Abstract
Immunohistochemistry for thyroid transcription factor-1 (TTF-1), thyroglobulin, and calcitonin was done in normal, hyperplastic, and neoplastic canine thyroid glands that had been fixed in formalin and embedded in paraffin. Prolonged fixation did not significantly alter the immunostaining for TTF-1. Staining for TTF-1 was always nuclear and usually strong. One of two C-cell adenomas, five of five follicular cell adenomas, 5 of 11 C-cell carcinomas, 38 of 42 follicular cell carcinomas, two of five cases of C-cell hyperplasia, two of two cases of follicular epithelial hyperplasia, one of two metastatic C-cell carcinomas, and three of four metastatic follicular carcinomas were positive for TTF-1. One follicular carcinoma that was positive for TTF-1 was negative for thyroglobulin. Thirty-nine of 42 follicular cell carcinomas were positive for thyroglobulin, including two cases that were negative for TTF-1. All C-cell lesions were positive for calcitonin. Of a variety of normal canine tissues and 278 nonthyroid tumors, only normal lung (airway and alveolar epithelial cells) and four of five pulmonary carcinomas were positive for TTF-1. TTF-1 is a good marker of thyroid differentiation and can be used in conjunction with thyroglobulin and calcitonin to increase the detection and differentiation of thyroid tumors and their metastases.
Keywords
Thyroid tumors are uncommon in the dog and account for 1.2–4% of all canine tumors in dogs.30 The thyroid gland has two main cell types: thyroglobulin-producing cells (follicular cells) and calcitonin-producing cells (C-cells or parafollicular cells). The most common thyroid tumors in domestic species are of follicular origin except in bulls, in which C-cell tumors are more common.5,6 In dogs, follicular cell carcinomas are more common than adenomas.18,30 Metastases are fairly common in thyroid neoplasms, mainly to the lungs and regional lymph nodes.18,26 Immunohistochemical characterization of thyroid cells is done with antibodies raised against hormones secreted by this gland (i.e., thyroglobulin, and calcitonin).7 Other markers have been used, including calcitonin gene-related peptide, chromogranin A, synaptophysin, cytokeratins, and S-100 protein.2,7,15,27 Several reports have been made of immunohistochemical characterization of canine thyroid neoplasms, both of thyroglobulin-producing cells,1,20,27,28 and calcitonin-producing cells.27,31 Calcitonin is present in 70–100% of canine medullary carcinomas and thyroglobulin is detected in 90–100% of thyroid carcinomas.27,28,31
Thyroid transcription factor-1 (TTF-1), also known as thyroid-specific enhancer-binding protein or NKX-2.1, is a member of the NKX-2 gene family of the homeodomain-containing transcription nuclear factors that plays an important role in tissue morphogenesis and cytodifferentiation.32 TTF-1 is a 38-kda nuclear protein that has been detected in the thyroid, brain, and lung during early embryogenesis and mainly in thyroid cells and bronchioloalveolar cells after birth.17,25,32,36 In the lung, TTF-1 activates surfactant proteins and Clara cell secretory protein gene promoters.3 In the thyroid gland, TTF-1 activates thyroglobulin, thyroperoxidase, thyrotropin (thyroid-stimulating hormone) receptor, thyroid peroxidase, major histocompatibility complex class I, and thyroid Na+/I− symporter genes transcription.8,9,12,24,35 TTF-1 has been detected in normal thyroglobulin-producing cells and C-cells from the thyroid and in parathyroid cells from different species.21–23,37,38 Recently, antibodies against TTF-1 have been used to characterize human thyroid follicular neoplasms by immunohistochemistry.2,13,21,23,29 TTF-1 has also been reported in human C-cell tumors.2,19,23
We hypothesized that TTF-1 would be a more sensitive marker of thyroid follicular tumors than is thyroglobulin. The purpose of this study was to characterize the immunoreactivity of monoclonal antibody (MoAb) 8G7G3/1 to TTF-1 in canine normal, hyperplastic, and neoplastic thyroid tissues fixed in formalin and embedded in paraffin and to compare TTF-1 immunoreactivity with that of other thyroid markers (thyroglobulin and calcitonin). In addition, the specificity of this antibody for thyroid cells and their tumors was determined by immunostaining a variety of nonthyroid canine neoplasms and normal tissues.
Materials and Methods
Selection of cases
A total of 60 dogs with a diagnosis of thyroid neoplasia were selected from the database of the University of Missouri–Veterinary Medical Diagnostic Laboratory. Tissues had been fixed in 10% neutral buffered formalin for an undetermined period of time and embedded in paraffin. Paraffin blocks had been stored from several days to more than 9 years. Hematoxylin and Eosin–stained sections of these tumors were examined and tumors were classified by one author (JAR) following established criteria for thyroid neoplasms without knowledge of the previous diagnosis (Table 1).6,31 Those cases in which the morphologic diagnosis was not definitive were reviewed by at least two other pathologists (MAM, GCJ, LWP) to establish consensus. When multiple samples of the same tumor were available, only one block was selected for immunohistochemical studies. An additional six cases of metastatic thyroid neoplasia and seven hyperplastic thyroid lesions were included in this study. To determine possible cross-reaction of MoAb to TTF-1, an additional 278 canine tumors not associated with the thyroid gland were immunohistochemically stained with MoAb to TTF-1. These tumors included 11 adrenocortical carcinomas, 2 adrenocortical hyperplasias, 3 apocrine gland carcinomas, 18 basal cell tumors, 4 chemodectomas, 3 fibrosarcomas, 3 granular cell tumors, 7 granulosa cell tumors, 5 hair follicle tumors, 2 hemangiopericytomas, 8 hemangiosarcomas, 1 hepatocellular adenoma, 17 hepatocellular carcinomas, 6 hepatoid gland tumors, 6 histiocytomas, 11 interstitial cell tumors of the testis, 17 intestinal carcinomas, 3 smooth muscle tumors, 1 islet cell tumor, 7 lymphosarcomas, 2 malignant fibrous histiocytomas, 7 malignant peripheral nerve sheath tumors, 2 mammary tumors, 11 mast cell tumors (various grades), 4 melanomas, 6 mesotheliomas, 6 metastatic carcinomas, 2 myxosarcomas, 1 nasal carcinoma, 6 osteosarcomas, 2 ovarian carcinomas, 3 pancreatic carcinomas, 2 pheochromocytomas, 6 plasmacytomas, 2 prostatic carcinomas, 5 pulmonary carcinomas, 17 rectal polyps, 9 salivary carcinomas, 7 sebaceous cell tumors, 10 seminomas, 10 Sertoli cell tumors of the testis, 6 soft tissue sarcomas, 7 squamous cell carcinomas, 1 stromal tumor of the intestine, and 9 transitional cell carcinomas. Canine normal tissues were also immunostained for TTF-1.
Reactivity of hyperplastic and neoplastic thyroid with antibodies to thyroid transcription factor-1 (TTF-1), thyroglobulin, and calcitonin.
Number of positive cases/total number tested. The number in parentheses indicates percentage of positive cases.
Reagents and immunohistochemical procedure
All thyroid tumors were immunohistochemically stained for thyroglobulin, calcitonin, and TTF-1. The following primary antibodies were used: antibody to calcitonin (A0576, rabbit polyclonal, diluted at 2.25 µg protein/m1, DAKO Corporation), antibody to thyroglobulin (A0251, rabbit polyclonal, diluted at 30 µg protein/ml, DAKO Corporation), and mouse MoAb to thyroid transcription factor-1 (M3675, diluted at 0.54 µg immunoglobulin G/ml, DAKO Corporation). Antigen retrieval for calcitonin employed steam heat with citrate buffer, pH 6.0, 20 minutes at 90–95 C.33 A 10-minute incubation at room temperature of slides with ready-to-use proteinase K was used for thyroglobulin.33 For TTF-1, steam heat on ethylenediametetraacetic acid buffer, pH 8.0, 40 minutes at 90–95 C, was the antigen retrieval method of choice after comparing results with proteinase K and steam heat on citrate buffer, pH 6.0, at different incubation times.34 Slides were cooled for 20 minutes after steam heat or were put directly in Tris buffer after proteinase K treatment. The remaining incubations were done at room temperature. The primary antibodies to thyroglobulin and calcitonin were incubated for 30 minutes; incubation with MoAb to TTF-1 was for 60 minutes. For calcitonin and thyroglobulin, a labeled streptavidin-biotin-peroxidase detection method (K0689, DAKO Corporation) was used; for TTF-1, EnVision+–peroxidase (K4000, DAKO Corporation) was used. Both detection methods followed manufacturer's recommendations. Diaminobenzidine was used as chromagen. For negative controls, an unrelated MoAb of the same isotype and similar protein concentration was used instead of the primary MoAb; for polyclonal antibodies, normal rabbit serum was used instead of the primary antibody.
Scoring of immunohistochemical results with MoAb to TTF-1
A semiquantitative (estimated) evaluation of the percentage of positive tumor cells was used in which 1 indicated 5–10% of positive cells, 2 was 11–50% of positive cells, 3 was 51–80% of positive cells, and 4 was more than 80% of positive cells. A subjective grading system was used to determine the intensity of the reaction for TTF-1 in which 0 indicated no reaction, 1 was a weak reaction, 2 was a moderate reaction, and 3 was an intense reaction.
Effect of prolonged fixation for detection of TTF-1
Because the fixation time of most tumors was unknown, normal thyroid tissue was fixed for 24 hours to determine the effects of prolonged fixation on immunohistochemistry. The tissue stained positively for TTF-1, mostly in follicular epithelium. Additional samples from this organ were fixed for 2, 3, 4, 5, 6, 7, 10, 13, 17, and 24 days before processing. Immunostaining was compared among samples by semiquantitative evaluation of percentage of positive cells and subjective grading of intensity for TTF-1.
Results
Cases selected
Sixty thyroid tumors were examined microscopically (Table 1), including 5 follicular adenomas (2 cystic), 42 follicular carcinomas (14 compact-cellular, 2 mixed cell type, 3 papillary, 11 follicular, 11 follicular-compact cellular, and 1 small-cell type), 2 C-cell adenomas (classic pattern), and 11 C-cell carcinomas (4 classic pattern, 2 trabecular, 4 mixed, and 1 clear). The mixed type of follicular carcinoma consisted of variable amounts of neoplastic follicular epithelium mixed with mesenchymal components, mainly cartilage, bone, or both. The classic pattern of C-cell (medullary) neoplasms was that of tight packets of polygonal cells with ample eosinophilic and finely granular cytoplasm, round nucleus with reticulated chromatin and nucleoli, and delicate stroma (neuroendocrine pattern).31 Some C-cell tumors had prominent trabeculae of connective tissue separating neoplastic cells. Attempts to demonstrate amyloid with special stains were not made. Mitotic activity in most of the tumors examined was nonexistent or less than one mitosis per high-power field. Also included in this study were two follicular cell hyperplasias, five C-cell hyperplasias, four metastatic follicular carcinomas (three lung and one diaphragm), and two metastatic C-cell carcinomas (lymph node). The tumor subtypes were similar in thyroid follicular carcinomas and their metastases (two follicular and two compact-cellular) and in C-cell carcinomas and their metastases (one clear cell and one trabecular type). Most tumors were encapsulated. Malignancy was defined by the presence of tumor invasion of the capsule, extracapsular neoplastic cells within vessels, regional or distant metastases, or a combination of these.
Immunohistochemical results on thyroid hyperplastic and neoplastic samples
The staining with TTF-1 was always diffusely nuclear, never cytoplasmic. Tumors reacting to MoAb to TTF-1 were five (100%) of five follicular cell adenomas (Fig. 1), one (50%) of two C-cell adenomas, five (45%) of 11 C-cell carcinomas, 38 (90.5%) of 42 follicular carcinomas (Fig. 2), one (50%) of two metastatic C-cell carcinomas, and three (75%) of four metastatic follicular carcinomas. Intravascular emboli of thyroid carcinoma were also positive for TTF-1. Also, two (100%) of two follicular cell hyperplasias and two (40%) of five C-cell hyperplasias reacted with MoAb to TTF-1. Thirty-one (74%) thyroid carcinomas had more than 50% positive cells; five (83%) of C-cell carcinomas had more than 50% positive cells for TTF-1. Four follicular carcinomas were negative for TTF-1, including one compact-cellular, two follicular, and one papillary cell types. The percent positive cells in metastatic follicular carcinomas was low but similar to that of the primary tumors except for one metastasis that was negative although the primary tumor was positive. More than 80% cells stained for TTF-1 in the only metastatic C-cell carcinoma, which was slightly higher than the primary tumor. Staining for TTF-1 did not always correlate with staining for thyroglobulin (thyroid follicular tumors) or for calcitonin (medullary tumors).
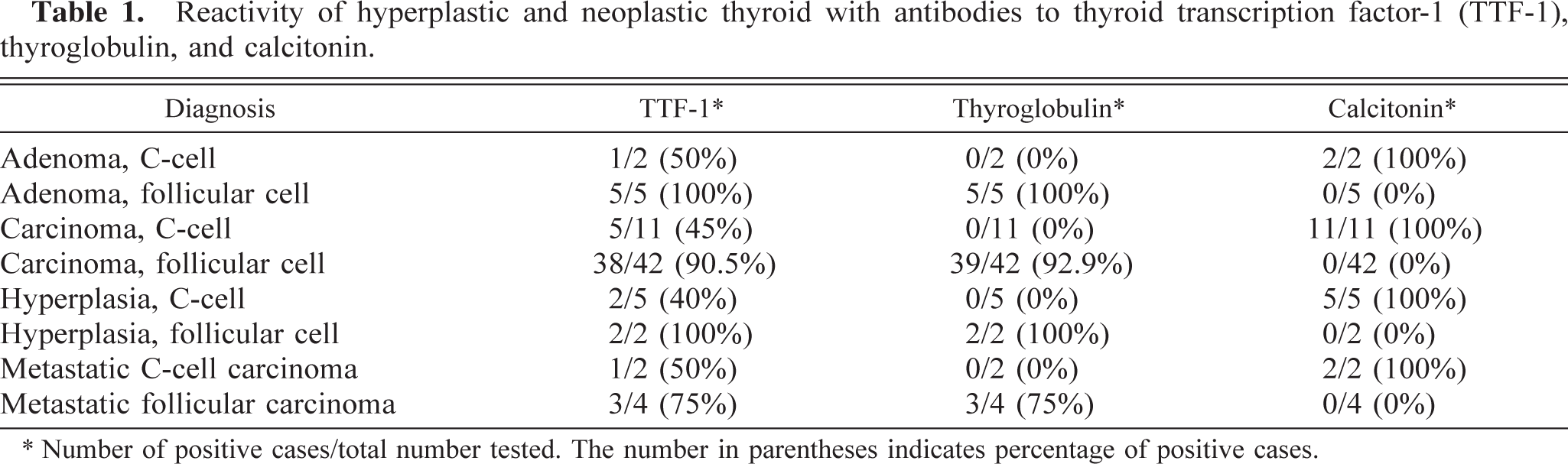
Thyroid; follicular adenoma; dog. Most neoplastic follicular cells have strong (dark) nuclear staining for thyroid transcription factor-1. Negative nuclei are light gray. EnVision+–peroxidase stain with Mayer's hematoxylin counterstain. Bar = 64 μm.
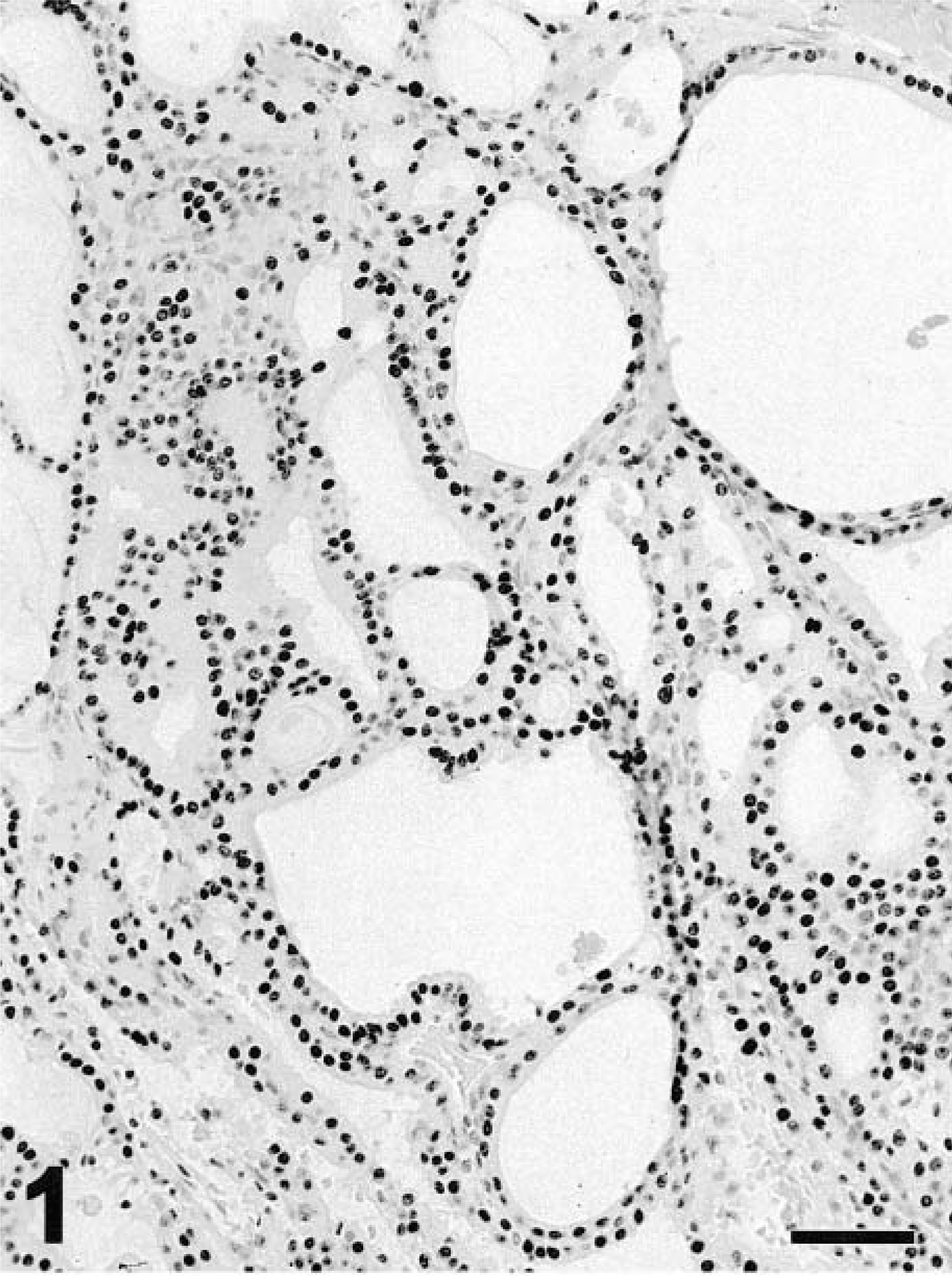
Thyroid; follicular carcinoma; dog. The stain for thyroid transcription factor-1 is nuclear and diffuse. EnVision+–peroxidase stain with Mayer's hematoxylin counterstain. Bar = 32 μm. Inset. Detail of nuclear staining. EnVision+–peroxidase stain. Bar = 12 μm.
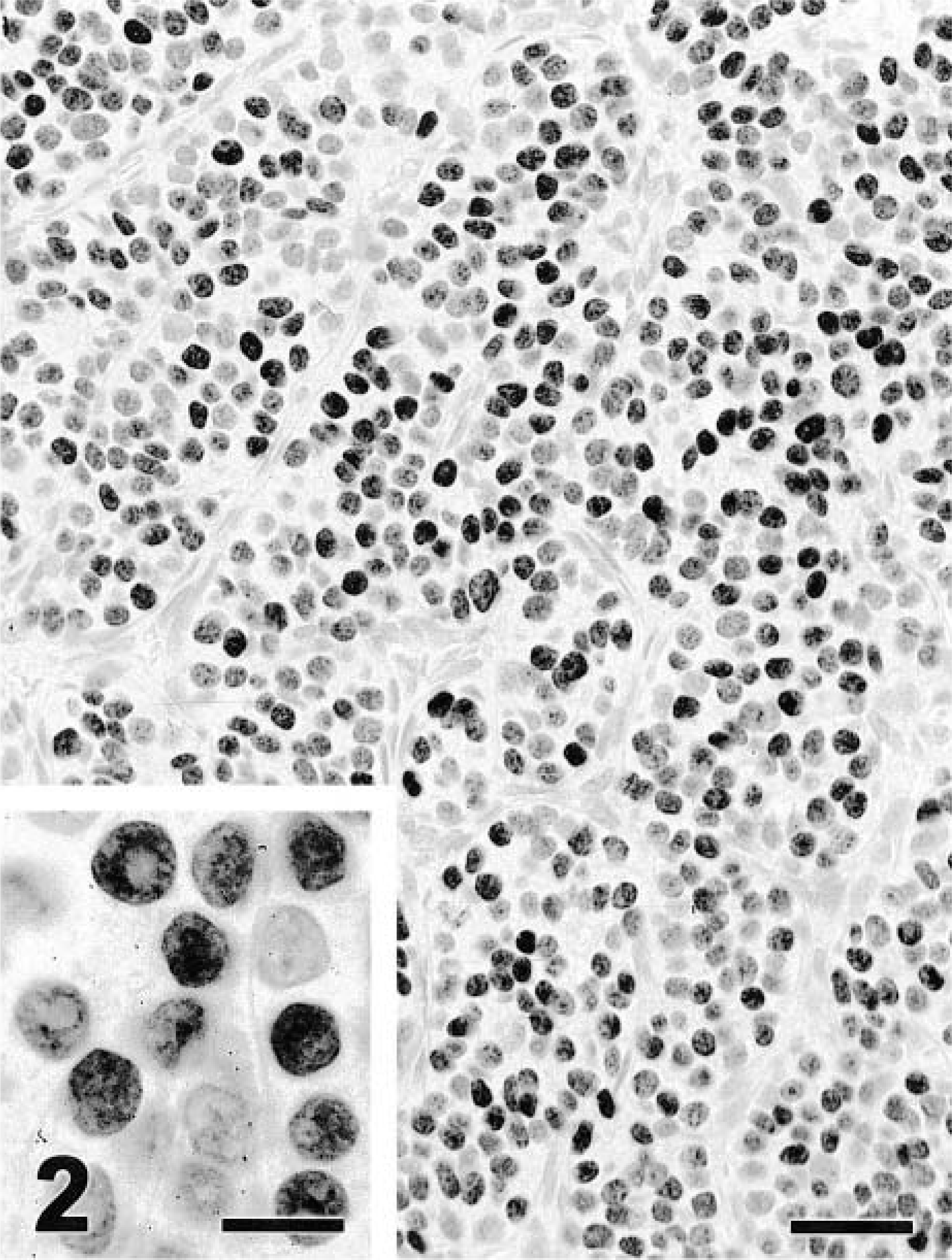
Staining for thyroglobulin was always cytoplasmic, and usually diffuse (Fig. 3), but occasionally the apical cytoplasm stained more strongly. Percent of thyroglobulin-positive cells was similar or higher to that for TTF-1 staining except in four cases. Two thyroid carcinomas negative for TTF-1 also were negative for thyroglobulin. However, another two TTF-1–negative thyroid carcinomas were strongly positive for thyroglobulin. In one carcinoma with follicular and solid components, TTF-1 was restricted mainly to solid areas, whereas thyroglobulin was limited almost exclusively to areas with follicular differentiation. Thyroglobulin antibody usually gave abundant background staining in the tumor stroma and vascular lumina.
Thyroid; follicular carcinoma; dog. Variable cytoplasmic staining for thyroglobulin. Streptavidin–peroxidase stain with Mayer's hematoxylin counterstain. Bar = 32 μm.
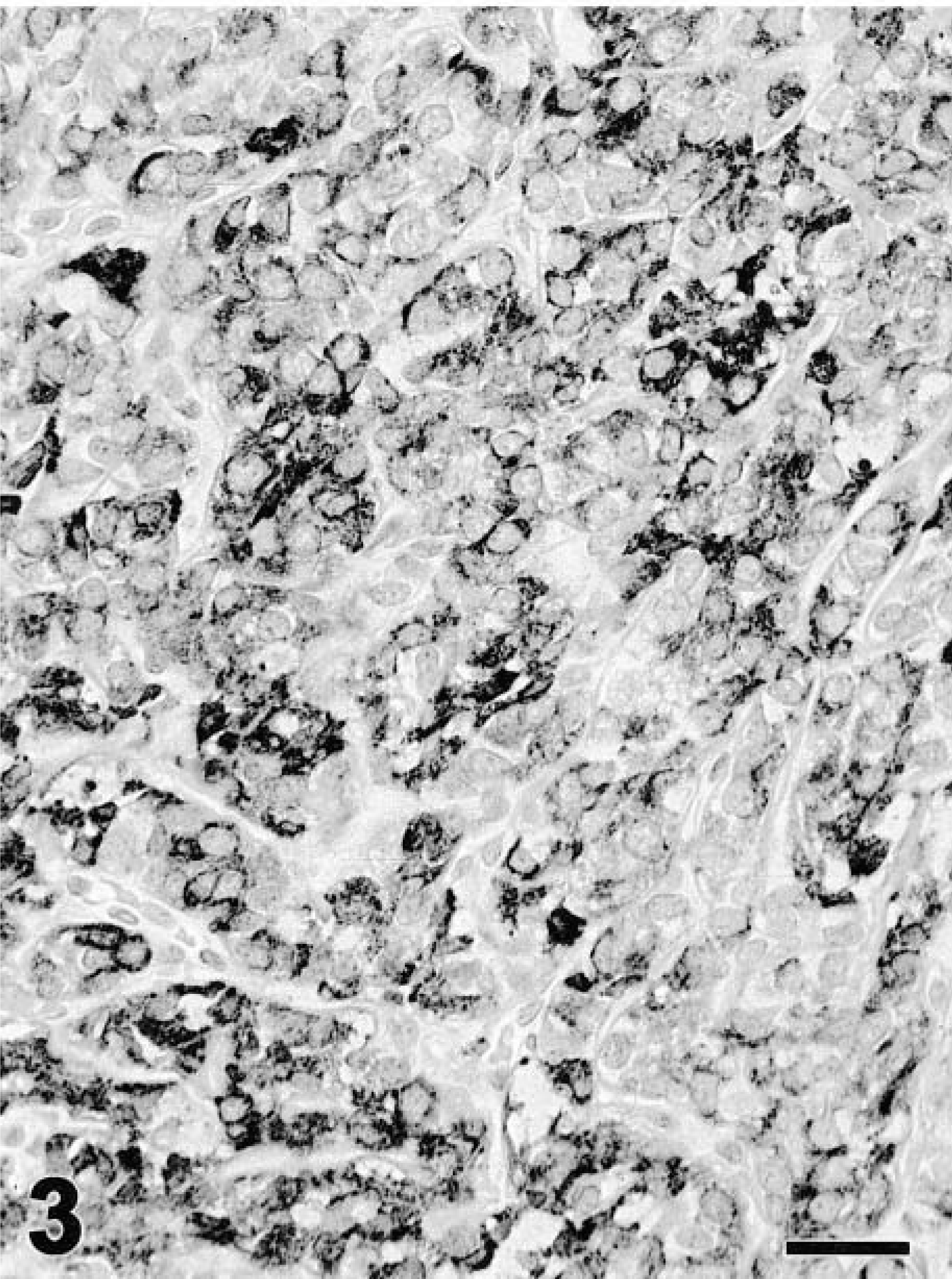
Staining for calcitonin usually was diffusely cytoplasmic and in all cases more than 50% of cells were positive. Staining intensity usually varied among cells within a tumor. Multifocally, one C-cell carcinoma had characteristic punctate cytoplasmic staining (Fig. 4). In two cases in which neoplastic cells formed large nodules, the neoplastic cells in the outer layer were perpendicularly oriented to the surrounding stroma and were the main cells labeled with antibody to calcitonin. When compared with TTF-1, calcitonin stained a similar or higher number of cells. Both TTF-1 and calcitonin antibodies stained more than 80% of cells in the metastatic C-cell carcinoma that was positive for both markers (Fig. 5). The other metastasis was strongly positive for calcitonin but negative for TTF-1.
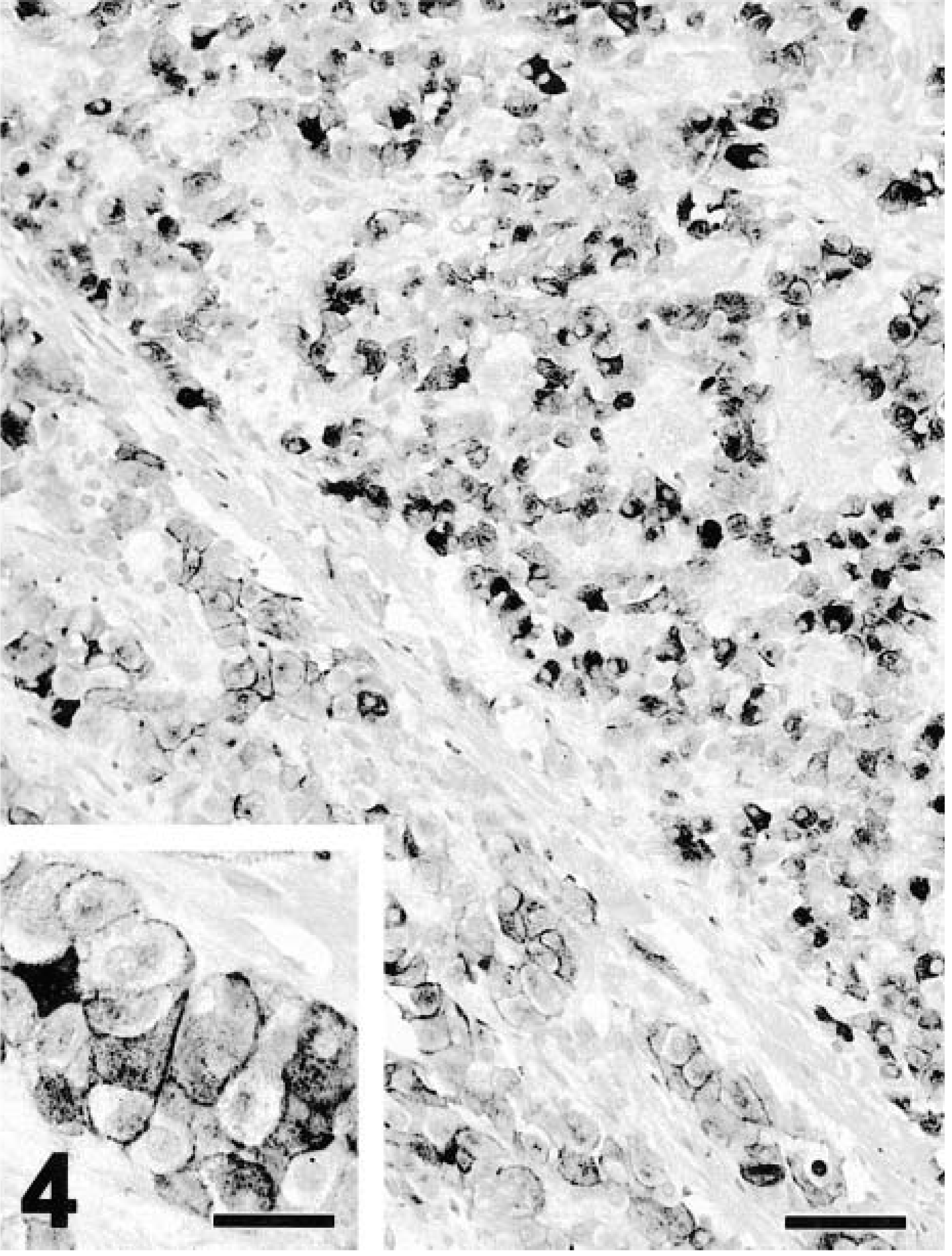
Thyroid; C-cell carcinoma; dog. The staining for calcitonin in the upper right half of the figure is cytoplasmic and diffuse; in the lower left half, it is polar. Streptavidin–peroxidase stain with Mayer's hematoxylin counterstain. Bar = 64 μm. Inset. Detail of the cytoplasmic staining. Streptavidin–peroxidase stain with Mayer's hematoxylin counterstain. Bar = 32 μm.
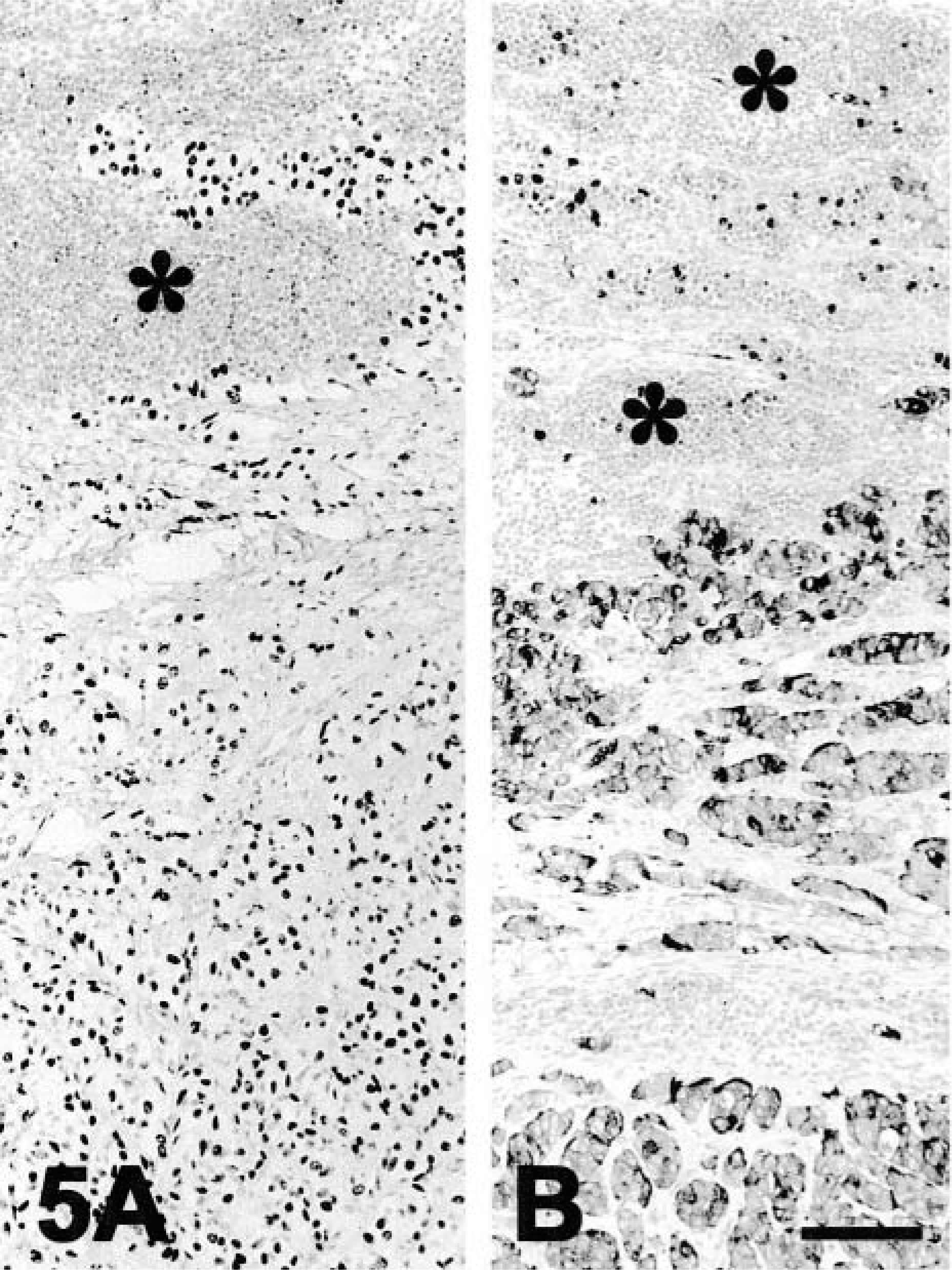
Cervical lymph node; metastasis of C-cell carcinoma; dog.
One tumor originally diagnosed as chemodectoma was classified as thyroid carcinoma based on its strong immunostaining for thyroglobulin. This tumor also was strongly positive for TTF-1. Another nine thyroid masses and one metastasis originally diagnosed as thyroid carcinoma were reclassified as C-cell carcinomas based on the strong staining (>50% cells) for calcitonin and no staining for thyroglobulin. Four of these tumors were positive for TTF-1.
Significant reduction in the number of immunostained cells with MoAb to TTF-1 was not observed with prolonged fixation for the duration of testing (24 days).
Immunohistochemistry of TTF-1 on nonthyroid neoplasms and normal tissues
Nonthyroid tumors included carcinomas and sarcomas from different organs as well as gonadal, endocrine, and nervous tumors. Of 278 nonthyroid tumors, only four (80%) of five pulmonary adenocarcinomas reacted with MoAb to TTF-1. Neoplastic cells had strong nuclear staining (Fig. 6). The number of positive cells ranged from 20% to almost 100%. Different regions of the same tumor had variable numbers of positive cells. Lung and thyroid were the only normal tissues with reactivity for TTF-1. In the lung, some bronchiolar and alveolar epithelial cells were strongly reactive for TTF-1. In the thyroid gland, most follicular epithelial cells were positive for TTF-1. Significant staining was seldom observed in thyroid C-cells.
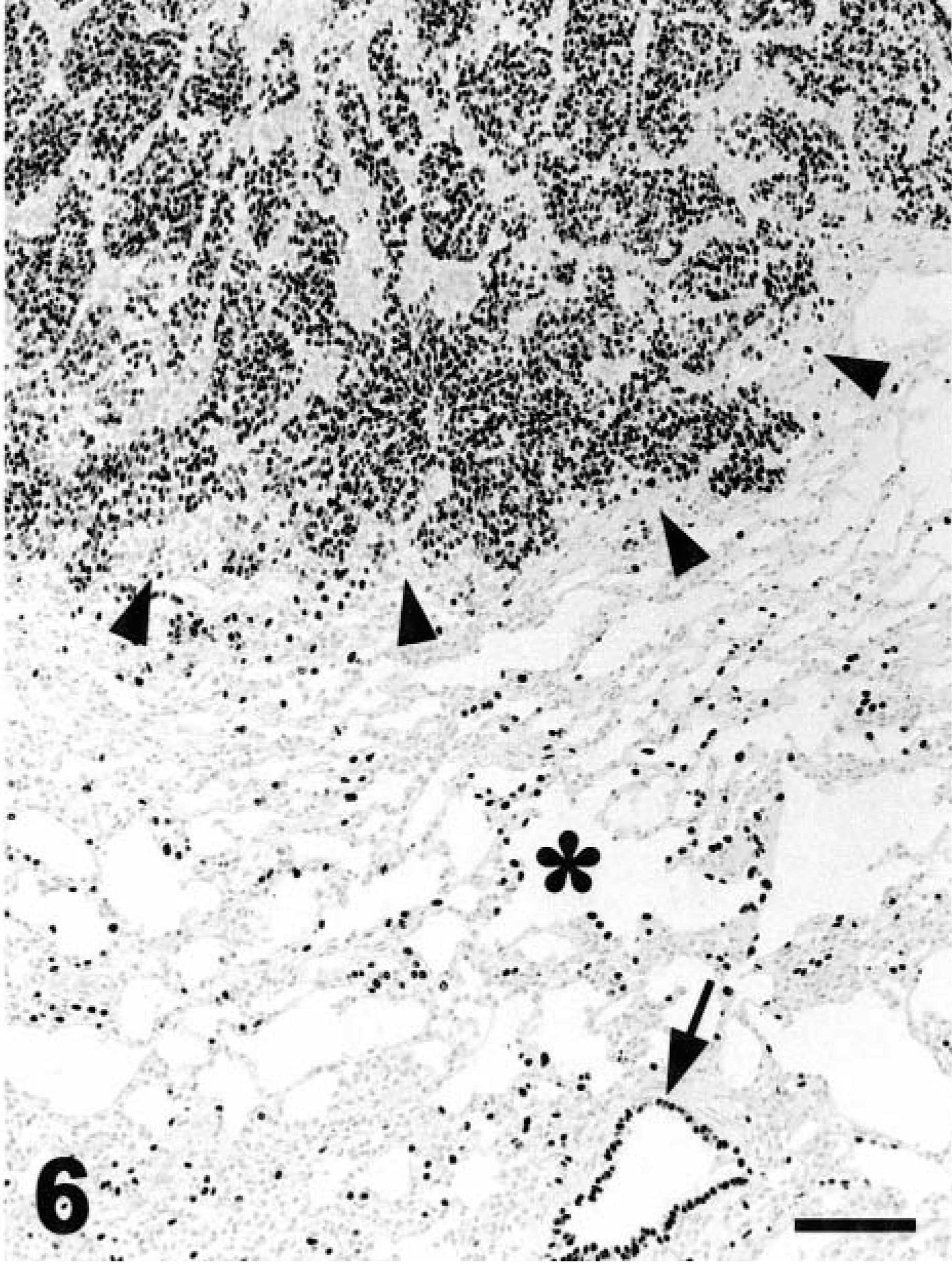
Lung; pulmonary carcinoma; dog. A pulmonary nodule (arrowheads) is strongly positive for thyroid transcription factor-1 (dark nuclei). Normal alveolar epithelium (asterisk) and bronchiolar epithelium (arrow) have TTF-1–positive cells. EnVision +–peroxidase stain with Mayer's hematoxylin counterstain. Bar = 130 μm.
Discussion
By using immunohistochemical methods, thyroid transcription factor-1 (TTF-1) was detected in formalin-fixed, paraffin-embedded normal, hyperplastic, and neoplastic canine thyroid samples. The reactivity in normal, hyperplastic, and benign neoplastic thyroid follicular epithelium was 100%. Ninety percent of thyroid follicular carcinomas expressed TTF-1. The percentage of human thyroid tumors expressing TTF-1 varies from 74 to 100%, with approximately 75% of metastatic carcinomas expressing TTF-1.2,13,21 Although two of the tumors negative for TTF-1 and thyroglobulin were anaplastic and had rare follicular structures, it was not clear whether the degree of differentiation of follicular carcinomas influenced the detection of TTF-1 because few anaplastic neoplasms were examined. The other tumors negative for TTF-1 were classified as follicular (one) and papillary (one) carcinomas. Human anaplastic thyroid carcinomas have minimal or no expression of TTF-1.2,13 As with some tumors in our series, TTF-1 and thyroglobulin markers do not colocalize in all human thyroid carcinomas.22
Normal, hyperplastic, and neoplastic canine C-cell epithelium also reacted with MoAb to TTF-1 but in fewer cases. Although originally TTF-1 was only reported in thyroid follicular cells and their tumors, different laboratories have described TTF-1 in C-cells and parathyroid cells by immunohistochemistry, in situ hybridization, and polymerase chain reaction.23,37 This finding has prompted the hypothesis that TTF-1 may play a role in the regulation of genes important in calcium homeostasis.37 In our series, 45% of C-cell carcinomas and 50% of C-cell carcinoma metastases reacted with MoAb to TTF-1. In human C-cell (medullary) carcinomas, demonstration of TTF-1 can reach 100% of cases.23 We did not detect expression of TTF-1 in the two normal or hyperplastic canine parathyroid glands examined.
When comparing the reactivity of TTF-1 with the other markers examined, thyroglobulin and calcitonin were more sensitivive in detecting thyroid follicular carcinomas and C-cell neoplasms, respectively. However, some thyroid carcinomas were not reactive for thyroglobulin but expressed TTF-1. The sensitivity for thyroglobulin and TTF-1 alone for thyroid carcinomas and their metastases was 90.5% and 92.9%, respectively. When combined, TTF-1 and thyroglobulin had a slightly increased sensitivity of 95.2%. This increased sensitivity whith dual markers has also been reported for human neoplasms2 and suggests that combined use of TTF-1 and thyroglobulin will increase the chances of detection of thyroid follicular neoplasms. The reported sensitivity of thyroglobulin to detect primary thyroid carcinomas is high (90–100%), although only 50% of metastases are detected by this marker.27,28 TTF-1 also was expressed by 46% canine C-cell neoplasms, which makes this marker unsuitable by itself to discriminate between these two tumor types. Calcitonin, on the other hand, was expressed in all C-cell carcinomas and their metastases (100% sensitivity) and was never detected in follicular tumors. These results are similar to those in the literature,31 although lower percentages (67%) have been reported.27 Background staining with antibodies to TTF-1 and calcitonin was not significant. Nuclear staining with TTF-1 made visualization of the reaction easy even when fewer positive cells were present. However, polyclonal antibody to thyroglobulin usually had extensive background staining that occasionally hampered visualization of the reaction. Inadequate or delayed fixation may allow difussion of thyroglobulin into the interstitium. It is noteworthy to mention that 10 thyroid tumors in this series were reclassified based on immunohistochemical results, emphasizing the difficulty of accurate diagnosis in some cases without immunohistochemistry.
The only nonthyroid neoplasm reacting with MoAb to TTF-1 was pulmonary carcinoma. Staining was nuclear in a variable number of cells but always distinct. Normal canine bronchiolar and alveolar epithelial cells also were consistently positive for TTF-1. The diagnostic relevance of this staining is under investigation in our laboratory. Reactivity of human pulmonary neoplasms with antibodies to TTF-1 varies depending on the tumor type.4,10,11,14,16,29,32
In summary, thyroid transcription factor-1 has been detected in thyroid epithelial cells (follicular and C-cell) and their tumors. The sensitivity of TTF-1 in detecting these tumors is slightly lower than with thyroglobulin for thyroid follicular tumors and lower with calcitonin for C-cell tumors but complements immunohistochemical characterization and increases the sensitivity of detection of follicular carcinomas, when used in conjunction with antibodies to thyroglobulin. Because TTF-1 present in pulmonary epithelial cells, this marker should be used in conjunction with thyroglobulin and calcitonin for thyroid carcinomas metastatic to the lung.
Footnotes
Acknowledgements
We thank H. Wilson and D. Connor for photographic preparations.
