoma; thus, these results are grouped as one diagnosis.
Expected immunohistochemical and histochemical results for canine cutaneous round cell tumors.∗

∗ MCT = mast cell tumor; HCT = histiocytoma; TLSA = T cell lymphosarcoma; BLSA = B cell lymphosarcoma; PCT = plasmacytoma; IHC = immunohistochemistry; HC = histochemistry; +, positive; −, negative.
Once a stain panel diagnosis, based on IHC and histochemistry, was made for each tumor, this was compared with the histologic diagnosis using a kappa statistic (Win Episcope 2.0, University of Edinburgh). 20 The level of agreement, using the kappa statistic, was also determined between stains in tumors in which both stains were expected to be positive (e.g. tryptase and chymase for mast cells). Guidelines for interpreting agreement are as follows: kappa less than 0.4, poor; 0.4–0.75, fair to good; greater than 0.75, very good to excellent. 22
Results
Chymase IHC stained both nontumor and tumor cells (within all tumor types), consistent with nonspecific staining. Because chymase IHC was not useful, chymase results refer to chymase activity using histochemistry (HC). Serotonin IHC only occasionally demonstrated focal staining of mast cell tumor cells, despite appropriate staining in control tissues. The human CD1a antibodies did not detect any canine cells expected to be CD1a positive. These antibodies were not diagnostically useful and were not considered in further comparisons. For the remaining IHC and HC stains, results are provided for all tumors in Table 2. None of these remaining stains demonstrated appreciable nonspecific staining.
Stain panel results (immunohistochemical and histochemical) for 72 canine cutaneous round cell tumors.∗
∗ MCT = mast cell tumor; HCT = histiocytoma; TLSA = T cell lymphosarcoma; BLSA = B cell lymphosarcoma; PCT = plasmacytoma; HC = histochemistry; +, positive; −, negative; ND = not done.
† Unable to determine if cells are neoplastic.
Mast cell tumors
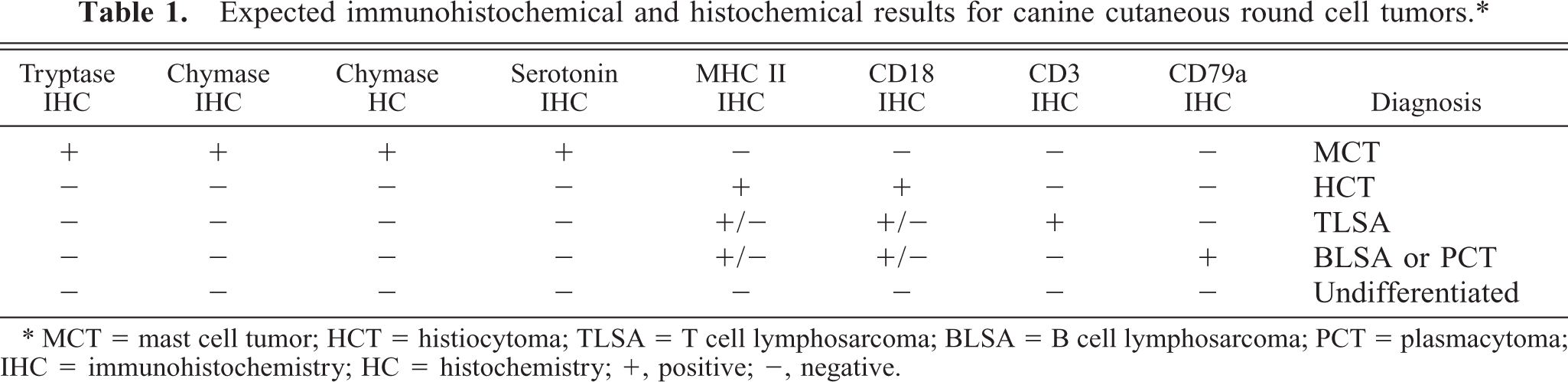
Of the 21 tumors that were diagnosed histologically as mast cell tumors (MCTs), 17 (80.9%) had staining patterns characteristic of MCTs. All (7/7; 100%) grade I MCTs, 6/7 (85.7%) grade II MCTs, and 3/7 (42.9%) grade III MCTs were positive for both tryptase antigen and chymase activity (i.e., chymase histochemical stain; Fig. 1a, b). One grade III MCT was positive for toluidine blue despite being negative for tryptase antigen and chymase activity. This tumor was also positive for CD18 (Fig. 2). The four remaining tumors included two that were positive for only CD18 and therefore designated as undetermined. These tumors were both histologically diagnosed as grade III MCT. All MCTs diagnosed by stain panel were negative for MHC class II; however, many MCTs contained numerous MCH class II–positive nontumor cells.
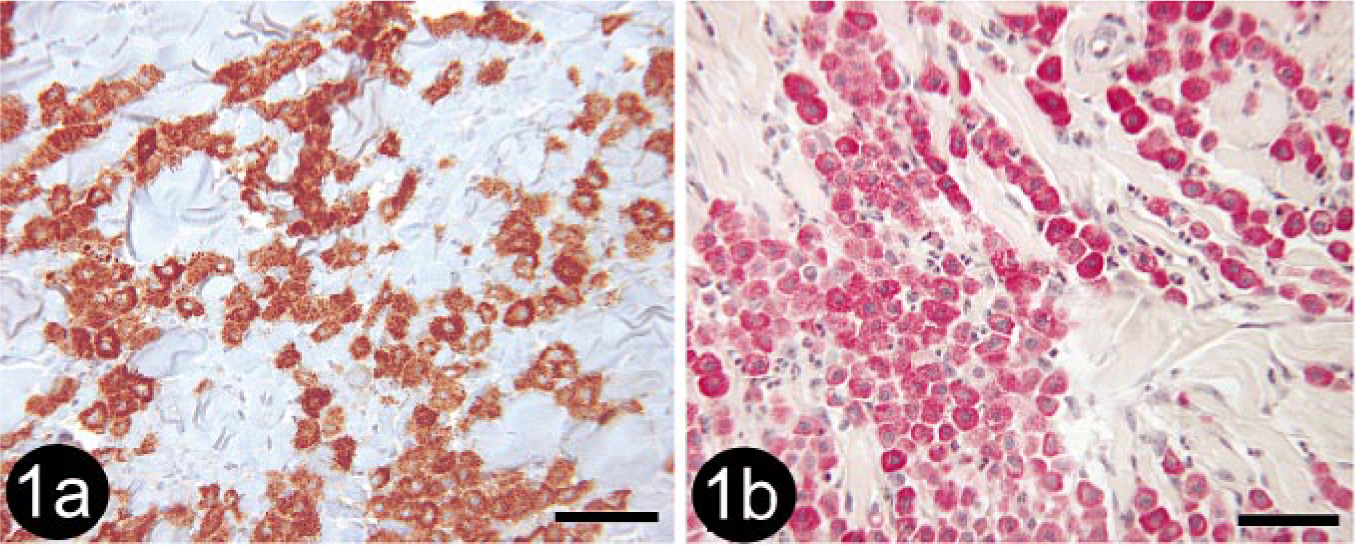
Skin; canine, mast cell tumor. Immunohistochemical staining for CD18 detection demonstrating numerous positive tumor cells (arrows). Note that staining is membranous rather than cytoplasmic. Cells demonstrating cytoplasmic staining are eosinophils (arrowheads). This tumor was also positive with toluidine blue, but negative for tryptase antigen and chymase activity. BMK iVIEW DAB Paraffin detection kit (Ventana Medical Systems Inc.) with primary antibody CA16.3C10 (1 : 4, P. Moore). Bar = 50 μm.
The agreement between the histologic diagnosis and the stain panel diagnosis was very good to excellent. Kappa (with 95% confidence intervals) was 0.78 (0.56–1.00). All tumors that were positive for tryptase antigen were also positive for chymase activity. The kappa statistic was 1.00 (0.77–1.23), which indicates excellent agreement between these two tests beyond what could be expected due to chance.
Histiocytoma
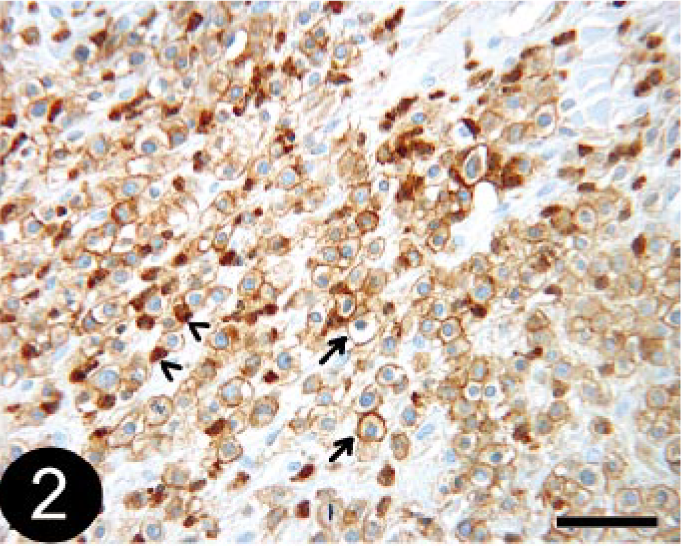
Of the 19 tumors that were diagnosed histologically as histiocytoma (HCT), 7 (36.8%) tumors were clearly identified as HCT using the stain panel (CD18 and MHC class II positive; Fig. 3a, b), 1 (5.7%) was diagnosed as B-cell lymphosarcoma (BLSA) or plasmacytomas (PCTs), and the remaining 11 (57.9%) could not be definitively diagnosed based on the stain panel alone and were designated as undetermined. While these tumors were positive for CD18 and MHC II, they also contained CD3- and/or CD79a-positive cells. It was impossible to determine whether the cells staining positively for CD3 and CD79a were infiltrating lymphocytes associated with HCT regression or whether they were neoplastic cells (Fig. 4). All of the three tumors histologically diagnosed as atypical HCT (HCTA) were designated as undetermined by the stain panel.
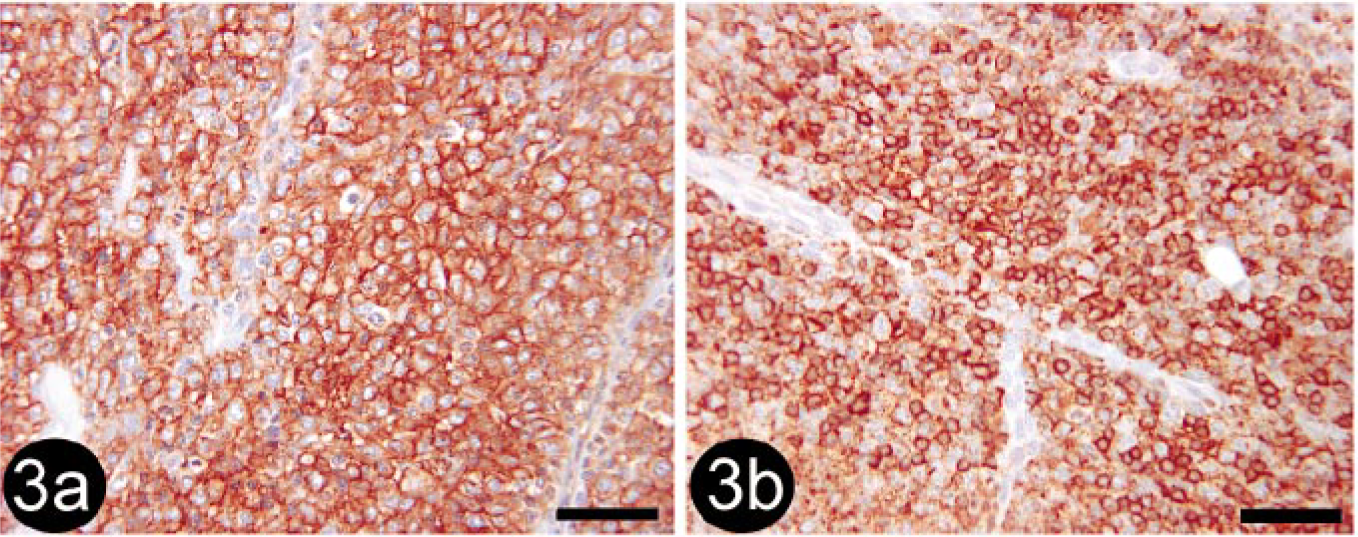
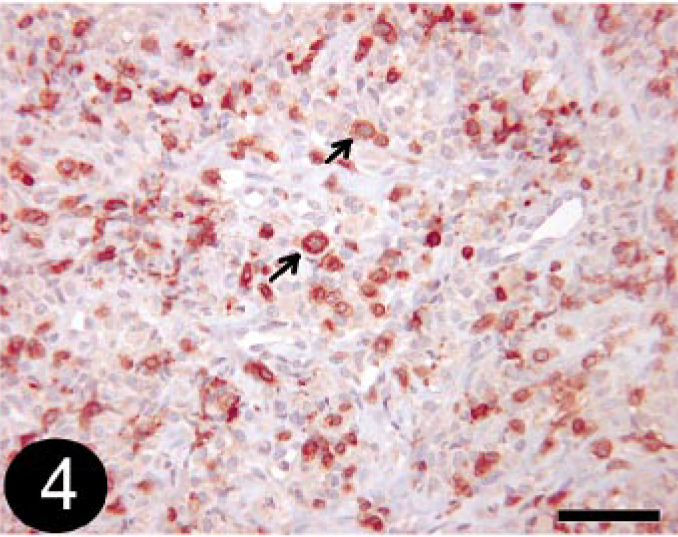
Skin; canine, regressing histiocytoma or T-cell lymphosarcoma. Immunohistochemical staining for CD3 detection. Note that positive cells do not have typical lymphocyte morphology (arrows). BMK iVIEW DAB Paraffin detection kit (Ventana Medical Systems Inc.) with primary antibody CD3-12 (1 : 25, Novocastra Laboratories Ltd.). Bar = 50 μm.
There were four tumors not histologically diagnosed as HCT that were clearly diagnosed as HCT by the stain panel: two PCTs, one MCT grade II, and one unidentified tumor.
The kappa statistic comparing agreement between the histologic diagnosis of HCT and the stain panel diagnosis was poor, at 0.29 (0.07–0.51). The agreement between MHC class II and CD18 was fair to good, at 0.50 (0.27–0.72), when the stains were compared across all tumors.
Lymphosarcoma and plasmacytoma
T-cell lymphosarcoma was the most common (10/13; 76.9%) stain-panel diagnosis for histologically diagnosed lymphosarcoma, based on positive CD3 staining (Fig. 5). Tumors that were positive for CD79a were diagnosed as BLSA or PCT because the stain-panel results are the same for these two tumors (our panel was not designed to differentiate these tumors; Fig. 6).
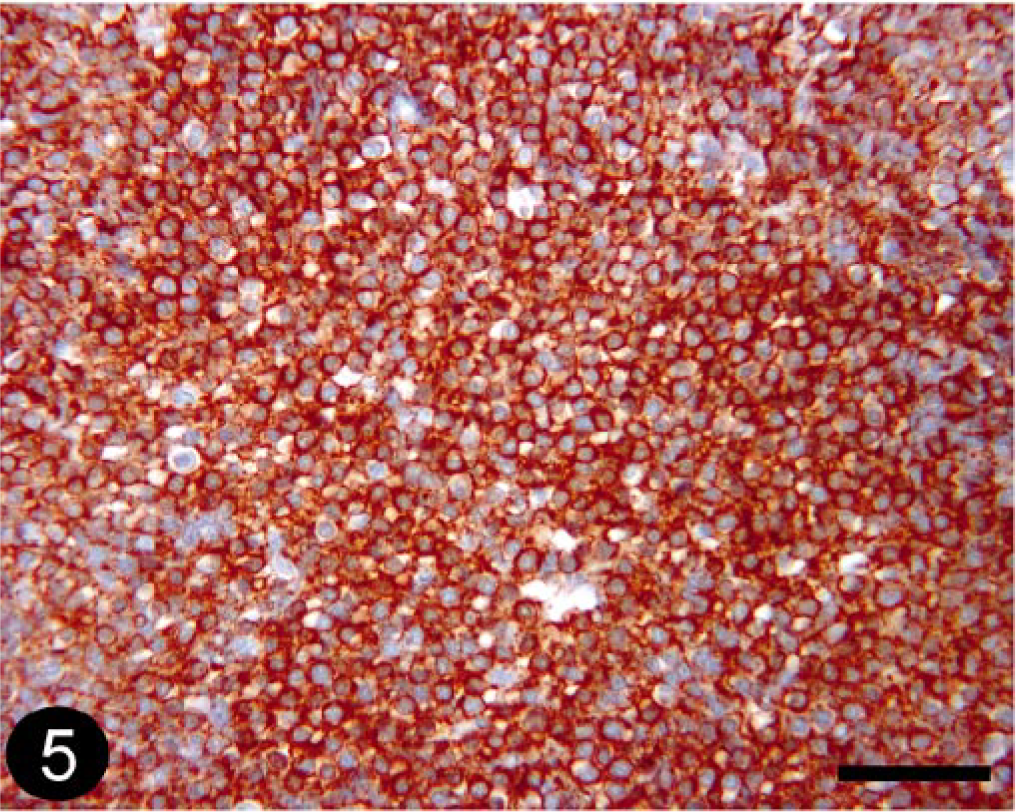
Skin; canine, T-cell lymphosarcoma. Immunohistochemical staining for CD3 detection. BMK iVIEW DAB Paraffin detection kit (Ventana Medical Systems Inc.) with primary antibody CD3-12 (1 : 25, Novocastra Laboratories Ltd.). Bar = 50 μm.
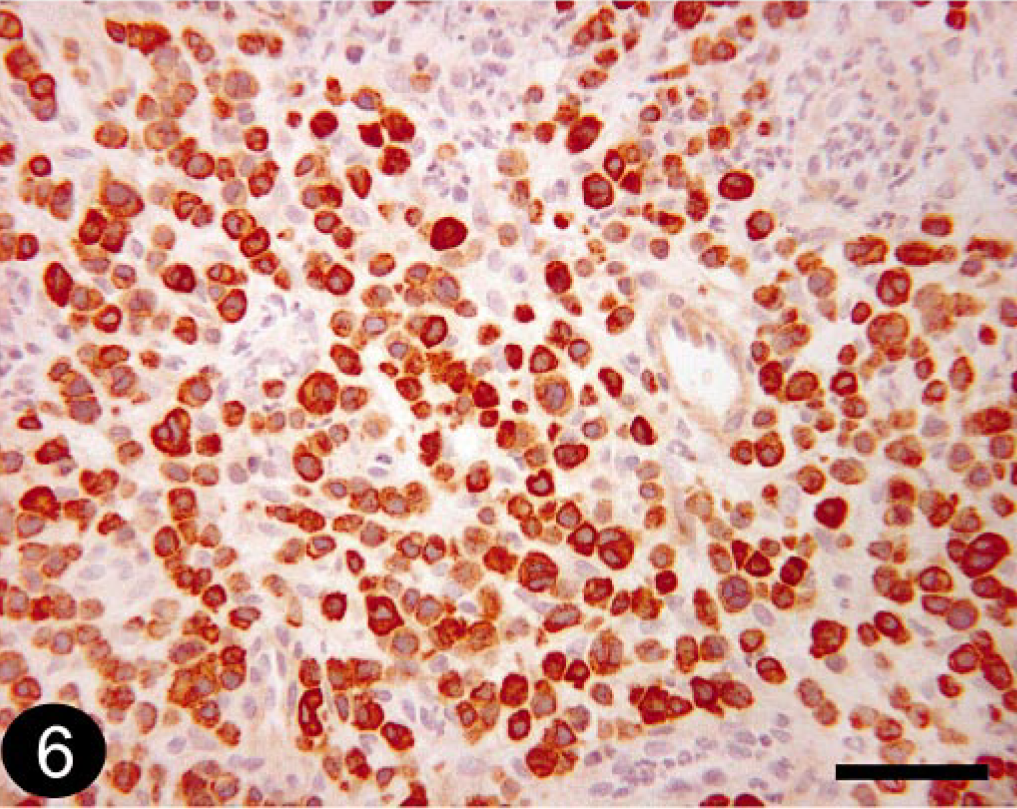
Skin; canine, B-cell lymphosarcoma or plasmacytoma. Immunohistochemical staining for CD79a detection. Avidin-biotin immunoperoxidase (Codon Histomatic slide stainer, Fisher Scientific Co.) with primary antibody HM47/A9 (1 : 2, NeoMarkers). Bar = 50 μm.
Unidentified round cell tumors
A diagnosis was obtained for 4/5 (80%) histologically unidentified round cell tumors. The tumor that was negative for all stains was designated as undifferentiated, but could also be a PCT because only about 80% of PCTs express CD79a. 15
Discussion
In this study, tryptase and chymase were examined extensively in canine cutaneous MCTs. Welle et al., 28 in their study of bovine cutaneous and tumor mast cells, examined one canine cutaneous MCT and found it to be positive for both tryptase and chymase. Our study agrees with this finding. When compared with the histologic diagnosis, tryptase antigen and chymase activity were detected in all the grade I MCTs, all but one of the grade II MCTs, and about half of the grade III MCTs. However, because the histologic diagnosis is not a gold standard, the lack of positive staining in some tumors may reflect either a failure of these two stains or an incorrect histologic diagnosis. Clinical follow-up would be useful in determining the correct diagnosis. Despite these differences, agreement (kappa) was good between the histologic and stain panel diagnosis. Ozaki et al. 18 reported that only 62% of gastrointestinal MCTs in dogs were positive for tryptase antigen, although all tumors were positive with toluidine blue. This is similar to our finding that 76% (16/21) of histologically diagnosed cutaneous MCTs were positive for tryptase antigen, although only 81% (17/21) were positive with toluidine blue. Tryptase antigen and chymase activity were not positive for any of the other tumors in the study (those not diagnosed histologically as MCTs), which supports the specificity of tryptase antigen and chymase activity for mast cells. Toluidine blue staining was not performed in all tumors; therefore, its specificity cannot be similarly assessed.
We found a strong agreement between tryptase IHC and chymase HC (kappa = 1.00). This suggests that either stain could be used to aid the diagnosis of a MCT. We did not evaluate toluidine blue staining with a kappa statistic; however, toluidine blue staining detected one more MCT than did either tryptase IHC or chymase HC, suggesting that it is as good or better for detecting MCTs. Also, toluidine blue staining is likely to be readily available in a diagnostic histopathology laboratory and is a good first choice for confirmation of a diagnosis of MCT in the dog.
The finding of equal detection of tryptase and chy-mase in MCTs in this study may reflect the predominant mast cell subtype in normal canine skin. 12, 27 A negative result with one or both of these stains does not definitively rule out MCT as a diagnosis, and stains that detect other mast cell–specific substances (e.g., to-luidine blue for sulfated glycosaminoglycans) may be useful for diagnosis. This may explain one case, which was negative for both tryptase antigen and chymase activity but positive for toluidine blue.
Of MCTs positive for tryptase antigen and chymase activity, 6/16 (37.5%) were also positive for CD18. Higher grade, less differentiated MCTs expressed CD18 more often than low-grade, well-differentiated ones. This result was unexpected; however, high levels of CD18 expression have been described in immature cultured human mast cells 24 and in a low percentage of normal human skin mast cells. 25 The tumors in our study that expressed only CD18 may have been poorly differentiated, immature MCTs that had not yet developed the mast cell–specific proteases tryptase and chy-mase within their granules.
None of the MCTs was positive for MHC class II expression. However, many of the MCTs contained nontumor, MHC class II–positive cells resembling normal dendritic cells, which has not been previously described.
One grade I MCT that was positive for tryptase antigen and chymase activity was also positive for CD3 expression. CD3 is closely associated with the T-cell receptor and is reported to be expressed only on the surface of mature T cells and thymocytes. 15 The staining pattern of CD3 in this tumor was granular and cytoplasmic rather than membranous, as would be expected in T cells. This suggests that these neoplastic cells were producing a molecule within their granules that was antigenically similar to CD3, and thus recognized by the CD3 monoclonal antibody. Ozaki et al. 18 examined 39 gastrointestinal MCTs using various monoclonal antibodies, including CD3 and CD79a. None of the tumors was positive for CD3, but 7/39 (17.9%) tumors were positive for CD79a.
Several authors have described the use of chymase IHC to detect mast cells in both canine MCTs 28 and human skin; 9 however, the chymase antibody that we used consistently stained both tumor cells and adjacent nontumor cells and was not useful. In our study, all tumors were negative for serotonin, including those that were positive for tryptase and chymase. The reason for the absence of serotonin staining in our MCTs is unclear. Mast cells in canine fundic mucosa do not contain detectable serotonin, 2 and this may be true of mast cells in canine skin as well.
Of 19 tumors histologically diagnosed as HCTs, 11 (57.9%) could not be definitively diagnosed based on the stain-panel results. The most significant difficulty in making a diagnosis of HCT using the stain panel was differentiating regressing histiocytoma from T-cell lymphosarcoma. Others have reported difficulty in distinguishing lymphocytes and neoplastic cells. 11 Tumors that were diagnosed as HCTs by the stain panel frequently contained CD3-positive nontumor cells with typical small mature lymphocyte morphology (i.e., round cells with small, round hyperchromatic nuclei and scant cytoplasm). In the undetermined cases, both small mature lymphocytes and larger cells with pleomorphic nuclei and abundant cytoplasm stained positively for CD3. It was impossible to determine, based on morphology, whether these pleomorphic CD3-positive cells were neoplastic or normal cells (i.e., lymphocytes). In these questionable cases, the histologic appearance of the tumor may be useful in reaching a diagnosis. The histologic appearance was not characteristic of T-cell lymphosarcoma, and in a diagnostic setting, these tumors likely would have been diagnosed as regressing HCTs without the aid of IHC. IHC may be more useful in aiding the diagnosis of HCTs that do not have a typical histologic appearance. The use of IHC for HCTs is probably best restricted to this situation.
There were also three tumors that contained CD79a-positive cells in addition to the CD3-positive cells, and these were similarly difficult to diagnose based only on the stain panel. Although regressing HCTs are generally infiltrated with CD3-positive T cells, B cells can occasionally be found within these tumors. 11, 17 A diagnosis of T cell–rich BLSA is unlikely in these cases because both normal-appearing small lymphocytes and pleomorphic cells were positive for CD3. In T cell–rich BLSA, IHC indicates T-cell origin of the small normal lymphocytes and B-cell origin of the large pleomorphic cells. 23
In summary, the most reliable criteria for immunohistochemical diagnosis of canine cutaneous HCT were CD18 and MHC class II positivity coupled with negative staining for CD3 and CD79a. However, as discussed above, IHC is not always useful or recommended. In this study, by definition, an HCT had to be positive for both CD18 and MHC class II, which was necessary because of the nonspecific nature of CD18 and MHC class II. If both of these antibodies were not used, the tumor that was positive for CD18 only could have been incorrectly classified as an HCT, when in fact it was most likely an MCT based on positive staining with toluidine blue. Novel diagnostic methods may be useful in such cases, for example, the development of a canine CD1a monoclonal antibody that is effective in FFPE tissues. CD1a is the most reliable marker for Langerhans' cell differentiation in the epidermis, the cell of origin of canine cutaneous HCT. 17 Currently, there is a canine-specific CD1a antibody available that is applicable only to fresh or frozen tissues and which detects an allotype of CD1a that is present in about 80% of dogs. 17 Also, the use of only toluidine blue and methyl green pyronine to rule out MCT and PCT, respectively, does not allow diagnosis of HCT by exclusion because lymphosarcoma remains a differential diagnosis. IHC is a necessary next step.
Although it was not the primary focus of our study, we found that one of the epitheliotropic lymphosarcomas was of B-cell rather than T-cell origin. This was unexpected, as epitheliotropic lymphosarcoma in the dog is reported to be of CD8+ T cell origin. 5, 16 Nonepitheliotropic lymphosarcoma is reported to be mostly of T-cell origin in the dog, although it can be of B-cell origin. 5 We found an equal distribution of T-cell and B-cell lymphosarcomas within our nonepithe-liotropic group; however, our sample size was small (n = 4). Diagnosis of non–T cell, non–B cell lymphosarcoma (of presumed natural killer cell origin) is rare in dogs; 21 however, its existence presents a potential problem in the use of solely CD3 and CD79a to diagnose lymphosarcoma or to rule out lymphosarcoma in the diagnosis of HCT. Morphology would likely be useful in establishing a diagnosis given the granularity of these cells, perhaps combined with additional immunohistochemical stains. 10
The stain-panel results were the same for both BLSA and PCT; however, the two could likely be distinguished based on morphology. Methyl green pyronine staining for RNA 6 or immunohistochemical staining for immunoglobulin and kappa and lambda light chains 8 could also be used to confirm the diagnosis. One histologically diagnosed PCT was designated as undetermined because it was positive for CD3 as well as CD79a.
Four of the five histologically unidentified tumors were given a final diagnosis based on the stain panel. The one tumor without a final diagnosis was negative for all stains. This may be because the tumor was poorly differentiated, or it may be one of the 20% of PCTs that are negative for CD79a. 15 Methyl green pyronine staining may have been useful in ruling out PCT.
In conclusion, a minimum of five adjunct stains is most useful for the diagnosis of cutaneous round cell tumors in dogs: tryptase IHC, chymase HC, or toluidine blue for mast cells, MHC class II and CD18 IHC for histiocytes, and CD3 and CD79a IHC for lymphocytes (Fig. 7). Additional stains may be required depending on the outcome of the initial panel, especially for suspected HCTs.
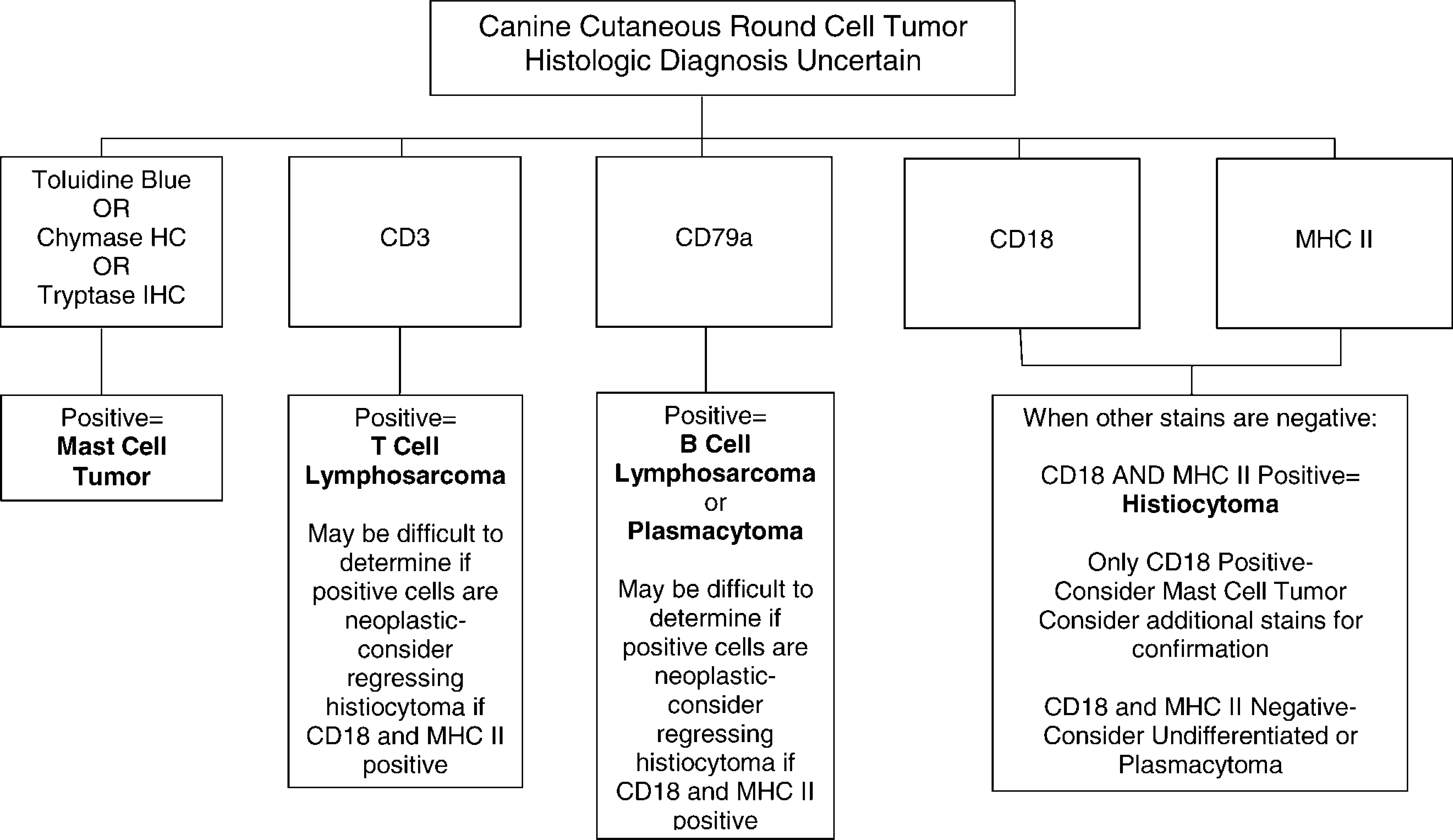
Recommended panel of adjunct stains for the diagnosis of canine cutaneous round cell tumors.
Footnotes
Acknowledgements
Thanks to Karen Moline, Brian Chelack, and Phil Dillman for technical assistance. Thanks also to Dr. Peter Moore for supplying the CD18 antibody. This study was funded, in part, by a grant from the WCVM Companion Animal Health Fund, Saskatoon, SK, Canada. Dr. Fernandez was supported by an Interprovincial Graduate Student Fellowship.
