Abstract
A dose of 1010 Mycobacterium avium subspecies paratuberculosis was administered orally on seven occasions to produce experimental paratuberculosis infection in 10 5-8-week-old goat kids. Bacteriological, immunological, and histopathological changes, their relationships, and the efficacy of the commonly used diagnostic methods were studied during the progressive disease up to 270 days postinfection (DPI). Significant lymphocyte proliferative responses in the peripheral blood of five goats were detected as early as 60 DPI. A lymphoproliferative test was also performed on lymphocytes purified from different compartments of the guts of five infected and five control goats. Significant proliferative responses were observed in lymphocytes of jejunal compartments of all five goats, of which four had also significant lymphocyte proliferation in the blood. The ileal lymphocytes from two goats, one each at 120 and 270 DPI, had significant proliferation. The histological lesions were mainly observed in the gut-associated lymphoid tissues of the ileocecal valve, the ileum, and the terminal jejunum. Acid-fast bacilli were demonstrated in the lesions of two goats at 60 and 210 DPI. Bacterial culture showed poor sensitivity, detecting positive results for only one goat in the fecal and tissue samples at 210 DPI, whereas polymerase chain reaction (PCR) detected one goat in fecal sample at 210 DPI and two goats in tissue samples at 60 and 210 DPIs, respectively. Enzyme-linked immunosorbent assay and agar gel immunodiffusion test were found to be 100% sensitive from 180 and 210 DPI onwards, respectively.
Keywords
Paratuberculosis (also called Johne disease) is an intestinal chronic granulomatous disease of domestic and wild ruminants. It is caused by a slow-growing, acid-fast bacterium, Mycobacterium avium subspecies paratuberculosis (M. a. paratuberculosis). Animals pick up the infection at the neonatal stage and, after a protracted incubation period of months to years, the clinical disease occurs in adults. 7, 8 In the small ruminants, especially goats, the disease is characterized clinically by progressive emaciation and soft or un-pelleted feces with occasional diarrhea at the terminal stages and pathologically by granulomatous enteritis with thickening and corrugation of the intestinal mucosa, regional lymphangitis, and lymphadenitis. 7, 29 The disease occurs worldwide and is responsible for huge financial losses. 24
The pathogenesis of paratuberculosis is largely unelucidated. Various studies on clinical cases related to bacteriology, immunology, histology, and their relationships have been widely reported. 5, 9, 41 Such studies are quite informative but do not provide information on sequential changes during progressive development of the disease. Experimental studies in goats are also meager in number. In the few experimental studies, the pattern of disease development appears to vary and depends on age of the animals and the dose rate. 3, 33, 19 Two recent experimental studies in goats with different dosage (100 and 250 mg bacteria) and different age groups (7–26 days and 5–9 weeks) did not result in definite disease pattern. 35, 37 No experimental studies have been performed on Indian goats till now.
Most previous studies are limited to examining peripheral immunity despite the fact that the initial host–pathogen interaction occurs at the gut level. Investigation of other mycobacterioses has demonstrated that peripheral and local changes may differ. 2 Following endocytosis by M cells of dome of the follicle-associated epithelium in the small intestine, 14, 34 M. a. paratuberculosis are then taken up by macrophages, where they interact with the gut-associated lymphoid tissue (GALT) and produce various local changes. Therefore, the outcome of M. a. paratuberculosis infection is likely to depend on the local immunological responses elicited by the lymphocytes in different layers of the intestine. 3, 5, 40 There is no study available on the early local lymphoproliferative responses in the experimentally infected goats, though a few studies on the lymphoproliferative response at the gut and peripheral levels in the naturally 5 and experimentally infected sheep 3 have been reported. Based on previous experimental studies in sheep 19, 21 and goats, 35, 37 the early immunopathogenetic mechanism may differ.
Diagnosis of paratuberculosis in early stages is difficult due to the long incubation period, the presence of a variable lag-phase associated with bacterial proliferation and multifocal distribution of slowly developing lesions. 8, 35 The efficacy of commonly used diagnostic methods has been less frequently evaluated 8, 24 in subclinical paratuberculosis. 35, 39
The present study, in an experimental model, describes the progressive changes at the gut and peripheral levels in relation to histological lesions. The ability of the commonly used diagnostic methods to detect M. a. paratuberculosis infection at early stages of infection was also assessed.
Materials and Methods
Animals and experimental design
Eighteen healthy crossbred goat kids, aged between 5 and 8 weeks of either sex testing negative to paratuberculosis infection in lymphocyte proliferation test (LPT), agar gel immunodiffusion test (AGID), enzyme-linked immunosorbent assay (ELISA), and fecal PCR were used in the study. After an initial adaptation in the experimental shed for 15 days, during which all animals were vaccinated against pestes des petits ruminants (PPR) virus infection and given anthelmintic (Albomar, Pfizer India, Mumbai, India) at the rate of 5–10 mg/kg body weight, the animals were divided into two groups: infected and control, comprising 10 (goats No. 1–10) and 8 (goats No. 11–18) animals, respectively. Animals of both groups were kept separately in different pens and maintained with standard care and management. Animals of the infected group were orally inoculated on seven occasions after every 2 days with triturated tissue material prepared from a clinical case (multibacillary) of a paratuberculosis-infected goat. Animals of the control group were inoculated with 5 ml of sterile phosphate buffer saline (PBS) (pH 7.4) on the same occasions. In the infected group, one animal each was euthanatized by intravenous administration of Pentobarbitone (Pfizer) at 15, 30, 60, 90, 120, 150, 180, 210, 240, and 270 days postinoculation (DPI). In the control group, only five animals were euthanatized, one each at 60, 120, 180, 240, and 270 DPI. All the experimental procedures on the animals were carried out in accordance with recommendations of the Institute Animal Ethics Committee and the Committee for the Purpose of Control and Supervision of Experiments on Animals. The DPI was counted after the last dose of inoculation. All animals in both groups were regularly observed for clinical signs. Fecal smears were prepared at monthly intervals and stained with the Ziehl Neelsen (ZN) staining method.
Preparation of inoculum and counting of bacteria
The intestinal segments from the midjejunum to the terminal ileum were collected from a multibacillary goat (a clinically advanced case) during necropsy and contents were flushed out using tap water, and finally washed in sterile distilled water. Large clusters of acid-fast bacilli were detected by ZN staining of smears from different locations of the segments. The mesentery and serosal tissue fat were removed as much as possible and intestinal segments were chopped into small pieces using sterile scissors and finally were minced in a sterile blender with a small amount of PBS containing penicillin (20,0000 IU/liter) (Himedia India Limited, Mumbai, India) and chloramphenicol (200 mg/liter) (Pfizer India Limited). The bacterial suspension was filtered through the sterile gauge to remove the debris and finally stored in sterile containers at −20°C. Five milliliters of the same filtered content was taken for counting the bacteria. The acid-fast bacilli (AFB) in the suspension were counted as described previously 21 and numbers adjusted to 2 × 109/ml. Five milliliters of the same was used to inoculate animals in the infected group. A portion of the same inoculum was tested by mycobactin J–dependent bacterial culture and the presence of IS900 sequence by PCR assay. 17
Bacteriological examination
Bacterial culture from feces was performed as described by Rajukumar et al. 32 The samples were decontaminated with hexadecyl pyridinium chloride (final concentration 0.75%) and inoculated onto four slants of Herrold egg yolk medium (HEYM, pH 7.1–7.4), two each with and without mycobactin J (Allied Monitor Inc., Fayette, Missouri, USA). Colonies appearing after 4 weeks in mycobactin J–supplemented tubes were considered positive. Mycobactin dependency was retested by subculturing the primary isolate on HEYM to confirm the isolates. 43 For culture of tissue samples, 2.5 g of infected tissue was collected in 15 ml of sterile distilled water, triturated, and allowed to settle for 30 minutes at room temperature. Five milliliters of supernatant was transferred in another 50-ml centrifuge tube and processed as described for the fecal sample.
Isolation of peripheral blood lymphocytes
The peripheral blood lymphocytes (PBLs) were prepared from each animal at 0, 60, 120, 180, 240, and 270 DPI as described previously. 4 Briefly, 10 ml of blood was collected in a tube containing 2.7% sterile ethylenediamine-tetraacetic acid (EDTA) in PBS (pH 7.4) in a ratio of 20 : 1. Blood was centrifuged at 2,000 rpm for 30 minutes at room temperature. Buffy coat was collected and added with an equal quantity of PBS (pH 7.4, Ca++, Mg++ free) and layered on a double quantity of Histopaque, density 1.077 (Sigma, St. Louis, MO, USA) for centrifugation at 2,500 rpm for 45 minutes. The interface ring was collected, washed three times in sterile PBS, and finally collected in Roswell Park Memorial Institute-1640 (RPMI) growth medium (Sigma) supplemented with 10% newborn calf serum (NBCS) (Sigma). The cell viability was found to be over 90% by Trypan blue–exclusion method.
Isolation of intraepithelial and lamina propria lymphocytes
The isolation of intraepithelials (IELs) and lamina propria lymphocytes (LPLs) was performed from five infected and five control goats as described previously 27 with slight modifications. Briefly, a 12-cm piece each from the distal ileum and midjejunum was collected from freshly euthanatized animals in ice-cold citrate buffer saline (CBS, pH 7.2). The blood vessels, mesentery, and fat were gently removed and intestinal segments were washed with sterile distilled water on a magnetic stirrer. Small pieces of 0.5 mm were cut and suspended in CBS containing 2 mM EDTA and 10 mM He-pes buffer (Sigma) and gently stirred for 1 hour at 37 C. The supernatant was collected into a fresh tube and the EDTA extraction was repeated twice. The supernatants were pooled and kept for 30 minutes at room temperature, passed through a sterile nylon mesh, and washed three times with RPMI. The remaining tissue fragments were incubated with RPMI-NBCS containing 45 U/ml collagenase and 50 U/ml hyaluronidase (Sigma) and gently stirred at 37°C for 4–5 hours. The supernatant was kept for 30 minutes at room temperature, passed through a sterile nylon mesh, and washed three times with RPMI.
Using a discontinous Percoll gradient, lymphocytes were purified from the clear supernatant. The suspension was mixed with 5 ml Percoll (density 1.055 g/ml) (Pharmacia P-L Biochemicals, Milwaukee, WI, USA) and layered on 3.5 ml ice-cold Percoll (density 1.079 g/ml), followed by layering of 3 ml RPMI on the top. The gradient was centrifuged at 650 × g for 20 minutes at 4°C and cell bands at the interface were collected and washed three times with RPMI. The cell viability was found to be approximately 80% both for IELs and LPLs by Trypan blue–exclusion method.
Isolation of lymphocytes from mesenteric lymph node, jejunum Peyer patch, and ileum Peyer patch
The mesenteric lymph node (MLN), jejunum Peyer's patch (JPP), and ileum Peyer's patch (IPP) were collected in sterile RPMI 1640. The fat, mesentery, and blood vessels were gently removed and cut into small pieces. The suspension was collected in a tube and kept for 30 minutes at room temperature. The supernatant was passed through a sterile nylon mesh and washed three times with RPMI. Lymphocytes were purified using a discontinuous Percoll gradient as described above. The cell viability as determined by Trypan blue–exclusion method was found to be 85, 80, and 80% for MLN, JPP, and IPP, respectively.
Lymphocyte proliferation test
Lymphocyte proliferation test (LPT) was performed on both peripheral blood lymphocytes and gut-associated lymphocytes, by MTT [3-(4,5 dimethylthiazole 2–4) 2,5-diphenyl tetrazolium bromide] dye (Sigma) as described previously. 21, 26 Briefly, 100 μl cell suspension (2 × 106 viable cells/ml) was incubated with johnin PPD (final concentration 10 μg/ml) 37 (Biological Product Division, Indian Veterinary Research Institute, Izatnagar, India) and Con A (Sigma) (final concentration 10 μg/ml) 37 in a 96-well, flat-bottom tissue culture plate (NUNC, Roskilde, Denmark) at 37 C having 5% CO2 concentration in a humidified atmosphere for 96 hours. Ten microliters of MTT (5 μg/ml) was added to each well and the plates were further incubated for 4 hours. After incubation, 100 μl of dimethyl sulfoxide was added to each well, mixed by vigorous pipetting, and incubated for 15 minutes at 37°C. The optical densities were measured at 492 nm with reference reduction of background color at 650 nm in the ELISA reader (Microscan, ECIL, Hyderabad, India). The stimulation indices (SI) were calculated as ratio of optical density of stimulated cultures to optical density of unstimulated cultures. The diagnostic cut-off value of LPT was taken as 33% above the mean values of control goats.
PCR assay
Feces from all goats collected on the day of euthanasia were subjected to DNA extraction using Invisorb stool DNA kit (Invitek GmbH, Berlin, Germany) as per manufacturer's instructions. Distal ileum, distal jejunum, and mesenteric lymph nodes were pooled and triturated, and DNA was extracted by using Tissue DNA kit (Peqlab Biotechnology GmbH, Erlangen, Germany) as per manufacturer's instructions.
The IS900 primer sequences: forward (MK 05), TTC TTG AAG GGT GTT CGG GGC C; and reverse (MK 06), GCG ATG ATC GCA GCG TCT TTG G, flanking a region of 518 bp was used for the PCR assay. 17 Five milliliters of DNA template and 45 μl of master mix (1.5 mM MgCl2, 1× PCR buffer [Invitrogen], 0.2 mM concentration of each deoxynucleotide triphosphate, 0.5 μM concentration of each primer, and Taq DNA polymerase [0.02 U/μl] [Invitrogen]) were added to each tube. After an initial denaturation step (94°C, 4 minutes), 40 PCR cycles (94°C, 30 seconds; 64°C, 30 seconds; and 72°C, 30 seconds) were performed, followed by a final extension step (72°C, 5 minutes). The PCR products were analyzed on a 1.5% agarose gel containing ethidium bromide (0.2 μg/ml). Positive and negative controls were always included.
Detection of humoral immune response
Serum was collected from the animals before infection and at monthly intervals thereafter until the termination of the experiment. Agar gel immunodiffusion (AGID) test and indirect ELISA were performed using sonicated antigen to estimate the antibody titer. Sonicated antigen was prepared from M. a. paratuberculosis (316 F strain) as described previously. 32 AGID was performed in 1% agarose gel prepared in NaCl solution (0.15 M) on the microslides. The presence of a precipitation line was considered as a positive result. Positive and negative controls were included in each slide.
ELISA was carried out after Rajukumar et al. 32 Briefly, 96-well, flat-bottom plates (Maxisorp, Nunc, Roskilde, Denmark) were coated with antigen at a concentration of 20 μg/ml. A serum dilution of 1 : 100 was used in duplicates and incubated at 37°C for 2 hours. Anti-goat IgG peroxidase conjugate (Sigma) at a dilution of 1 : 25,000 was added to all wells and incubated for 1 hour at 37°C. Substrate containing orthophenylene diamine and buffered urea H2O2 (Sigma) was added to all wells and incubated for 30 minutes at 37°C. Reaction was stopped by 50 μl of 1 M HCl and absorbance was read at 492 nm in an ELISA reader (Microscan). Positive and negative sera and antigen and antibody controls were always included in each plate.
Pathology
The euthanatized animals were subjected to thorough necropsy examination. Tissues for histological examination were collected from the ileocecal valve (ICV); distal, middle, and proximal portions of the ileum; four portions 5 feet apart from proximal to distal region of the jejunum; mesenteric lymph node (MLN), ileocecal lymph node (ICLN), cecum, colon, rectum, liver, kidneys, lungs, heart, tonsils, and aorta. The tissues were processed conventionally and embedded in paraffin, and sections were cut at 5 microns. All sections were stained with hematoxylin and eosin (HE) and ZN methods. Small portions (2–3 cm) from distal ileum, midjejunum, MLN, and ICLN adjacent to those collected for histology examination were also collected separately in sterile vials for bacterial culture and genomic detection of M. a. paratuberculosis by PCR.
Statistical analysis
The data were compared with unpaired t-test at 95% confidence level using GraphPad QuickCalcs. P values less than 0.05 were considered significant. Significance of stimulation responses in peripheral blood lymphocytes of each infected goat was calculated against average response of control goats at respective DPI.
Results
Clinical signs
Distinct clinical signs were not observed in any of the infected goats until 150 DPI. Four goats, Nos. 7, 8, 9, and 10, started showing variable degrees of loss of appetite, reduction in body weight, and occasional loose feces. All the control goats were healthy throughout the experiment. Fecal-smear examination detected AFB in one goat, No. 7, at 150 DPI; two goats, Nos. 8 and 10, at 180 DPI; and one goat, No. 8, at 210 DPI.
Bacteriology and PCR analysis
The results of bacteriological examination and PCR assay in feces and tissues are presented in Table 1. One goat, No. 8, was detected positive by culture and PCR in both fecal and tissue samples. One goat, No. 3, was positive only by tissue PCR.
Results of different diagnostic tests in M. a. paratuberculosis-infected goats.∗
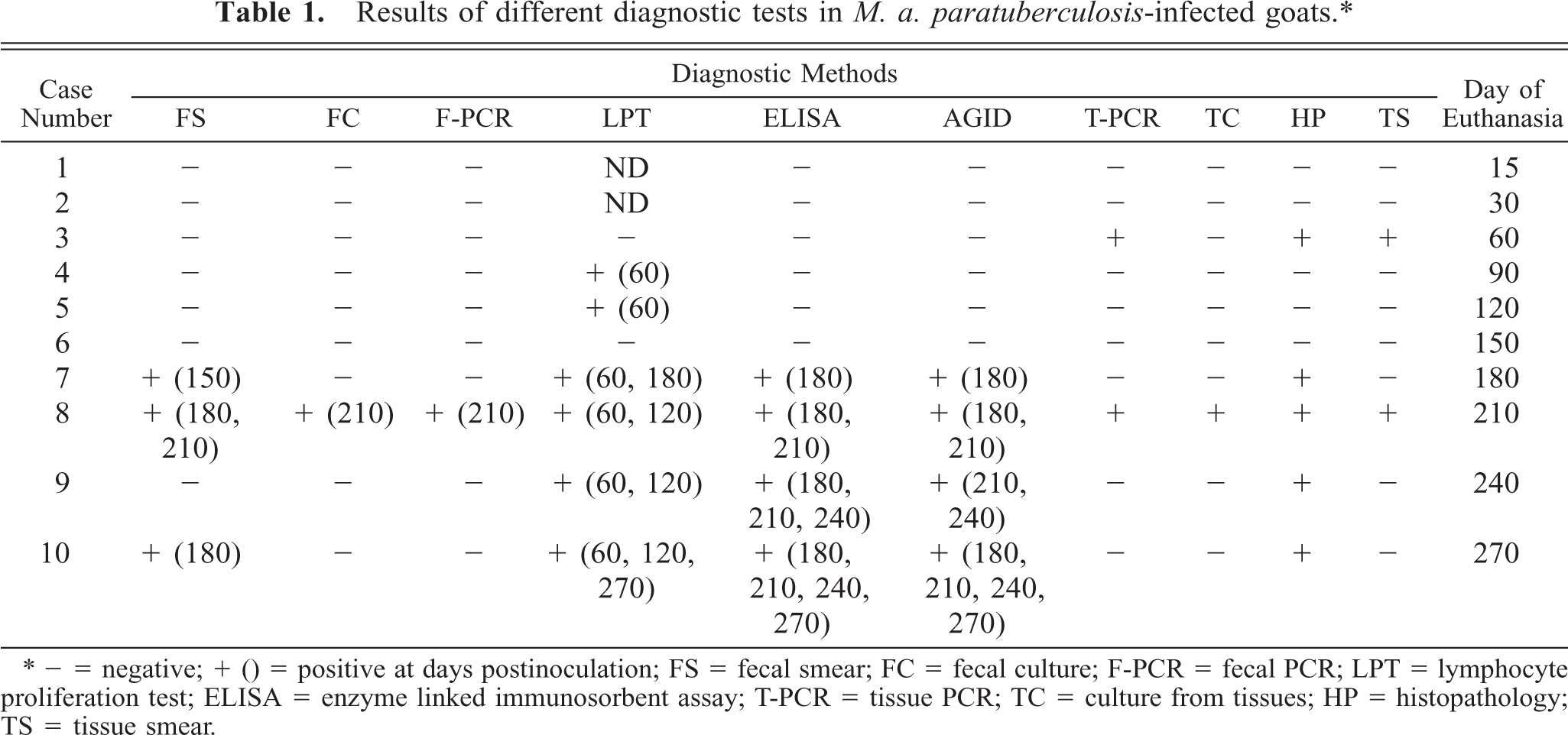
∗ − = negative; + () = positive at days postinoculation; FS = fecal smear; FC = fecal culture; F-PCR = fecal PCR; LPT = lymphocyte proliferation test; ELISA = enzyme linked immunosorbent assay; T-PCR = tissue PCR; TC = culture from tissues; HP = histopathology; TS = tissue smear.
Cellular and humoral-mediated immune responses
Stimulation indices with Con A in the peripheral and gut lymphocytes were found to vary at different stages, but no difference was observed between infected and control groups (data not shown). Lympho-proliferative responses after in vitro stimulation with johnin PPD in peripheral blood of infected and control goats at different intervals postinfection (PI) are shown in Fig. 1. Significant increases in proliferative responses were observed in four goats at 60 and 120 DPI, which decreased to one goat at 180 and 270 DPI. Proliferative responses after in vitro stimulation with johnin PPD in different compartments of gut lymphocytes of the infected and control goats at different intervals PI are shown in Figs. 2–6. At 60 DPI, jejunum IELs and lymphocytes of MLN of goat No. 3 had significant proliferation against its control (goat No. 17) (Fig. 2). At 120 DPI, lymphocytes of jejunum lamina propria (LP), jejunum Peyer patches (PP), ileum PP, and MLN of goat No. 5 had significant proliferative responses against the control (goat No. 12) (Fig. 3). At 180 DPI, lymphocytes from two compartments of GALT (jejunum IELs and jejunum PP) of goat No. 7 had significant proliferative responses against the control (goat No. 15) (Fig. 4). Lymphocytes of two goats, Nos. 9 and 10, at 240 and 270 DPI had significantly increased proliferation in three compartments (jejunum IELs, jejunum LP, jejunum PP) and four compartments (jejunum IELs, ileum LP, jejunum LP, jejunum PP) of GALT, respectively (Figs. 5, 6). The AGID detected precipitating antibodies in three goats at 180 DPI, whereas ELISA detected four goats positive at 180 DPI. At 210, 240, and 270 DPI, both methods detected three, two, and one of the goats positive, respectively (Table 1).
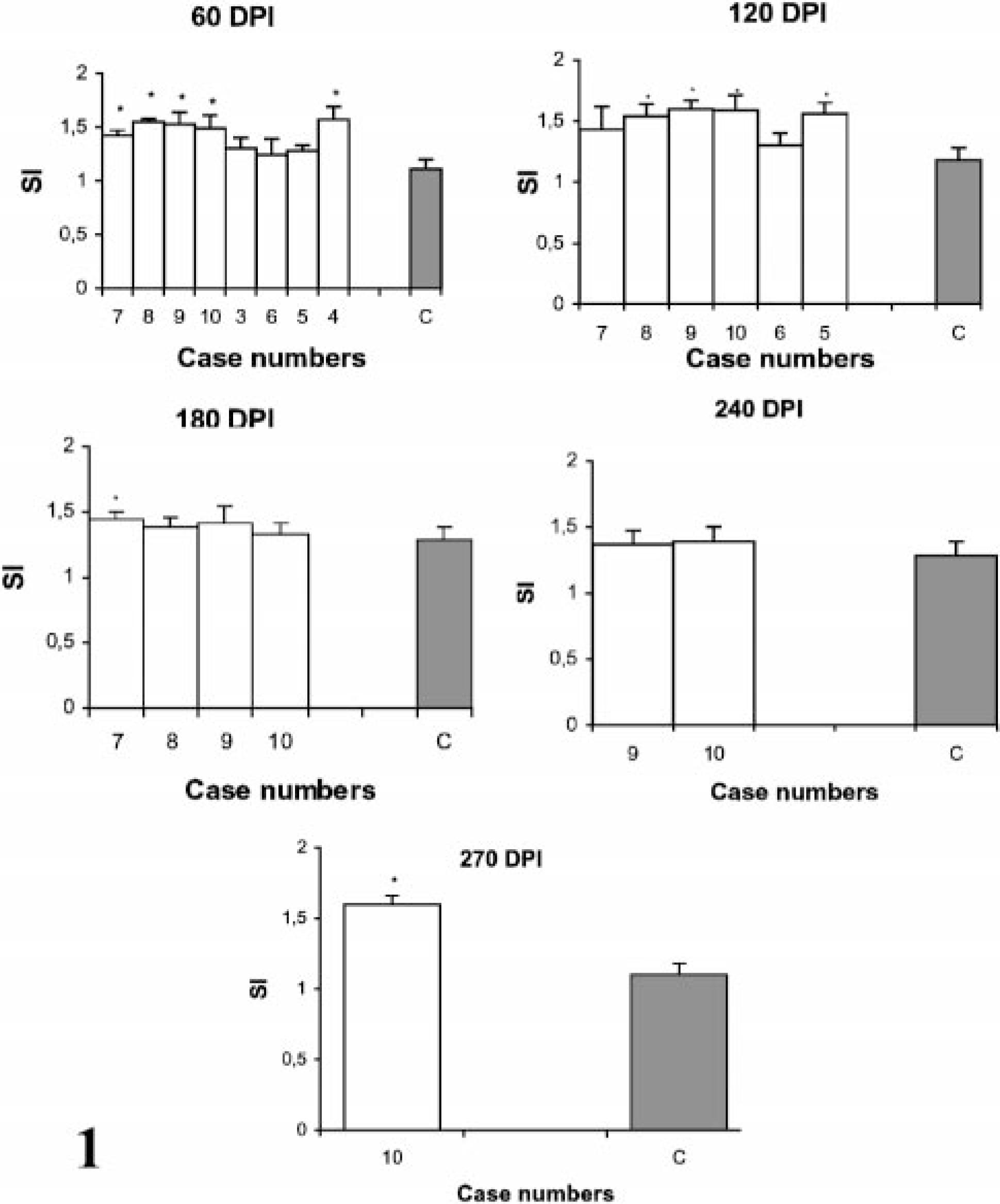
Lymphocyte proliferation responses (SI) after in vitro stimulation with johnin PPD in peripheral blood lymphocytes of paratuberculosis-infected and control goats at 60, 120, 180, 240, and 270 DPI. C = Average of all control; SI = stimulation indices; ∗ = significantly different from control (P < 0.05).
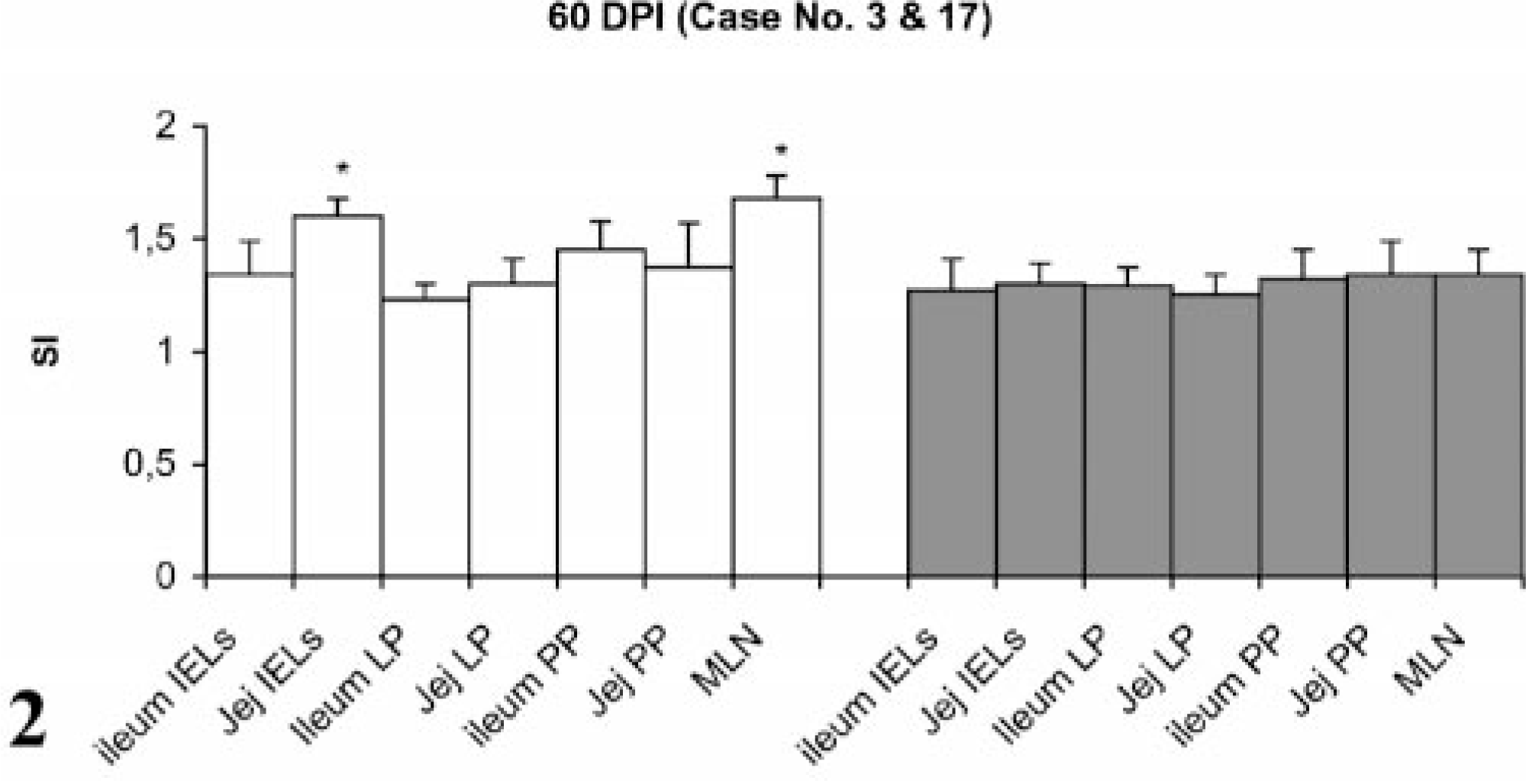
Lymphocyte proliferation responses (SI) after in vitro stimulation with johnin PPD of lymphocytes from different compartments of gut in paratuberculosis-infected and control goats at 60 DPI. SI = stimulation indices; IELs = intestinal epithelial lymphocytes; LP = lamina propria; PP = Peyer patches; MLN = mesenteric lymph nodes; Jej = Jejunum; PI = postinfection; ∗ = significantly different from control (P < 0.05).
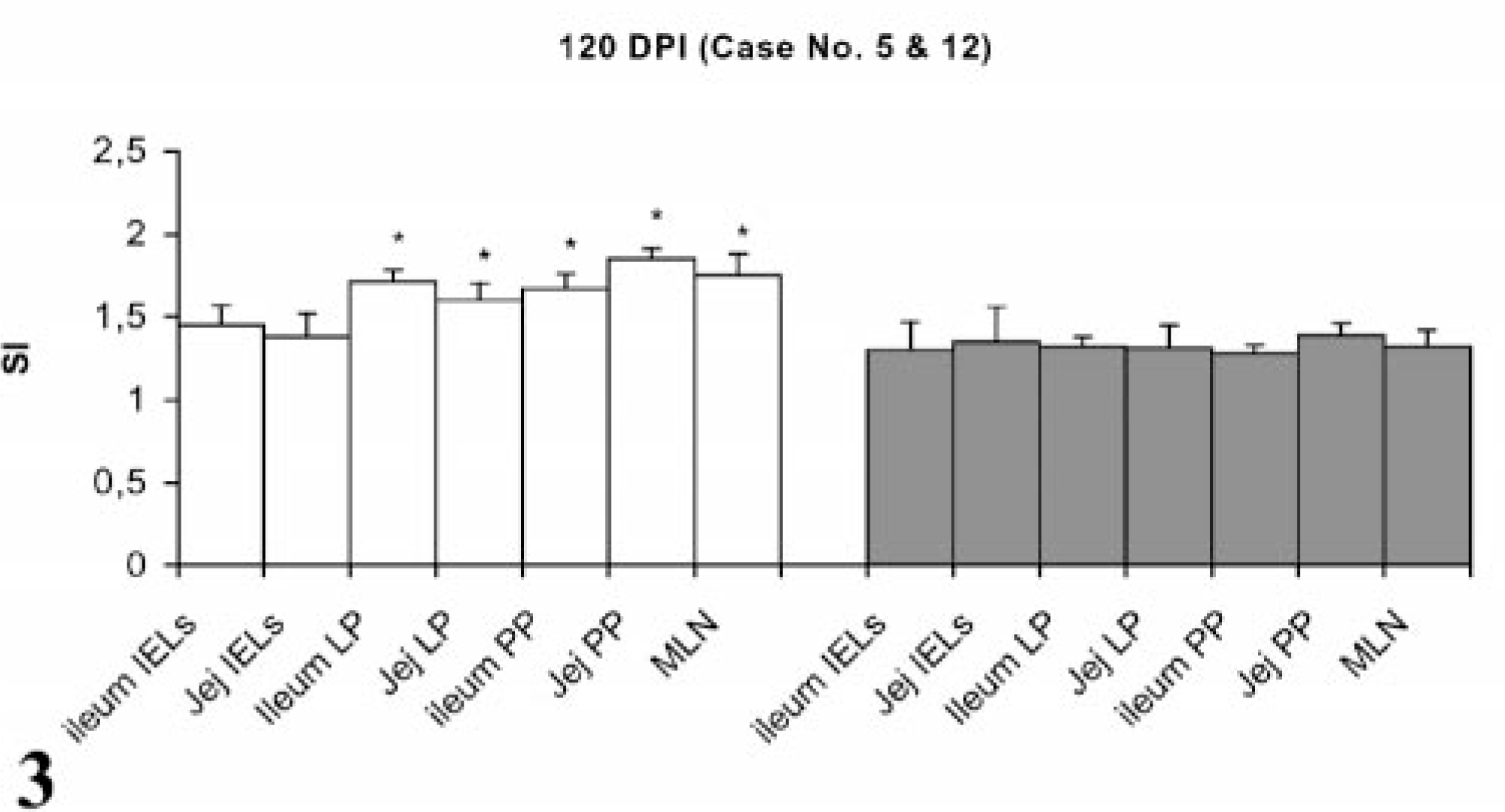
Lymphocyte proliferation responses (SI) after in vitro stimulation with johnin PPD of lymphocytes from different compartments of gut in paratuberculosis-infected and control goats at 120 DPI. SI = stimulation indices; IELs = intestinal epithelial lymphocytes; LP = lamina propria; PP = Peyer patches; MLN = mesenteric lymph nodes; Jej = Jejunum; PI = postinfection; ∗ = significantly different from control (P < 0.05).
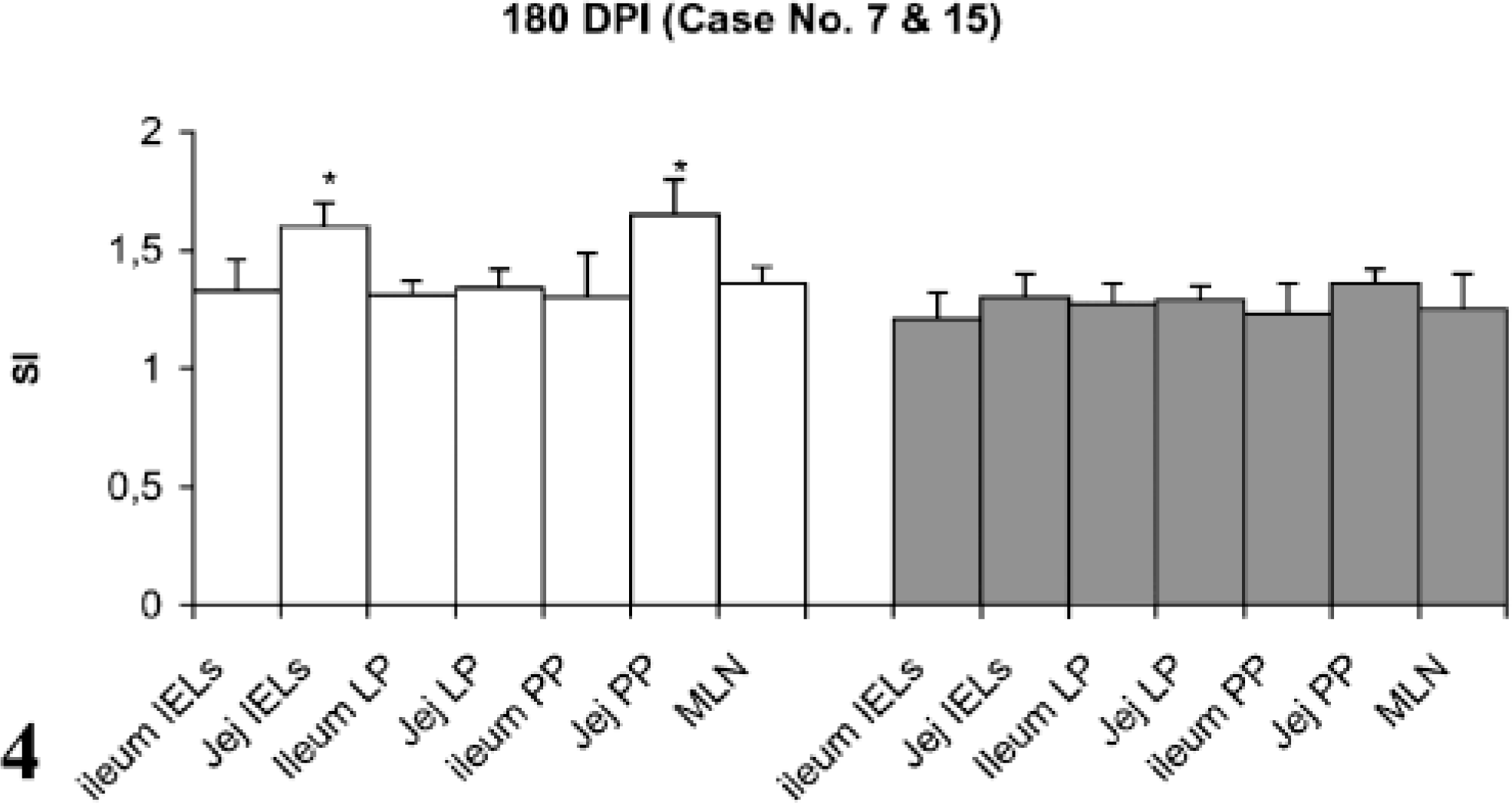
Lymphocyte proliferation responses (SI) after in vitro stimulation with johnin PPD of lymphocytes from different compartments of gut in paratuberculosis-infected and control goats at 180 DPI. SI = stimulation indices; IELs = intestinal epithelial lymphocytes; LP = lamina propria; PP = Peyer patches; MLN = mesenteric lymph nodes; Jej = Jejunum; PI = postinfection; ∗ = significantly different from control (P < 0.05).
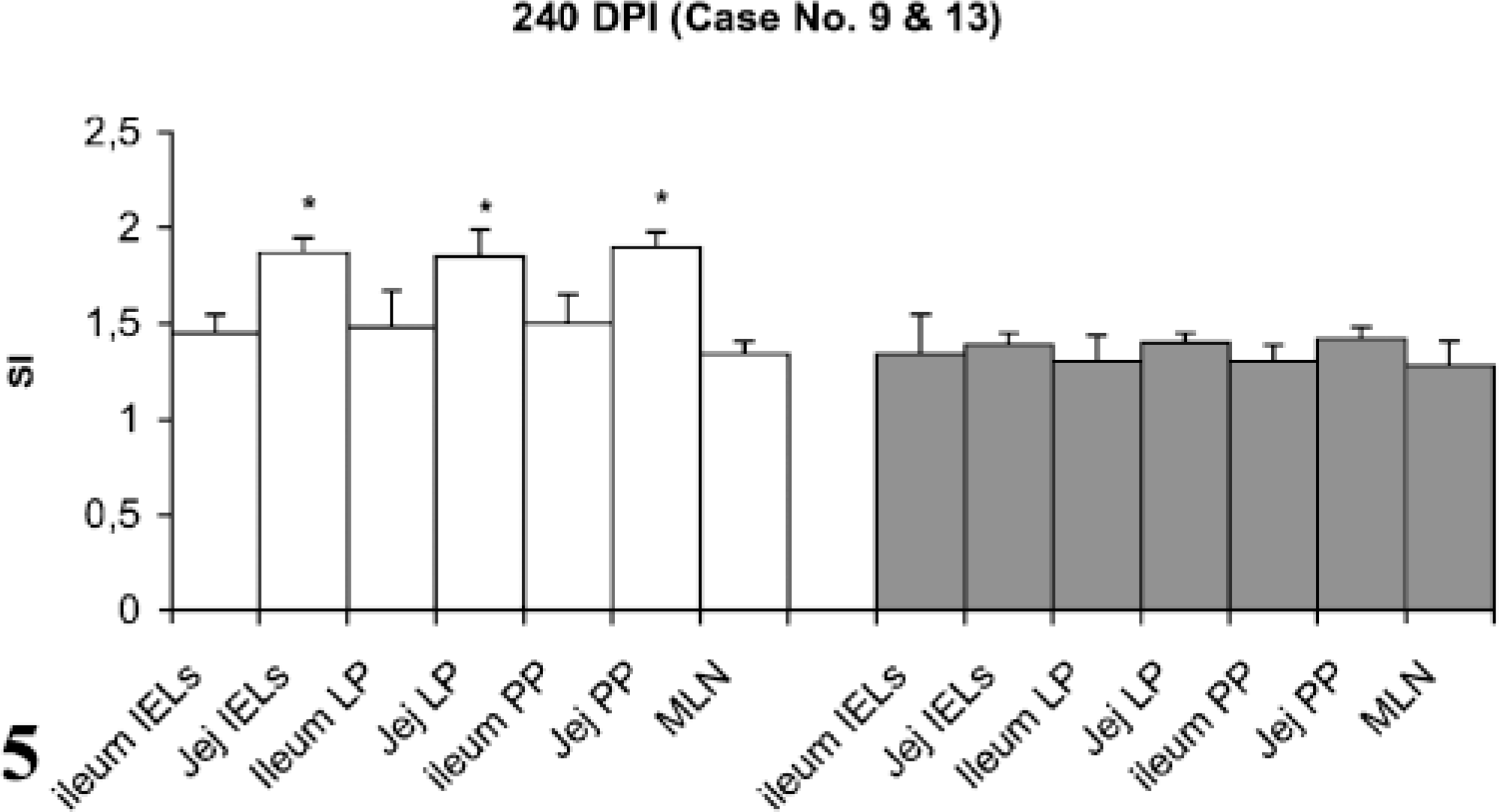
Lymphocyte proliferation responses (SI) after in vitro stimulation with johnin PPD of lymphocytes from different compartments of gut in paratuberculosis-infected and control goats at 240 DPI. SI = stimulation indices; IELs = intestinal epithelial lymphocytes; LP = lamina propria; PP = Peyer patches; MLN = mesenteric lymph nodes; Jej = Jejunum; PI = postinfection; ∗ = significantly different from control (P < 0.05).
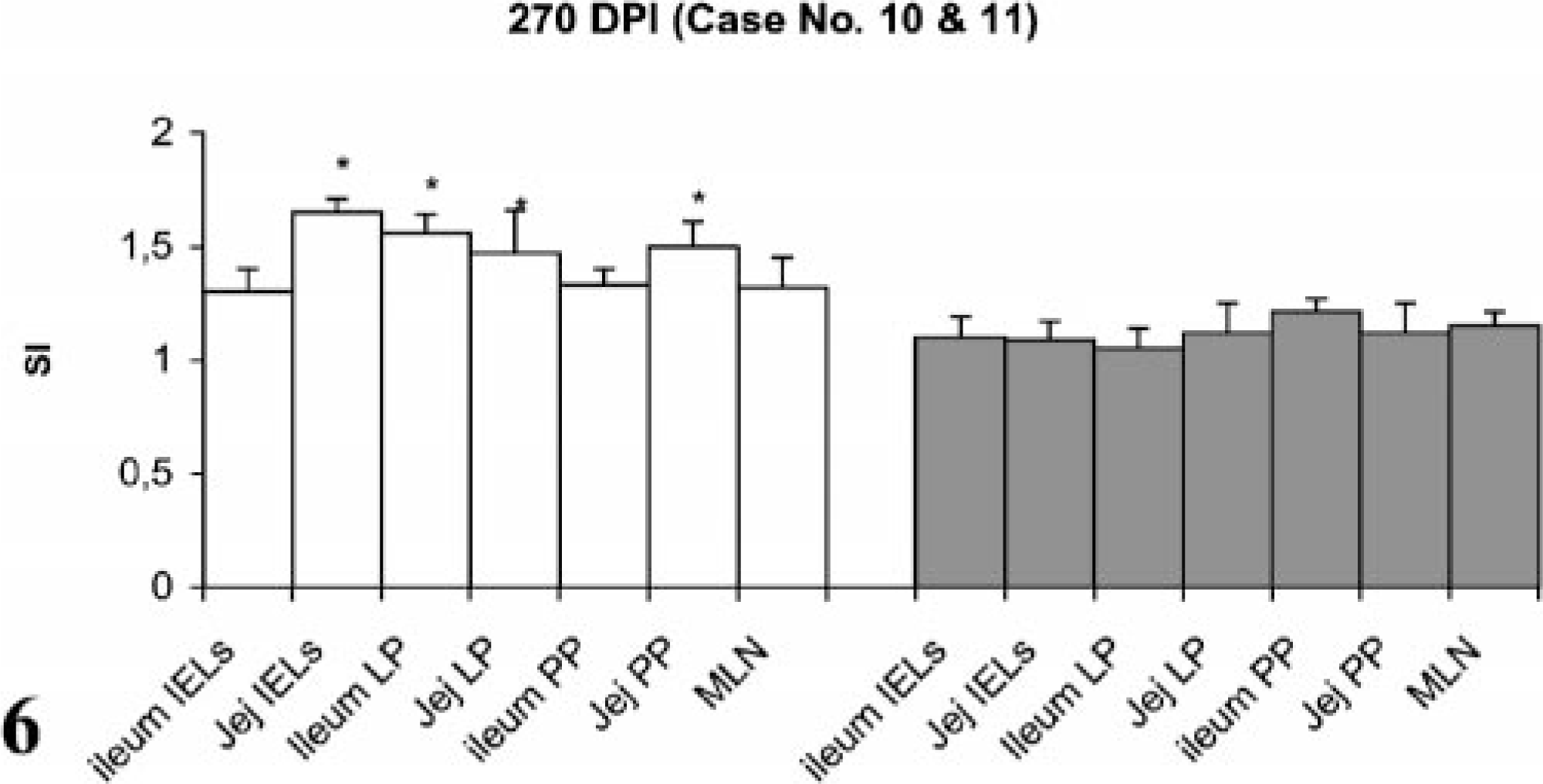
Lymphocyte proliferation responses (SI) after in vitro stimulation with johnin PPD of lymphocytes from different compartments of gut in paratuberculosis-infected and control goats at 270 DPI. SI = stimulation indices; IELs = intestinal epithelial lymphocytes; LP = lamina propria; PP = Peyer patches; MLN = mesenteric lymph nodes; Jej = Jejunum; PI = postinfection; ∗ = significantly different from control (P < 0.05).
Tissue studies
Grossly, no changes were observed in control and infected goats euthanatized at 15 and 30 DPI. Goats euthanatized at 60, 90, 120, 150, 240, and 270 DPI had a slight thickening of ileal mucosa extending anteriorly to the distal jejunum and a slight enlargement of ICLN. The lesions in goats euthanatized at 180 and 210 DPI consisted of corrugated mucosa of the ileum and jejunum and slight enlargement of MLN and ICLN. Depletion of fat deposits and lymphatic cording were observed in a goat euthanatized at 210 DPI.
Microscopically, none of the control and infected goats euthanatized at 15 and 30 DPI exhibited any lesions. The goat euthanatized at 60 DPI exhibited an increase in thickness of the intestinal villi and their tips appeared flat and wide. The LP and PP revealed loose aggregates of epithelioid macrophages with many AFB in the jejunum (Fig. 7) and ileum. The villous and crypt areas of LP were infiltrated with mononuclear cells consisting of aggregates of epithelioid macrophages and lymphocytes (Fig. 8). The lesions in goats euthanatized at 90, 120, 150, 180, 240, and 270 DPI consisted of scattered aggregates of epithelioid macrophages in the interfollicular areas of the ileum and jejunum. In addition, villi were broader, more blunt, and thickened and infiltrated with lymphocytes and epithelioid macrophages without demonstration of acid-fast bacilli. The ileocecal and mesenteric lymph nodes revealed infiltration with epithelioid macrophages and lymphocytes in the interfollicular areas of cortex and paracortex. Multinucleated giant cells (MNGC) along with heavy infiltration of epithelioid macrophages were observed in the PP area of the ileum and jejunum in goats euthanatized at 180 DPI. MNGC were also observed in the cortical area of the MLN of a goat euthanatized at 240 DPI (Fig. 9), with few granulomatous foci.
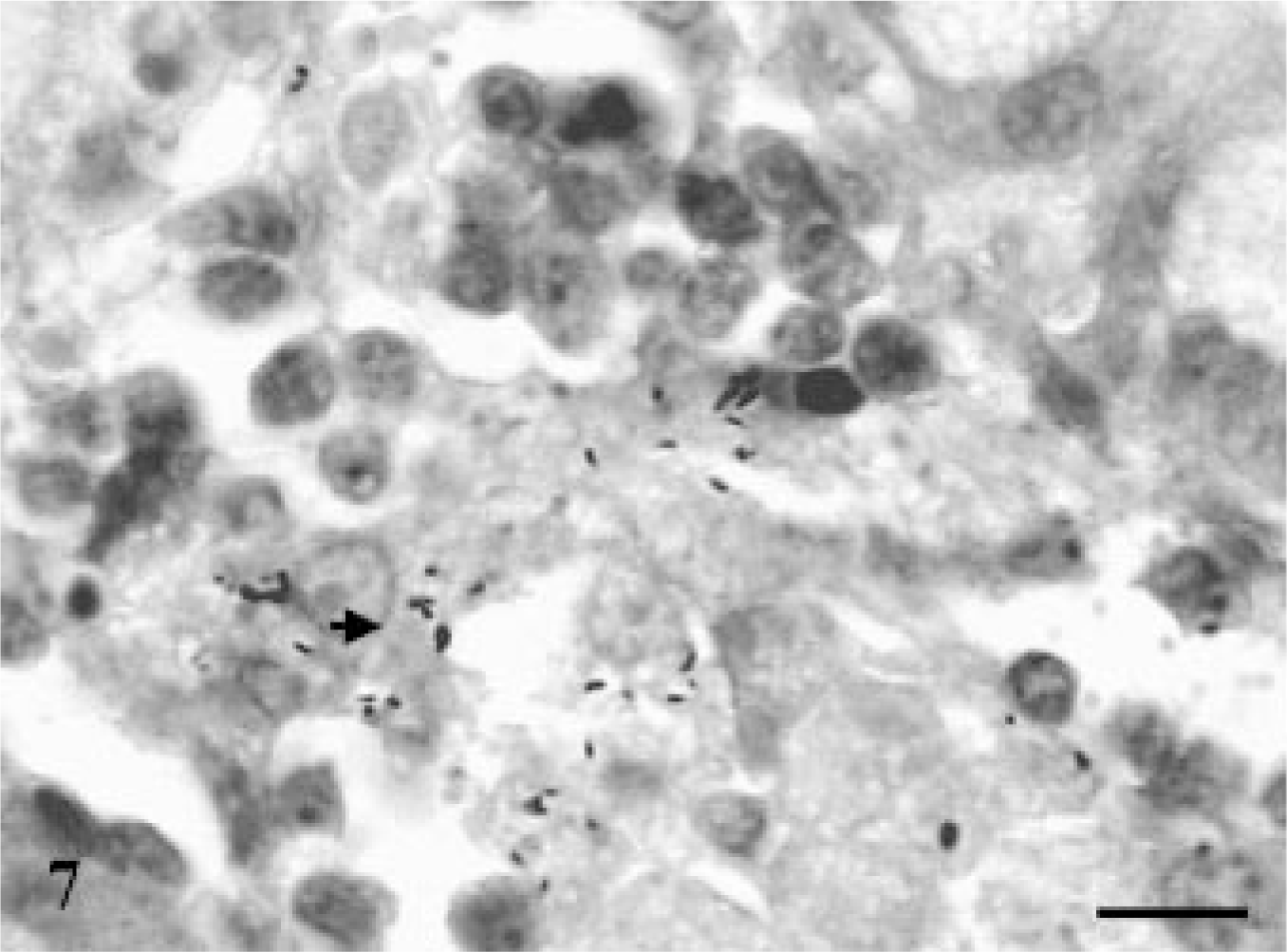
Jejunum; goat No. 3. Presence of acid-fast bacilli in the lamina propria of jejunum at 60 DPI. ZN. Bar = 100 μm.
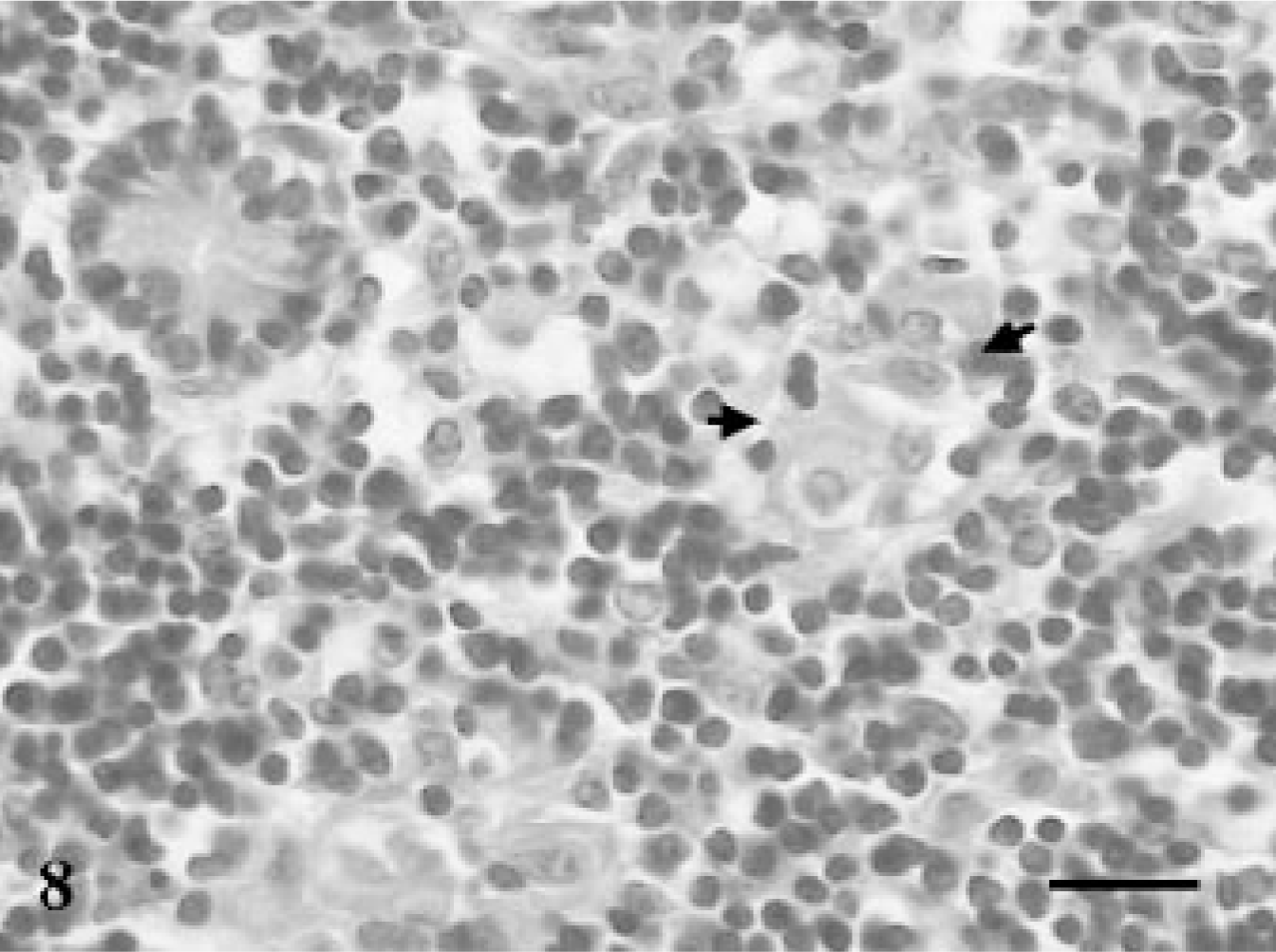
Jejunum; goat No. 8. Small aggregates of epithelioid macrophages among large number of lymphocytes in the crypt areas at 60 DPI. HE. Bar = 100 μm.
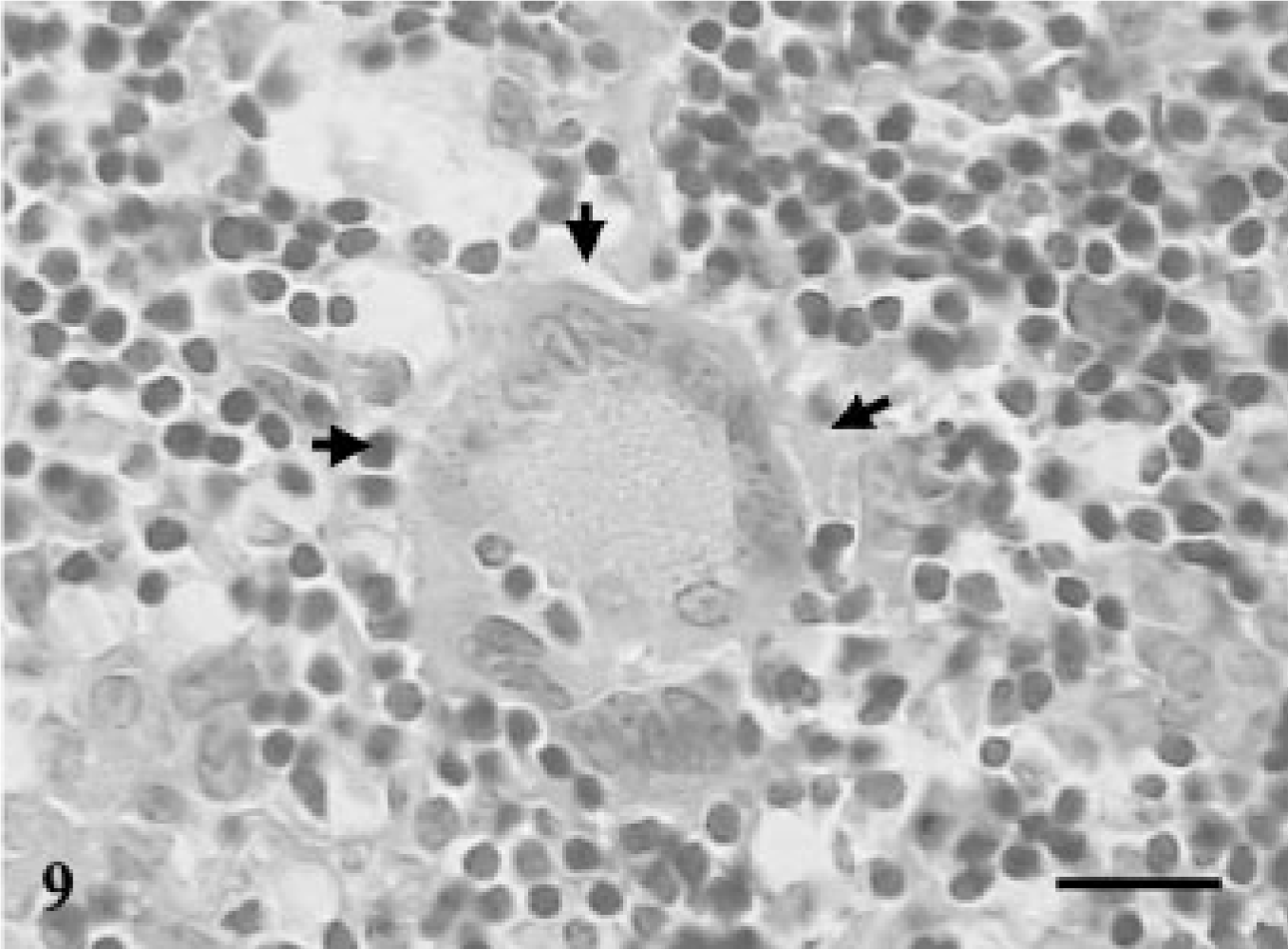
Mesenteric lymph node; goat No. 9. Presence of multinucleated giant cells in the section of the cortical areas at 240 DPI. HE. Bar = 100 μm.
Goats euthanatized at 210 DPI contained heavy infiltration with epithelioid macrophages in PP as well as non–Peyer patch areas of the ileum and jejunum. Few acid-fast bacilli were observed in the epithelioid macrophages in the lamina proprial region of the jejunum. The submucosal region contained lymphocyte and plasma cell infiltrates. A few lymphocytes and macrophages were also observed around lymphatics.
Discussion
This study demonstrates successful production of experimental paratuberculosis infection in Indian goats. The dose rate of 1010 mycobacteria on seven occasions is similar to a natural infection, where healthy goat kids remain in the company of multibaccillary adult goats shedding up to 5 × 1012 mycobacteria per day. 7 The clinical signs were not characteristic of paratuberculosis, as diarrhea was not observed in any of the goats. It is difficult to identify early signs of caprine paratuberculosis since animals generally remain bright and alert with good appetite. As the disease progresses, weight loss becomes evident. 8
Cell-mediated immunity (CMI) has inhibitory effects on the survival of intracellular bacteria and is considered to be the principal mechanism for clearing infection. 5, 15, 31 Significant proliferative responses in PBLs of four infected goats were observed at an early stage of 60 and 120 DPI, which decreased to one goat at 180 DPI, suggesting a reduction in CMI with the progression of infection that led to the proliferation of mycobacteria. Detectable and significant proliferative responses with johnin PPD in PBLs have been reported in goats from 9 weeks onwards in subclinical paratuberculosis following experimental infection. 37
In the present study, proliferative responses in gut were mainly observed in compartments of the jejunum as compared with the ileum. At 60 DPI, goat No. 3 showed significant proliferation in two compartments of gut (jejunum IELs and MLN), but no proliferation was observed in the peripheral blood. The same goat showed heavy infiltration of epithelioid macrophages laden with AFB in LP and PP areas of the small intestine. At 120 DPI, goat No. 5 had significant lymphocyte proliferation in four compartments of GALT and in PBLs, with few histological lesions, without demonstration of AFB. At 180 DPI, significant increases in SI of lymphocytes from jejunum and PBLs of goat No. 7 were observed. The presence of MNGC without demonstration of AFB was observed in a few goats, a characteristic feature of tuberculoid paratuberculosis. 9 Tuberculoid pathology with strong CMI has been reported in sheep and goat paratuberculosis. 9, 28 This shows that the CD4+ lymphocytes of IPP, JPP, and MLN might have acted strongly against the mycobacteria and were able to remove them from the gut of two goats, Nos. 5 and 7. CD4+ T lymphocytes are mainly responsible for protection against M. a. paratuberculosis. 23, 28 JPP has large interfollicular areas with a larger population of CD4+ lymphocytes, 22 which could produce a Th 1–type response. 23 This activates macrophages and results in efficient intracellular killing of the organisms. 26
Recruitment of macrophages at the site of paratuberculosis infection is dependent on various factors, including IL-1α and TNF. 1 Deficiency of these factors in goat No. 3 might have downregulated the recruitment and intracellular killing ability of macrophages, resulting in survival of AFB in the tissues.
At 210 days PI, goat No. 8 had severe lesions with the presence of a few AFB, but lymphocyte proliferative experiments in gut could not be performed in this goat. At 240 and 270 DPI, jejunal lymphocytes of goats (Case Nos. 9, 10) showed significant proliferative responses. Absence of AFB at histology could afford a similar explanation as for the above goats, Nos. 5 and 7. When results of LPT were compared between gut and PBLs, it was found that proliferative responses in lymphocytes of jejunal compartments were correlated well with PBL responses as compared with ileal and MLN lymphocytes, where only one or two of five infected goats had significant proliferation. A similar study is not available in goats for comparison. Based on the results of this study and of Burrells et al. 5 in sheep, reporting positive correlation between MLN and gut lymphocytes with PBLs, it is plausible to speculate that proliferative responses, especially of the jejunal compartments, may be reflected peripherally in the blood. This is most likely because of the morphology, phenotypic composition, and functional differences of the ileum and jejunum PP. The jejunal PP have large T cell–rich interfollicular areas with more mature T cells, especially the CD4+ subpopulation, whereas the ileal PP has densely packed lymphoid follicles with scant interfollicular T-cell areas containing small numbers of CD4+ T cells. 12, 22 CD4+ T cells are mainly responsible for production of Th 1 responses, high proliferative responses, and protection against M. a. paratuberculosis. 23, 28 For this reason, proliferative responses in lymphocytes of jejunum had correlated well with proliferative responses in peripheral blood.
However, the individual variation with regard to host–pathogen interaction and the use of one animal per time point in the present study warrants further investigation to establish the validity of our results. Similar correlation would have been expected between lymphoid tissue of ICV and peripheral blood because of functional and phenotypic similarities between lymphoid tissue of ICV and jejunal PP. 12, 40 But the proliferative responses of lymphocytes of ICV were not performed in the present study.
The histological lesions were mainly dominant in the lymphoid tissues of ICV, ileum, terminal jejunum, and the draining lymph nodes. Association of lesions to gut-associated lymphoid tissue has been reported previously in subclinical and early stages of paratuberculosis in goat 40 and sheep. 30 The lesions consisted of loose aggregates of macrophages in different portions of the ileum and jejunum; however, no clear granuloma was observed in any sections of the ileum or jejunum. A similar observation has been previously reported during the early subclinical phase of the experimental infection of goat kids with M. a. paratuberculosis. 35 In another study in goats that terminated 2 years after experimental infection, lesions were mainly associated with the T cell–rich interfollicular areas of the organized lymphoid tissue of midjejunum, distal ileum, and ICV. 40 Other investigators have reported lesions in sheep as early as 30–45 DPI, consisting of epithelioid cell aggregates and microgranulomas in the lymphoid follicles of the jejunum and ileum. 3, 19, 20, 21 In most experimental studies of goats, investigators have not examined animals until several months to over a year after infection. 18, 37, 40, 41 Our study indicates a difference in the pattern of development of lesions in sheep and goats in early stages of paratuberculosis infection.
Diagnosis of paratuberculosis in the subclinical stage is still considered a major problem. Three goats euthanatized at 30, 90, and 120 DPI, respectively, without detectable histological lesions did not react in any of the tests used in the present investigation. This could possibly be due to high resistance of animals eliminating infection soon after the entry of mycobacteria, as reported previously. 30, 16 Genetic resistance has been reported in murine studies. 13, 38 Fecal smear, though nonspecific, detected more goats as positive than bacterial culture and PCR. The PCR test was found to be better than bacterial culture and detected the IS900 gene in the fecal sample of one goat and tissue samples of two goats. Organisms may be present in the feces in insufficient numbers to be detected by current culture methods. 42 Isolation of bacteria from a smaller number of animals may also be due to difficulty in cultivation of ovine and caprine strains. 6, 10 The low sensitivity of the fecal culture method is well known. 7, 11, 44 Even then, it is still considered as a gold standard and is used for comparison assessment of the efficacy of other diagnostic tests, including PCR, ELISA, and AGID. PCR has been widely accepted as a rapid test with high specificity and sensitivity for the diagnosis of paratuberculosis. 36, 42
Animals in the early stages of infection often do not elicit an immune response (cell mediated or humoral) detectable by currently available tests. 9, 24 In the present study, LPT on blood samples was found to be a sensitive test that detected animals as early as 60 DPI. LPT has been well recognized for diagnosis of paratuberculosis in early stages by other workers. 21, 25 ELISA and AGID were found to be 100% sensitive from 180 and 210 DPI onward, respectively. The ELISA has been found to be more sensitive than AGID; 11 however, sensitivity depends on the stage of infection and fecal shedding. Histological lesions in paratuberculosis are characteristic, and, if present, confirm the diagnosis. 8, 30 Five goats were detected positive by histological examination, where lesions were primarily limited to the gastrointestinal tract and associated lymph nodes. Two goats were confirmed positive by the presence of a few solitary MNGC. 16, 39 In conclusion, in the early stages of experimental paratuberculosis infection in goats, lesions were dominant in the lymphoid follicles of ICV, ileum, terminal portions of the jejunum, and adjoining lymph nodes without demonstration of clear granuloma. The reflection of lymphoproliferative responses in different compartments of gut, especially the jejunum, was observed in the peripheral blood. The individual variation with regard to host–pathogen interaction and the use of a small number of animals in the present study calls for further confirmation. PCR was found to be a better method in comparison with the gold standard, the bacterial culture for the diagnosis of paratuberculosis in the early stages of infection. ELISA was found to be better than AGID, and it is the cheapest and easiest test available for the diagnosis of paratuberculosis.
Footnotes
Acknowledgements
We are grateful to the Director of the Institute for providing facilities to carry out this work. We thank Shri RamBujh for technical assistance.
