Abstract
Cryptosporidiosis is a common opportunistic infection in the gastrointestinal tract of human and nonhuman primates with AIDS. Pulmonary infection associated with Cryptosporidium spp. has not been previously reported in monkeys. Two macaques experimentally infected with simian immunodeficiency virus (SIV) had lesions containing cryptosporidial organisms involving the trachea, lungs, bile ducts, pancreas, and intestine. The pulmonary sections revealed moderate to severe bronchopneumonia associated with cryptosporidiosis. Numerous 2-4 μm oval Cryptosporidium spp. organisms were present in the cytoplasm of alveolar macrophages, multinucleated giant cells, and intestinal epithelial cells. Giant cells were positive for SIV by in situ hybridization. These are the first reported cases of cryptosporidiosis with involvement of pulmonary parenchyma in SIV-infected macaques.
Cryptosporidium species are protozoan parasites that cause diarrhea in a wide variety of mammals. 1 5 16 Cryptosporidiosis is a common opportunistic infection in immunosuppressed people, 9 11 including patients with AIDS. 7 8 The disease has been frequently observed in the biliary system, stomach, and intestine. 8 Respiratory cryptosporidiosis involving the trachea, bronchi, and, rarely, the pulmonary parenchyma has been seen in immunosuppressed human patients with respiratory failure. 4 6 7 11 13 In simian immunodeficiency virus (SIV)-infected macaques, Cryptosporidium spp. has been frequently observed in the small intestine, gallbladder, and biliary and pancreatic ducts. 2 3 12 14 15 17 18 Although respiratory cryptosporidiosis involving the trachea and bronchi has been found in association with intestinal involvement in humans, 13 involvement of pulmonary parenchyma has not been reported in macaques. Here, we report an unusual bronchopneumonia associated with Cryptosporidium spp. in two SIV-infected rhesus macaques.
The macaques were born and raised at the New England Regional Primate Research Center (NERPRC); after inoculation with SIV, they were housed individually in high-energy–particulate air-filtered stainless steel cages in an Association for Assessment and Accreditation of Laboratory Animal Care-approved Biosafety level 3 animal facility. Room temperature was maintained at 20–24 C; the relative humidity was 40–70%, with 12–15 changes of 100% fresh air per hour. A 12-hour light/dark cycle was maintained. The diet consisted of commercial chow (Old World Monkey Chow 5045, Purina, St. Louis, MO) supplemented with fresh washed fruit once daily, and water was provided ad libitum.
A male rhesus macaque (macaque no. 1) was inoculated with SIVmac239, a pathogenic clone of SIV, intravenously at 16 months of age. Approximately 7 years after inoculation, the animal showed persistent diarrhea and marked weight loss and was euthanatized. Macaque no. 2 was a male rhesus macaque that was infected intrarectally with SIVmac239 at 14 months of age. Approximately 5 months after inoculation, macaque no. 2 developed dyspnea, anorexia, diarrhea, and generalized wasting; the animal collapsed and was euthanatized.
At necropsy, macaque no. 1 showed moderate emaciation, extensive alopecia, and evidence of diarrhea. The lung lobes demonstrated moderate irregular consolidation, especially in the right middle and accessory lobes. The spleen and the peripheral lymph nodes were small. The small intestine was distended with liquid contents, and the large intestine was dilated with semiliquid, tan feces. Macaque no. 2 was moderately emaciated. The lungs had irregular consolidated areas involving the right middle, accessory, and left cranial lobes. The liver had prominent intrahepatic bile ducts; the gallbladder had a markedly thickened wall with reduced luminal space containing a small amount of clear, yellow bile. The small and large intestines contained a moderate amount of watery material. The spleen was moderately enlarged with prominent white pulp, and the mesenteric and iliac nodes were moderately enlarged.
Tissues were collected and fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). Immunohistochemistry was performed on prepared slides using the avidin–biotin–horseradish peroxidase (ABC) method with primary antibodies against Cryptosporidium spp. (Biogenesis, Kingston, NH), Pneumocystis carinii (Dako Corporation, Carpinteria, CA), rhesus cytomegalovirus (CMV; provided by Dr. Peter Barry, University of California, Davis, CA) and adenovirus (Chemicon, Temecula, CA). In situ hybridization for SIV was performed as previously described, 10 using DNA probes labeled with digoxigenin by random priming (Boehringer Mannheim, Indianapolis, IN). The SIV probe was obtained from Dr. Ronald Desrosiers and Dean Regier, NERPRC.
Histopathologically, both macaques had severe disseminated cryptosporidiosis involving the digestive and respiratory systems (Table 1). Both animals had moderate to severe bronchopneumonia characterized by multifocal suppurative or subacute inflammation (Fig. 1). Cryptosporidium spp. were identified as weakly basophilic, oval, 2–4-μm organisms in the alveoli. The bronchial epithelium and an occasional peribronchiolar gland were hyperplastic, with marked diffuse squamous metaplasia and many Cryptosporidium spp. organisms attached to the luminal surface; various amounts of suppurative exudate were seen in the lumen. Affected alveoli contained a large number of neutrophils and macrophages, with moderate mucus accumulation. Alveolar septa were moderately thickened with an infiltrate of macrophages and lymphocytes. Numerous Cryptosporidium spp. organisms were found free in alveoli and within alveolar macrophages. Macaque no. 2 had more extensive lesions in lobular bronchi and terminal bronchioles than macaque no. 1 (Fig. 1); these lesions consisted of multifocal aggregates of multinucleated giant cells (MNGC; Fig. 2) that contained a few Cryptosporidium spp. organisms (Fig. 3). MNGC were positive for SIV by in situ hybridization (Fig. 3, inset). Additionally, macaque no. 2 had multifocal areas of eosinophilic foamy exudate characteristic of Pneumocystis carinii (PC) infection admixed with the Cryptosporidium spp. organisms. The presence of PC was confirmed by immunohistochemistry (Fig. 4).
Pathological features of pulmonary cryptosporidiosis in two SIV-infected rhesus macaques.∗

− = negative; + = slight; ++ = moderate; +++ = severe; IH = immunohistochemistry; CMV = rhesus cytomegalovirus.
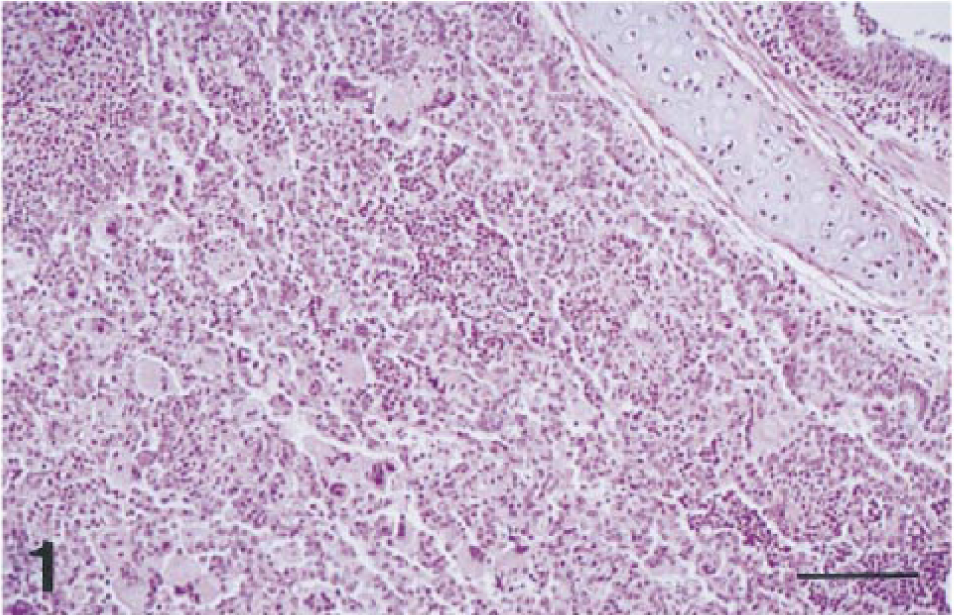
Rhesus macaque no. 2. Pulmonary cryptosporidiosis (right middle lobe). The lobular bronchus (upper right) has marked squamous metaplasia. Numerous multinucleated giant cells admixed with macrophages and neutrophils are seen in the lesion. HE. Bar = 160 μm.
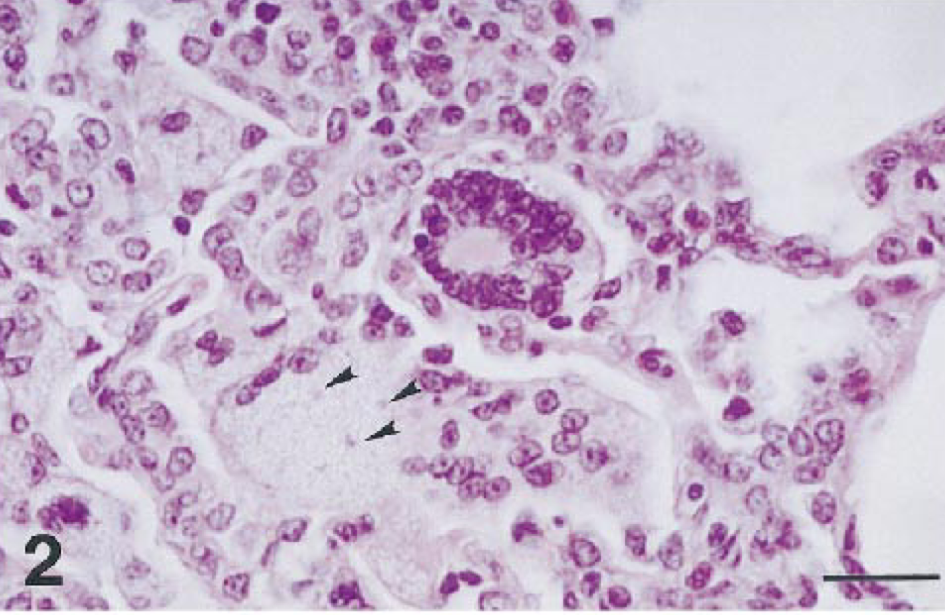
Rhesus macaque no. 2. Pulmonary cryptosporidiosis. The multinucleated giant cells contain subtle basophilic, oval Cryptosporidium spp. organisms in the cytoplasm (arrowheads). HE. Bar = 32 μm.
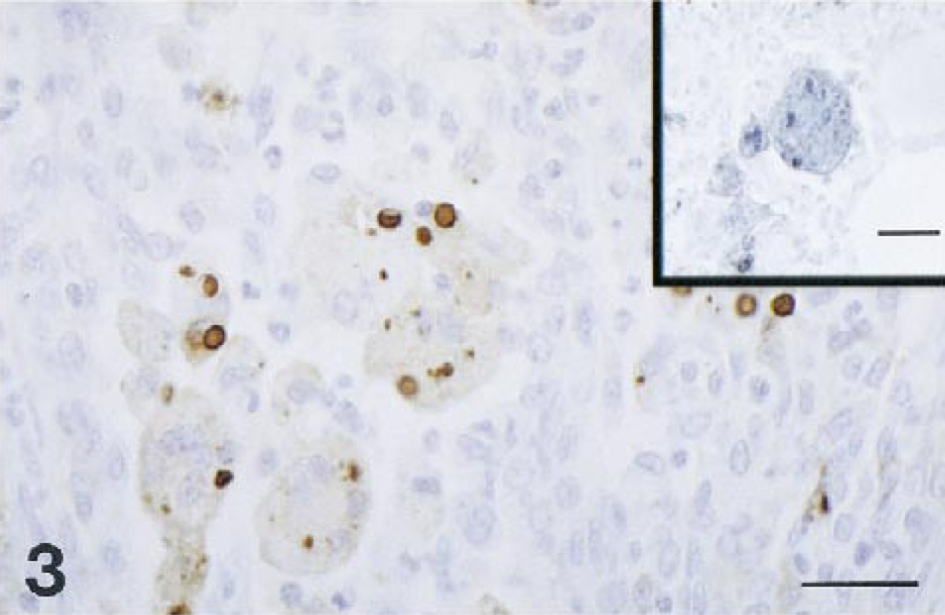
Rhesus macaque no. 2. Pulmonary cryptosporidiosis. Immunohistochemistry for Cryptosporidium spp. by ABC method. Bar = 32 μm. Inset: SIV in situ hybridization demonstrates positive multinucleated giant cells in alveoli. Bar = 28 μm.
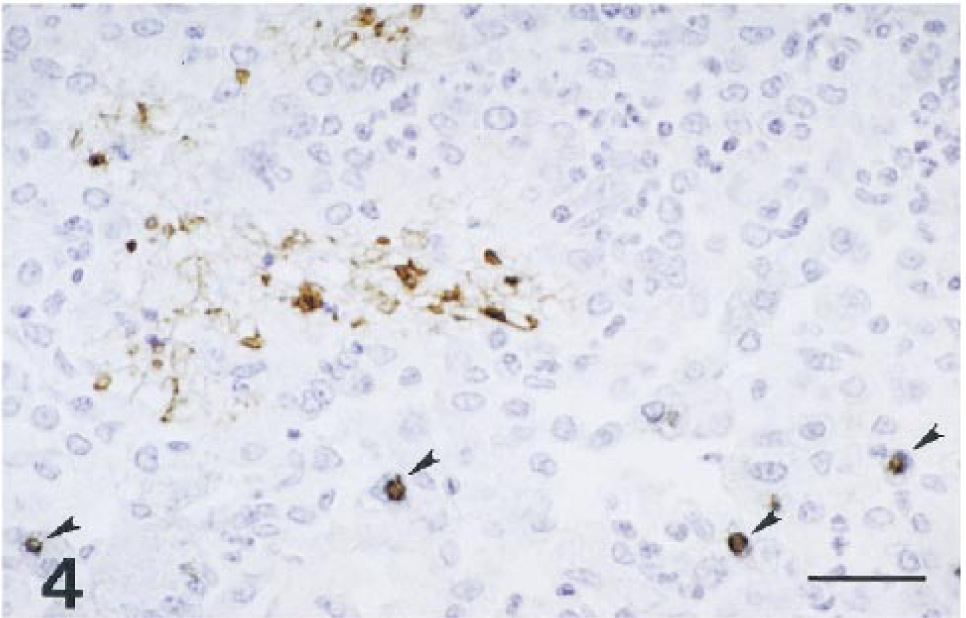
Rhesus macaque no. 2. Double immunohistochemical staining for Cryptosporidium and Pneumocystis carinii. The photomicrograph demonstrates positive reactions for both Cryptosporidium (brown-blue; arrowheads) and Pneumocystis carinii (brown). ABC method. Bar = 32 μm.
Other lesions were noted in these SIV-infected macaques. There was severe tracheitis with marked epithelial squamous metaplasia and submucosal lymphocytic infiltration. Large- to medium-sized pancreatic ducts demonstrated severe epithelial hyperplasia with varying degrees of duct proliferation; inflammatory infiltrates composed of lymphocytes, eosinophils, and neutrophils extended into small ducts. Cryptosporidium spp. organisms were noted at the apical surfaces of epithelial cells in the pancreatic ducts. In the liver of macaque no. 2, there was severe multifocal cholangiohepatitis and cholecystitis characterized by prominent inflammatory cell infiltrates and fibrosis in the portal areas with varying numbers of Cryptosporidium spp. organisms in bile ducts. In the intestine, macaque no. 1 had severe diffuse subacute enteritis with myriad Cryptosporidium spp. organisms both on the surface epithelium and deep in the crypts throughout the entire small intestine. Macaque no. 2 had moderate multifocal cryptosporidiosis with frequent crypt abscesses in the rectum.
AIDS-related lymphoid alterations were seen in both animals. Macaque no. 1 had generalized lymphoid depletion. Macaque no. 2 showed moderate general lymphoid hyperplasia due to paracortical expansion; mesenteric nodes had multifocal aggregates of SIV-positive MNGCs in sinuses. These syncytial giant cells are most common in the lungs, brain, and secondary lymphoid organs of SIV-infected macaques late in disease when the immune system can no longer restrict viral replication. 12 Other opportunistic infections included intestinal trichomoniasis, adenoviral inclusions in the jejunal epithelium, and CMV-positive inclusions in the tubular epithelium in the kidney. The lymphoid changes, including lymphoid depletion and hyperplasia, plus a number of opportunistic infections, are symptoms of AIDS due to SIV infection in rhesus macaques. 12 17
Cryptosporidium spp. associated diarrheal illness has been reported in several species, including cows, sheep, monkeys, and humans. 5 In immunosuppressed populations, especially AIDS patients 7 and SIV-infected macaques, 3 disseminated cryptosporidiosis in digestive organs with severe persistent diarrhea can be life threatening. 8
Respiratory cryptosporidiosis has been reported in immunosuppressed human patients such as those with AIDS. 4 6 13 From 1983 to 1996, approximately 13 cases of pulmonary cryptosporidiosis involving only the upper tracheobronchial region were reported in human patients. 13 Cryptosporidium spp. was detected microscopically in the lung parenchyma in only two cases, one involving a patient with AIDS 4 and the other a patient with acute nonlymphatic leukemia. 9 Two of four SIV/Delta experimentally infected macaques demonstrated cryptosporidial organisms in the trachea and bronchioles. 3 One of these SIV/Delta–infected animals had concurrent Pneumocystis carinii infection. 3 There have been no previous reports of pulmonary cryptosporidiosis extensively involving the alveoli in SIV-infected macaques.
The route of exposure and pathogenesis of these cases of pulmonary cryptosporidiosis is uncertain. Inhalation of infective oocysts from feces is considered to be most likely in macaques as in humans. 13 Further studies may clarify the route of infection and pathogenesis of pulmonary cryptosporidiosis in SIV-infected macaques and in AIDS patients.
Footnotes
Acknowledgements
We thank Ms. Heather Knight and Mr. William Kennedy for in situ hybridization, Mr. Douglas Pauley for immunohistochemistry and proofreading of the manuscript, Ms. Kristen Toohey for excellent photos, and Ron Desrosiers for use of tissues collected from these animals. This study was supported by Public Health Service grants RR00168 and RR07000 and Japanese Educational Ministry grant 10306020.
