Abstract
Plasmodium falciparum remains one of the world’s deadliest diseases and with ongoing concerns of evolving drug resistance, there is a need for continued refinement of the Plasmodium coatneyi infection model in macaques to study severe malaria. As such, the systemic ultrastructural lesions associated with P. coatneyi infection in splenectomized rhesus macaques was evaluated in 6 animals. Autopsy samples from multiple areas of the central nervous system (CNS), kidneys, heart, liver, and lungs of all 6 animals were processed for electron microscopy. A systematic analysis of the ultrastructural changes associated with the plasmodium was undertaken by multiple pathologists to ensure consensus. All tissues exhibited marked sequestration of infected red blood cells comprised either of cytoadherence to endothelium or rosette formation, associated with variable degrees of host cell damage in a range of tissues that in severe cases resulted in necrosis. This is the first complete systemic evaluation of ultrastructural tissue lesions in P. coatneyi–infected rhesus macaques, and the findings have important implications evaluating of the use of this model for the study of severe malaria caused by P. falciparum in humans.
There were approximately 229 million cases of malaria infection in humans reported in 2019. Of these, 99% of the cases in Africa, and 94% of the cases globally were due to Plasmodium falciparum. 67 While there has been a marked decrease in the overall number of cases in the Mekong Delta, South East Asia remains the epicenter of P. falciparum drug resistance, underscoring the importance of continued development of antimalarial drugs. 67 Plasmodium coatneyi is one of the number of species of plasmodium causing malaria in non-human primates.4,14,26,35,40 It infects several macaque species and has been used as an animal model for P. falciparum which induces severe and cerebral malaria in humans. The parasite and the associated lesions have become the focus of renewed interest based on significant debate as to the relevancy of animal models such as the murine and non-human primate models of severe and cerebral malaria in humans.14,17,28,35,37,66
Prior to this study, there has not been a comprehensive description of P. coatneyi–induced lesions in this animal model. Histopathological examinations of selected autopsy samples from P. coatneyi–infected rhesus macaques have been reported in the literature. However, with the exception of recent work examining the dyserythropoeisis in the bone marrow in a non-splenectomized macaque model 44 and several elegant studies of placental pathology,15,16 these descriptions were typically quite limited in scope. They were conducted in small numbers of animals and were targeted to specific organs, such as the brain and the heart, to examine and draw parallels with particular aspects of the pathophysiology of human disease. These studies aimed to examine pathophysiological mechanisms such as rosette formation2,31,60,64 and deep vascular sequestration,19,55 and the subsequent patterns of tissue injury proposed to result from adhesion to endothelial or other cell types mediated by knob formation on the infected red cell.2,29,30,32,60,64 Knob formation consists of electron-dense structures that develop on the surface of infected erythrocytes and that are purported to contribute to the cytoadhesion observed in malaria infections. Furthermore, these reports were complicated by the heterogeneous genetic and immunologic backgrounds of experimental animals, differing levels of parasitemia, splenic status, and whether curative treatment had been given prior to euthanasia.
To provide a more comprehensive and systematic examination of lesions in this model, a larger-scale examination was conducted using multiple tissues and emphasizing relative uniformity of parameters, as an important step in characterizing the ultrastructural tissue changes in the splenectomized rhesus macaque/P. coatneyi model of severe malaria.
This was conducted with comparisons against age-related and non-disease-associated pathological findings in control animals (n = 15) and examined by multiple independent pathologists (n = 3) to validate the disease-associated findings.
In addition, this study included tissues, such as the spinal cord, in which the lesions of P. coatneyi infection had not been previously evaluated, and yet may be important in the pathophysiology and comparisons with human disease, if the model is to be successfully used in interventional studies such as vaccine, antimalarial, or adjuvant therapy investigations. Herein, the ultrastructural lesions of P. coatneyi infection in the rhesus macaque are described in an organ-specific manner, thus allowing a more comprehensive comparison with the lesions found in human patients with P. falciparum–induced disease.
Materials and Methods
Study Design
Six splenectomized Indian-origin rhesus macaques were infected with P. coatneyi in a protocol conducted at the Armed Forces Research Institute of Medical Sciences (AFRIMS, Bangkok, Thailand) in 2013 and 2014 as part of a P. coatneyi pathophysiology and model refinement study. The experimental animals were 4 adult males (mean 11 years and 5 kg) and 2 adult females (mean 11 years and 5 kg). The rhesus macaques in the AFRIMS colony underwent an annual physical examination, including hematological and clinical chemistry evaluation as part of routine management, and all animals underwent similar evaluation criteria prior to being selected for inclusion in any study. Animals with any abnormalities were excluded from involvement in infectious disease research. All animals included in this study were interpreted as being within normal limits regarding clinical chemistry and hematology. In all instances, the experimental animals were inoculated from one donor rhesus macaque through a blood challenge of P. coatneyi–infected erythrocytes. The donor was initially infected by intravenous inoculation of freshly thawed cryopreserved P. coatneyi–infected blood. The stabilate stock used was the Hackeri strain of P. coatneyi archived at AFRIMS, thereby ensuring that the strain of plasmodium was homogeneous and standardized across the experimental animals. Once the parasitemia in donor animals had reached >50,000 parasitized erythrocytes per microliter, 10 to 20 mL of blood was drawn into a heparinized tube, diluted with sterile phosphate-buffered saline solution to a concentration of 5 × 106 infected erythrocytes per 1 mL suspension, and divided into equal 1-mL aliquots. The 1-mL aliquots were subsequently used to infect the study animals. Research was conducted in compliance with the Animal Welfare Act and other US federal statutes and regulations relating to animals and experiments involving animals, and adhered to principles stated in the Guide for the Care and Use of Laboratory Animals, NRC Publication, 2011 edition. 45 All animal use was approved by the AFRIMS Institutional Animal Care and Use Committee (IACUC) Protocol Number 12-08.
Tissues used in this study were derived from animals used in 2 arms of the main study. The initial 3 animals were planned as part of a drug dosing study based on the historical animal model. Under that study design, the animals were assessed 3 times daily for parasitemia. Unfortunately, that proved to be insufficient to identify the steep spike in parasitemia and as such the protocol was amended for the subsequent 3 positive control animals as follows: a daily blood smear was collected 3 times daily, from day −1 to day 5 relative to inoculation with infected blood. In all animals, blood smears were collected 3 times daily, at 0700, 1100, and 1900. On study day 6, blood smear collection was increased to 12 times (every 2 hours). On study days 7 and 8, blood smears were collected every hour once parasitemia reached 6% and continued on that schedule until euthanasia. Thick and thin blood smears were prepared from capillary blood samples collected from each monkey by sterile pediatric lancet puncture of the lateral auricular vein. In the majority of cases, parasitemia, temperatures, clinical signs were consistent and correlated. Terminal parasitemias from the refined model ranged from 34.2% to 39.2% (mean = 36.1%, ie, parasites in 36% of erythrocytes).
The criteria for euthanasia were parasitemia levels of 20% or greater, and/or at least 3 blood values and clinical signs meeting defined benchmarks of severe malaria. These included evidence of severe acute tubular injury (evidenced as both hemoglobinuria/hematuria combined with elevated serum creatinine and urea); hepatic or hepatobiliary injury (as evidenced by jaundice and significantly elevated alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase [ALP], gamma-glutamyl transferase [GGT], and decreased albumin); severe anemia defined by a hematocrit of 15% or less; coagulopathy based on improper clotting as well as cutaneous petechiae, and ecchymoses; anorexia lasting 3 meals; continued recumbency combined with severe lethargy and depression; neurological signs such as pupillary dilation, lack of a menace response, marked depression, and/or severe weakness characterized by an inability to sit or perch; persistent vomiting; persistent diarrhea with either frank blood or melena; persistent tachycardia or bradycardia; marked and persistent dyspnea/tachypnea; excessive vocalizing or other signs of significant distress; persistent fever of 3°C above baseline or 2°C below baseline; ataxia as a feature of neurologic disease (but not specifically due to weakness); and/or progressive cutaneous necrosis. By day 7 postinfection, all animals were euthanized based on these criteria.
Electron Microscopy
Complete autopsies of all 6 animals were conducted immediately postmortem, with collection and processing of tissues for histopathological, immunohistochemical, and ultrastructural analyses using a standardized tissue sampling protocol. This included samples from multiple areas of the CNS (cerebrum, cerebellum, brain stem, and spinal cord), kidneys, and heart in all 6 experimental animals and in the control animals. In addition, sections of liver, lung, and retina were sampled from the experimental animals. Retina was collected but was unsuitable for analysis. These tissues were selected based on the lesions most commonly reported in humans infected with P. falciparum. At autopsy, tissue samples were collected from target organs, trimmed into ~1 mm4 blocks which were immersed within 1 minute of collection in 2.5% glutaraldehyde in 0.1M phosphate buffer and subsequently processed for routine transmission electron microscopy (TEM). Samples were postfixed in osmium tetroxide, dehydrated, and embedded in Spurr’s epoxy resin. Thin sections of areas of interest were cut and stained with uranyl acetate and lead citrate, and grids were examined in a JEOL 1200EX electron microscope.
Light Microscopy
Similar tissue samples from multiple sites were collected for light microscopic examination, including multiple areas of the brain and spinal cord, fixed in formalin, processed routinely, and stained with hematoxylin and eosin. Sections were compared with archival tissue from normal control rhesus macaques (n = 15), with attention paid to changes which may represent normal aging or background variation.10,52 Control tissues were harvested postmortem through the tissue sharing program in association with ongoing IACUC-approved infectious disease protocols at AFRIMS, including renal transplantation studies, Shigella sp. drug trials, simian-human immunodeficiency virus chimera pathogenesis studies, Plasmodium cynomolgi drug trials, and routine attrition within the macaque colony due to spontaneous neoplasms or other lesions not associated with experimental protocols. In all cases, the control animals were euthanized and did not have an acute infectious cause of death. The control animals were 9 males and 6 females, with mean ages of 10 and 11 years, respectively.
Results
Lesions in multiple organs
All light microscopic and ultrastructural lesions observed and described were noted only in the experimental animals.
All tissues had microvascular sludging with parasitized erythrocytes and hemozoin pigment, which appeared to be causing vascular obstruction. Many vessels had increased caliber (suggesting congestion), and there were an increased number of vessels per unit area, suggesting opening of collateral vessels.
In addition, infected erythrocytes contained either late ring-stage trophozoites or schizont stages of parasite development. In the case of later-stage parasites, food vacuoles containing brown-gold crystalline hemozoin pigment were clearly seen even at low magnification.
Erythrocytic changes appeared to occlude microvessels in all tissues due to sequestration of infected erythrocytes through cytoadherance of infected erythrocytes to other infected red cells, to uninfected erythrocytes (rosette formation), or to the vascular endothelium (margination). Infected erythrocytes had ornamentation on the cell membrane (knobs and caveolae). There was a lack of regularity of the electron-dense knobs on the surface of trophozoite-infected erythrocytes and infected erythrocytes were also observed to be adorned by high numbers of caveolae.
In all tissues, endothelial cells occasionally had swollen mitochondria, and dilatation of the cisternae in the endoplasmic reticulum, and fragmentation that formed vacuoles (Fig. 1). Sporadically, pseudopod-like structures from endothelial cells interacted with distorted infected erythrocytes.
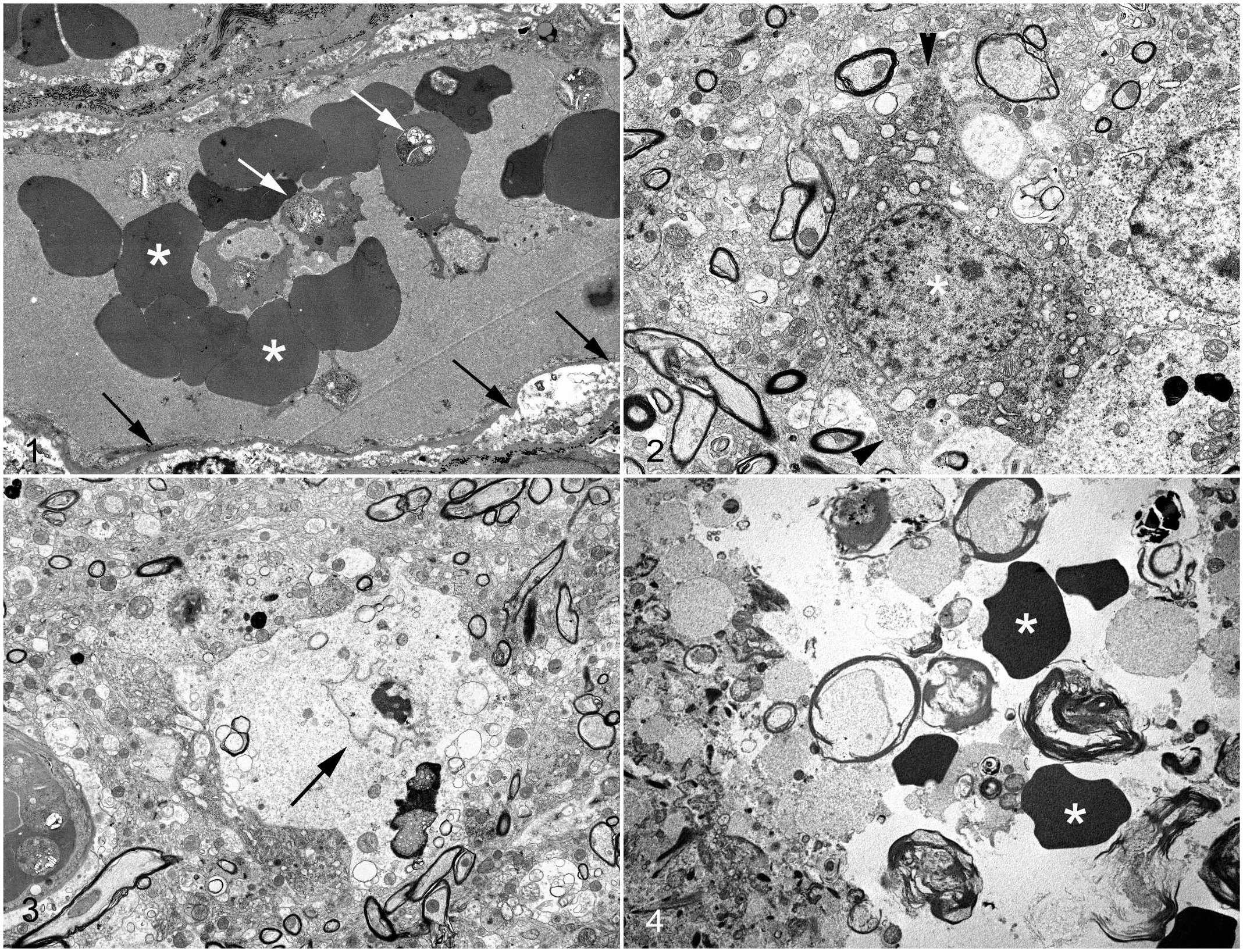
Plasmodium coatneyi infection, brain, rhesus macaque. Transmission electron microscopy.
Central nervous system
In rare instances, there was evidence suggesting irreversible acute cellular swelling, including loss of the nucleolus and chromatin, and angular consolidation of the nucleus (Fig. 2). Cytoplasmic changes consistent with necrosis included residual bodies, lipid droplets and vacuoles, and necrotic debris. (Fig. 3). The plasma membrane, when identifiable, was undulant and fragmented. In rare instances, hemorrhage was observed within the cerebrum in 2/6 animals, in which extravasated erythrocytes separated and surrounded the myelin fibers and disrupted the neuropil (Fig. 4). Histopathological examination showed hemorrhage in the CNS of the P. coatneyi experimental animals, predominantly in the spinal cord (5/6 animals).
Kidney
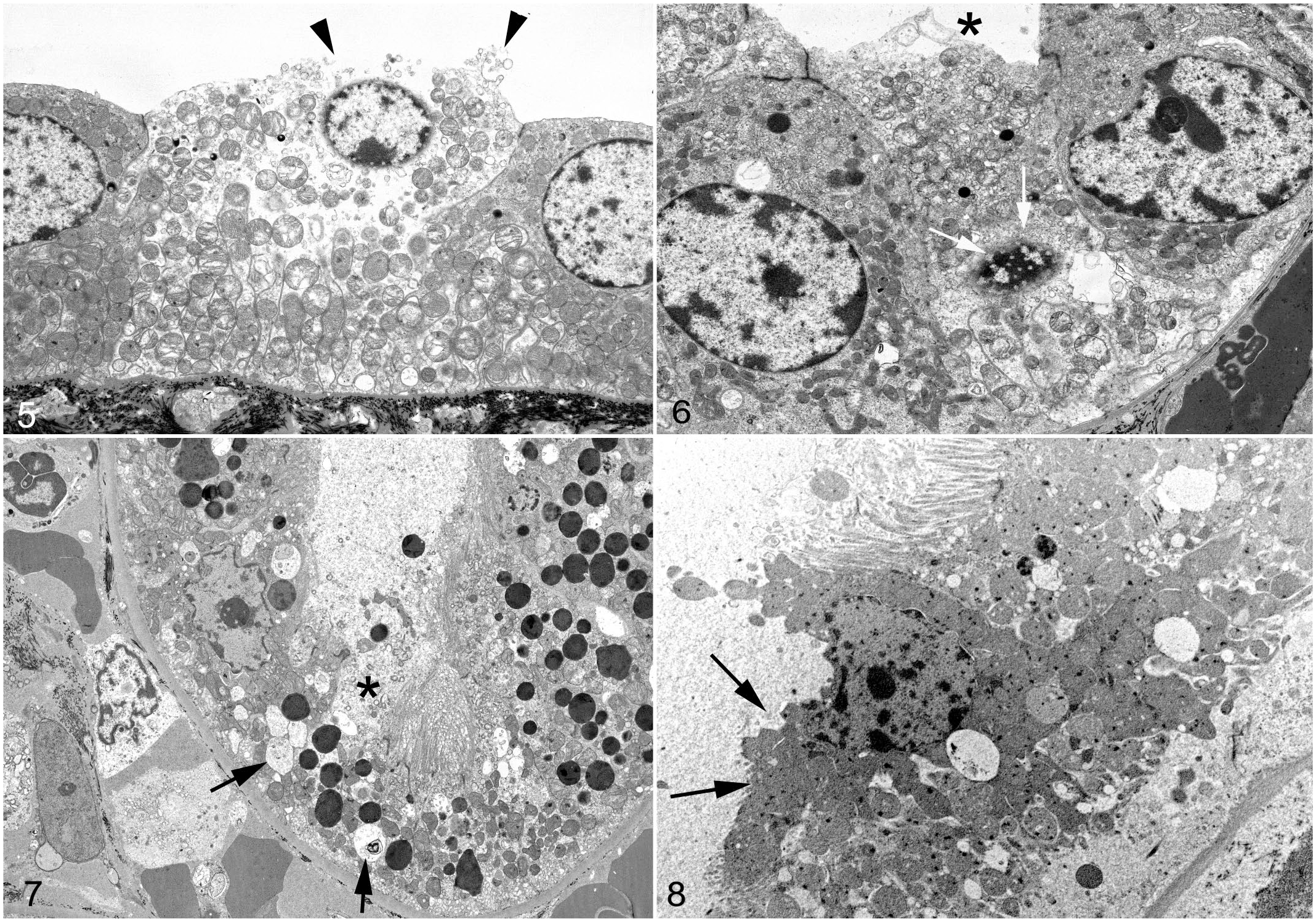
Ultrastructural lesions included the erythrocyte changes described above, in addition to injury to distal and proximal tubules. In the distal tubules, lesions of individual or aggregates of tubular epithelial cells were consistent with degeneration or necrosis and included cytoplasmic swelling, lysis of cytoplasmic elements, swelling of the mitochondria, chromatolysis and condensation of nuclei, and fragmentation of the plasma membrane with extrusion of organelles into the lumen of the distal tubule (Figs. 5–6). Lesions of proximal tubules were similar; in addition, the condensed and fragmented (necrotic) cells had blunting and fusion or complete loss of the apical microvilli. The affected proximal tubular epithelial cells had cytoplasmic and organelle swelling and some lesions had nuclear condensation, margination of condensed chromatin, nuclear fragmentation, extrusion of cellular elements into the tubular lumen in addition to the formation of structures consistent with apoptotic bodies (Figs. 7–8).
Plasmodium coatneyi infection, kidney, rhesus macaque. Transmission electron microscopy.
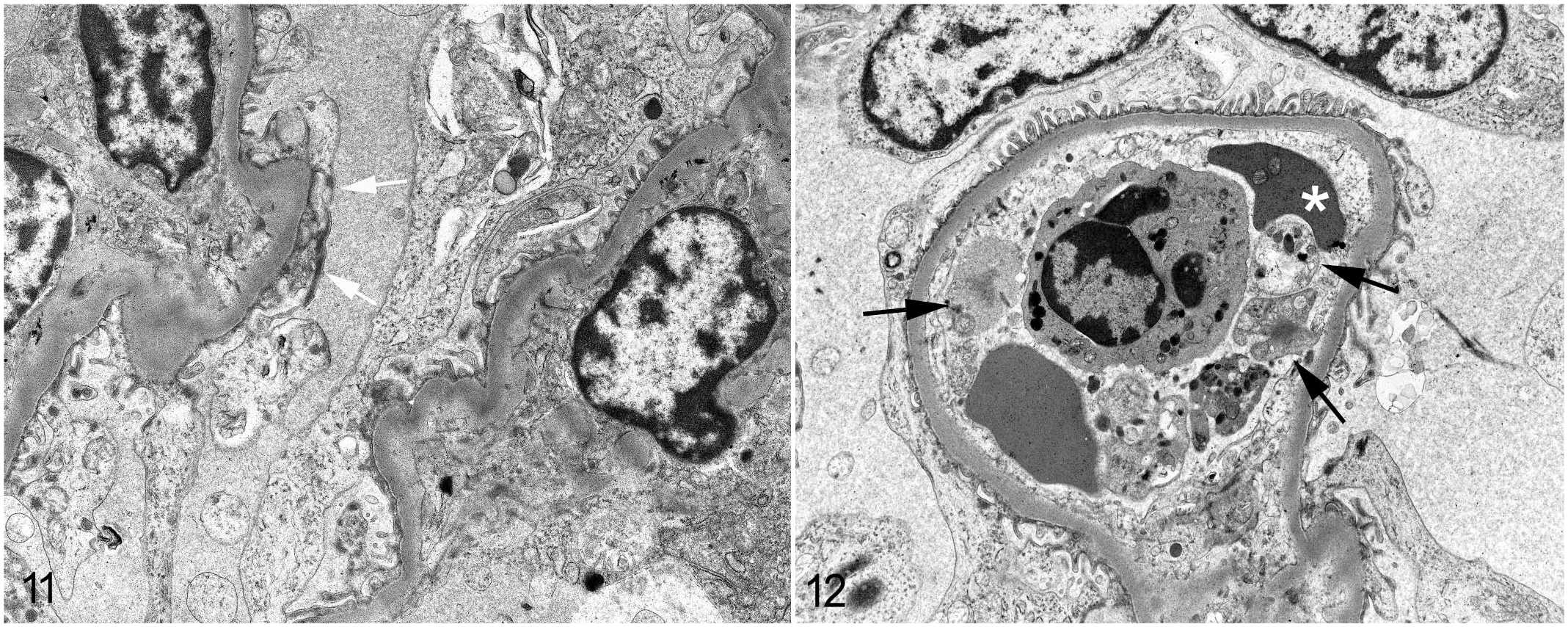
Ultrastructural changes in the glomeruli were subtle. Histopathologically, the glomeruli were often hypercellular and occasionally hypersegmented, and glomerular capillaries were often occluded by infected erythrocytes, high numbers of neutrophils, and frequent macrophages containing phagocytosed cellular debris and hemozoin fragments (suggesting clearance of erythrocyte ghosts, a feature described in P. falciparum–induced disease in humans, 50 in which the “ghosted” or “ghost” erythrocytes stain poorly due to lysis or an absence of hemoglobin subsequent to infection by plasmodiae). In addition, under light microscopy, there were synechial attachments of the glomerular tuft to the glomerular membrane (Figs. 9–10, equivalent to tip lesions seen in proteinuria or minimal change disease), without other evidence of segmental glomerular collapse. Occasionally the foot processes of the podocytes were mildly hypertrophic with evidence of fusion (Fig. 11).
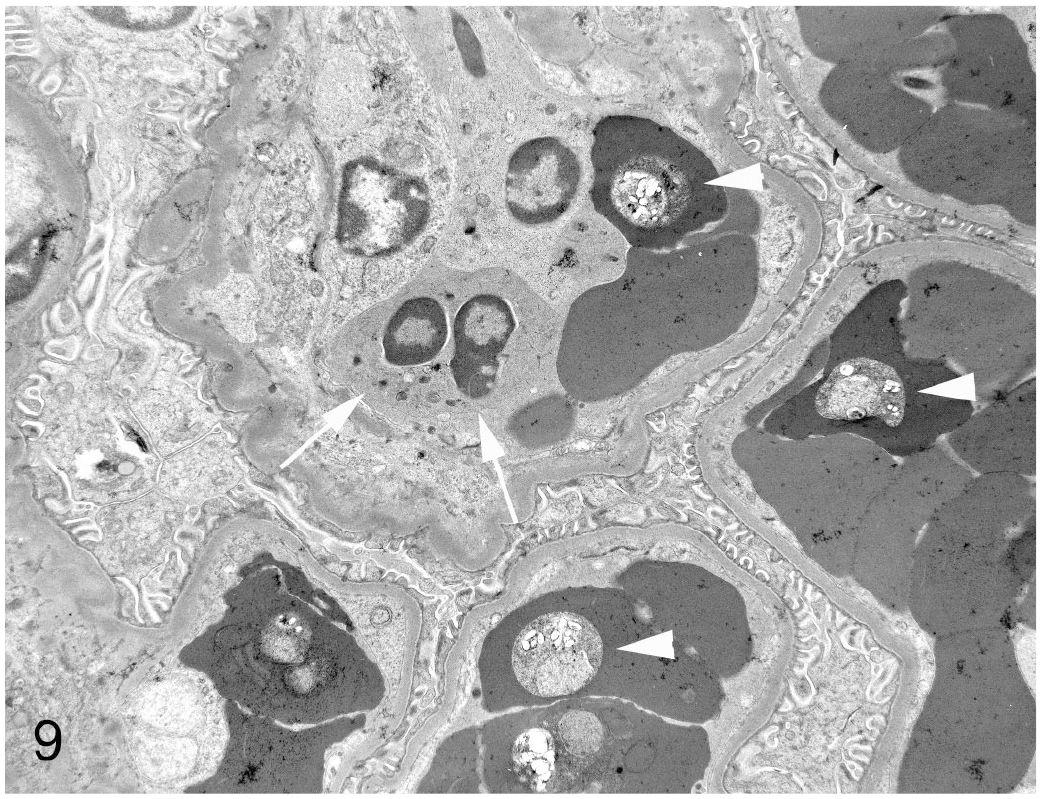
Plasmodium coatneyi infection, kidney, rhesus macaque. A glomerular capillary contains a neutrophil (arrow) and infected erythrocytes containing plasmodial trophozoites (arrowheads). Transmission electron microscopy.
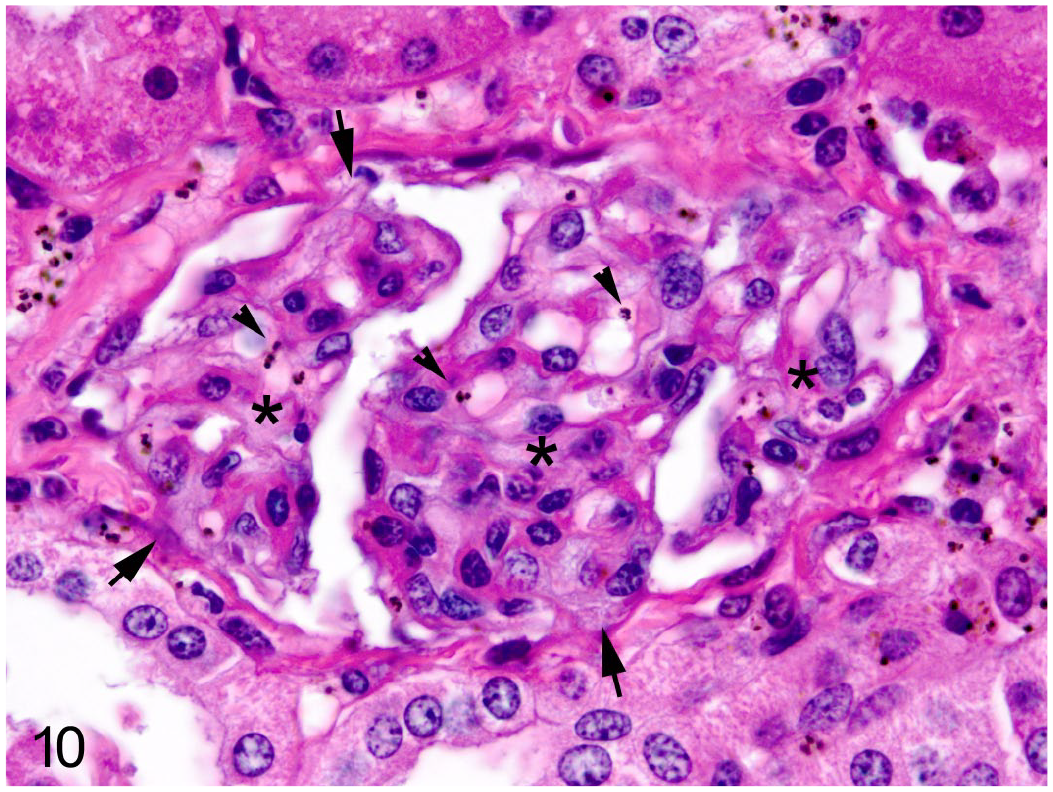
Plasmodium coatneyi infection, kidney, rhesus macaque. A glomerulus has hypersegmentation (asterisks), frequent synechiae (arrows), and infected erythrocytes and hemozoin (arrowheads). Hematoxylin and eosin.
Plasmodium coatneyi infection, kidney, rhesus macaque. Transmission electron microscopy.
There was no evidence of immune complex deposition in the subendothelial tissues. Rather, the basement membrane underlying the podocyte foot processes was typically uniform in thickness and in electron density (Fig. 12).
Heart
Ultrastructural lesions suggested degeneration and necrosis of the cardiomyocytes. There was accumulation of interstitial electron-lucent edema and debris, occlusion and sequestration of the parasitized erythrocytes within the microvasculature, without a significant inflammatory response.
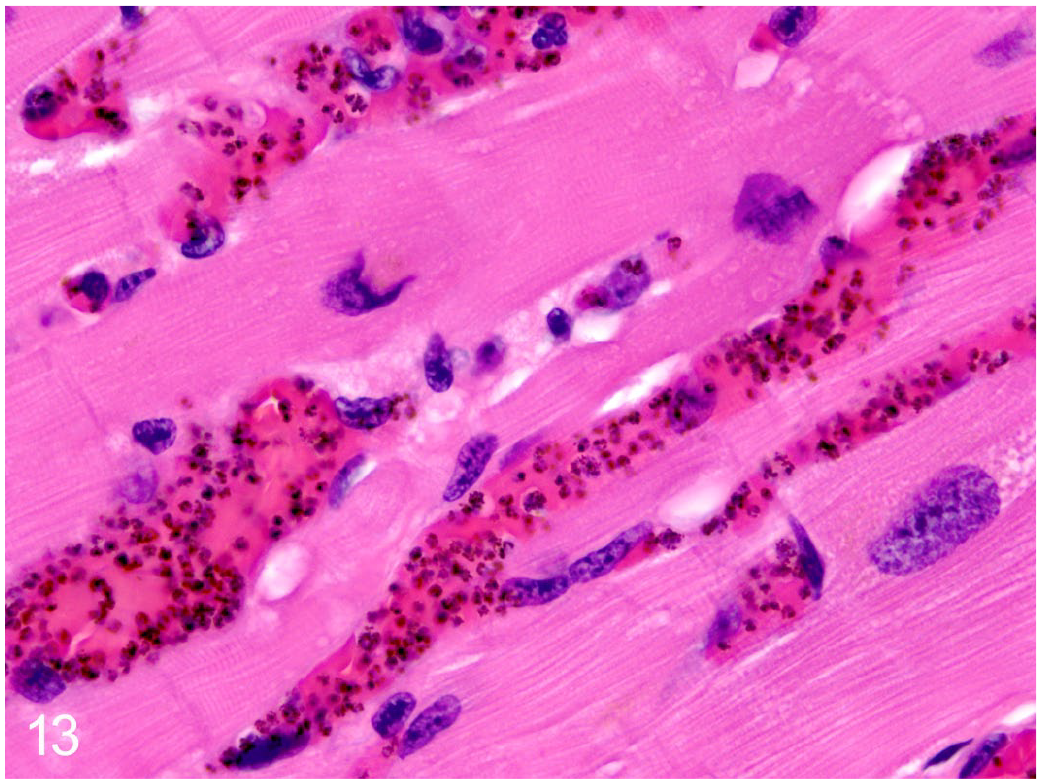
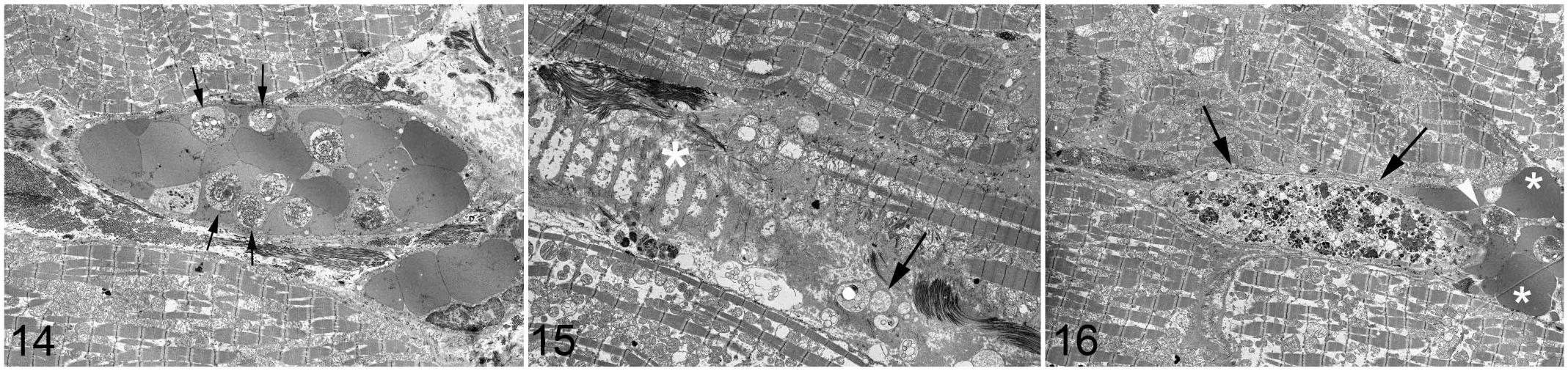
Almost 100% of visible vessels were occluded (microvascular sequestration, Fig. 13). Ultrastructurally, the parasitized erythrocytes were frequently marginated, suggesting cytoadhesion to the endothelium (Fig. 14). The cardiac interstitium, especially in proximity to occluded vessels contained varying amounts of edema. Cardiomyocytes appeared hypertrophied and had degenerative changes including loss of thick (myosin) filaments, and streaming and clumping of Z bands. With increased severity, affected cells became condensed and fragmented, with extensive lysis of myofibrils, loss of T-tubules and mitochondria and the sarcoplasmic reticulum were swollen and electron-lucent. Occasional cardiomyocytes contained electron-dense bodies and were separated from adjacent cells, having lost intracellular connections (Figs. 15–16).
Plasmodium coatneyi infection, heart, rhesus macaque. In the myocardium, the microvasculature has abundant hemozoin and sequestration of infected erythrocytes. Hematoxylin and eosin.
Plasmodium coatneyi infection, heart, rhesus macaque. Transmission electron microscopy.
Lung
In the interstitium, the microvasculature was occluded by infected erythrocytes admixed with frequent monocytes. There was no discernible inflammatory response, alveolar edema, or abnormalities of the alveolar macrophages.
Liver
The ultrastructural lesions were difficult to interpret due to dissociation, degeneration, and necrosis of the hepatocytes. Individual hepatocytes often were expanded by lipid and intracytoplasmic vacuoles. Lesions suggested a progression from degeneration to necrosis with hepatocellular vacuolation, rupture of the plasma membrane, karyolysis, and extrusion of cellular contents. These findings were largely concentrated in centrilobular regions. Hepatic stellate macrophages (Küpffer cells) were increased in number and often contained phagocytosed hemozoin pigment.
Discussion
The pathogenesis of P. falciparum and P. coatneyi infections is proposed to be dependent on the capability of the parasite to remodel the surface membrane of the infected erythrocyte and in doing so induce a range of adhesion proteins that are crucial to survival and pathogenicity of the parasites. P. falciparum infection in humans has been associated with a range of severe life-threatening complications including cerebral malaria, severe anemia, metabolic acidosis, hepatic injury with accompanying jaundice, acute kidney injury, acute respiratory distress syndrome, disseminated intravascular coagulation, malaria retinopathy, and cardiac disease. 68 These conditions are almost universally seen as part of a spectrum of lesions that are interconnected rather than independently responsible for disease.
The search for an accurate animal model that recapitulates the pathology of human disease and mirrors the heterogeneity of human disease remains critically important. The use of a mouse model has been controversial because of differences in pathophysiology, host–parasite interactions, and lesions in rodent and human malarias.8,9,14,28,35,37,66 Although non-human primates have been studied for over 50 years, the parasite–host interaction has only been described in limited numbers of heterogeneous animals and under variable experimental conditions,2,18,19,26,30,54 and lesions have not been described in multiple organs. Furthermore, research on non-human primates infected with P. coatneyi has typically concentrated on a single tissue such as the brain, and none of these studies conducted a systematic analysis of multiple body systems. Similarly, lesions in multiple tissues such as the spinal cord have not been described in human autopsy studies, although this would be critical for comparison with the animal model.
In humans infected with P. falciparum, the kidneys have been reported with acute tubular injury, mild endocapillary (but not immune complex-mediated) glomerulonephritis, and interstitial leukocyte infiltrates, although without classical tubulointerstitial nephritis.7,23,46,61,63 Also in humans, ultrastructural examination of the glomeruli suggested that the basement membrane was either multifocally absent or asymmetrical in thickness. In one study of human infections, mesangial cells were described as being proliferative and containing electron-dense deposits suggesting immune complex nephritis, 24 whereas another study concluded an absence of immune complex glomerulonephritis in cases of acute renal failure. 46 A third study suggested that glomerulopathy was only observed in 20% of autopsied victims of P. falciparum and described prominent mesangial proliferation with only minimal basement membrane ultrastructural changes. 7 Both the histopathological and ultrastructural features of renal disease were closely recapitulated in the model in the current study, including acute kidney injury in some cases. 48
The brevity of the experimental infection (approximately 6–8 days) in the rhesus macaque may not have permitted adequate time for the formation of the subepithelial deposits, or the disease may simply not involve immune complex deposition as a significant factor. Considering that there is still some debate as to the presence or absence of an immune complex pathogenesis in acute renal failure in P. falciparum infection in humans, the lack of lesions in the macaques may simply be a characteristic of both P. coatneyi–driven and P. falciparum–driven diseases. The rhesus macaques of the current study had acute tubular injury with a proliferative glomerulopathy but lacked a significant inflammatory response. This raises the question of variability in disease presentation and suggests that no perfect model may exist.
An example of this variability can be seen in the lungs. Pulmonary lesions are strongly associated with morbidity in severe P. falciparum–induced malaria in humans,21,59 so the absence of significant lung lesions in the rhesus macaque model is an important discrepancy in applying this animal model to human disease. 22
This is the first study to directly focus on cardiac tissue in P. coatneyi infections. There are only scant ultrastructural descriptions of cardiac injury in P. falciparum–induced disease, and those reports typically concentrate on the microvasculature rather than on the cardiomyocytes themselves.39,49 Reports based on single cases describe ischemic lesions secondary to sequestration and occlusion of cardiac microvasculature, 11 a lymphocytic myocarditis in humans, or undiagnosed cardiac disease based on ante-mortem clinical findings.13,43 Given the high levels of sequestration of parasitized erythrocytes and the clinical picture of terminal metabolic derangement and shock, it may be that parasite-induced occlusion of cardiac vessels caused ischemic injury to the cardiomyocytes and contributed to death in this model, via the mechanisms of shock similar to those postulated over a century ago in human postmortem studies of “algid” malaria. 22 Malaria-associated cardiac lesions have been described historically in humans, but are not considered to be a consistent feature of disease.25,56 Erythrocyte sequestration in capillaries in P. falciparum infections is posited to occlude the microvasculature and cause a degree of ischemic cardiomyopathy; however, there are only scant references to this in the current literature.11,13,43
P. coatneyi–infected rhesus and Japanese macaques have been used as animal models for the severe and cerebral human malaria caused by P. falciparum.2,16,29,38,44 Disease caused by P. coatneyi shares a range of pathophysiological, pathological, and ultrastructural features with that caused by P. falciparum, as well as several distinguishing phenotypic characteristics. Both species of plasmodium have 48-hour intraerythrocytic development cycles of asexual multiplication (schizogony) resulting in merozoite formation. The newly released merozoites undergo reinvasion of uninfected erythrocytes and the host has a resultant febrile spike. Both P. falciparum and P. coatneyi develop rosettes comprising one or multiple infected erythrocytes and multiple uninfected erythrocytes.20,37,38,44 They both have well-documented microvascular cytoadherence and both parasites appear to share some variation of knob-like structure on the cell surface hypothesized to be involved in cytoadhesion. 38 Our ultrastructural study focused primarily on the brain, kidneys, and heart and reinforced the fact that P. coatneyi in rhesus macaques undergoes cytoadherence to vascular endothelium and other cells in vivo, and appears to cause microvascular obstruction similar to that seen in human studies of P. falciparum infection, and as such is a strong model of sequestration. There was evidence that suggested infected erythrocytes adhered to the endothelium through binding of the surface ornamentation on the infected erytrhocytes, as well as through a process of interdigitation and pseudopodia formation. Furthermore, there was abundant evidence of autoagglutination and rosette formation. Of importance was the ultrastructural finding that the surface ornamentation of P. coatneyi differed from that of P. falciparum by the lack of regularity of the electron-dense knobs scattered on the surface of erythrocytes infected with mature trophozoites, and the fact that P. coatneyi–infected erythrocytes were also adorned by high numbers of caveolae (a feature more in common with Plasmodium vivax).1,4,6,38 Although the present study represents a single time point in the development of the parasites, caveolae appear to be the predominant surface ornamentation observed with P. coatneyi.1,4,6,38 These are described either as caveolae or as caveola–vesicle complex structures and are also described in P. cynomolgi, P. vivax, and Plasmodium ovale.1,3,5,6,41
P. coatneyi and P. vivax share a propensity for rosette formation.12,38,60 This may reflect phenotypic similarities in caveolae, which have been shown in P. vivax to co-localize with CD71 (transferrin receptor) expression on the infected reticulocyte surface and to mediate adhesion to CD236 (glycophorin) on uninfected erythrocytes during rosette formation. 36 Finally, instead of having Maurer’s clefts, which are P. falciparum–associated membranous structures remodeled into the host cell cytosol and which are involved in a parasite-derived protein trafficking system, P. coatneyi–infected erythrocytes contained single lamellar coiled vesicular structures throughout the cytoplasm. 38 All of these findings were confirmed in samples from spleen-intact monkeys. 38
One of the main pathophysiological questions about severe P. falciparum malaria in humans is the relative role of parasitized erythrocyte sequestration in causing end organ damage. Some investigators believe this to be an epiphenomenon, and that severe disease is a consequence of the host immune response,27,34,57 whereas others support alternative mechanisms such as an apoptosis-driven pathogenesis.33,47,62 Most supporting data arise from murine models of cerebral or severe malaria,14,53,58,65,66,68 although the relevance of this model to human cerebral malaria is questioned due to the lack of sequestration of infected erythrocytes in the mouse brain. In addition, neurological signs following Plasmodium berghei, Plasmodium yoellii, and Plasmodium chabaudi infections in a variety of host murine strains are clearly related to an inflammatory and immune-mediated encephalitis which does not recapitulate the pathological features of human disease.
While the P. coatneyi rhesus macaque model appeared to be appropriate for the study of multi-organ involvement, there were differences from human P. falciparum malaria. Specifically, there was a lack of pneumonitis and pulmonary edema, and there was only rare cerebral (ring) hemorrhage (which is typically concentrated in white matter and in the subcortical white-gray matter vascular watershed areas). The latter is important because the literature concerning P. falciparum has described perivascular edema in association with sequestration and alterations of the cerebral microvascular endothelium, 42 and cerebral disease in humans is marked by typical ring hemorrhages due to microvascular rupture.50,51 Conversely, there were additional findings in the rhesus macaque that were not typical of human disease, such as the severity of hepatic and cardiac injury.
This study is the first systematic ultrastructural survey of tissue lesions associated with P. coatneyi infection in a non-human primate model and underscores both its strengths and weaknesses as a model of P. falciparum–induced tissue injury in humans.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E. D. Lombardini is a Colonel in the US Army. The opinions or assertions herein are those of the authors and do not necessarily reflect the views of the US Department of the Army or Department of Defense.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
