Abstract
A pregnant 4-year-old rhesus monkey (Macaca mulatta) was presented with a history of acute vaginal bleeding. Physical examination revealed an open cervix. An ultrasound scan demonstrated a viable early third-trimester fetus, approximately 16 weeks of gestational age. Hematology results showed that the monkey was anemic, with a normal leukogram and Döhle bodies. A subsequent cervical culture was positive for Campylobacter fetus. The fetus died 3 days later, and a necropsy of the fetus and placenta was performed. Microscopic examination of the placenta revealed villitis, perivillitis, and deciduitis with S-shaped and gull wing-shaped bacteria. C. fetus was considered the cause of the placental lesions and fetal death; however, the pathogenesis of the infection (hematogenous vs. ascending from the maternal genital tract) was not clear. This is the first report of a Campylobacter-induced fetal death in the rhesus monkey. Because macaques can be asymptomatic carriers and Campylobacter-induced diarrhea is common, this finding has implications for breeding success in nonhuman primate breeding colonies.
Although members of the genus Campylobacter have frequently been associated with infertility, abortion, and gastroenteritis in animals and humans1–3 we found only one report of possible Campylobacter-induced abortions in three cynomolgus macaques (Macaca fascicularis) shortly after importation.13,14 Because Campylobacter may be prevalent in macaques1,4,5 and because of the importance of fetal loss in a nonhuman primate breeding colony, we report here a case of Campylobacter-induced fetal death in a rhesus monkey (Macaca mulatta).
A pregnant 4-year-old multiparous rhesus monkey from the M. D. Anderson Cancer Center (Bastrop, TX) breeding colony was presented with a history of acute vaginal bleeding. The animal was housed in a five-member harem group. No animals within this group had a history of illness. This female had given birth to a healthy female infant 10 months earlier. Physical examination revealed an open cervix and vaginal blood originating from the uterus. Ultrasound showed a viable early third-trimester fetus, approximately 16 weeks of gestational age, with normal heart rate and conformation. The placenta had numerous small hyperechoic areas interpreted as calcification. The hemogram showed that the female was anemic. The leukogram was normal, but Döhle bodies, suggestive of sepsis or a severe inflammatory disorder, were present in most neutrophilic white blood cells. A cervical culture was taken, and the monkey began to receive antibiotics. The culture was positive for Campylobacter fetus. For 3 days, there was sporadic vaginal bleeding, and on the third day no fetal heartbeat could be visualized on ultrasound. Ultrasonography also revealed hyperechoic uterine sections bordered by hypoechoic areas, suggestive of hematoma formation and probable grade 1 or grade 2 abruptio placentae. The dead fetus was removed by Cesarean section. Following surgery, the female fully recovered over a 2–3 week period. Hemograms returned to normal, and a cervical culture 3 weeks postsurgery was negative for organisms.
A necropsy was performed on the mildly autolytic 176-g female fetus with attached umbilical cord and placenta. The fetus had no abnormalities. The placenta had the usual two disks (3 cm and 5 cm in diameter) and weighed 76 g. Both disks were pale tan on cross section, and the larger disk had a 0.5-cm band of hemorrhage in the peripheral subchorionic area. Blood clots were present on the maternal (decidual) side of the disks. The placental membranes (amnion and chorion) were opaque, brown, and mildly thickened near the insertion with the disks. The umbilical cord was 16 cm long and inserted near the center of the larger disk. Measurements for the fetus, umbilical cord, and placenta were consistent with an early third-trimester pregnancy. All tissues were immersion fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Placental tissues examined included membrane rolls, sections of the central and peripheral parts of each disk with maternal and fetal surfaces, and cross sections of the umbilical cord at the midpoint and near the insertion on the larger disk. Tissues were sectioned at a thickness of 4–5 μm and stained with hematoxylin and eosin (HE), Gram's stain, periodic acid–Schiff, and modified Warthin-Starry stain (WS).
Histopathologic examination of selected fetal tissues (lung, liver, and kidney) was unremarkable except for the prematurity of the organs and autolysis. Cross sections of the umbilical cord and reflected membranes away from the disks showed no changes. The amnionic and chorionic membranes at the disk insertion and the peripheral chorionic plate were elevated from the underlying placental tissue by large amounts of irregularly laminated fibrin containing random foci of mineralization and blood. In other areas, the chorionic plate was edematous and had occasional blood vessels with small numbers of neutrophils in the wall. The placental parenchyma contained several large foci (10–20% of the section area) of necrosis with numerous neutrophils, fibrin, and mineralization (Fig. 1). Small numbers of S-shaped and gull wing–shaped bacteria were observed in necrotic areas stained with the WS (Fig. 2). The decidua contained a more intense infiltrate of neutrophils but no necrosis or bacteria. Histologic diagnoses were villitis, perivillitis, and deciduitis with bacteria.
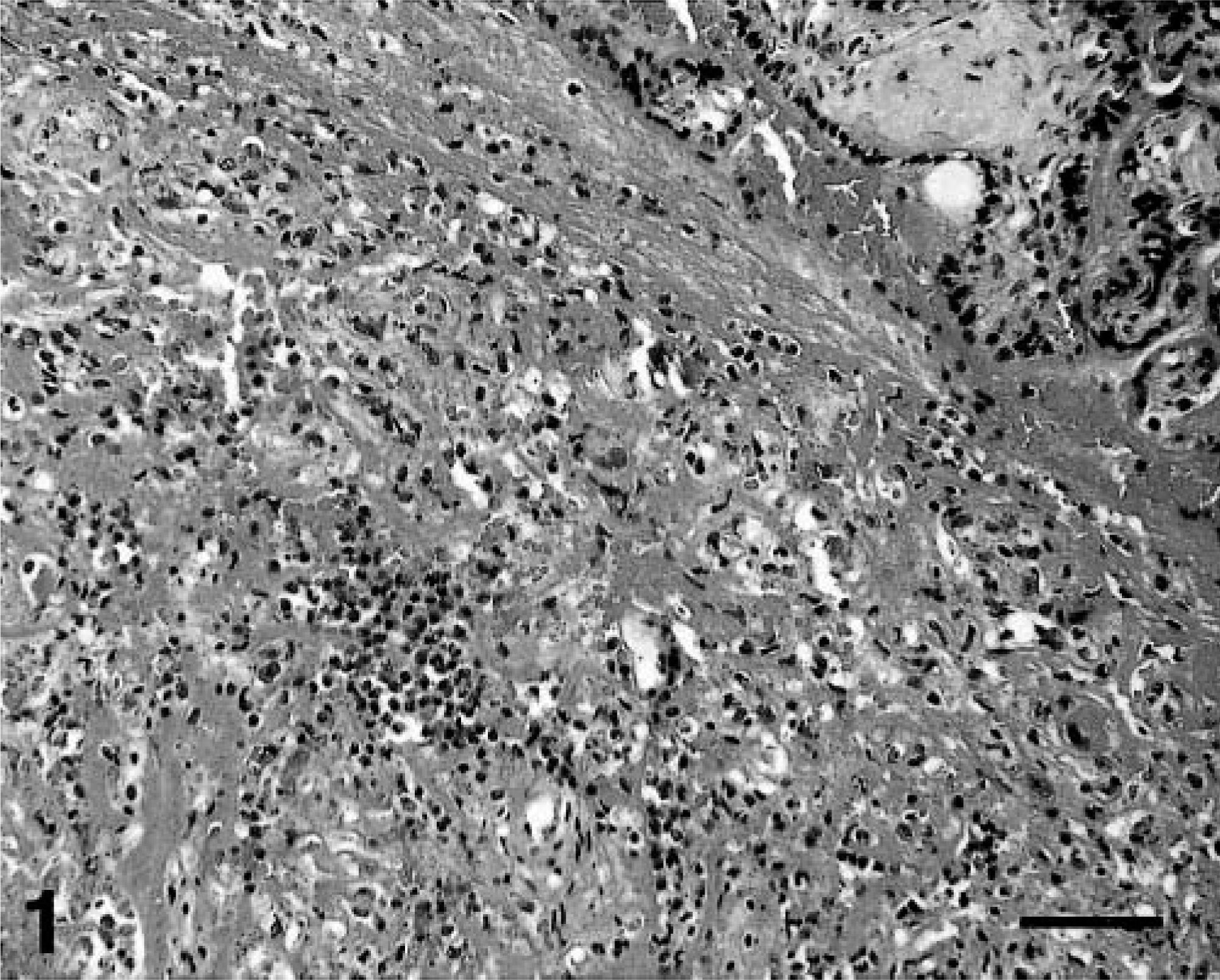
Placenta; rhesus monkey. Necrosis and neutrophilic infiltrates in the parenchyma. HE. Bar = 150 μm.
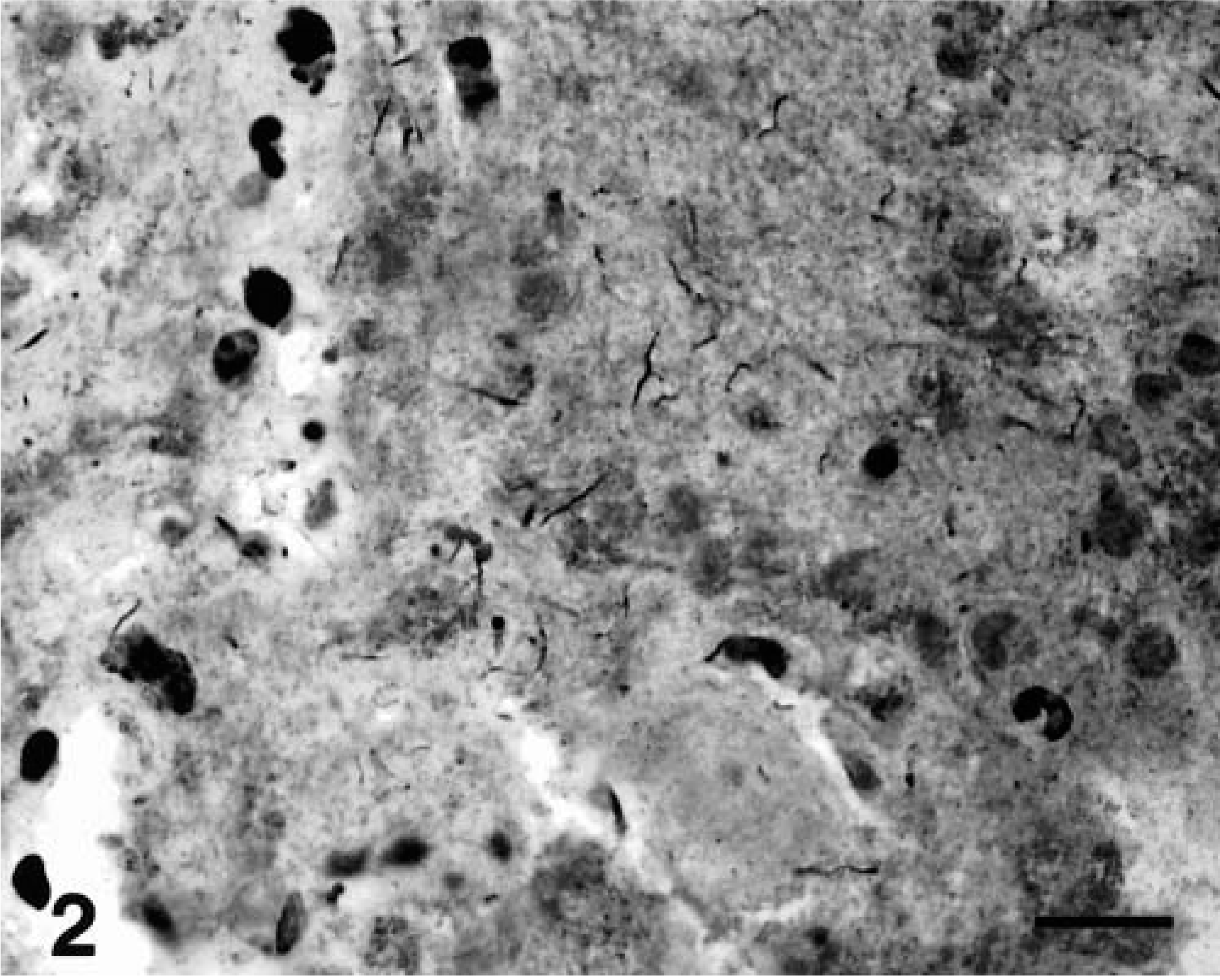
Placenta; rhesus monkey. S-shaped and gull wing-shaped bacteria within a necrotic parenchymal area. WS. Bar = 15 μm.
Based on the cervical culture and histologic findings, C. fetus was considered the cause of the placental lesions and fetal death in this case. In our experience, most cases of fetal death (abortion or stillbirth) are due to an ascending bacterial infection from the maternal genital tract. Over the last 2 years, we have identified a variety of bacterial organisms; however, gram-positive cocci (e.g., coagulase-positive staphylococcus, α-hemolytic streptococcus) have been the most common, and no cultures have been positive for Campylobacter. Organisms were usually cultured from the fetal membranes or maternal genital tract and were associated with acute inflammation of the fetal membranes (chorioamnionitis), premature delivery, and fetal death. In some instances, fetal pneumonia and septicemia were present.
In this monkey, in addition to being unusual because of the identification of C. fetus, histologic changes were primarily confined to the parenchyma of the placenta (villitis, perivillitis) and the decidua (deciduitis), with little involvement of the fetal membranes. Such lesions are most consistent with the hematogenous spread of bacteria to the placenta or with bacterial spread from the adjacent endometrium, not from an ascending infection.6,7,12 However, because the organisms were cultured from the cervix, the pathogenesis of this infection is not clear.
In humans, an ascending infection is normally associated with fetal membrane lesions (chorioamnionitis), but villitis may occur secondarily if there is fetal septicemia.6,7,9 In contrast, chorioamnionitis is almost always present with villitis.12 In this monkey, no fetal lesions or significant membrane lesions were detected. Early reports of Campylobacter-induced gastrointestinal infections in humans and monkeys indicated that organisms appeared to penetrate the mucosa and enter the blood.2 In humans, because the organisms were rarely recovered from the genital tract, localization in the placenta during bacteremia was considered the likely pathogenesis. More recently, a report of C. jejuni diarrheal disease in a pregnant woman suggested ascending infection may also be an important route of infection and a cause of septic abortion.3
This is the first report of Campylobacter-induced fetal death in a rhesus monkey and only the second report of such an occurrence in macaques. C. fetus causes abortions and infertility in animals,2,8 but in humans the organism is usually associated with bacteremia and extraintestinal infections such as abortions, arthritis, abscesses, peritonitis, endocarditis, and salpingitis.10 However, C. fetus can cause gastroenteritis in humans, and the incidence of infection may be underestimated because of the routine fecal culture techniques used.10 That is, the organism does not grow well at 42 C, and C. fetus is usually susceptible to an antimicrobial agent (cephalothin) present in some selective media used for stool cultures in humans and animals. C. jejuni and C. coli are most often associated with gastrointestinal infections and diarrheal diseases in animals and humans.10 But, C. jejuni was linked to septic abortion in a woman with diarrheal disease.3
The scarcity of reports of Campylobacter-induced fetal death in macaques suggests that the problem is rare. However, the finding in this female rhesus monkey has implications for breeding success in nonhuman primate breeding colonies. This finding may be particularly important because macaques can be asymptomatic carriers and Campylobacter-induced diarrhea is a common problem.11
Footnotes
Acknowledgements
We thank Gerald Costello for necropsy and histology technical assistance and Dennis Winkler and Debbie Herring for microbiology support.
