Abstract
A 12-year-old, neutered, male Belgian Malinois/Great Dane cross dog presented with a 5-month history of weakness and lack of endurance followed by acute onset of rear limb ataxia. At autopsy, a 9 x 16 mm, multilobular, firm, white to tan, expansile mass was found in the cerebellum. Mild dilatation of the lateral ventricles was also noted. Histologically, there was a well-demarcated glial neoplasm composed of medium-sized astrocytic elements that had homogeneous cytoplasm, sometimes with globular eosinophilic inclusions, irregular peripherally located nuclei with a single nucleolus, and short cytoplasmic processes. Prominent peri-vascular pseudorosettes with cellular processes in contact with blood vessels were present. Some blood vessels exhibited hyalinized walls. Mitotic figures were not observed. Immunohistochemically, neoplastic cells expressed glial fibrillary acidic protein and vimentin. These features are consistent with an astroblastoma. This is the first clinicopathologic correlation and detailed description of a low-grade glial tumor with features of astroblastoma in a dog.
Astroblastoma is a rare, often well-circumscribed, and slightly lobulated glial neoplasm that was first described in humans by Bailey and Cushing in 1926. 1 Since then, there has been debate on its existence as a distinct entity 5 because astrocytomas and ependymomas can have focal “astroblastic” differentiation. However, when restrictive diagnostic criteria are considered, astroblastomas have been shown to have characteristic clinical, radiographic, and histopathologic features. 3–5 In humans, astroblastomas usually occur in children and young adults 1–5 and have been associated with certain chromosomal abnormalities that are not found in other glial tumors such as astrocytomas or ependymomas. 4 Radiographically, the tumors are contrast enhancing and solid or solid with cystic areas. 4, 10 Histologically, all astroblastomas are characterized by astroblastic pseudorosettes, and most have prominent vascular hyalinization. The radiating perivascular neoplastic cells express glial fibrillary acidic protein (GFAP) with broad, nontapering processes that extend to a central hyalinized blood vessel. 1–5, 13 The cell of origin of astroblastomas, the primary considerations being astrocytic or ependymal (possibly tanycytic), is still subject to debate. 5, 9, 11, 13 The 2000 World Health Organization's International Classification of Tumors of the CNS classifies astroblastoma as a neuroepithelial tumor of uncertain origin. 9
This report documents the gross, histologic, immunohistochemical, and ultrastructural features of a canine tumor that are strikingly similar to those of astroblastoma in humans.
A 12-year-old, neutered, male Belgian Malinois/Great Dane cross dog had a 5-month history of weakness and lack of endurance followed by acute onset of rear limb ataxia. Disk disease was ruled out clinically. The dog was euthanatized.
At necropsy, there was a 9 × 16 mm multilobular, firm, white to tan mass in the cerebellar vermis, centered slightly to the left of midline that compressed the surrounding tissue. The tumor was not continuous with the ventricular system (Fig. 1). Mild dilatation of the lateral ventricles was also noted. There were no other significant gross findings.
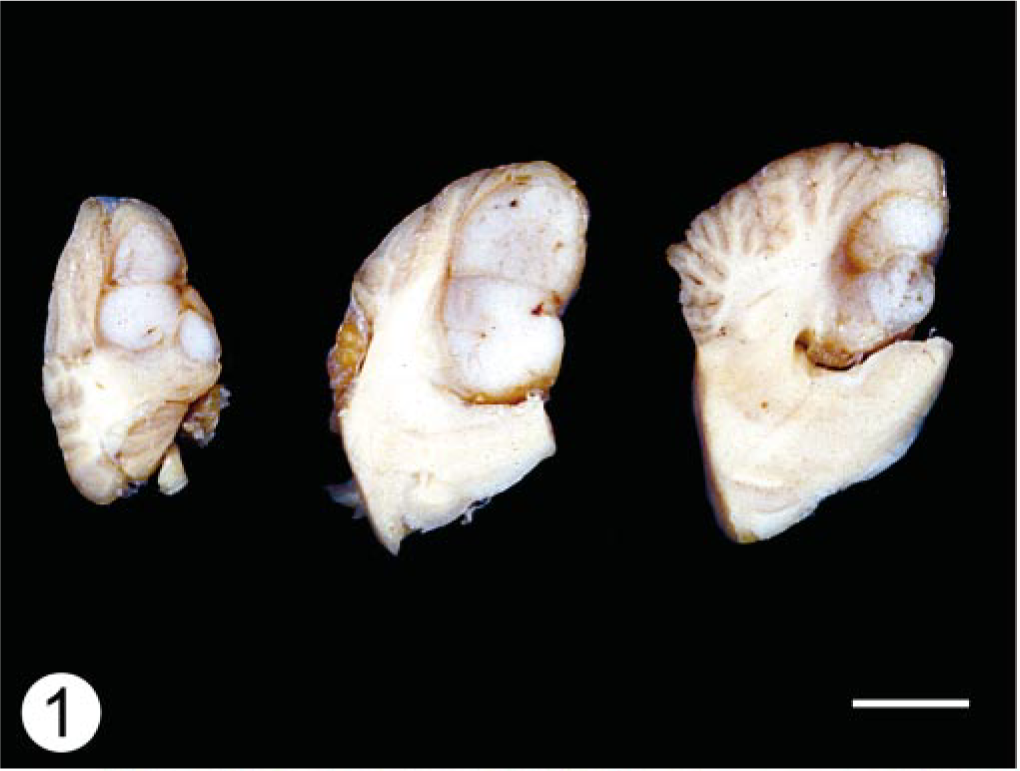
Cerebellum; dog. Sagittal sections demonstrate a well-circumscribed, lobulated mass in the caudal aspect of the cerebellum. Note the displacement and compression of the adjacent folia.
A complete set of tissues including brain was fixed in 10% neutral buffered formalin for histopathologic examination. Tissues were processed routinely and embedded in paraffin blocks. Five-micrometer-thick sections were cut, mounted on positively charged glass slides (Superfrost Plus), and stained with hematoxylin and eosin (HE).
Samples of formalin-fixed tissue were postfixed in 1% osmium tetroxide, dehydrated, and embedded in effepoxy plastic (Ernest F. Fullam), sectioned at 800 Å, and stained with 4% uranyl acetate and Reynolds lead citrate for ultrastructure examination. Sections were examined and photographed on a Zeiss EM10 electron microscope (Leo Electron Microscope Inc., Thornwood, NY).
Additional sections were immunostained for reactivity to vimentin and GFAP by the avidin–biotin–peroxidase complex (ABC) method. Sections were also stained under identical conditions with normal rabbit or normal mouse serum to serve as negative controls. All immunohistochemical stains were counterstained with Mayer's hematoxylin.
Histologically, expanding the cerebellar cortex and white matter and distant from the ventricle, there was a well-demarcated, compact cellular neoplasm composed of polygonal cells arranged in nests and perivascular pseudorosettes. The perivascular pattern occupied approximately 60% of the tumor in the examined sections. Neoplastic cells had uniform, irregularly round to oval nuclei, stippled to clumped chromatin, single nucleoli, and variably distinct cell margins. The perivascular cells had broad, short cellular processes that contacted a centrally located blood vessel (Fig. 2). Many intervascular neoplastic cells contained an eosinophilic globular cytoplasmic inclusion that displaced and indented the nucleus (Fig. 3). The mitotic index was less than one per 10 high-power fields. Frequently, blood vessels had mildly to moderately thickened and hyalinized walls (Fig. 4). Focally along the margin of the tumor, there were several lymphocytes and occasional plasma cells. Within the cerebral white matter of the frontal lobe was a 1-mm-diameter area of necrosis, with reactive astrocytes and macrophages.
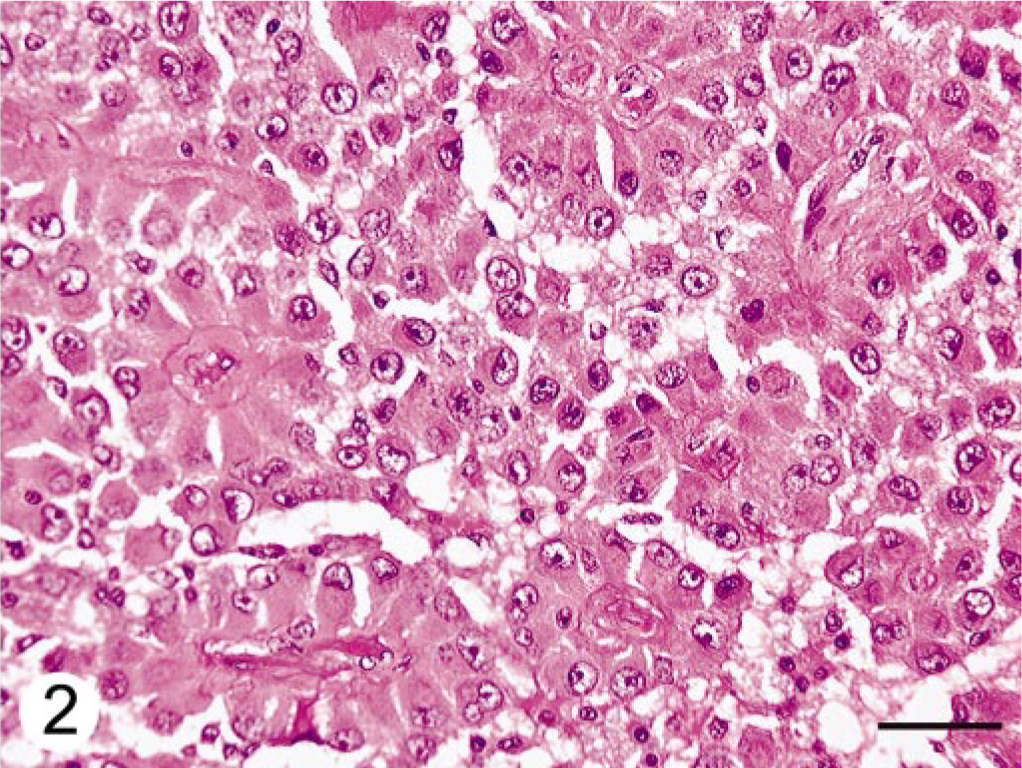
Cerebellar neoplasm; dog. Neoplastic cells are arranged in pseudorosettes. Broad, short cellular processes contact a centrally located blood vessel that often has a thick hyalinized wall. HE. Bar = 50 μm
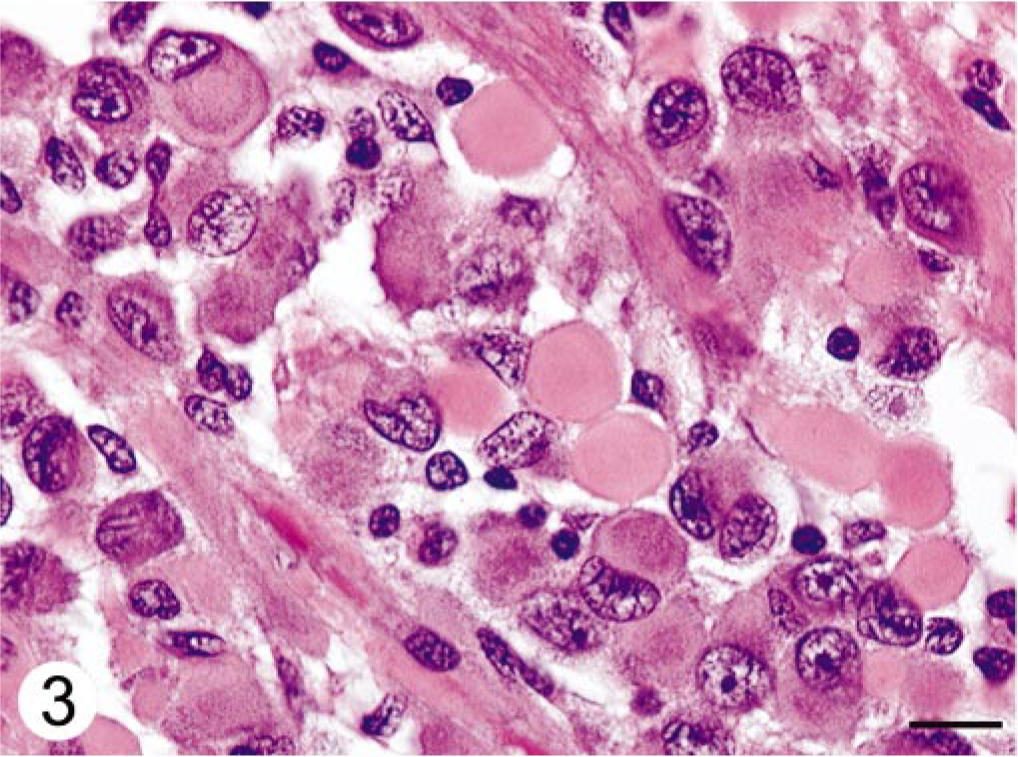
Cerebellar neoplasm; dog. Globular cytoplasmic inclusions within intervascular neoplastic cells. The globular inclusions peripheralize and indent the nucleus. HE. Bar = 10 μm
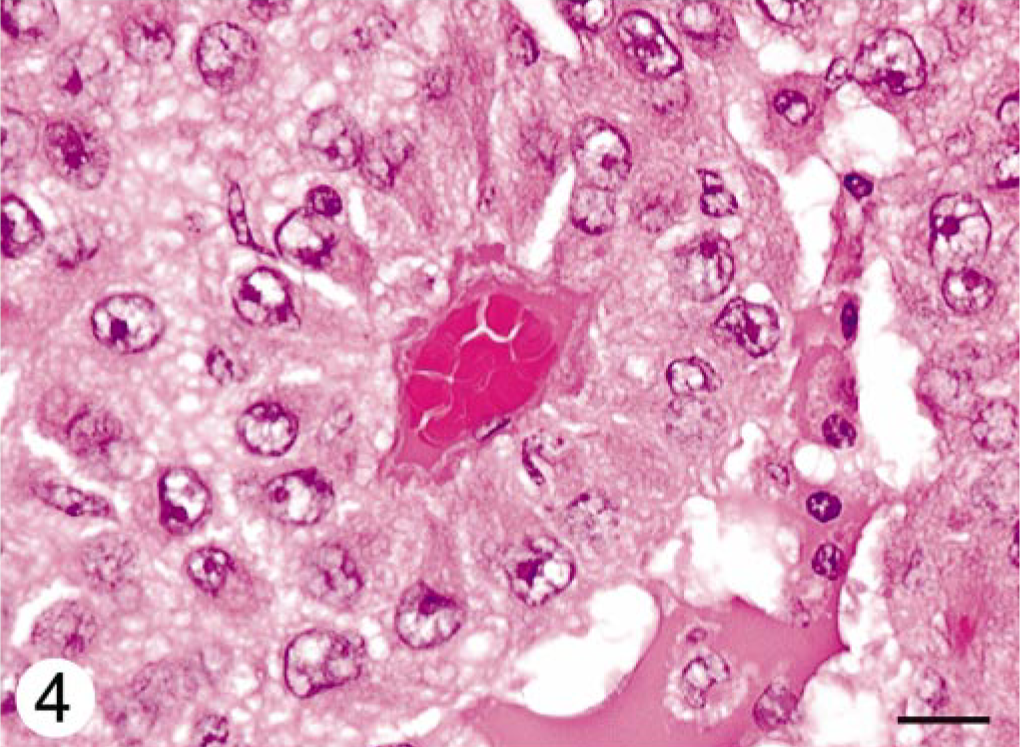
Cerebellar neoplasm; dog. A perivascular pseudorosette with neoplastic cells radiating from a centrally located hyalinized blood vessel. HE. Bar = 10 μm
Immunohistochemically, neoplastic cells forming the perivascular rosettes expressed strong reactivity for GFAP, with mild to moderate positivity of the intervascular cells (Fig. 5). Neoplastic cells including intracytoplasmic inclusions exhibited diffuse strong staining for vimentin (Fig. 6).
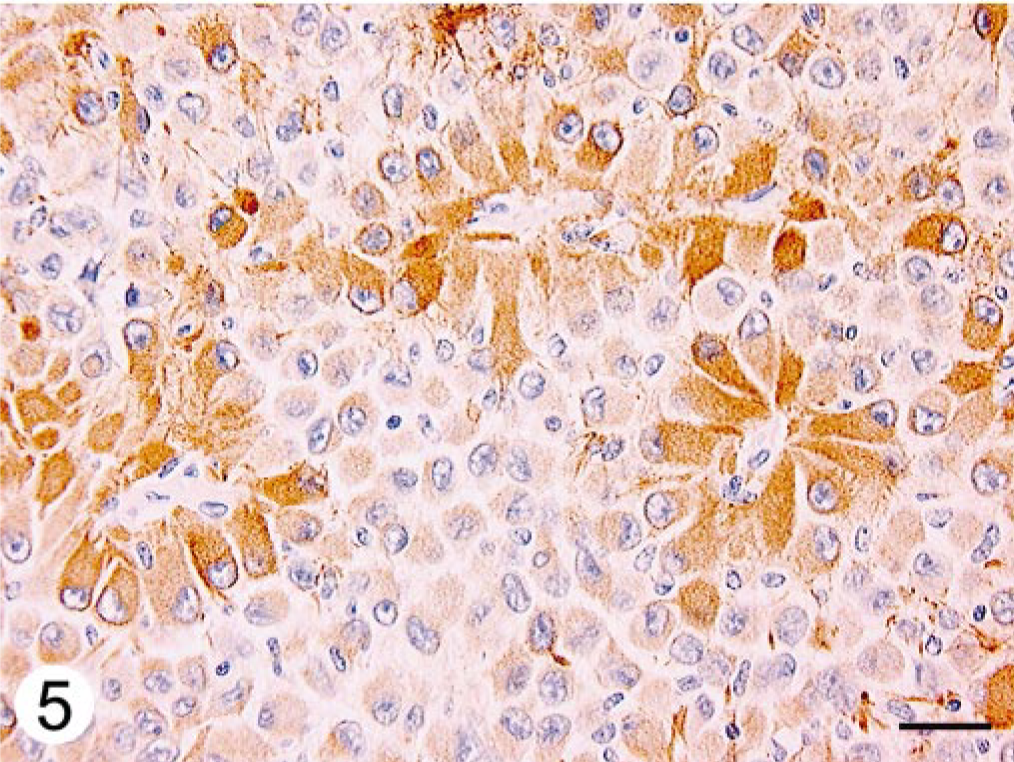
Cerebellar neoplasm; dog. Immunohistochemical staining for GFAP is strongly positive in perivascular neoplastic cells with mild to moderate positivity of the intervascular cells and intracytoplasmic inclusions. ABC immunostain, Mayer's hematoxylin counterstain. Bar = 50 μm
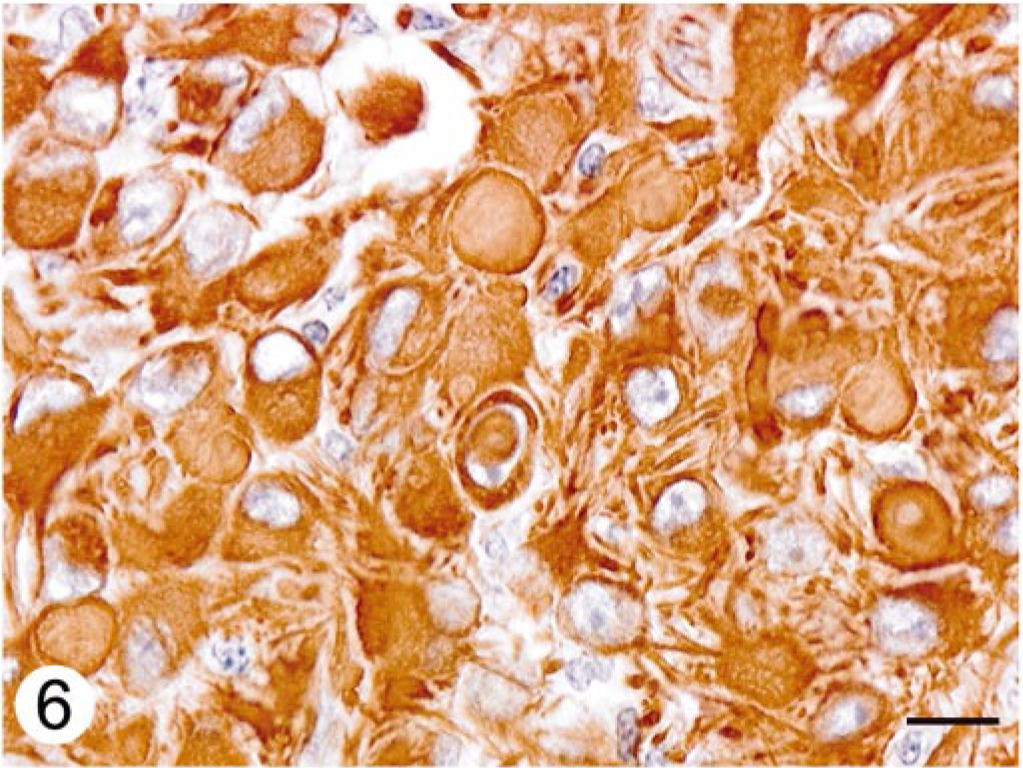
Cerebellar neoplasm; dog. Neoplastic cells including cytoplasmic inclusions are strongly positive for vimentin. ABC immunostain, Mayer's hematoxylin counterstain. Bar = 10 μm.
Ultrastructural characteristics of astroblastoma in humans include neoplastic cells resting upon a continuous basal lamina, surface microvilli extending from a narrowed “purse-string” base, pleating of the cytoplasmic membrane forming interdigitations with occasional intercellular junctions, and variable distributions and numbers of intermediate filaments. 5, 11 The tissue was preserved suboptimally for electron microscopic examination. The cytoplasmic inclusions were composed of paranuclear aggregates of intermediate filaments averaging 14 nm in diameter and arranged in whorls (Fig. 7). The presence of cytoplasmic inclusions composed of aggregates of intermediate filaments and immunoreactive for vimentin and GFAP is compatible with astrocytic elements. No other distinguishing features could be identified.
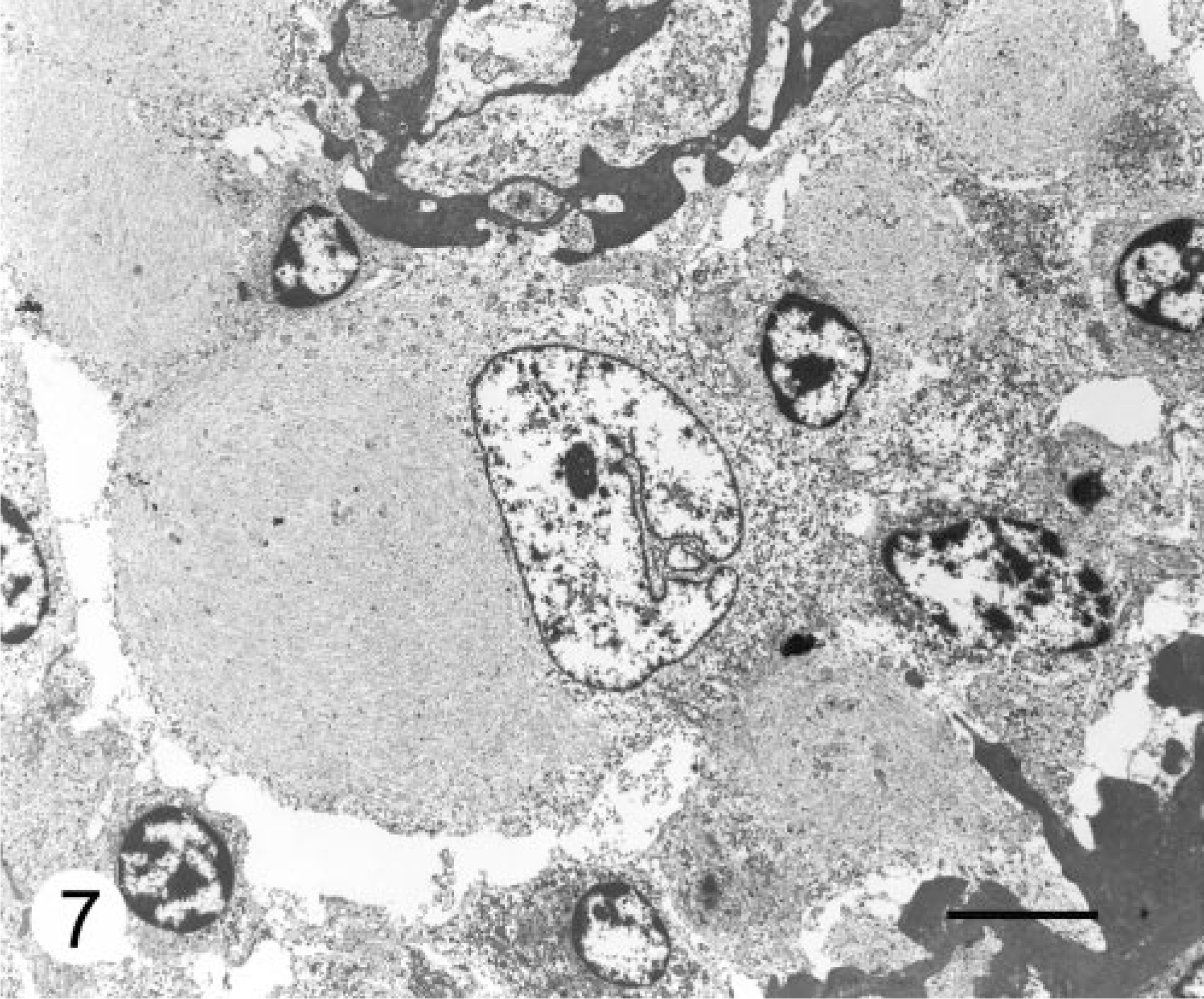
Cerebellar neoplasm; dog. Paranuclear cytoplasmic inclusions composed of aggregates of intermediate filaments, averaging 14 nm in diameter, arranged in whorls. Transmission electron micrograph. Bar = 5 μm.
The differential diagnosis in this dog included astrocytoma, ependymoma, and subependymal giant cell astrocytoma (SEGA).
The well-demarcated nature of this tumor and the uniform nuclei help differentiate it from most astrocytomas. Gemistocytic astrocytomas have more hyperchromatic nuclei, are diffusely infiltrative, and have a corona of processes that are highlighted with GFAP. 5 Although high-grade astrocytomas (glioblastoma multiforme) may be well circumscribed and do exhibit pseudorosettes, the pseudorosettes form around foci of necrosis and not around blood vessels. 5, 8, 13 Furthermore, necrosis and glomeruloid vascular proliferation, characteristic of high-grade astrocytomas, 5, 8, 13 are not present in the tumor of this dog.
The perivascular pseudorosettes and compact cellular nature of this tumor resemble ependymoma; however, the broad and coarse perivascular cytoplasmic processes are distinctly different from the narrow tapering processes of ependymomas. 5, 8, 13 In addition, ependymomas in the posterior fossa are usually ventricular tumors and do not typically exhibit the vascular sclerosis that is present in this tumor. 5
The well-demarcated nature of this tumor and perivascular pseudorosettes are also compatible with SEGA. However, SEGAs are composed of a heterogenous cellular population including ganglion and gemistocytic-like cells that express immunoreactivity for neuronal and glial markers. SEGAs frequently show calcification and are most commonly found along the walls of the lateral ventricles. 5, 7
Two astroblastomas, one from a 10-year-old male Boxer and one from a 6-year-old female Rottweiler, were included in a recent report on the morphology, immunohistochemistry, and genetic alterations of 31 astrocytic tumors in dogs. 12 Both of these previously reported astroblastomas were located in the left cerebral hemisphere. The described histologic features included a papillary growth pattern, elongated neoplastic cells radially arranged around a central vessel, and vascular sclerosis. Immunohistochemically, the neoplastic cells stained positively for GFAP and S-100 protein and negative for p53 and epithelial growth factor receptor. No mitotic activity was reported.
The histologic and immunohistochemical findings of the two previously reported astroblastomas are similar to this dog; however, we did not observe a papillary growth pattern, and in this dog, the tumor was located in the cerebellum.
The clinical significance of this tumor is not known. The reported rear leg ataxia may be related to the presence of the brain tumor, if there were signs of cerebellar ataxia, such as dysmetria. Whereas weakness was also reported and is not a feature of cerebellar ataxia, weakness may be confused for the inability to stand when severely ataxic. 6 The cause of the focal cerebral necrosis is not apparent, but the lesion is suggestive of vascular compromise.
Although most astroblastomas in humans occur in children and young adults and are usually supratentorial, 1–5 they may occur in older adults, i.e., more than 45 years of age, and in other regions of the central nervous system including the cerebellum. 3, 4 Astroblastomas in humans have been associated with abnormalities in chromosomes 20q and 19, which have not been described with other glial neoplasms. 4 Chromosomal analysis was not performed in this dog.
In addition to the histomorphology, features supporting a diagnosis of astroblastoma in this dog include the macroscopic finding of a well-demarcated, lobulated, and solid tumor, presence of abundant cytoplasmic intermediate filaments, and immunohistochemical reactivity pattern. On the basis of this report and a previous report, 12 astroblastoma should be considered in the differential diagnosis for low-grade glial tumors in dogs.
Footnotes
Acknowledgements
We thank Mr. Doug Landry and Mr. Andy Morataya for photographic support, Mr. John Jenkins, for electron microscope service, LTC Dana Scott for assistance in interpretation of the electron micrographs, and MAJ Joy Atkin for technical assistance. The opinions or assertions contained in this study are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army or the Department of Defense.
