Abstract
Thyroid transcription factor-1 (TTF-1) is a specific and sensitive marker for canine pulmonary tumors but is also expressed in thyroid carcinomas, which commonly metastasize to lung. Napsin A and surfactant protein A (SP-A) are used in the histologic diagnosis of non-small-cell lung cancer in humans but have not been thoroughly evaluated in neoplasms of dogs. The objective of this study was to compare the efficacy of immunohistochemistry for SP-A, napsin A, and TTF-1 in the diagnosis of canine pulmonary carcinomas. TTF-1, napsin A, and SP-A antibodies were applied to 67 formalin-fixed, paraffin-embedded canine pulmonary tumors. Although each marker had good sensitivity, only 3% (2/67) of lung tumors were negative for SP-A compared with 7% (5/67) and 9% (6/67) for napsin A and TTF-1, respectively. Each antigen was detected in a greater percentage of cells of tumors with acinar or papillary patterns compared with those with squamous differentiation. SP-A immunoreactivity was absent in all 113 nonpulmonary tumors tested. Of 108 normal tissues, SP-A was detected only in lung and in 1 of 6 adrenal, 1 of 3 endometrial, and 1 of 4 hepatic sections. Based on these findings, SP-A and napsin A are useful markers of canine lung epithelial neoplasia. Of these, SP-A is the most sensitive and specific (a possible pitfall is the need to distinguish entrapped normal pulmonary epithelial cells or alveolar macrophages from neoplastic cells) and can be used in combination with TTF-1 or napsin A to improve detection and differentiation of pulmonary carcinomas from metastatic tumors in the canine lung.
Keywords
The incidence of canine lung cancer increases with age and has a prevalence ranging from 25% in a research colony of aged dogs to 0.1%, based on the total number of dog autopsies in another institution. 6,13,39 Pulmonary carcinomas have a wide array of histologic patterns from acinar to papillary to squamous. 5,7,39 The lung is also a common location for metastatic tumors, many of which can resemble primary neoplasms, further complicating definitive diagnosis. 7 The importance of distinguishing metastatic tumors in the lung from pulmonary carcinoma is underscored by the differences in prognosis and treatment. In dogs with primary pulmonary tumors, lobectomy can result in remission for greater than 1 year. 24 In contrast, metastatic disease of unknown origin requires more intensive diagnostics and treatment and carries a poorer prognosis with an average survival of only 3 months. 28
In people, the development of treatments targeting specific lung tumor types necessitates precise diagnosis. 11,33,36 For example, treatment with tyrosine kinase inhibitors improves survival of patients with lung cancer with certain epidermal growth factor receptor (EGFR) mutations. 17 Mutated tumors frequently express either thyroid transcription factor-1 (TTF-1) or surfactant protein A (SP-A), both of which have been suggested to be surrogate immunohistochemical markers for EGFR mutations. 19,33,36 The EGFR pathway is also reportedly overexpressed in approximately 75% of canine lung tumors, many of which also express TTF-1. 26,29
A combination of immunohistochemical markers, including TTF-1, napsin A, and SP-A, is used to diagnose human pulmonary tumors. 12,18,20 –22,30,34,42 Expressed in normal lung and thyroid, TTF-1 is a nuclear transcription factor detected in more than 70% of human lung tumors. 18,20 –22,30,42 Napsin A, an aspartic proteinase involved in surfactant protein B processing, is detected in 70% to 90% of lung tumors, with higher expression associated with longer survival. 12,18,20,30,34 Immunoreactivity for SP-A is observed in surfactant and in type II pneumocytes and in 45% to 60% of human pulmonary carcinomas. 34,42 Although SP-A is reportedly less sensitive than TTF-1 or napsin A, it was highly specific in 1 study, with immunoreactivity detected in only 1 of 6 colorectal carcinomas of a total of 55 nonpulmonary tumors evaluated. 42
Of the antibodies used in human tissues, TTF-1 is the best characterized in dogs 26 ; however, immunoreactivity is absent in up to 36% of canine pulmonary carcinomas. In addition, TTF-1 is detected in more than 80% of canine thyroid carcinomas, a tumor that commonly metastasizes to the lung. 26,27 Recently, napsin A immunoreactivity was detected in 5 of 5 canine pulmonary carcinomas, normal type II pneumocytes, and some bronchiolar epithelial cells. 25 In humans, napsin A is detected in > 90% of primary pulmonary carcinomas, with reactivity of 100% in adenocarcinomas and no reactivity in squamous cell carcinomas; 16 in dogs, napsin A is expressed in 62% of thyroid neoplasms and in approximately 60% of renal cell carcinomas. 23,25 Immunoreactivity for SP-A has been reported in a single canine pulmonary large-cell carcinoma and in 50% of feline pulmonary carcinomas. 3,5 The purpose of the current study was to evaluate the sensitivity of napsin A and SP-A in comparison with that of TTF-1 in the diagnosis of canine pulmonary carcinomas and to evaluate the immunoreactivity of SP-A in normal canine tissues and nonpulmonary neoplasms.
Materials and Methods
Selection and Classification of Canine Pulmonary Tumors
The databases of Purdue University Animal Disease Diagnostic Laboratory and Colorado State University Veterinary Diagnostic Laboratory were searched for canine cases with a histologic diagnosis of pulmonary carcinoma. Only cases with no masses outside of the lungs were selected for this study. A total of 67 cases were selected based on the quality of samples available for immunohistochemical evaluation. Tissues had been fixed in 10% neutral-buffered formalin for an undetermined period and were embedded in paraffin. Hematoxylin and eosin-stained sections of these tumors were reviewed and reclassified by 4 authors (JB, CF, MAM, JARV) according to adapted criteria (Table 1). 4 This classification system is based on a predominant histologic pattern 31 and, in human lung cancer, is more reproducible and has greater prognostic and therapeutic value. 14,37,41
Predominant Histologic Patterns in 67 Cases of Canine Pulmonary Carcinoma.

Immunohistochemistry
Immunohistochemistry for TTF-1, napsin A, and SP-A was applied to 67 canine pulmonary tumors. The details of antibodies, dilutions, and incubation times are in Table 2. Antigen retrieval for all markers was performed using Diva Decloaker Solution in Medical’s Decloaking Chamber (Biocare Medical, Concord, CA, USA) for 30 seconds at 125ºC and between 18 and 24 PSI. Antibodies for TTF-1, napsin A, and SP-A were mouse monoclonal, rabbit monoclonal, and goat polyclonal, respectively. All incubations were at room temperature. The chromogen for TTF-1 and SP-A was diaminobenzidine (DAB), and for napsin A, DAB with Sparkle (Biocare Medical). In addition to single immunostaining, sequential application of TTF-1 and napsin A antibodies was performed, as reported in human lung cancer. 10 To differentiate TTF-1 and napsin A immunoreactivity in double-stained slides, the chromogens were DAB and Red Warp, respectively. For each of the 3 antibodies used, negative controls were randomly selected sections of pulmonary carcinoma stained with the Biocare Medical polymer negative control serum in place of the primary antibody (Supplemental Figures S1-S3).
Antibody Reagents, Antigen Retrieval, and Detection Systems Used in Immunohistochemistry.

Immunoreactivity Scoring
Immunoreactivity for TTF-1 (nuclear and diffuse) and for napsin A and SP-A (cytoplasmic and granular) was scored as percentage of labeled cells and reaction intensity simultaneously by 3 authors (JB, MAM, JARV). The percentage for each case was scored in the areas with the highest density of positive cells. Typically, evaluation was done with the 10× or 20× objective, using a 10× eyepiece. This evaluation was semiquantitative without a specific number of cells counted. The percentage of labeled tumor cells was scored as 0, 1 (1%–15%), 2 (16%–50%), or 3 (≥ 51%). Reaction intensity was scored as 0 (no reaction), 1 (weak), 2 (moderate), or 3 (intense). A mean score for percentage of cells and for reaction intensity was recorded for each antibody. A score of < 1.0 in both indices (intensity, % cells), consistent with either absent or weak reactivity in rare cells, was considered negative.
Immunoreactivity of SP-A in Nonpulmonary Neoplasms and Normal Canine Tissues
The reactivity of SP-A was investigated in 113 nonpulmonary tumors: adrenocortical carcinoma (3), apocrine carcinoma of the anal sac glands (4), ceruminous carcinoma (4), hepatocellular carcinoma (8), large intestinal carcinoma (6), small intestinal carcinoma (5), mammary carcinoma (6), pancreatic carcinoma (5), parathyroid carcinoma (2), salivary carcinoma (2), sebaceous carcinoma (4), squamous cell carcinoma (4), gastric carcinoma (8), thyroid carcinoma (1), urothelial carcinoma (5), adrenocortical adenoma (1), dysgerminoma (1), granulosa cell tumor (2), hemangiosarcoma (5), histiocytic sarcoma (3), interstitial (Leydig) cell tumor (2), lymphoma (8), melanoma (5), mesothelioma (3), osteosarcoma (3), pheochromocytoma (2), seminoma (4), Sertoli cell tumor (5), thymoma (1), and trichoblastoma (1). In addition, SP-A reactivity was investigated in 108 normal tissues, including adrenal gland, fetal tissues, gall bladder, heart, large intestine, liver, small intestine, lymph node, lung, mammary gland, ovary, pancreas, parathyroid gland, pituitary, prostate, salivary gland, skin, spleen, stomach, testis, thymus, thyroid, tonsil, urinary bladder, and uterus (multiple samples from each tissue).
Effect of Prolonged Fixation on SP-A Immunoreactivity
Because the fixation time of most tumors was unknown, the effects of fixation time on SP-A immunoreactivity were evaluated. Normal canine lung tissue was fixed for 2 to 21 days, using a reported technique. 25,26,38 Immunoreactivity was compared among samples by semiquantitative evaluation of percentage of positive cells. The effect of fixation duration on TTF-1 and napsin A immunoreactivity has already been examined. 25,26
Results
Immunohistochemistry—Normal Tissues
All 3 markers were expressed in normal canine pulmonary tissue. Details of reactivity to napsin A and TTF1 have been reported. 25 –27 Immunoreactivity to SP-A was observed in intraluminal secretions along the apical surface of alveolar cells, within the cytoplasm of bronchiolar epithelial cells and type II pneumocytes, and within alveolar macrophages (Figs. 1, 2). Reactivity of TTF-1 was observed in alveolar and bronchiolar epithelial cells. Napsin A was detected in type II pneumocytes, terminal bronchiolar epithelial cells, and alveolar macrophages. In normal nonpulmonary canine tissues, SP-A immunoreactivity was weak but diffuse in the adrenal medulla (1/6) and endometrium (1/3), weak in a few bronchiolar epithelial cells of the fetal lung (1/1), and cytoplasmic or polar in a few scattered hepatocytes (1/4). When investigating the effects of prolonged fixation on SP-A immunoreactivity, no reduction in the number of SP-A immunoreactive cells was apparent in normal lung tissue fixed in formalin for up to 3 weeks compared with tissues fixed for 2 days.
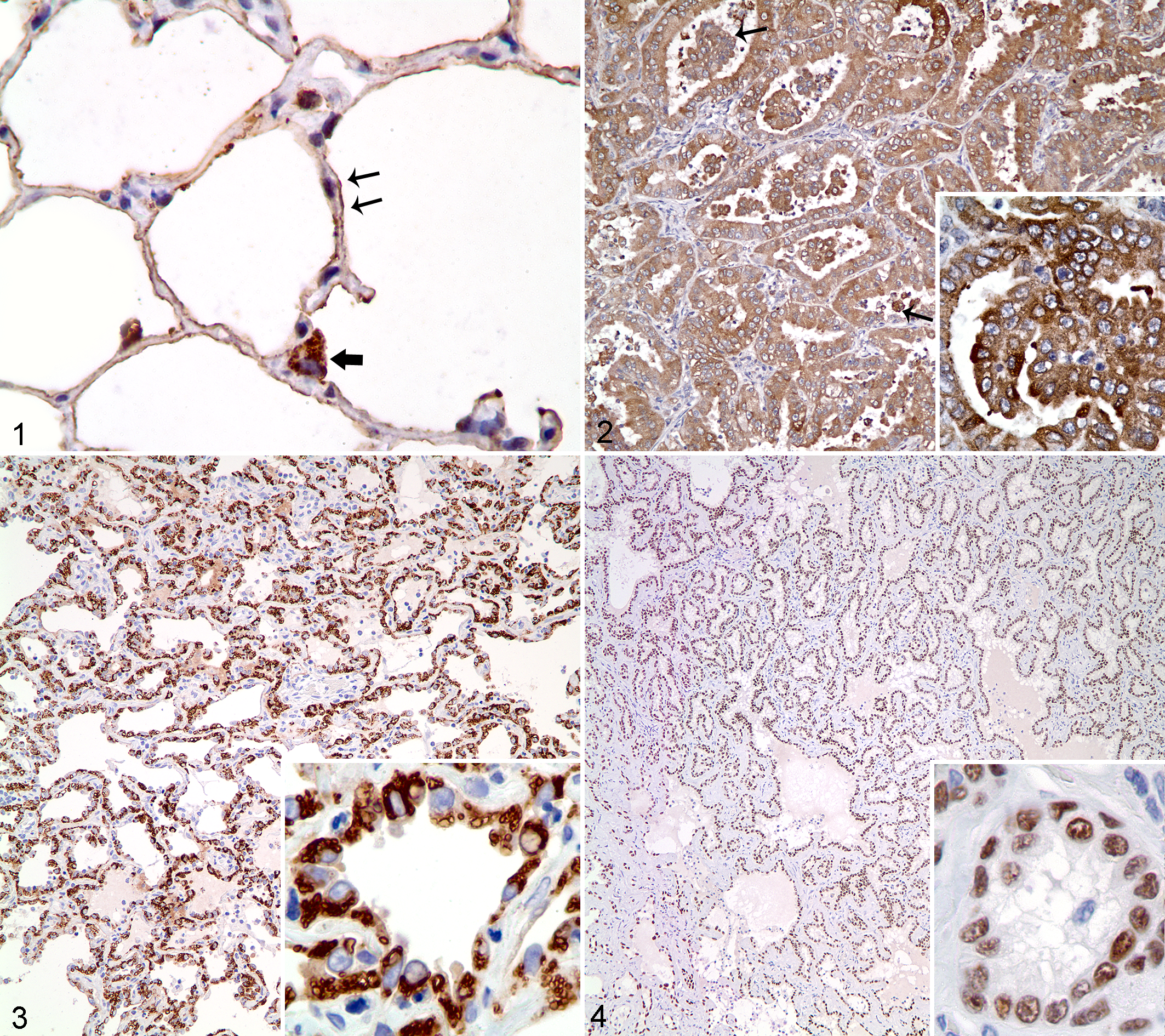
Immunohistochemistry—Neoplastic Tissues
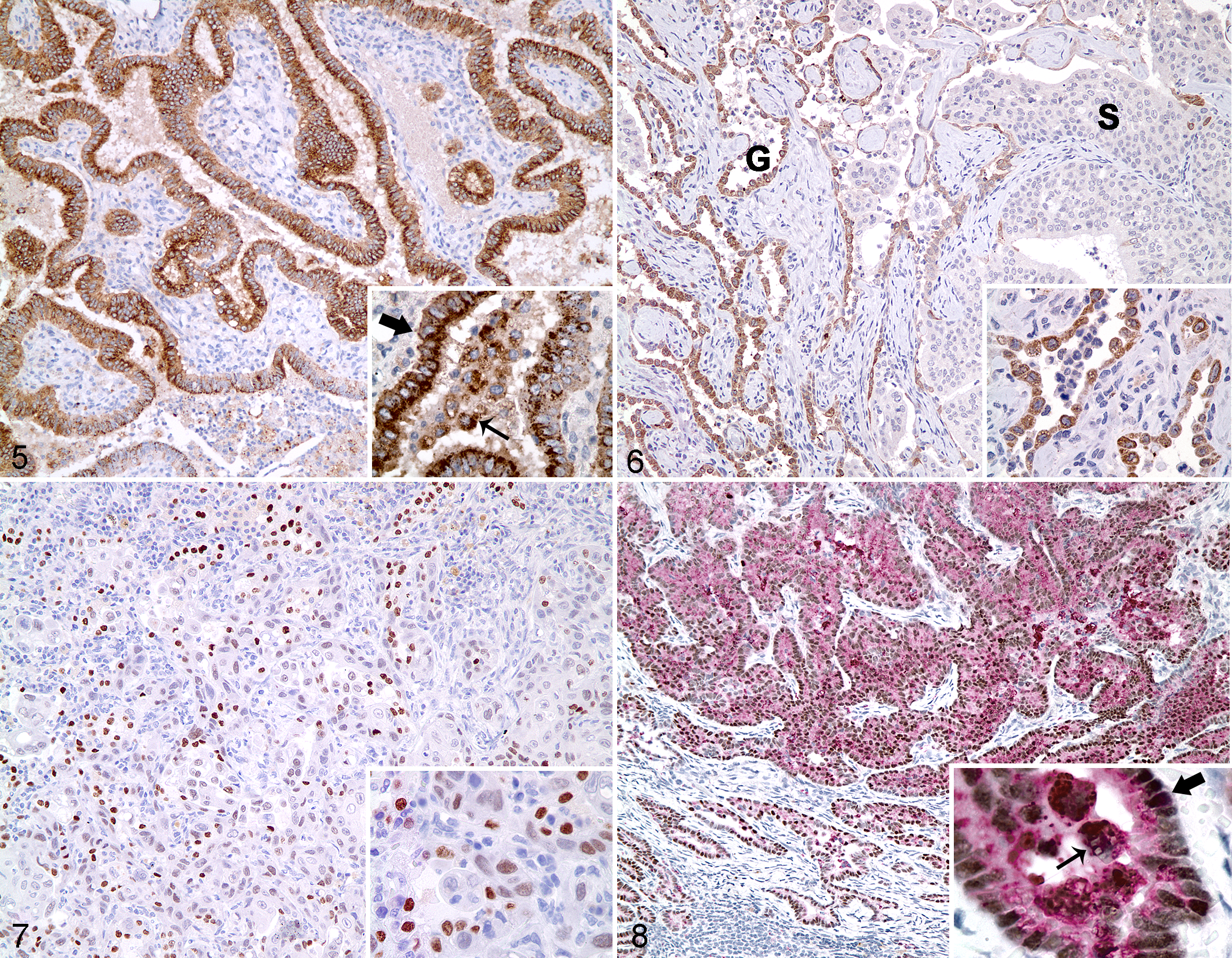
Canine pulmonary carcinomas (n = 67; Table 1) displayed various histologic phenotypes. Immunolabeling was cytoplasmic and granular for SP-A (Fig. 2) and napsin A (Fig. 3), and nuclear and diffuse for TTF-1 (Fig. 4). Reactivity for TTF-1, napsin A, and SP-A varied among histologic patterns and was detected in a higher percentage of cells in papillary (Fig. 5) and lepidic adenocarcinomas, compared with tumors with squamous differentiation (Fig. 6, Table 3). In the single squamous cell carcinoma, moderate TTF-1 reactivity was observed in less than 50% of cells (Fig. 7), whereas napsin A and SP-A were weakly expressed in rare tumor cells. Areas of squamous differentiation in the adenosquamous tumors also had reduced immunoreactivity of SP-A and napsin A.
Immunoreactivity for Surfactant Protein A, Napsin A, and Thyroid Transcription Factor-1 in Canine Pulmonary Carcinomas.
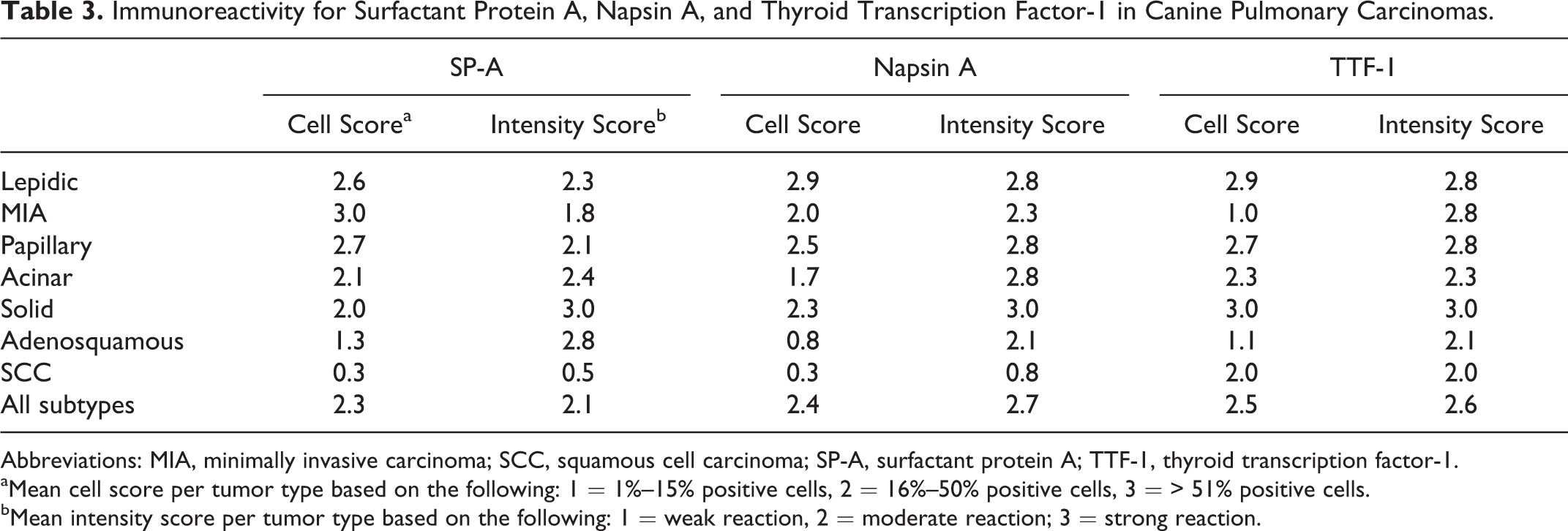
Abbreviations: MIA, minimally invasive carcinoma; SCC, squamous cell carcinoma; SP-A, surfactant protein A; TTF-1, thyroid transcription factor-1.
aMean cell score per tumor type based on the following: 1 = 1%–15% positive cells, 2 = 16%–50% positive cells, 3 = > 51% positive cells.
bMean intensity score per tumor type based on the following: 1 = weak reaction, 2 = moderate reaction; 3 = strong reaction.
The most sensitive marker was SP-A, with detection in 65 of 67 (97%) pulmonary carcinomas. The vast majority of tumors also expressed napsin A (92%; 62/67) and TTF-1 (91%; 61/67). Only 1 pulmonary carcinoma was immunohistochemically negative for all 3 markers. Most tumors expressed all 3 markers (88%; 59/67). Eight tumors were considered negative for at least 1 marker based on scores of < 1.0 (Table 4). Of the tumors that did not express all 3 markers, 6 of 8 (75%), 3 of 8 (38%), and 2 of 8 (25%) were immunoreactive for SP-A, napsin A, and TTF-1, respectively, further demonstrating the slightly higher sensitivity of SP-A compared with napsin A or TTF-1. The combined use of napsin A and TTF-1 increased the sensitivity for pulmonary carcinoma to 95% (64/67) from 91% (61/67) when TTF-1 was used alone. All but 1 tumor (98%) had immunoreactivity for SP-A or TTF-1. The addition of napsin A to an immunohistochemical panel of SP-A and TTF-1 did not further increase the sensitivity.
Eight Canine Pulmonary Carcinomas Negative by Immunohistochemistry for SP-A, Napsin A, and/or TTF-1.a
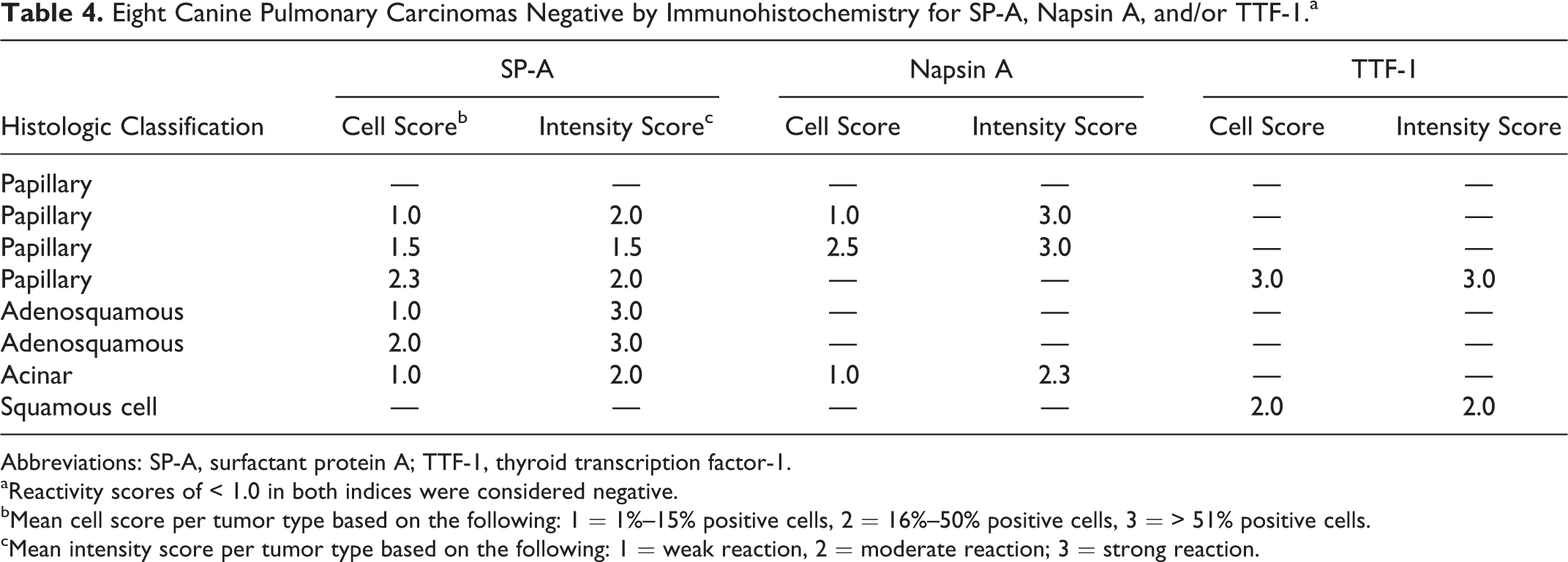
Abbreviations: SP-A, surfactant protein A; TTF-1, thyroid transcription factor-1.
aReactivity scores of < 1.0 in both indices were considered negative.
bMean cell score per tumor type based on the following: 1 = 1%–15% positive cells, 2 = 16%–50% positive cells, 3 = > 51% positive cells.
cMean intensity score per tumor type based on the following: 1 = weak reaction, 2 = moderate reaction; 3 = strong reaction.
Five canine lung tumors expressed either napsin A (n = 3) or TTF-1 (n = 2). In the tumor that was double immunolabeled for napsin A and TTF1, most neoplastic cells coexpressed both markers (Fig. 8).
Variable immunoreactivity within a pulmonary tumor was occasionally observed in areas of necrosis or regions of poor fixation toward the center of large samples. The deleterious effects of necrosis and autolysis on immunoreactivity were most striking for TTF-1.
In normal canine tissues, rare SP-A reactivity was detected in the adrenal gland and liver; however, none of the adrenal gland tumors (n = 4), hepatic tumors (n = 8), or the other 101 nonpulmonary tumors tested expressed SP-A.
Discussion
Detection of TTF-1 has been documented in both human and canine primary pulmonary tumors. 21,26,32,40,42 However, reactivity for TTF-1 in thyroid tumors has prompted the search for additional markers, specifically napsin A and SP-A, with a goal of improved differentiation of primary from metastatic carcinomas in the canine lung. 26 In this study, we characterized the immunoreactivity of SP-A and napsin A in canine pulmonary tumors and compared the sensitivity and specificity of these markers with that of TTF-1.
Consistent with previous reports, 5,25 –27,30,34,42 immunoreactivity for all 3 antibodies was observed in normal canine lung tissue. Immunoreactivity of these antibodies within pulmonary epithelium was used as an internal control, confirming appropriate labeling in examined tissue sections. In addition to reactivity within the pulmonary epithelium, intraluminal SP-A reactivity was interpreted as labeling the surfactant. In several cases, SP-A was also observed in alveolar macrophages, interpreted as phagocytosis of surfactant. Human alveolar macrophages play an important role in surfactant turnover. 1,15 Because both pulmonary epithelium and alveolar macrophages expressed these markers, care is required to distinguish entrapped normal pulmonary epithelial cells or alveolar macrophages from neoplastic cells. This is particularly important near the periphery of neoplasms and in foci of inflammation where nonneoplastic airway epithelium and macrophages were often entrapped in the pulmonary tumors.
Based on this study, TTF-1, napsin A, and SP-A are sensitive markers of canine lung cancer with immunoreactivity of all 3 markers detected in more than 90% of evaluated lung tumors. Only 8 of 67 pulmonary tumors were negative for at least 1 marker. In these 8 tumors, SP-A had the highest sensitivity with detection in 6 tumors.
Combining immunohistochemical markers that target similar cells enhances the detection of primary canine lung tumors. Detection of SP-A is highly specific with no reactivity in nonpulmonary neoplasms and only rare immunoreactivity in normal tissues. In contrast, both TTF-1 and napsin A are expressed in some nonpulmonary tumors. 23,25 It is important that TTF-1 is expressed in most thyroid tumors, which can metastasize to the lung. 27 Similarly, napsin A reactivity was detected in 80% of thyroid tumors (84/105) and in 2 of 5 renal carcinomas in dogs. 23,25 Napsin A reactivity in renal tissue has also been documented in humans, with expression in approximately 40% of clear cell renal carcinomas and in more than 80% of papillary renal carcinoma subtypes. 9,43 In dogs, both of these renal tumor subtypes commonly metastasize to the lung. 8
The majority of tumors expressed all 3 markers. Detection of SP-A within a TTF-1–positive tumor was not surprising as TTF-1 has been found to induce SP-A transcription in murine pneumocytes. 2 The role of napsin A in surfactant maturation suggests that its expression would correlate with that of TTF-1 35 ; this hypothesis is supported by the expression of both markers in most canine pulmonary carcinomas. Sequential application of napsin A and TTF-1 antibodies was performed with concurrent detection of immunoreactivity in the cytoplasm and nucleus of tumor cells, respectively. This technique, albeit more laborious, reduces technical time and tissue requirements for immunohistochemistry and diagnosis. Applying TTF-1 and napsin A to the same human tissue section increases accuracy in lung cancer diagnosis, when compared with using TTF-1 alone. 10 Using both in combination in canine tissues can also increase specificity by distinguishing a napsin A–positive metastatic tumor of renal origin from a pulmonary carcinoma.
Although the reactivity of TTF-1 and napsin A in nonlung tissues is limited, the detection of both in thyroid tumors and of napsin A in renal tumors underscores the importance of using a lung-specific marker to differentiate metastatic thyroid and renal tumors from primary lung tumors. In this study, SP-A immunoreactivity was not observed in normal or neoplastic renal or thyroid tissues, making SP-A a useful marker to complement napsin A and TTF-1. The significance of rare labeled cells with SP-A in the adrenal gland and liver is unknown, and a spurious reaction could not be ruled out. In summary, the sensitivity for pulmonary carcinoma of the 3 markers used (napsin A, SP-A, and TTF-1) is comparable, with a slight superiority for SP-A; however, when this and previous studies are compared, 25 –27 SP-A is the most specific marker for canine pulmonary carcinoma. Based on these findings, SP-A is a good marker of lung origin in the dog and can be used either alone or in combination with TTF-1 and napsin A to improve the differentiation of primary tumors from metastatic carcinomas in the lung.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
