Abstract
In a previous study, we characterized the reactivity of monoclonal antibody (MoAb) 8G7G3/1 to thyroid transcription factor-1 (TTF-1) in canine thyroid tumors. In this study, we have examined the reactivity of this antibody in 120 canine pulmonary tumors, including 78 primary epithelial tumors. Tissues had been fixed in formalin and routinely processed for histopathology. Nuclear staining for TTF-1 was detected in 64.2% of primary pulmonary epithelial tumors. The most common TTF-1-reactive tumor types were bronchioloalveolar carcinomas and bronchogenic carcinomas. Staining was diffuse, heterogeneous, or patchy. Nonpulmonary, metastatic epithelial tumors, except two of two thyroid carcinomas, did not react with antibody 8G7G3/1. Mesotheliomas and other mesenchymal tumors were also negative for this marker. A reduction or loss of reactivity was apparent in pulmonary epithelial tumors archived in paraffin blocks for 7-8 years. There was slight reduction in the number of positive cells or the intensity of the reaction in control tissues fixed longer than 1 week. On the basis of our limited studies and the human literature, it appears that MoAb 8G7G3/1 to TTF-1 is a highly specific (with the exception of thyroid tumors) and moderately sensitive marker for canine pulmonary epithelial tumors.
Pulmonary neoplasms are uncommon in dogs, with a prevalence of 0.1–0.9% in the overall canine population; however, when only aging dogs are considered, the percentage of animals with pulmonary tumors may reach 25%.
41
The lung is a common place for metastases of nonpulmonary neoplasms. Metastatic tumors can resemble primary pulmonary tumors microscopically,
9
making it challenging to determine the origin of the tumor on the basis of routine histopathology. Thyroid transcription factor-1 (TTF-1) is a 38-kDa homeo-domain containing nuclear protein that is a member of the
Materials and Methods
Selection of cases
A total of 120 dogs with a histologic diagnosis of pulmonary neoplasia were selected from the databases of the University of Missouri, Veterinary Medical Diagnostic Laboratory, and the Purdue University, Animal Disease Diagnostic Laboratory. Tissues had been fixed in 10% neutral-buffered formalin for an undetermined period of time and embedded in paraffin. Hematoxylin and eosin–stained sections of these tumors were classified by one author (J. A. Ramos-Vara) following a slightly modified (Table 1) established criteria for pulmonary neoplasms 41, without knowledge of the previous diagnosis. In brief, when exact classification was not possible, primary pulmonary tumors (tumors with no clinical history or necropsy findings of extrapulmonary masses) were categorized as carcinoma or adenocarcinoma. Those cases in which the morphologic diagnosis or tissue origin was not definitive were reviewed by another pathologist (M. A. Miller). Immunohistochemistry was also used in selected cases to determine the tissue origin of some tumors of uncertain origin (see Reagents and Immunohistochemical Procedure). When multiple samples of the same tumor were available, only one block from each tumor was selected for immunohistochemical studies.
Reactivity of canine primary and metastatic pulmonary neoplasms with MoAb to TTF-1.
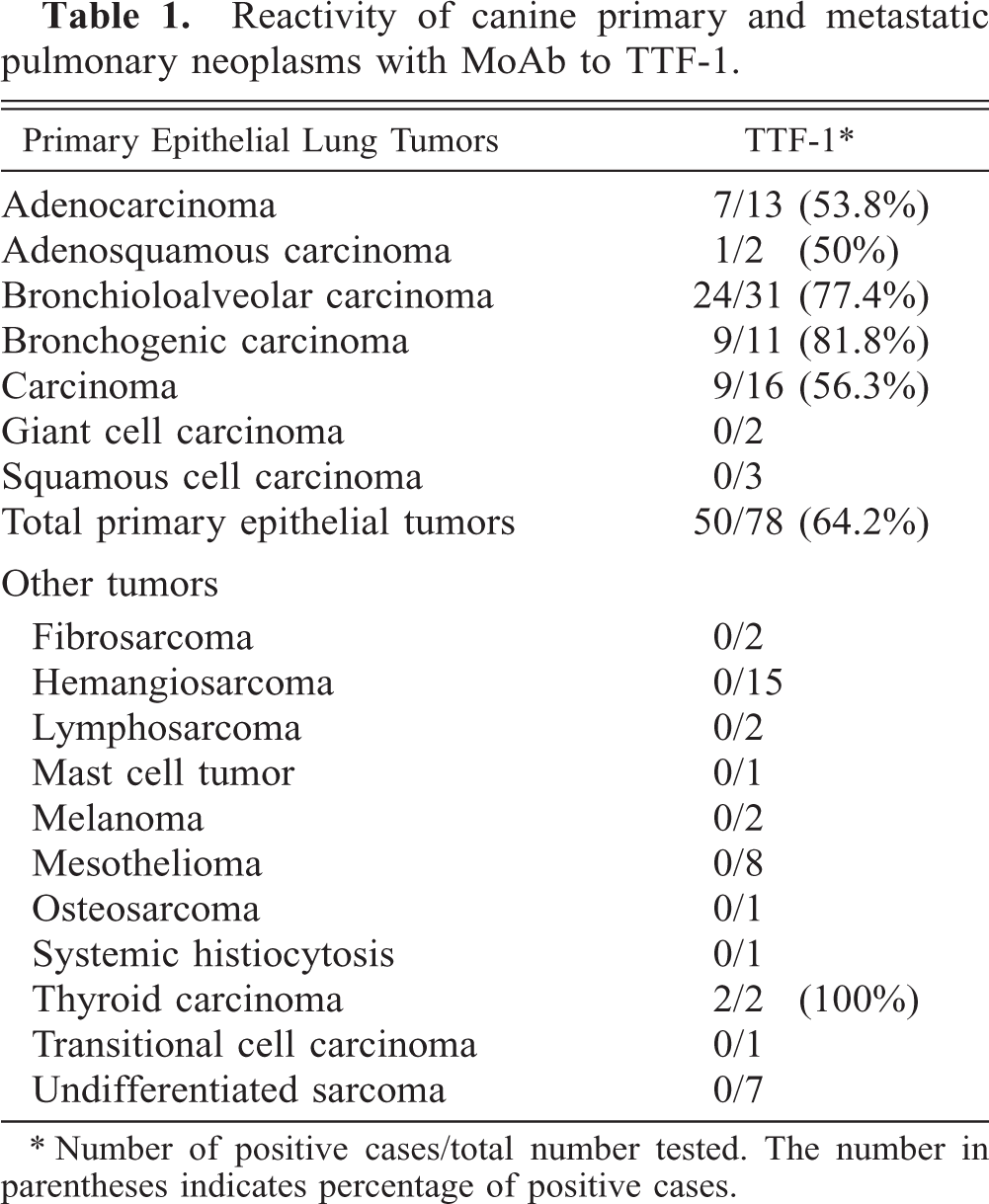
∗ Number of positive cases/total number tested. The number in parentheses indicates percentage of positive cases.
Reagents and immunohistochemical procedure
All the pulmonary tumors were immunohistochemically stained for TTF-1. On selected cases, antibodies to CD3, CD45, CD79a, chromogranin-A, cytokeratins, cytokeratin 7, factor VIII–related antigen, HepPar 1, Melan A, uroplakin III, and vimentin were used according to the methods reported previously. 28–31 We used a mouse MoAb against recombinant rat TTF-1 (M3675, clone 8G7G3/1, diluted at 0.54 μg protein/ml, DakoCytomation, Carpinteria, CA) incubated at room temperature for 60 minutes. 32 The antigen-retrieval method for TTF-1 was steam heat in ethylenediamine tetraacetic acid buffer, pH 8.0, 40 minutes at 90–95° C. For TTF-1, a non-avidin–biotin–peroxidase detection method (EnVision+, K4000, DakoCytomation) was used according to manufacturer's recommendations. Diaminobenzidine was used as chromagen. For negative serum control, we used an unrelated MoAb of the same isotype and similar protein concentration or normal rabbit serum, instead of the primary MoAb or polyclonal antibody, respectively. The scoring of immunohistochemical reaction with MoAb to TTF-1 and the effects of prolonged fixation on the detection of TTF-1 on lung tissue were determined, as reported previously. 32
Results
Seventy-eight primary pulmonary epithelial tumors were examined microscopically (Table 1), including 13 adenocarcinomas, 2 adenosquamous carcinomas, 31 bronchioloalveolar carcinomas, 11 bronchogenic carcinomas, 16 carcinomas, 2 giant cell carcinomas, and 3 squamous cell carcinomas. In addition, pulmonary masses comprising 3 nonpulmonary metastatic carcinomas, 8 mesotheliomas, 25 spindle cell sarcomas, 2 lymphosarcomas, 2 melanomas, 1 mast cell tumor, and 1 systemic histiocytosis were included. As has been reported in the literature, 41 the most common primary pulmonary epithelial tumor in our series was bronchioloalveolar carcinoma.
In the normal lung, MoAb to TTF-1 reacted with the nucleus of alveolar and occasional bronchiolar epithelial cells. The staining was usually of strong intensity, and distribution of positive cells was sometimes heterogeneous within a given section. Cytoplasmic reaction was never detected in normal or neoplastic cells.
TTF-1 immunoreactivity in primary pulmonary epithelial tumors varied among tumor types and was detected in order of decreasing frequency in bronchogenic carcinoma (9 of 11, 81.8%), bronchioloalveolar carcinoma (24 of 31, 77.4%), carcinoma (9 of 16, 56.3%), adenocarcinoma (7 of 13, 53.8%), and adenosquamous carcinomas (one of two, 50%). Giant cell carcinomas (zero of two) and squamous cell carcinomas (zero of three) did not have detectable TTF-1. The overall reactivity for primary epithelial pulmonary tumors was 64.2% (Figs. 1–3). Most bronchogenic carcinomas, bronchioloalveolar carcinomas, and pulmonary carcinomas had moderate to strong immunostaining for TTF-1, but cases with weak or fewer than 10% positive cells were also observed. In cases with weak or reduced staining, careful examination distinguished neoplastic cells from entrapped normal pulmonary epithelium, a common finding especially in tumor margins. The staining in some tumors occurred in the periphery of the mass and, in other tumors, was diffuse. In the single adenosquamous carcinoma positive for TTF-1, only the glandular part of the tumor had reactivity. Markedly atypical cells were sometimes strongly labeled. Intravascular neoplastic cells were usually positive to TTF-1 (Fig. 4). Nonpulmonary carcinomas (with the exception of two of two thyroid carcinomas), mesotheliomas, and mesenchymal tumors did not react with MoAb 8G7G3/1 to TTF-1 (Figs. 5, 6).
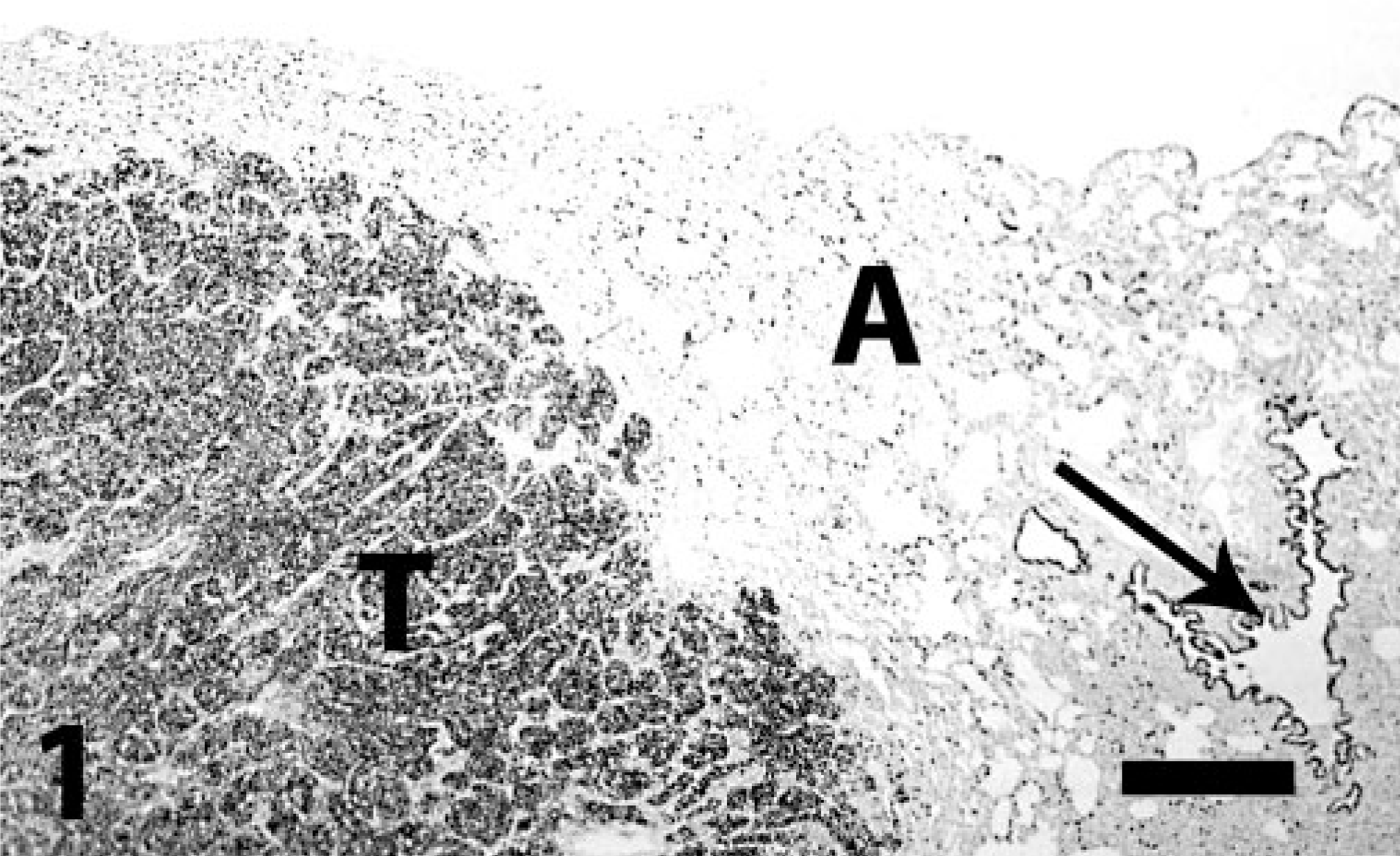
Lung; pulmonary carcinoma; dog. Strong staining for TTF-1 in the neoplastic nodule (T), alveolar epithelial cells (A), and in bronchiolar epithelial cells (arrow). Immunoperoxidase-DAB for TTF-1. Bar = 260 μm.
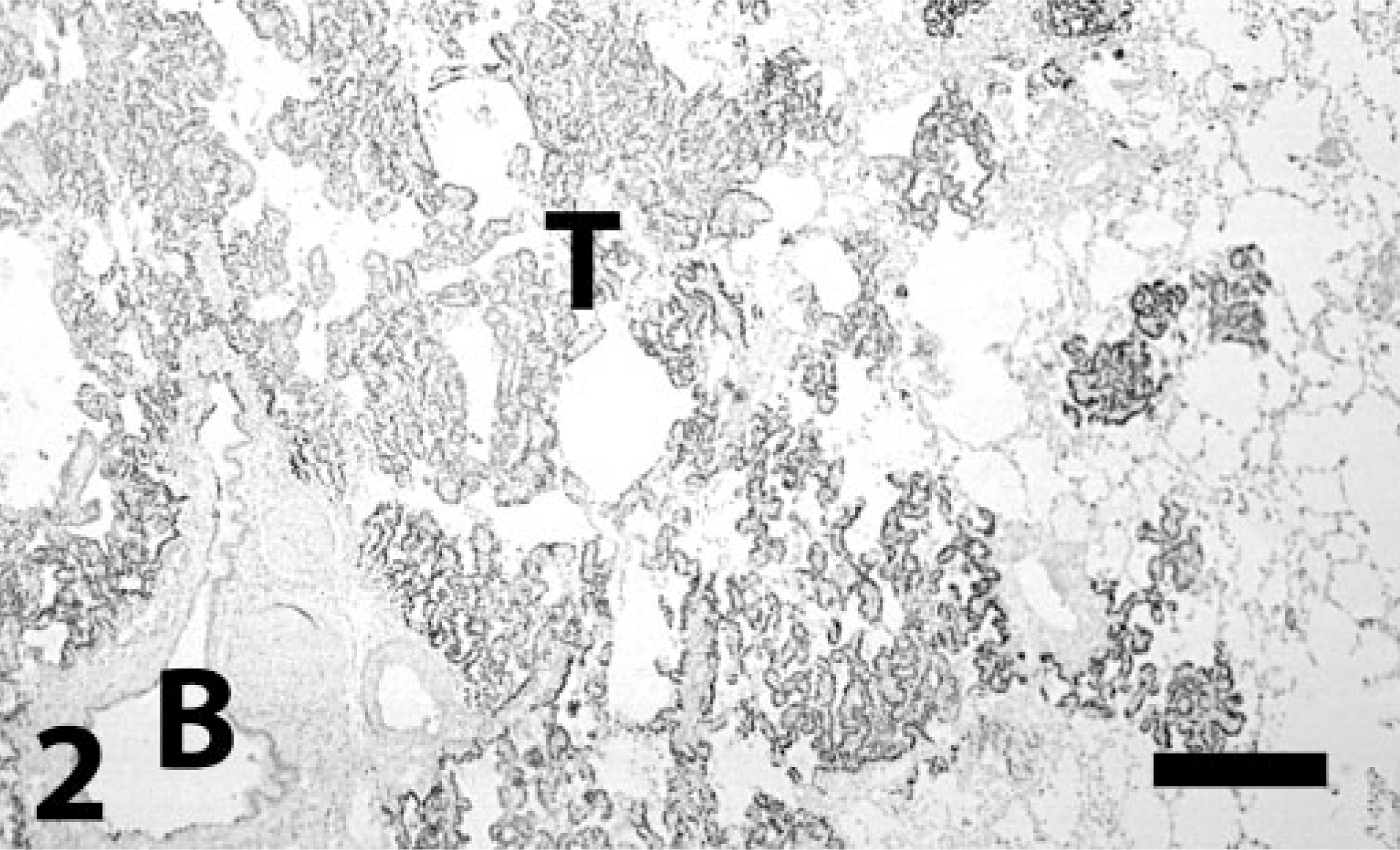
Lung; bronchioloalveolar carcinoma; dog. Typical growth pattern of neoplastic cells that are strongly reactive for TTF-1 (T). B = bronchiole with positive cells. Immunoperoxidase-DAB for TTF-1. Bar = 260 μm.
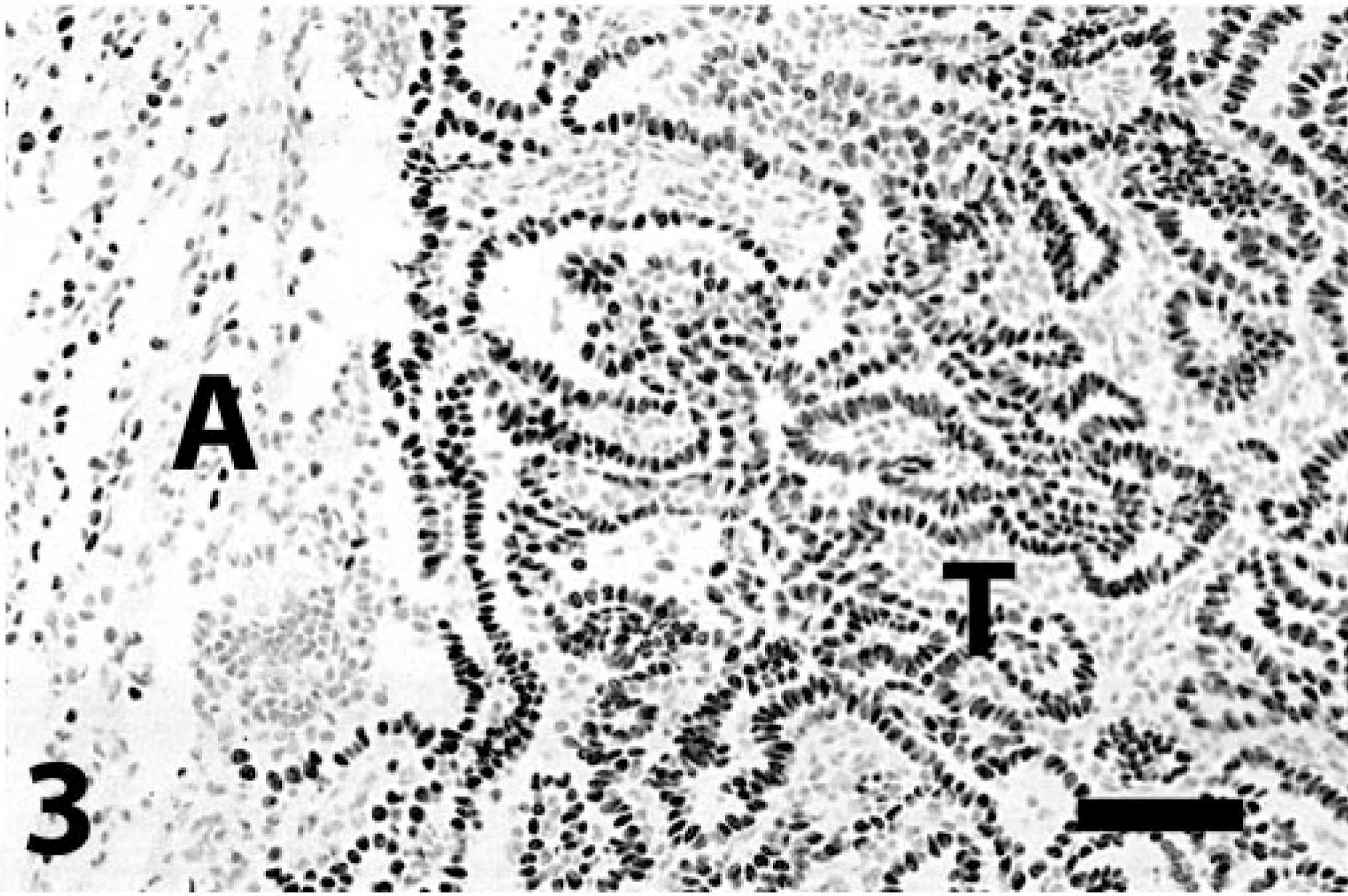
Lung; bronchioloalveolar carcinoma; dog. Detail of Fig. 2. T = tumor cells. Immunoreactivity is also present in alveolar epithelium (A). Immunoperoxidase-DAB for TTF-1. Bar = 65 μm.
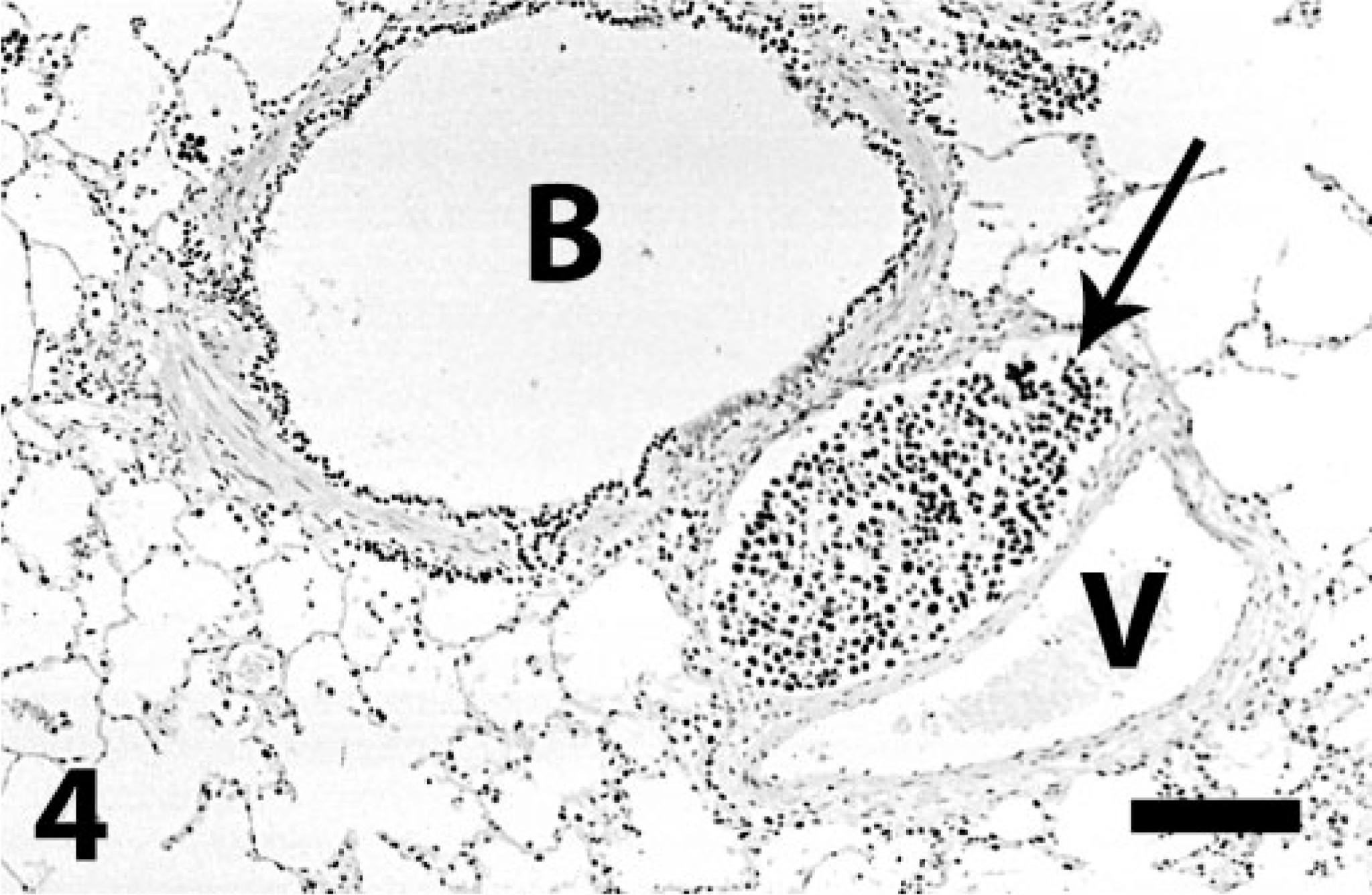
Lung; dog. Intravascular neoplastic emboli (arrow) of pulmonary carcinoma highly reactive to TTF-1. Positive epithelial cells in bronchiole (B). Note adjacent vein (V). Immunoperoxidase for TTF-1. Bar = 130 μm.
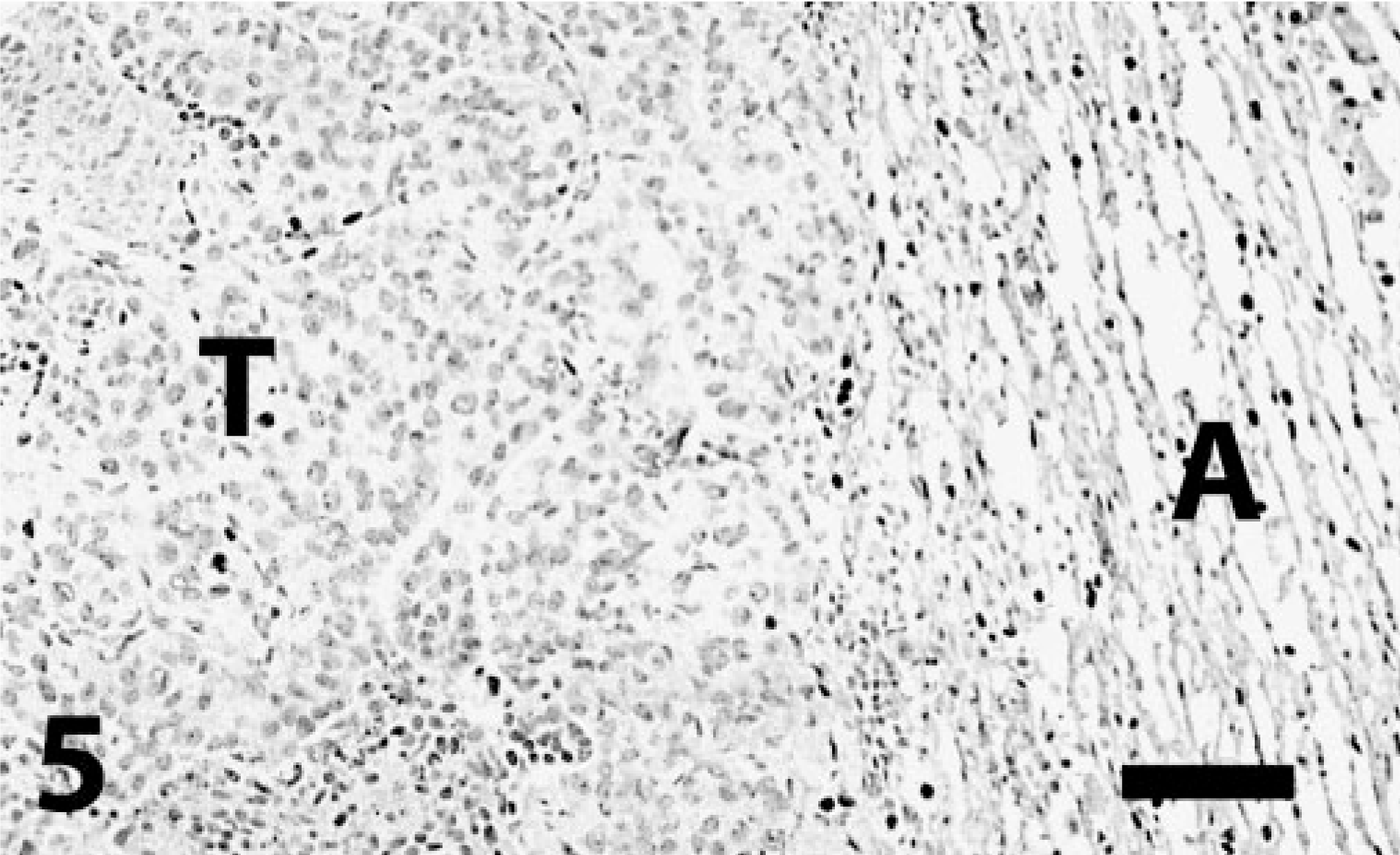
Lung; transitional cell carcinoma metastatic from urinary bladder; dog. Neoplastic cells (T) are not reactive to TTF-1 antibody. Some entrapped alveolar cells and alveolar epithelial cells in the nonneoplastic adjacent pulmonary parenchyma (A) have nuclear staining. Immunoperoxidase-DAB for TTF-1. Bar = 130 μm.
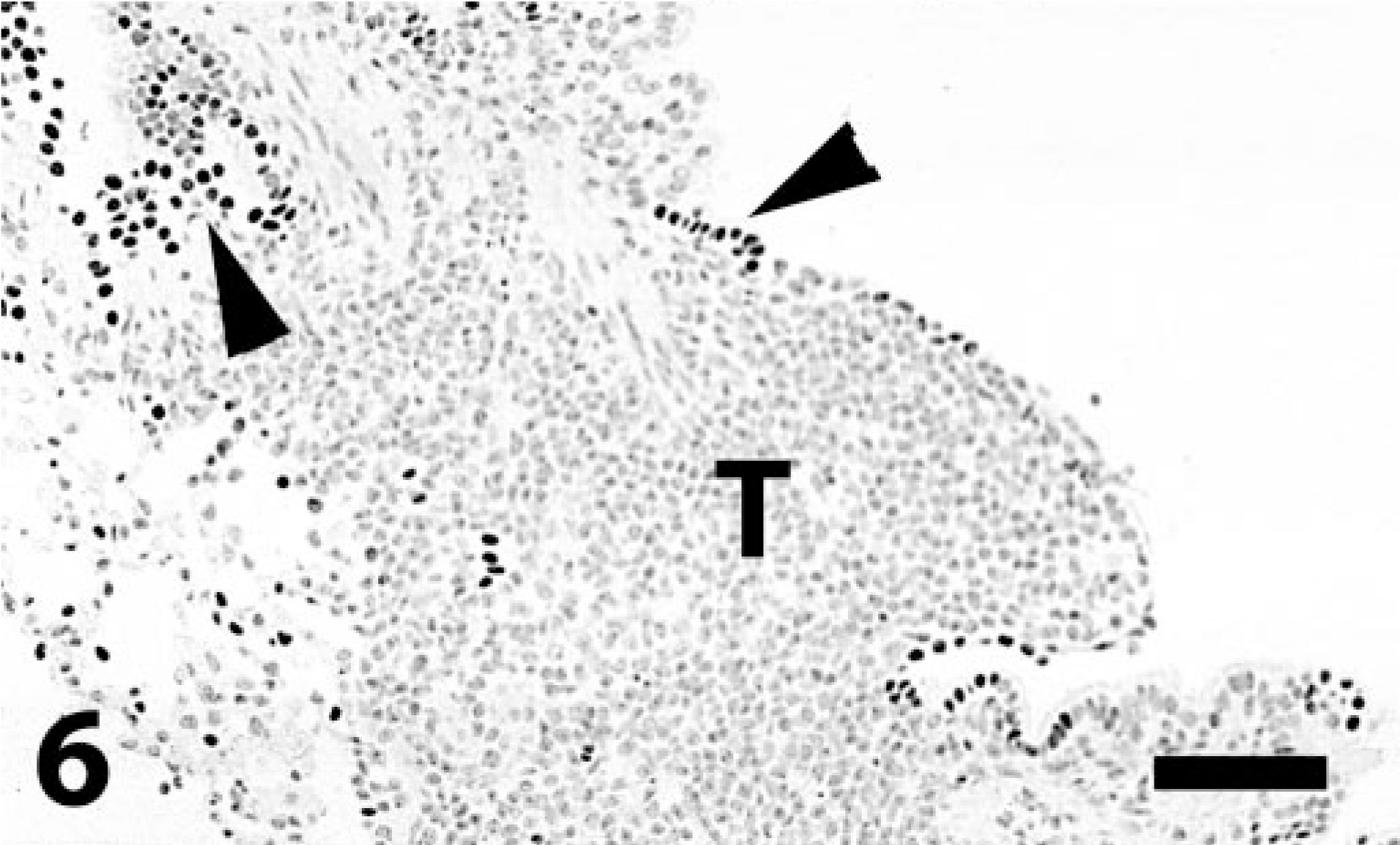
Lung; lymphosarcoma; dog. Tumor cells (T) abutting an airway are negative. Occasional cells in the respiratory epithelium (arrowheads) are strongly positive. Immunoperoxidase-DAB for TTF-1. Bar = 130 μm.
There was no significant reduction in the number of immunostained cells with MoAb to TTF-1 after prolonged fixation up to 24 days, although both the number of positive cells and the intensity of the reaction diminished slightly after seven days of fixation. Eight of 11 primary pulmonary epithelial tumors stored in paraffin for 7–8 years (the oldest blocks used in this study) did not react with MoAb to TTF-1 regardless of histologic classification.
Discussion
We examined the immunoreactivity of a MoAb to TTF-1 in formalin-fixed, paraffin-embedded canine pulmonary primary and metastatic neoplasms. We used a mouse MoAb to rat TTF-1 proven to react with human pulmonary neoplasms. 15 Reactivity with canine lung tissues was expected on the basis of our experience with TTF-1 in canine thyroid tumors and the fact that human, canine, and rat TTF-1 have 98% homology. 16 TTF-1 is expressed in some bronchiolar respiratory epithelial (Clara) cells and pneumocytes, particularly type II. 10, 16, 20, 21, 36, 42 TTF-1 is also expressed as early as the 11th week of gestation in the human fetus in which it is localized in bronchial and nonciliated bronchiolar epithelial cells. 16, 22 TTF-1 immunohistochemistry is a highly specific and sensitive method in the differential diagnosis of primary lung carcinomas and metastatic carcinomas to the lung in humans. 14, 17, 21, 24, 25, 33, 34 TTF-1 immunohistochemistry is more sensitive than other pulmonary markers such as surfactant proteins. 3, 20 However, reported TTF-1 reactivity varies depending on the specific tumor type and the author. 21 Lung adenocarcinomas: 62–100%; 1, 3, 8, 14, 17, 24, 26, 33, 42, 43 non–small cell carcinomas: 24–49%; 3, 10, 37 small cell carcinoma: 81–100%; 8, 10, 11, 19, 37, 43 squamous cell carcinoma: 0–18%; 8, 14, 26, 37, 43 carcinoid: 25–35%; 11, 14 and mucinous carcinoma: 25%. 12 As reported in humans, 26 we only detected TTF-1 immunoreactivity in the glandular component of the sole positive adenosquamous carcinoma in our series. Although there are differences between the histologic classification of human and canine pulmonary tumors, on the basis of literature on humans and our results, it appears that pulmonary adenocarcinomas (particularly bronchogenic and bronchioloalveolar types) express TTF-1 protein much more commonly than nonglandular epithelial tumors. We cannot completely rule out that some of the tumors classified as pulmonary carcinoma or adenocarcinoma were metastases from extrapulmonary sites; the assessment of primary pulmonary neoplasia was based on the lack of extrapulmonary masses.
TTF-1 immunoreactivity is similar between human primary and metastatic pulmonary carcinomas 25, 42 and correlates with the histologic subtype of the primary tumor. 35 We only examined one metastatic pulmonary carcinoma to a lymph node, its reactivity was slightly inferior to that observed in the primary tumor.
None of the canine nonpulmonary metastatic carcinomas, with the exception of thyroid carcinomas, or any of the mesenchymal tumors examined were positive for TTF-1. The human literature reports lack of TTF-1 immunoreactivity for nonpulmonary metastatic carcinomas. 17, 21, 24, 33 Only rarely has TTF-1 immunoreactivity been observed in metastatic nonpulmonary tumors including ovarian carcinoma, 14 small cell carcinomas, 2, 19 and carcinoids. 19 Thyroid carcinomas are consistently immunoreactive for TTF-1. 24 Mesotheliomas were consistently negative for TTF-1 in our study in agreement with the lack of staining for TTF-1 in human mesotheliomas. 1, 14, 20, 21, 43
TTF-1 is a nuclear protein. However, TTF-1 immunohistochemical cytoplasmic staining has been reported in both pulmonary and nonpulmonary adenocarcinomas. 4, 14, 24, 40 Cytoplasmic reactivity is usually considered nonsignificant although it is a consistent finding in hepatocellular carcinomas and more frequently detected than more specific hepatocellular markers such as hepatocyte antigen. 40 TTF-1 cytoplasmic staining has been proposed as a method to distinguish primary hepatocellular tumors from those metastatic to the liver. 40 In our study, we did not identify cytoplasmic staining even in cases with strong nuclear reactivity for TTF-1.
A salient finding in this study was the loss of reactivity for MoAb to TTF-1 in 7- to 8-year-old paraffin blocks. Loss of immunoreactivity in archival tissue is rarely examined, but there is evidence that, at least for some nuclear antigens, it can happen. 7 Additional studies with more tissues stored for prolonged periods of time need to be performed to determine the significance of our observations.
In summary, MoAb 8G7G3/1 to TTF-1 is highly specific (with the exception of thyroid tumors) and moderately sensitive for the immunohistochemical characterization of routinely processed canine primary pulmonary neoplasms, particularly of bronchogenic and bronchioloalveolar origin.
