Abstract
Understanding pathogenesis during progressive stages of infection by Mycobacterium avium subsp. paratuberculosis (MAP) and finding suitable methods for its diagnosis are key to the control of Johne's disease in animals. Paratuberculosis was experimentally produced in 20 crossbred lambs by oral administration of MAP to study the sequential development of lesions between 10 and 330 days postinfection and to assess commonly used diagnostic methods such as bacterial culture, lymphocyte stimulation test (LST), and enzyme-linked immunosorbent assay (ELISA) during progressive stages of infection. Histologic lesions were classified into four grades from grade 1 (least severe) to grade 4 (most severe) on the basis of location of granulomatous lesions in different regions and layers of intestines, their association with intestinal lymphoid tissues, pattern and distribution of lesions, types of cellular infiltration, and presence of acid-fast bacilli. It is evident that infection first establishes in lymphoid tissues of the small intestine, possibly at multiple sites, producing segmental lesions and from there spreads to lamina propria and local lymph nodes. Wide variability in the histologic lesions in relation to postinfection periods and initial tropism of MAP to the intestinal lymphoid tissues (Peyer's patches) suggests a differential susceptibility of young animals, possibly because of compositional phenotypic variation of Peyer's patches influencing subsequent course of infection. Histopathology was found to be a better indicator of paratuberculous infection than bacteriology in sheep. The LST (reflecting the cellular immune response) and ELISA (reflecting the humoral immune response) had overall sensitivities of 65% (11 of 17) and 42% (8 of 19), respectively, in sheep with different types of pathology but when employed together could detect about 88% of infected animals.
Keywords
Introduction
Paratuberculosis continues to be a major disease of domestic ruminants and is responsible for considerable economic losses all over the world. The disease is a chronic infectious granulomatous enteritis caused by Mycobacterium avium subsp. paratuberculosis (M. a. paratuberculosis), an acid-fast, slow-growing, and fastidious bacterium requiring mycobactin for its in vitro growth. The disease is clinically characterized by untreatable diarrhea accompanied by progressive weight loss. 8,9,18 The etiologic agent has also been implicated as a possible cause of Crohn's disease in humans. 38
In an endemic flock, the infection is usually initiated soon after birth, but the clinical disease is observed only in adults. The pathogenic mechanisms involved in the progression of the disease during the prolonged incubation period to clinically evident disease are poorly understood. 9 The organism is generally believed to cross from the lumen of the small intestine into the Peyer's patch area via M cells of the follicle-associated epithelium. 22,34 The bacteria are taken up by macrophages and with the passage of time cause severe granulomatous inflammation in the intestine and in the draining lymph nodes. 9 A few of the previous experimental studies with MAP have described focal granulomas made up of epithelioid cells in the ileal and jejunal Peyer's patches, which may spread to other layers of intestines. 3,16 The initial lesions exhibit very few acid-fast bacilli (AFB) that increase in number as lesions progress with time.
These findings have been derived from a few of the experimental studies, with different sets of experimental designs using different strains of bacteria. The results of these studies revealed great variation in disease induction in terms of incubation period and clinicopathologic spectra in experimental sheep. Also, these experiments were conducted with different objectives such as the effect of vaccination 14,16 and drugs, 1 evaluation of serologic tests, 15,28 and determination of early immunopathologic events. 3 These studies have not focused on the sequential development of the lesions over an extended period of observation in any single experimental setup. However, in a few of the recent studies on the natural cases of paratuberculosis in sheep 10 and goats, 12 different types of lesions have been reported between two extremes, i.e., ‘paucibacillary (tuberculoid)’ and ‘multibacillary (lepromatous)’ forms. These studies are definitely informative, but it is difficult to assess the time and course of infection, which remain largely speculative rather than confirmative.
Diagnosis of subclinical cases of paratuberculosis is still a problem. The most commonly used diagnostic assays include johnin test, bacteriologic examination of feces, and immunologic methods measuring humoral and cell-mediated immune responses. 11 Because of major variations in disease progression and immunologic responses to paratuberculous infection, a combination of two or more diagnostic methods has been suggested. Lymphocyte stimulation test (LST), an in vitro correlate of cell-mediated immunity (CMI), is reported to be sensitive for the detection of early stages of paratuberculous infection in cattle 4,20 and sheep. 5 The absorbed enzyme-linked immunosorbent assay (Ab-ELISA) that is considered sensitive and specific for clinical disease in bovines has been less extensively evaluated in ovine paratuberculosis. 11,15,16 The bacteriologic culture method considered to be the most definitive diagnostic test in cattle has shown low sensitivity, especially in sheep. 8
The present study aims to evaluate sequential development of histologic lesions in experimentally infected lambs and to comparatively assess different methods for the diagnosis of Johne's disease in infected animals at various stages of disease development, as categorized on the basis of clinical signs and histologic lesions.
Materials and Methods
Animals
A total of 25 crossbred lambs of either sex, aged 8–12 weeks, were used for the experiment. The lambs were negative to paratuberculous infection, as detected by agar gel immunodiffusion test, Ab-ELISA and fecal smear, and culture examination. Before the start of the experiment, animals were treated with anthelmintics (Albendazole, Glaxo India Ltd., Mumbai, India) and anticoccidiostat (Amprolium hydrochloride, Glaxo India Ltd.), as per recommended doses. Animals were fed green fodder and concentrate ration, as per standard nutritional requirements. Infected and control animals were maintained separately and allowed to graze for 4–5 hours in a restricted fenced area in the vicinity of the experimental shed. The experimental procedures on the animals were carried out in accordance with the Institute Animal Ethics Committee guidelines after approval of the Staff Research Council of the Institute.
Preparation of inoculum
Small intestines from midjejunum to the terminal ileum of two advanced clinical cases of paratuberculous sheep shedding large number of bacilli (‘multibacillary’) were collected at necropsy and the contents were flushed using tap water and finally washed in sterile distilled water. Mesentery and serosa were removed completely and tissues were chopped into small pieces using sterile scissors and minced in a blender with a small amount of phosphate-buffered saline (PBS) containing penicillin (200,000 IU/liter) and chloramphenicol (200 mg/liter). The material was then filtered through sterile gauze to remove tissue debris and to recover the bacterial suspension. The approximate number of organisms in the suspension was found to be 1.8 × 109 AFB/ml. 33 The inoculum was stored in screw-capped plastic containers in 50 ml aliquots at −20 C. The suspension was subjected to culture on Herrold's egg yolk medium (HEYM), and mycobactin J–dependent MAP was isolated. The identity of the strain was also confirmed by the demonstration of the IS 900 sequence by polymerase chain reaction. 2,13
Experimental design
Twenty-five lambs were randomly divided into two groups, infected (n = 20) and control (n = 5). In the infected group, animals were orally administered 5 ml of bacterial suspension on eight occasions at 3-day intervals within 1 month. Control animals received 5 ml of sterile PBS similarly. Animals were picked randomly for euthanasia by intravenous injection of thiopentone (Abbot Laboratory (India) Ltd., Mumbai, India), as per the schedule shown in Table 1.
Schedule of euthanasia and results of fecal examination, immune responses, and histological lesion development in sheep experimentally infected with Mycobacterium avium subsp. paratuberculosis.∗.
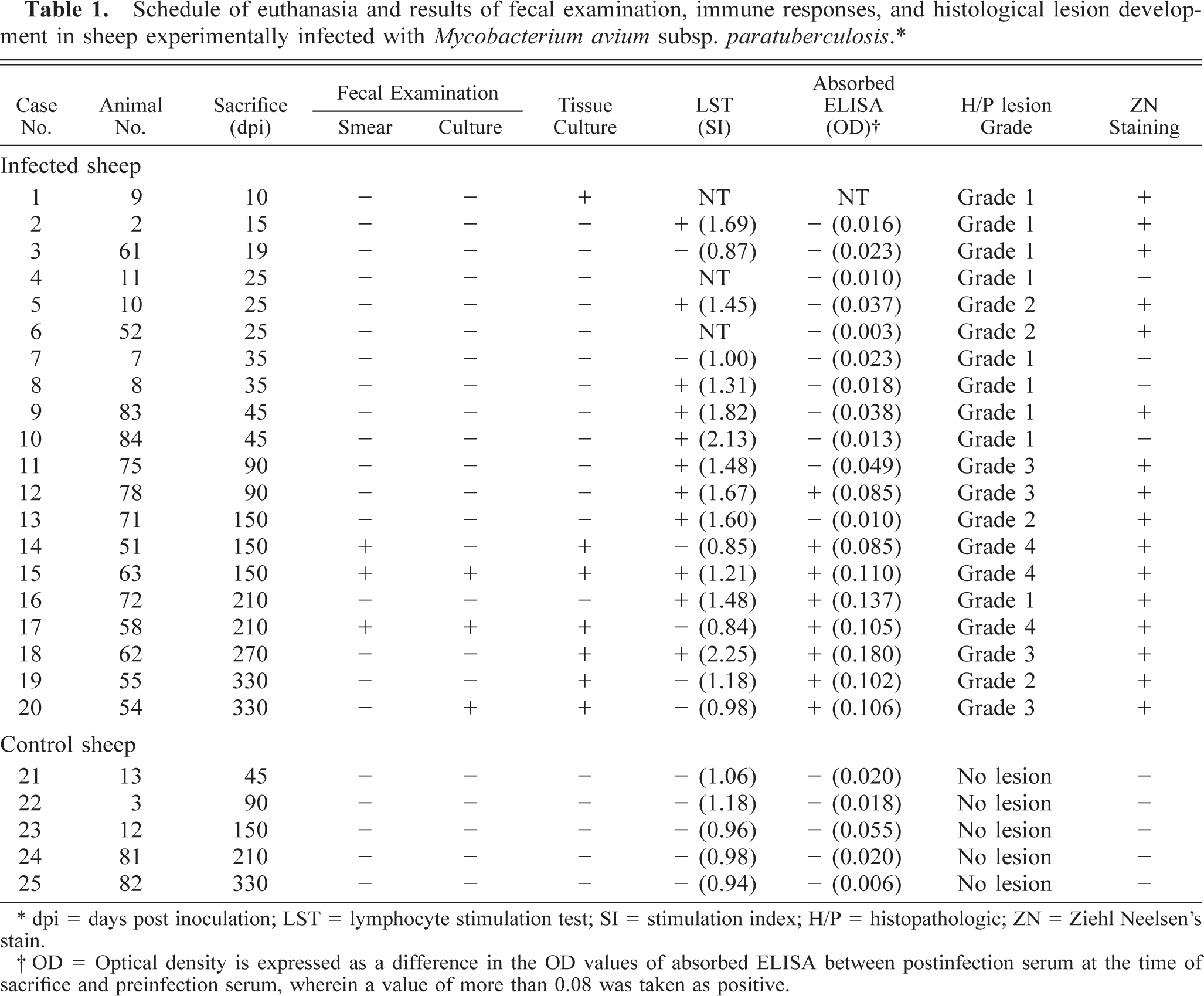
∗ dpi = days post inoculation; LST = lymphocyte stimulation test; SI = stimulation index; H/P = histopathologic; ZN = Ziehl Neelsen's stain.
† OD = Optical density is expressed as a difference in the OD values of absorbed ELISA between postinfection serum at the time of sacrifice and preinfection serum, wherein a value of more than 0.08 was taken as positive.
Clinical status
All animals in both groups were regularly observed for clinical signs. Body weights were recorded at monthly intervals, and the body conditions of the animals were scored as follows: 1) decreasing body condition, 2) stable, and 3) gaining body condition. The types of feces were recorded as ‘pelleted,’ ‘soft,’ or ‘liquid.’
Fecal collection
Fecal samples were collected at monthly intervals and before euthanasia directly from the rectum in sterile test tubes and were processed on the same day or stored at 4 C and processed within 1 week.
Fecal smear examination
One gram of fecal sample was mixed in 10 ml of distilled water and kept overnight for settling. The supernatant was centrifuged at 5,000 × g for 30 minutes. Smears prepared from the sediment, after heat fixation, were stained by Ziehl–Neelsen's (ZN) method and examined under oil immersion. The presence of a clump of three or more small strongly AFB was recorded as positive. 25
Bacterial culture
The culture procedures already standardized in our laboratory for feces and tissues were adopted. 40,41 Briefly, 2 g of feces was mixed thoroughly with 20 ml of sterile distilled water in a sterile test tube. The fecal suspension was allowed to settle for 30 minutes at room temperature. Similarly, 2.5 g of pooled tissues from ileum and mesenteric lymph nodes (MLN) from each of the necropsied animals was triturated in 15 ml sterile distilled water and ground in a sterile pestle and mortar. The triturated tissue was allowed to settle for 30 minutes in a test tube. Five milliliters of the supernatant (from fecal or tissue samples) was decontaminated with 0.9% hexadecyl pyridinium chloride and inoculated onto four slants of HEYM, two with and two without mycobactin J (Allied Monitor Inc., Fayette, MO), and isolates were identified and confirmed as described previously. 30,39,42
Collection of blood samples
Ten milliliters of blood was collected from the jugular vein in a vacutainer containing 2.7% ethylene diamine tetraacetic acid for LST. Another 5 ml of blood was collected from each animal using presterilized disposable syringes for serum just before euthanasia.
Lymphocyte stimulation test
The peripheral blood mononuclear cells (PBMCs) were separated from blood by density gradient centrifugation using Histopaque (density 1.077, Sigma, St. Louis, MO), as per standard methods.
37
The cells were finally collected in Roswell Park Memorial Institute–1640 growth medium (Sigma) containing 10% fetal calf serum (Sigma). The cell viability, ascertained with trypan blue dye exclusion method, was usually greater than 95%. The cell concentration was adjusted to 5 × 106 viable cells/ml. The lymphoproliferative assay was performed using 3-(4,5-dimethylthiazole-2-yl)-2,5-biphenyl tetrazolium bromide (MTT) dye (E Merck, Ltd., Mumbia, India), according to the method described by Mosmann
23
with some modifications. PBMC suspension (100 µl) from each animal was added to nine wells in a 96-well flat-bottom tissue culture plate (Nunc, Roskilde, Denmark). A total of 100 µl of medium alone or medium containing optimum concentrations of johnin purified protein derivatives (PPD) (Biological Products Division, Indian Veterinary Research Institute, Izatnagar, India) (20 µg/ml) or Con A (10 µg/ml) was added each in triplicate wells containing cells from each animal. The plates were incubated at 37 C for 96 hours in a humidified atmosphere containing 5% CO2 (Nuaire, Plymouth, MN). A 100 µl aliquot of supernatant was removed from each well, and 10 µl of MTT (5 mg/ml) solution in sterile PBS was added to each well. The plates were incubated for an additional 4 hours, and then 100 µl of dimethyl sulfoxide was added to each well. Formasan crystals formed by the metabolization of MTT by proliferating cells were dissolved by vigorous pipetting and the plates were incubated for 15 minutes at 37 C. Optical density was recorded at 492 nm with reference reduction of background color at 650 nm in an ELISA reader (Micro Scan, ECIL, Hyderabad, India). The stimulation index was calculated as follows:

The criterion for LST positivity was set at SI of 1.2, which was 33% more than the mean SI of uninfected control animals. The mean SI of uninfected experimental sheep against johnin PPD was found to be 0.90 (range 0.67–1.10).
Absorbed enzyme-linked immunosorbent assay
An indirect Ab-ELISA was carried out using sonicated antigen prepared from MAP (strain TEPS) on sera preabsorbed with Mycobacterium phlei (40 mg/ml of diluted serum), as described previously. 30 The optimum concentrations and dilutions of antigen (20 µg/ml), antibody (1 : 100), and antisheep immunoglobulin G-peroxidase conjugate (Sigma) (1 : 25,000) were determined by checkerboard titration. After antigen coating and incubation with test sera, bound antibodies were detected by orthophenylene diamine and buffered urea-H2O2 (Sigma). Absorbance was read at 492 nm in an ELISA reader (Micro Scan, ECIL). Positive and negative sera and antigen and antibody controls were always included in each plate.
Interplate variations in the absorbance values taken on different days were minimized by applying a correction factor as described previously. 30 An increase of 0.08 in the absorbance value of the postinfection serum over the absorbance value of preinfection serum was considered positive, which was always higher than the highest absorbance shown by the known negative and preinculation sera.
Necropsy and histopathology
After euthanasia, each animal was subjected to thorough necropsy examination. The gross changes were noted, and representative samples from various areas of intestines (three sections each of duodenum, ileum, caecum, colon, and rectum, six sections of jejunum; two sections of ileocaecal valve); ileocaecal lymph node (ICLN) and MLN, liver, adrenals, kidneys, spleen, pancreas, brachial and sciatic nerve plexuses, heart, lungs, and aorta were collected and preserved in 10% buffered formalin. The fixed tissues were processed conventionally to sections of 4 µm thickness and stained with hematoxylin and eosin. Adjacent sections from all tissues including intestines and lymph nodes were also stained with ZN staining method for AFB demonstration.
Grading of histologic lesions
Histologic lesions were classified into four grades on the basis of the location of granulomatous lesions in different regions and layers of the intestine, their association with intestinal lymphoid tissues (Peyer's patches [PP]), pattern and distribution of lesions, types of cellular infiltration, and presence of AFB.
Lesions found exclusively in the ileal or ileocaecal PP, or both, consisting of scattered microgranulomas in follicular and interfollicular areas, were categorized as grade 1 (mildest/earliest) lesions (Fig. 1). The microgranulomas were composed predominantly of large macrophages with occasional neutrophilic infiltration. The mucosal changes were limited to a mild increase in the number of lymphoid cells, plasma cells, and macrophages, as compared with uninfected controls. A few or no AFB could be demonstrated in a few microgranulomas of grade 1 lesions. Grade 2 lesions were those with small focal granulomas found also in the mucosa associated with PP (Fig. 2). Villous and crypt areas were infiltrated with moderate number of lymphocytes, macrophages, and a few plasma cells. A few AFB could be demonstrated in the grade 2 lesions. Grade 3 lesions consisted of multiple granulomas of varying size and occasionally diffuse granulomatous lesions in the PP and jejunal and ileal mucosae associated or not associated with the PP. The mucosa was diffusely infiltrated with lymphoid cells. Grade 3 lesions revealed a few to many AFB. Grade 4 lesions (most severe) were characterized by diffuse granulomatous enteritis comprising large sheets of macrophages and epithelioid cells (Fig. 3), loaded with abundant AFB (Fig. 4), in the jejunal and ileal lamina propriae and cryptal areas. Lymphoid cell infiltration was less numerous. Lesions in PP were much more extensive than those of grade 3.
Ileum; Case No. 9. A granuloma consisting of macrophages (arrows) in a lymphoid follicle in Peyer's patch (grade 1 lesion) at 45 dpi. HE. Bar = 42 µm.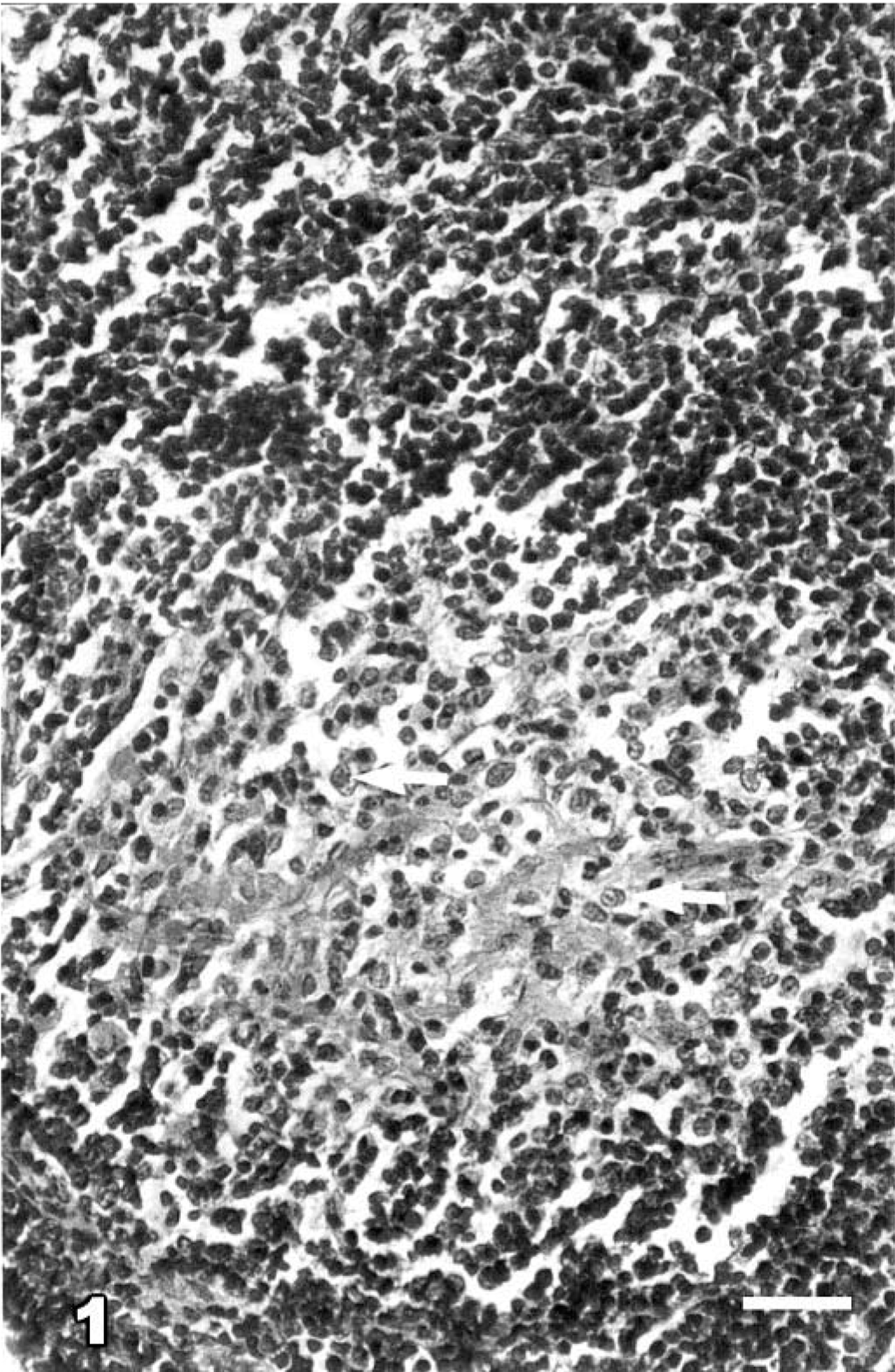
Ileum; Case No. 19. Focal small granuloma (arrows) amongst predominantly lymphoid cells infiltrated lamina propria (grade 2 lesion) at 330 dpi. HE. Bar = 55 µm.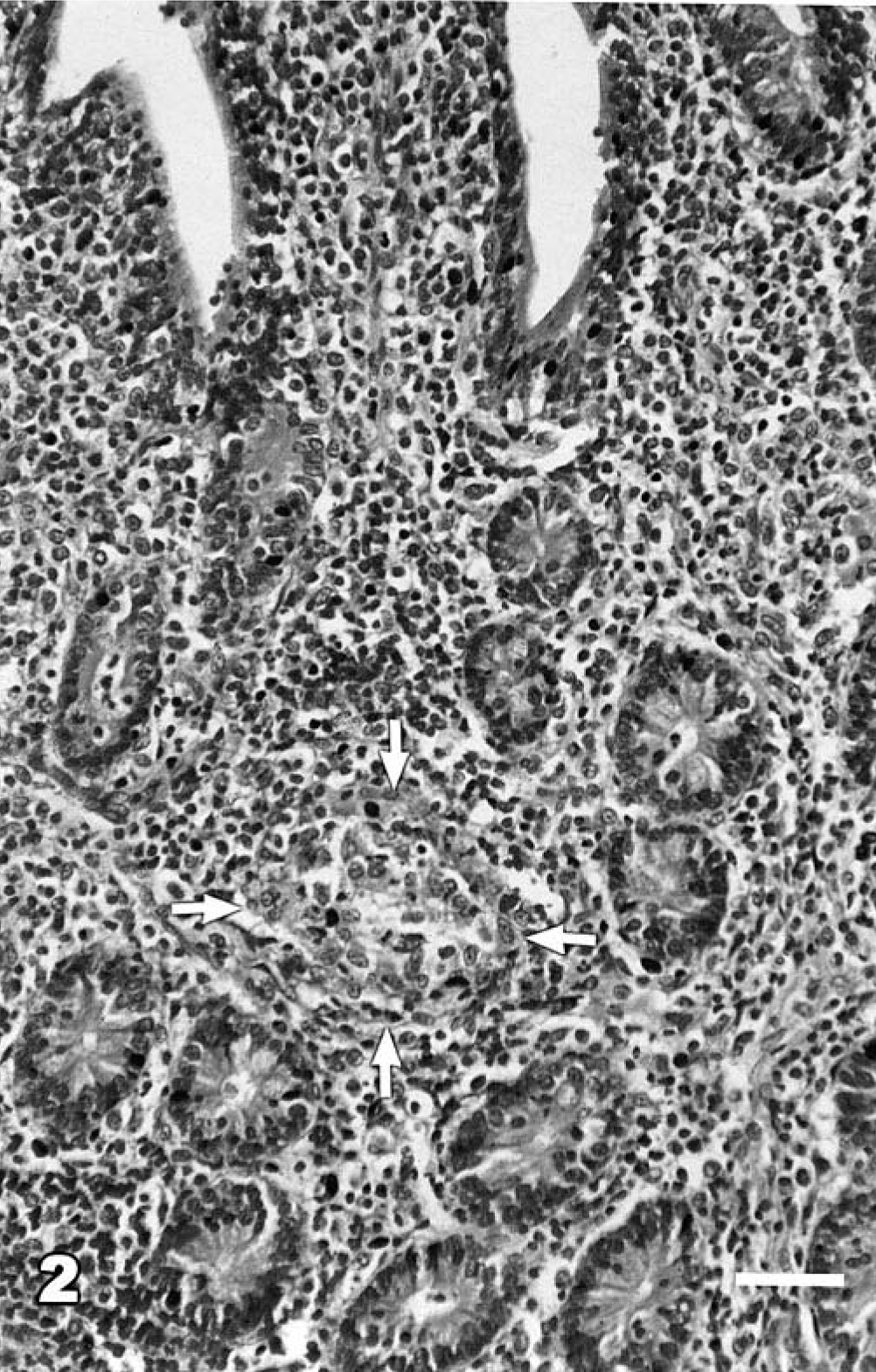
Ileum; Case No. 15. Atrophic villi, with sloughed epithelial lining, showing diffuse infiltration with epithelioid cells (arrows) forming sheet in the lamina propria. (grade 4 lesion) at 150 dpi. HE. Bar = 42 µm.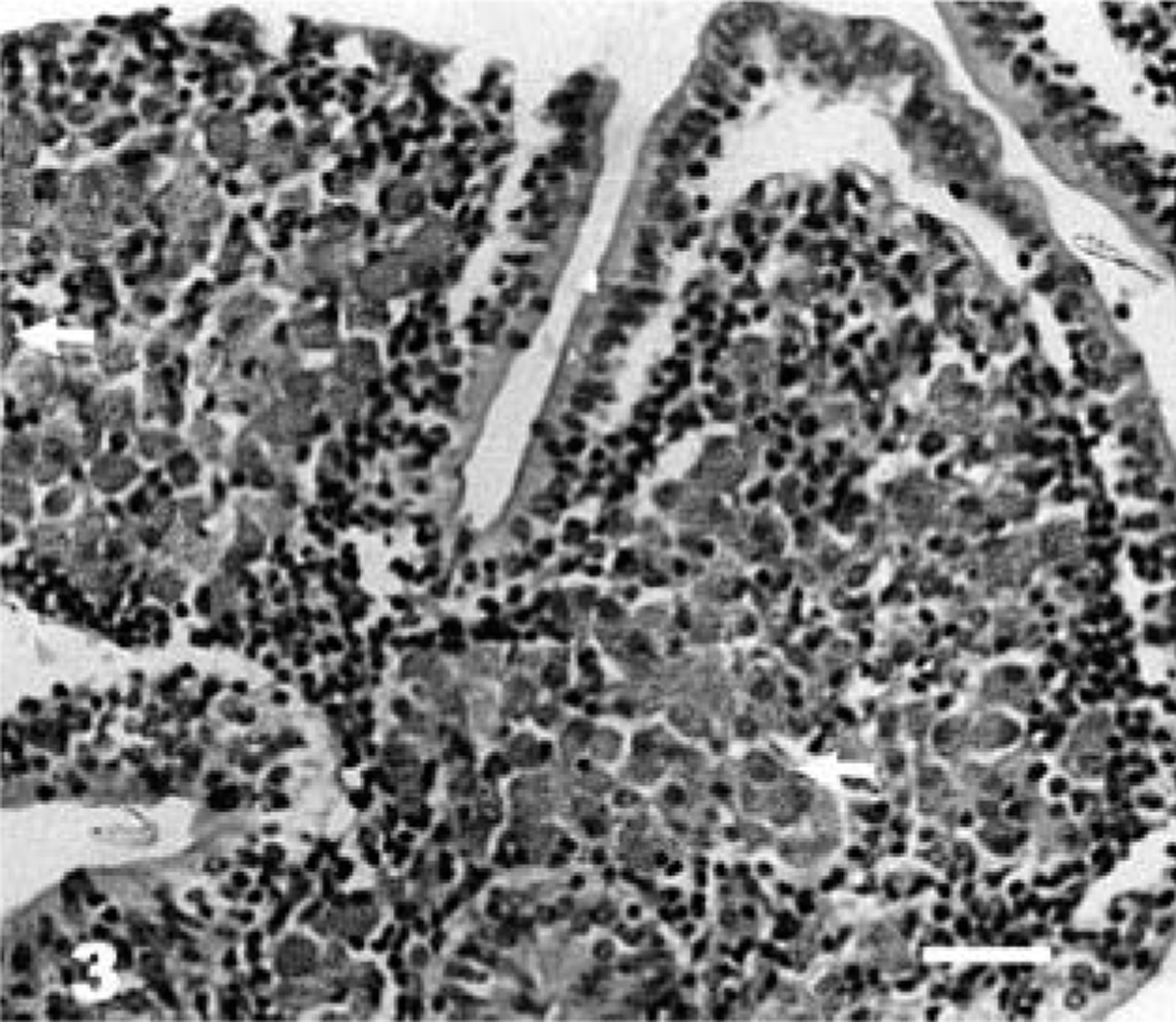
Jejunum; Case No. 15. Clusters of AFB (arrows) in lamina propria from non-Peyer's patch area (grade 4 lesion) at 150 dpi. ZN. Bar = 48 µm.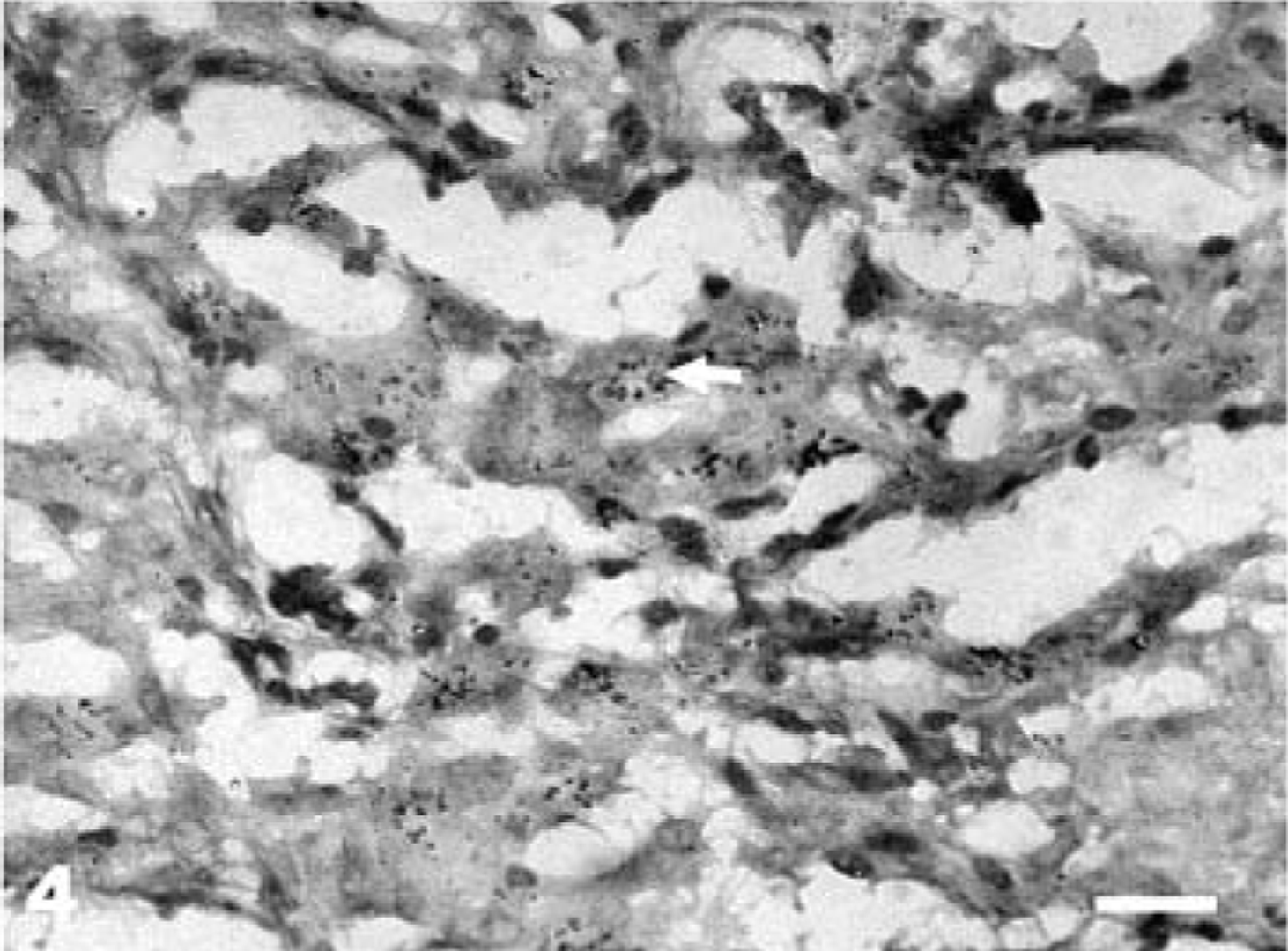
Results
Clinical signs
There was an inverse relationship between severity of the histologic lesions and the body condition score (Fig. 5). The animals with grade 1 lesions had mean body condition scores of 3.0, and the animals with grade 4 lesions had a minimum mean score of 1.33. None of the grade 1, 2, and 3 animals had diarrhea, whereas all the three sheep with grade 4 lesions passed soft unpelletted feces or had diarrhea around the time of their sacrifice.
Relationship of body condition score (mean ± SD) and grades of lesions in sheep experimentally infected with MAP.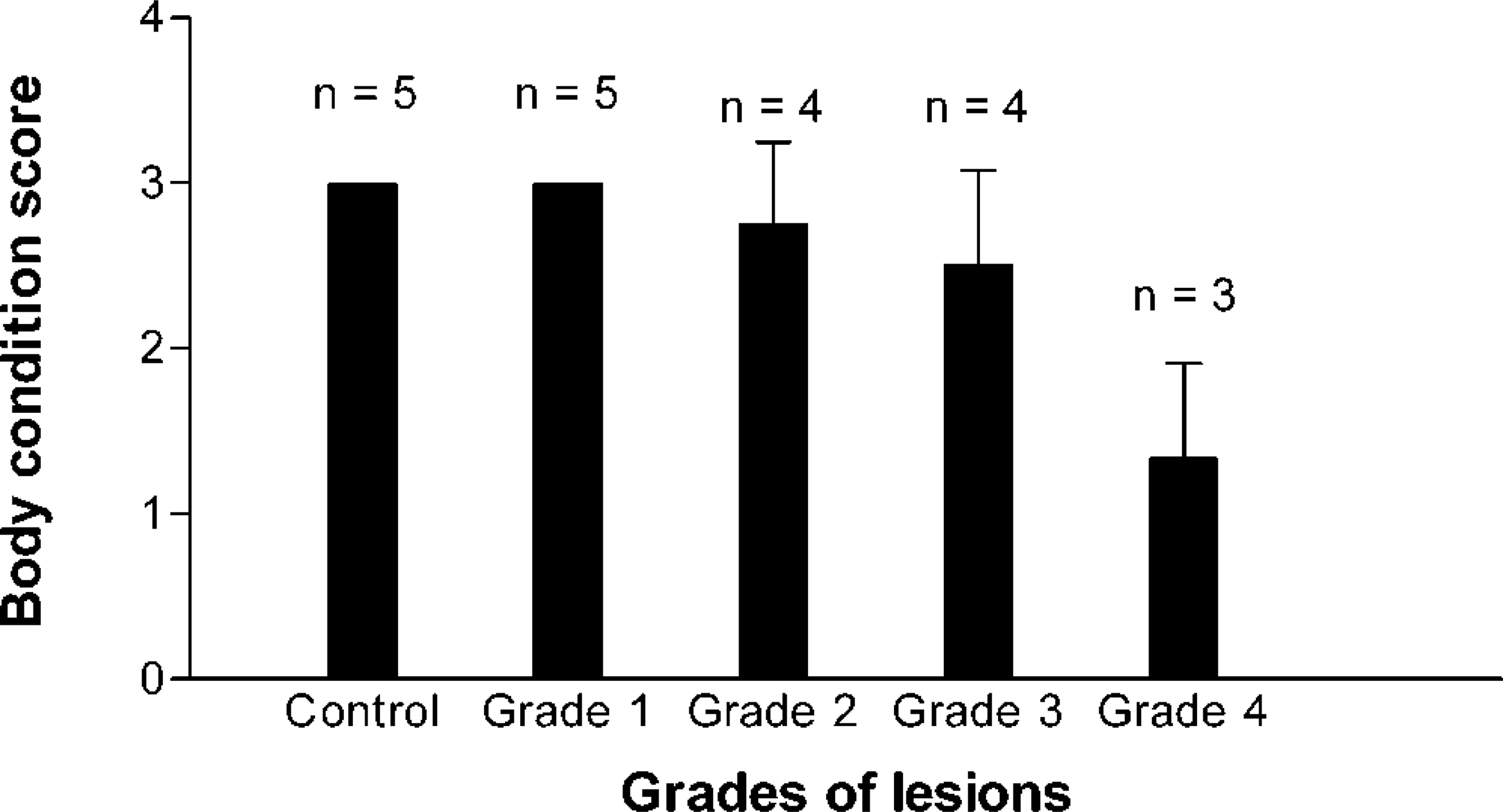
Fecal examination
None of the grade 1 sheep showed the presence of AFB in the fecal smears stained by ZN method (Table 1). One animal with grade 2 and all the animals with grade 3 lesions were positive for AFB at various intervals but were negative at the time of sacrifice. The sheep with grade 2 lesions was positive during the terminal part of the experiment at 240 and 270 days postinfection (dpi). One of the sheep with grade 3 lesions was positive at 30 dpi, and the other three were positive at 60 dpi and then intermittently throughout the experimental period (data of monthly fecal examination not shown). None of the grade 3 sheep showed presence of AFB in smears at 90 dpi, whereas the remaining two sheep were subsequently found positive only at 120 and 210 dpi. All the grade 4 sheep, which were intermittent shedders initially, became regular shedders in smears in the terminal 2–3 months before euthanasia. None of the uninfected sheep showed presence of AFB in the ZN-stained smears.
Fecal and tissue culture
Fecal samples of only three infected sheep (15%), one each at 150, 210, and 330 dpi, yielded MAP in pure culture. These included two grade 4 and one grade 3 sheep. Detectable growth of MAP was observed in seven (30%) of 20 tissue samples from the infected sheep and none of the five tissue samples from uninfected control sheep processed. These included one animal each with grade 1 and grade 2, two animals with grade 3, and three animals with grade 4 lesions (Table 1).
Immune responses
Results of cell-mediated and humoral immune responses, as detected by LST and ELISA, respectively, are shown in Table 1. The overall sensitivities of LST and ELISA were about 65 and 42%, respectively. The lymphoproliferative response was significant and of diagnostic value in most cases with grade 1, 2, and 3 lesions (71.4%). The response was reduced significantly in sheep with severe lesions of grade 4, and only 33% of them showed LST positivity.
Antibody response of diagnostic significance could be detected by ELISA from 90 dpi onward (Fig. 6). Although Ab-ELISA failed to detect most of the sheep with grade 1 and 2 lesions (16.7%), it was successful in detecting sheep with more advanced lesions (grades 3 and 4) (85.7%). When LST and ELISA were used together, they could diagnose up to 88% of the infected animals. There was an inverse relationship between the lymphoproliferative and antibody responses during the course of the experimental infection in sheep depending on the grade of lesion involved (Figs. 6, 7).
Detection of infection in sheep (%) experimentally infected with MAP at various days after inoculation with Ab-ELISA and LST.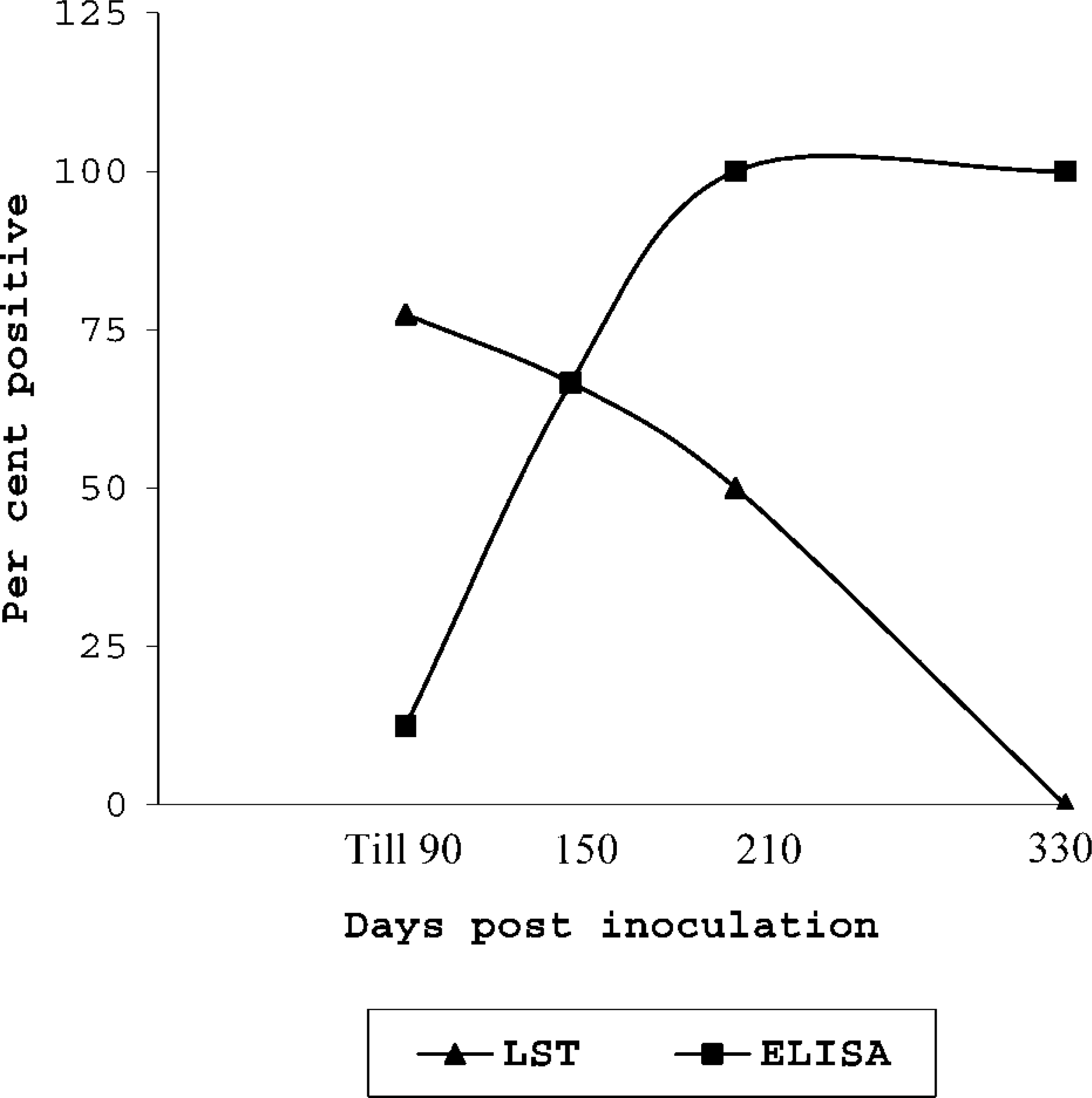
Detection of infection in sheep experimentally infected with MAP with different grades of lesions by Ab-ELISA and LST.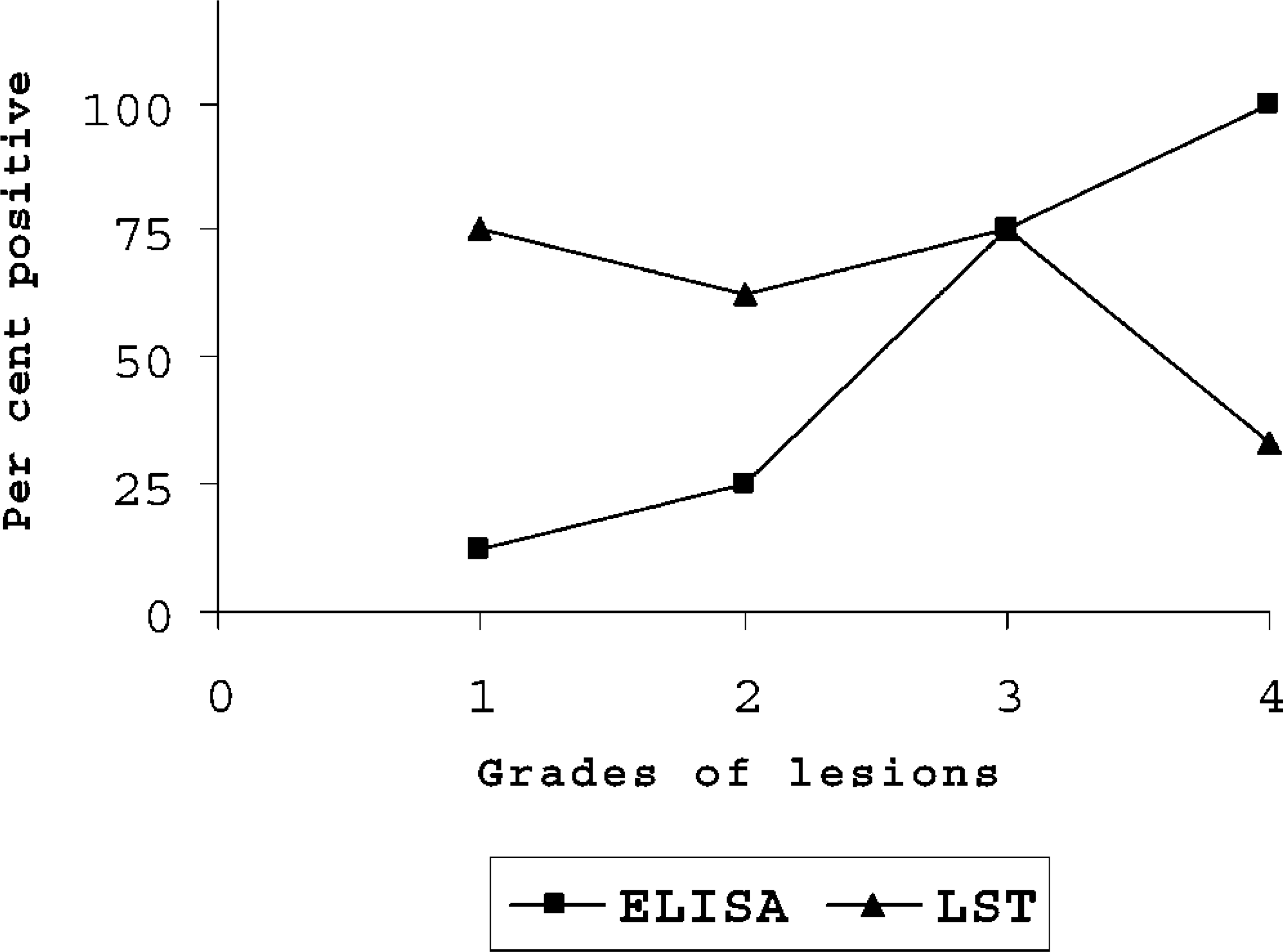
Gross and histopathologic findings
Of the 20 infected sheep, grade 1, 2, 3, and 4 lesions were observed in nine (45%), four (20%), four (20%), and three (15%) animals, respectively. Histologic grading of lesions in each animal euthanatized at different days after infection is shown in Table 1. None of the 10 sheep euthanatized up to 45 days had any gross abnormality of the intestine and associated lymph nodes. Histologically, lesions of grade 1 were present in eight of these animals, of which a few AFB could be demonstrated in intestinal sections of four sheep. Two of these also showed a few AFB in MLN sections. Two other animals (case Nos. 8 and 10) had only acid-fast debris, without any intact AFB. Lesions of grade 2 were also observed in two lambs sacrificed within the first 45 days (at 25 dpi).
At 90 dpi, segmental thickening of the ileal and jejunal mucosae with the appearance of corrugation of ileal mucosa could be observed. The ICLN and MLN were slightly enlarged and edematous. Histologic lesions were of grade 3. Ileal lesions were more severe than those involving jejunum. Villous changes comprised flattening, fusion, and sloughing of epithelium at places. Diffuse infiltration of lymphoid cells caused atrophy of crypt epithelium. One sheep (case No. 12) had follicular lymphoid infiltrates, with multifocal granulomas in the areas adjacent to PP in the ileum. The ICLN and MLN had a few granulomas containing a few AFB in the interfollicular areas of cortex and paracortex.
One of three sheep killed at 150 dpi had mild gross thickening of the jejunal and ileal mucosae with lesions of grade 2. The other two sheep exhibited more pronounced gross changes including severe emaciation, pallor of mucous membranes, velvety and corrugated thickening of the intestinal mucosa, and enlargement of ICLN and MLN. Case No. 15 was moribund at the time of euthanasia and had shown excess peritoneal fluid, mesenteric depot fat depletion, lymphatic cording, and thickened cecal mucosa. Histologically, these two lambs had grade 4 histologic lesion, with abundant AFB (Fig. 4). In non-PP areas of jejunum, lesions with abundant AFB were more often distributed in the superficial portions of the villi. The submucosa was edematous and loosely infiltrated with lymphoid cells, plasma cells, and a few aggregates of macrophages. In case No. 15, necrotic foci were observed in the intestinal mucosa with occasional calcification. The macrophages in these areas contained granular acid-fast material in the cytoplasm along with a few AFB. ICLN and MLN of both sheep showed multifocal granulomas or sheets of epithelioid cells (or both) with numerous AFB in paracortical and interfollicular areas. Subcapsular sinuses frequently revealed infiltration with macrophages.
Grade 1 lesions were observed in one of the two sheep euthanatized at 210 dpi. A few AFB could be demonstrated only in the MLN sections. The other sheep had grade 4 lesions along with perivascular and perilymphatic infiltration of mononuclear cells in the intestinal serosa. A few of these infiltrating cells had AFB. Both ICLN and MLN of grade 4 sheep had a large number of macrophages and giant cells laden with AFB. Caseation and calcification were seen in a granulomatous lesion with a few AFB in ICLN.
One sheep each euthanatized at 270 and 330 dpi showed grade 3 lesions similar to those observed at 90 dpi, with a slight difference in that the jejunal and ileal lesions with macrophage and epithelioid cell infiltration, besides being multifocal, showed a tendency toward diffuse distribution and sheet formation. Another sheep sacrificed at 330 dpi had lesions of grade 2.
None of the other segments of the intestine such as duodenum or large intestine or other organs including adrenals, kidneys, spleen, pancreas, brachial and sciatic nerve plexuses, heart, lungs, and aorta revealed histologic changes characteristic of paratuberculosis, except in one case (No. 14) in which the cecum revealed the presence of small granulomas in the lymphoid tissues and the overlying mucosa. Liver sections of a few cases occasionally revealed microgranulomas without demonstrable AFB. None of the uninfected sheep euthanatized at different days after infection revealed granulomatous lesions characteristic of paratuberculosis.
Discussion
In the past, various experimental designs have been adopted to induce infection with MAP in small ruminants with varying results. 12,14,16 The success of the present experimental design in producing histologic or bacteriologic evidence (or both) of paratuberculous infection 3,9,16 in all 20 infected sheep could possibly be attributed to the effective schedule and dose of infection, the virulence of the strain of MAP used, or the susceptibility of animals used (or all). 17
The current investigation mimicked a typical endemic flock situation, where animals were found in various stages of infection with immunologic, bacteriologic, and histologic spectra and provided an opportunity for the study of the pathogenesis and comparative assessment of certain commonly used diagnostic methods for paratuberculosis.
Histologically, the grade 1 lesions observed in the present study represent the earliest signs of infection described in ovine experimental paratuberculosis. The confinement of these lesions exclusively to the ileal and ileocaecal Peyer's patches 3,16 supports the suggestion that MAP were transported through M cells in the dome of PP, 22 where a relatively low CMI 9 may have assisted in the establishment of the infection. 21 The grade 2 lesions resembled those described earlier in experimental 16 and natural 27 ovine paratuberculosis and might be considered the next stage in the pathogenesis, where infection had spread from PP lesions to the adjacent mucosa. Grade 3 lesions in our study have been described previously as mild 6 or paucibacillary 10 or type 3a 27 lesions in natural cases. The presence of two distinct patterns of lesions, i.e., tuberculoid (diffuse lymphoid cell infiltration and strong CMI) (at 90 dpi) and lepromatous (diffuse macrophage infiltration with low CMI) (at 270 and 330 dpi), as described earlier by Clarke and Little, 10 and mixed types of immune responses in sheep with grade 3 lesions suggests that it can be regarded as the crucial transition stage between tuberculoid and lepromatous tendencies in the pathogenesis of paratuberculosis in sheep. Thus, sheep with grade 3 lesions would be suitable subjects to study the shifts between subsets of Th cytokines (Th1 or Th2), which have been postulated to determine the lesion type in mycobacterioses, 9,18 to understand the cellular and molecular mechanisms of lesion development in paratuberculosis. The most severe grade 4 lesions observed in our experiment corresponded to the classical type of pathology described frequently in natural cases of paratuberculosis. 6,10,31 Necrosis and calcification were observed in two sheep without the evidence of parasitic infection, unlike previous reports. 10,31 Given that the closest relative of MAP, M. avium, causes necrosis and calcification in its susceptible hosts, 18 it would be logical that MAP can also cause these changes.
It was interesting to note that the grades of lesions did not correlate well with the duration of the infection. Encountering early lesions (grades 1 and 2) with demonstrable AFB in three clinically normal sheep after several months of infection suggests either wide variability in the susceptibility of individual animals to MAP infection or latency or persistence of the infection. A possible variation in the phenotypic composition of T lymphocyte subsets (CD4+ & CD8+) and T and B cell ratios in the ileal and jejunal PP may determine an individual's susceptibility to MAP infection. 3,24,36 During latency or persistence, the bacteria may have existed in a quiescent state or in metabolically active viable forms, 26,27 multiplying very slowly in the hostile environment of the gut immunity. It would be worthwhile to study the response of such apparently healthy sheep to stress 8 and immunosuppression conditions, which would enable MAP to multiply heavily, leading to progressive disease and excretion of MAP. This would have important implications for the control of paratuberculosis.
The spread of infection to mucosa not associated with PP and the presence of segmental lesions in the small intestine support the contention that the infection spreads along the length of the intestinal tract after establishing initial foci of infection in the gut lymphoid tissue. In view of the presence of free MAP in the villous region, 34 whether a direct entry of bacteria through the absorptive villous epithelium, as reported in M. avium infection, 32 would establish foci of infection in the villous region remains unclear. A more extensive histopathologic protocol and loop experiments involving both PP and non-PP areas would give more information on the pathogenesis of villous lesions in nonlymphoid areas of the intestine.
Amongst the diagnostic tests for paratuberculosis, the lower sensitivities of bacterial culture of feces (15%) and infected tissues (30%) as compared with histologic demonstration of bacilli (80%) (Table 1) may be explained by the fact that at least 65% of the lambs had focal lesions (grades 1 and 2) containing either no or only a few AFB confined mainly to the submucosal region. It may also be attributed to the existing limitations of conventional culture method, especially in small ruminants. 6,35,43
Although diagnosis of paratuberculosis based on the immune responses to MAP infection has been the trend for many years, only in a few studies has it been correlated with the type of lesions and the duration of the infection. LST could detect about 65% of the infected sheep, mostly in the subclinical stages of the infection, and was in line with the earlier observations that CMI-based assay is sensitive in diagnosing subclinical cases. 8,11,29 Low sensitivity of LST in detecting animals with grade 4 lesions encountered in our study is not surprising in view of the fact that the CMI response wanes during the chronic course of the disease 29,36 and often induces antigen-specific anergy in advanced cases. 7 On the other hand, antibody response correlated well with the grade of lesion and the postinfection period. ELISA positivity in animals with early lesions much later in the course (at 210 and 330 dpi) suggests that even small number of MAP present for several months can elicit specific antibody response. Thus, ELISA can successfully detect even nonshedders, which may have escaped detection by fecal culture method. Hence, ELISA-positive cases that fail to yield positive culture should not be considered false positives based on the culture results alone. 19 Instead, these animals may be kept under observation until confirmation by other methods. The higher sensitivity obtained when both LST and ELISA were used concurrently, suggests that CMI and antibody response–based tests can never substitute each other. Hence, they should be used simultaneously to detect most of the infected animals at any time during the prolonged course of infection.
In conclusion, it is evident from our study that infection first establishes in lymphoid tissues of the small intestine, possibly at multiple sites producing segmental lesions. From gut-associated lymphoid tissue the infection spreads to lamina propriae to become diffuse lesions. Wide variability in the histologic lesions and initial tropism of MAP to the intestinal lymphoid tissues suggest a differential susceptibility of young animals, possibly because of the compositional phenotypic variation of lymphocytes in PP, which influences the subsequent course of infection. Sheep with grade 3 lesions may be suitable candidates for future exploration of shifts in cytokine responses in the complex immunobiologic environment of the gut, which determine the nature of lesions in paratuberculosis. The results of the current study suggest that both LST and ELISA should be used simultaneously to detect most of the infected sheep.
Footnotes
Acknowledgements
We are grateful to the Head, Division of Pathology, and Joint Director (Research) and Director of the Institute for providing facilities to carry out this work. We thank Shri. Rambujh for excellent technical assistance and Shri. Anirudh for preparation of photographs.
